Mushroom Poisoning—A 17 Year Retrospective Study at a Level I University Emergency Department in Switzerland
Abstract
:1. Introduction
2. Materials and Methods
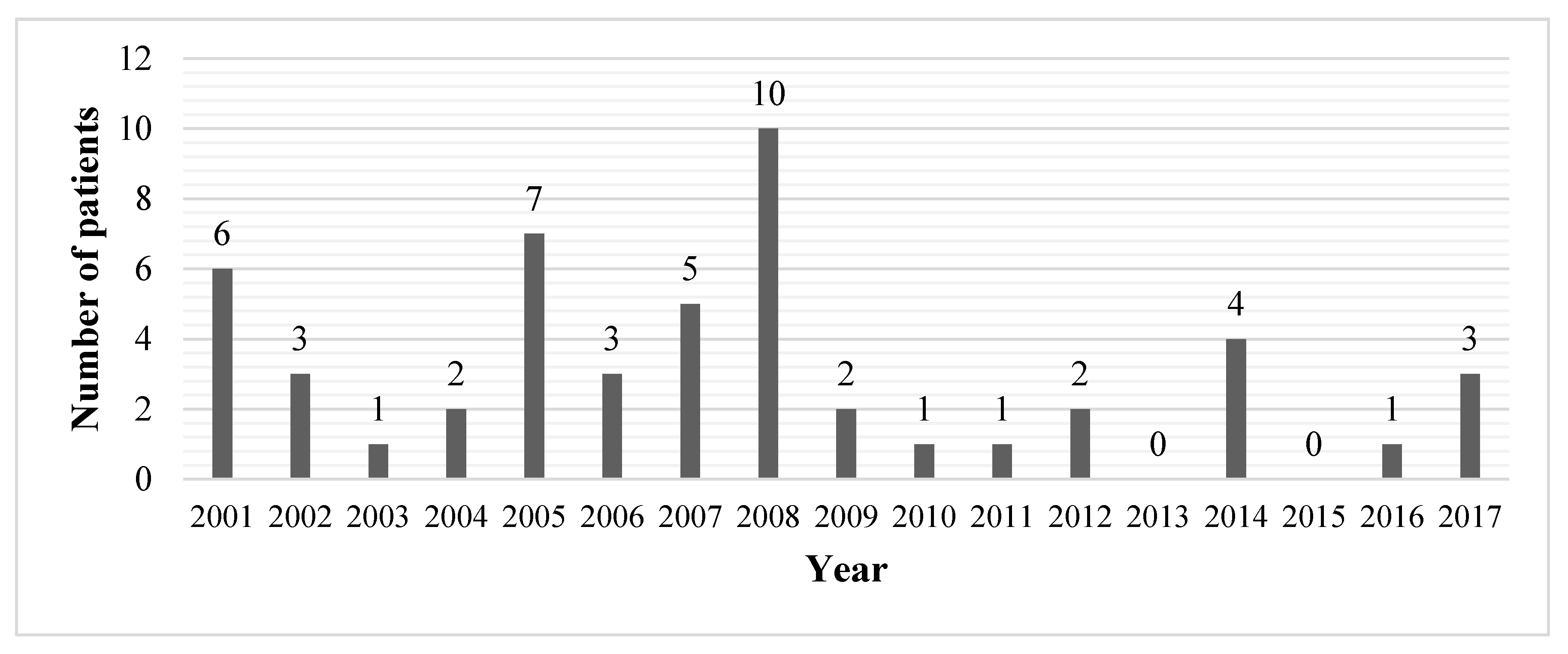
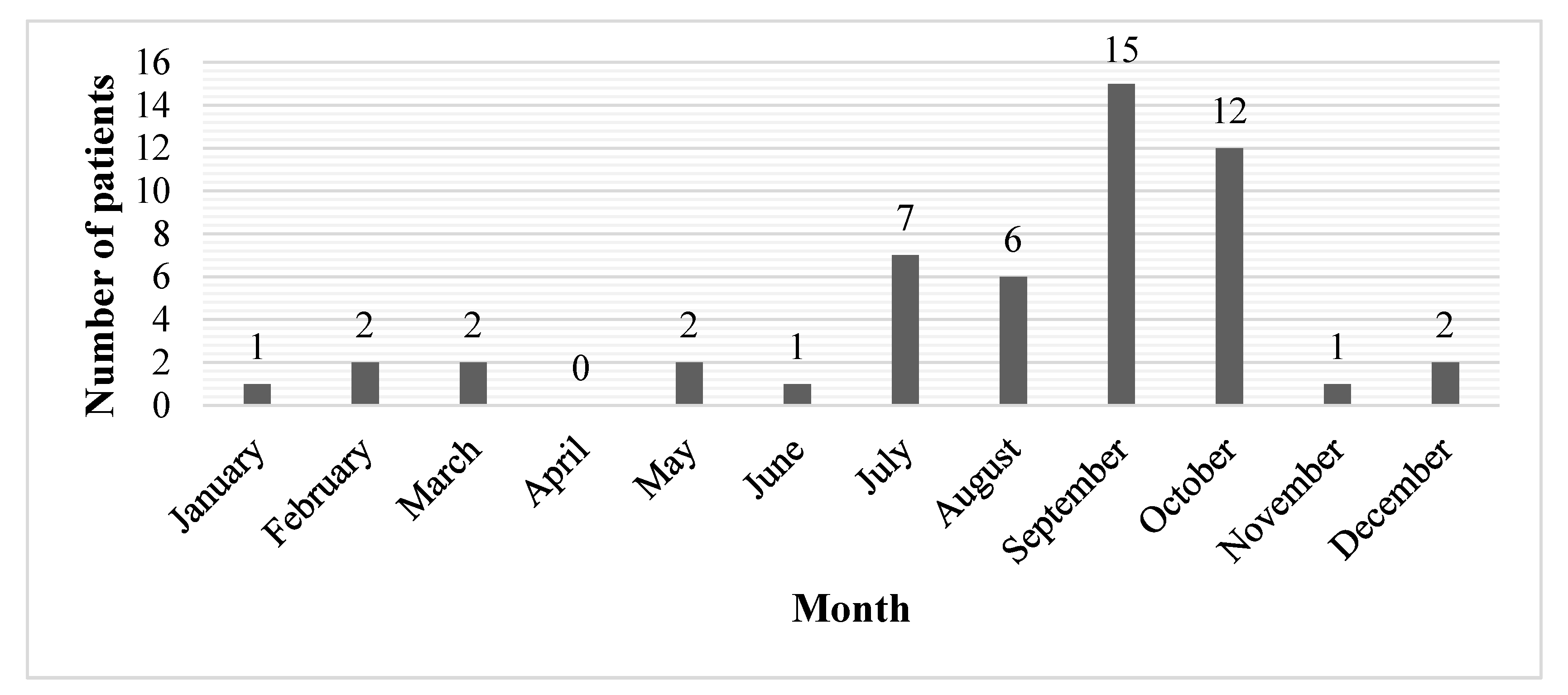
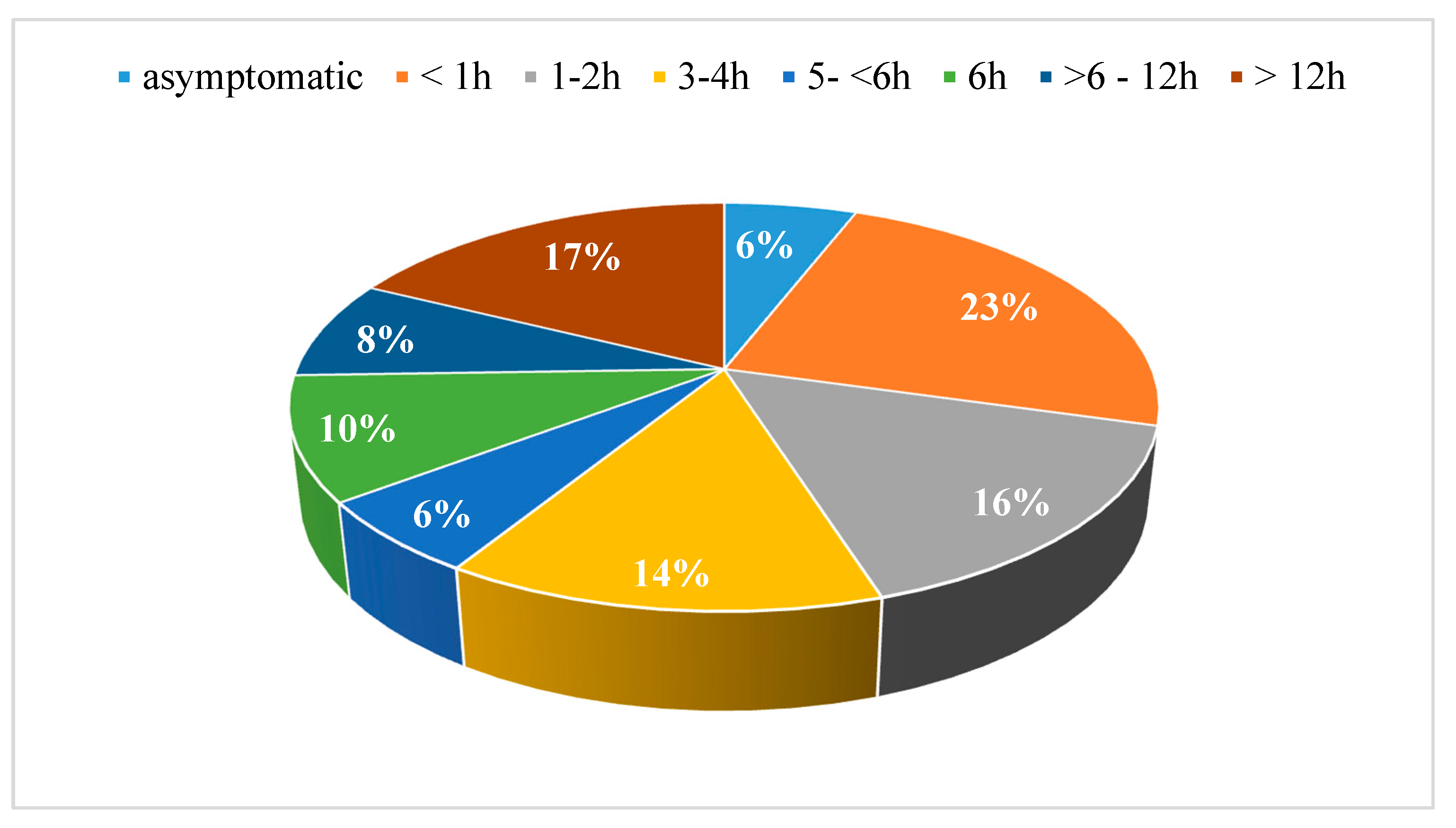
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
Abbreviations
| ALAT | alanine aminotransferase |
| AP | alkaline phosphatase |
| ASAT | aspartate aminotransferase |
| CRP | C-reactive protein |
| DIC | disseminated intravascular coagulation |
| ED | emergency department |
| ELISA | enzyme-linked immunoassay |
| GFR | glomerular filtration rate |
| GGT | gamma-glutamyl transpeptidase |
| h | hour(s) |
| INR | international normalized ratio |
| LC-MS/MS | liquid chromatography-tandem mass spectrometry |
| LDH | lactate dehydrogenase |
| M | man |
| n.a. | not applicable |
| OATP | organic anion transporting polypeptides |
| SD | standard deviation |
| W | woman |
| WBC | white blood cell |
Appendix A
| Questions/Measures | Comments | |
|---|---|---|
| Patient history | Where were the mushrooms collected? | Many toxic mushrooms tend to grow in woodlands; growing season and geographic variation may help identify particular mushrooms. |
| What type of tree was the mushroom near/on? | Amanita phalloides may grow under/near oak or pine trees. | |
| More than one type of mushroom collected? | ||
| What did the mushroom look like? | Description might suggest potential for serious toxicity; pictures of mushrooms can be shown. | |
| How long after ingestion did the symptoms begin? | Symptoms that develop later than six hours after ingestion are often associated with potentially lethal mushroom toxins (exceptions are possible). | |
| How much was eaten? | Information about the amount may be useful in predicting the course and degree of poisoning. | |
| Were the mushrooms eaten at more than one meal? | Symptoms may be the result of an earlier rather than the most recent ingestion. | |
| Were the mushrooms cooked/boiled? | ||
| Duration and type of mushroom storage? | Most mushroom poisonings are caused by edible mushrooms and are due to incorrect processing after harvesting, e.g., long-term storage of mushroom dishes, mushroom storage in plastic bags. | |
| Is everyone who ate the mushrooms ill? Is anyone who did not eat the mushrooms ill? | Help differentiate mushroom poisoning from other types of food poisoning. | |
| Were the mushrooms ingested for recreational purposes? | ||
| Was any ethanol co-ingested? | Certain mushroom species (e.g., Coprinus) can cause a disulfiram-like reaction when ethanol is ingested up to 48–72 h after consumption. | |
| Physical examination | Classification of poisoning based upon clinical presentation (e.g., gastroenteritis, liver failure, seizures, cholinergic poisoning, hallucinations, renal failure) helpful in guiding further treatment | |
| Asservation of mushrooms for analysis | Whenever possible, samples of all ingested mushrooms should be obtained for potential identification by a trained mycologist. Asservation of remainders of dishes, fresh mushrooms, peeling and waste. | Whole mushrooms are preferred, but identification can be made on parts of the mushroom, especially the cap. Further storage is facilitated by wrapping the mushrooms in wax paper, placing it in a paper bag, and refrigerating the sample. Storage in plastic bags should be avoided. |
| Contact with local poison control centre/mycologist | Consultation with a medical toxicologist/ mycologist is advised to determine likely species ingested based on clinical findings, identification of mushrooms, and specific treatment. | For Switzerland: www.toxinfo.ch, www.vapko.ch |
| Laboratory | Complete blood count, prothrombin time (PT), partial thromboplastin time (PTT), electrolytes, calcium, phosphate, urea nitrogen, creatinine, creatine kinase, liver enzymes, albumin, cholestatic parameters | Additionally in symptomatic patients:
|
| Therapy | Emergency and supportive measures:
| Specific drugs and antidotes:Seizures:
|
| Other | If amatoxin-poisoning suspected: identification and hospitalisation of the table companions (even if asymptomatic) |
References
- Berger, K.J.; Guss, D.A. Mycotoxins revisited: Part I. J. Emerg. Med. 2005, 28, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Schenk-Jaeger, K.M.; Rauber-Lüthy, C.; Bodmer, M.; Kupferschmidt, H.; Kullak-Ublick, G.A.; Ceschi, A. Mushroom poisoning: A study on circumstances of exposure and patterns of toxicity. Eur. J. Int. Med. 2012, 23, e85–e91. [Google Scholar] [CrossRef] [PubMed]
- Schmutz, M.; Carron, P.N.; Yersin, B.; Trueb, L. Mushroom poisoning: A retrospective study concerning 11-years of admissions in a Swiss Emergency Department. Intern. Emerg. Med. 2018, 13, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Eren, S.H.; Demirel, Y.; Ugurlu, S.; Korkmaz, I.; Aktas, C.; Guven, F.M. Mushroom poisoning: Retrospective analysis of 294 cases. Clinics 2010, 65, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wang, T. Mushroom poisoning. Ann. Disaster Med. 2004, 3, S8–S11. [Google Scholar]
- Ukwuru, M.U.; Muritala, A.; Eze, L.U. Edible and non-edible wild mushrooms: Nutrition, toxicity and strategies for recognition. J. Clin. Nutr. Metab. 2018, 2. Available online: https://www.scitechnol.com/peer-review/edible-and-nonedible-wild-mushrooms-nutrition-toxicity-and-strategies-for-recognition-dY0s.php?article_id=7395 (accessed on 1 October 2018).
- U.S. Food & Drug Administration, Center for Food Safety & Applied Nutrition. Mushroom Toxins. Available online: https://www.med.navy.mil/sites/nmcphc/Documents/nepmu-6/Epidemiology/FDA-Food-Borne-Pathogens/Natural-Toxins/Mushroom-toxins.pdf (accessed on 1 October 2018).
- Diaz, J.H. Syndromic diagnosis and management of confirmed mushroom poisonings. Crit. Care Med. 2005, 33, 427–436. [Google Scholar] [CrossRef]
- Trueb, L.; Carron, P.N.; Saviuc, P. Intoxication par les champignons. Rev. Med. Suisse 2013, 9, 1465–1472. [Google Scholar]
- Tox Info Suisse: Annual Report 2016. Available online: http://toxinfo.ch/customer/files/638/Tox_JB-2016_140817_DE_ES.pdf (accessed on 1 May 2018).
- Pajoumand, A.; Shadnia, S.; Efricheh, H.; Mandegary, A.; Hassanian-Moghadam, H.; Abdollahi, M. A retro-spective study of mushroom poisoning in Iran. Hum. Exp. Toxicol. 2005, 24, 609–613. [Google Scholar] [CrossRef]
- Unluoglu, I.; Tayfur, M. Mushroom poisoning: An analysis of the data between 1996 and 2000. Eur. J. Emerg. Med. 2003, 10, 23–26. [Google Scholar] [CrossRef]
- Schenk-Jäger, K.M.; Egli, S.; Hanimann, D.; Senn-Irlet, B.; Kupferschmidt, H.; Büntgen, U. Introducing Mushroom Fruiting Patterns from the Swiss National Poisons Information Centre. PLoS ONE 2016, 11, e0162314. [Google Scholar] [CrossRef] [PubMed]
- Tox Info Suisse: Annual Report 2016 (Appendix). Available online: http://toxinfo.ch/jahresberichte-neu_de (accessed on 1 May 2018).
- Ferréol, J.-Y. Champignons Cueillis et Utilisés Pour la Consommation Personnelle—Contrôles Effectués en 2014. Swiss Mushroom Control Association VAPKO. Available online: http://www.vapko.ch/phocadownload/public/FR/La_Vapko/contrle%20des%20rcoltes%20prives%202014%20-%20synthse%20internet.pdf (accessed on 1 July 2018).
- Cervellin, G.; Comelli, I.; Rastelli, G.; Sanchis-Gomar, F.; Negri, F.; De Luca, C.; Lippi, G. Epidemiology and clinics of mushroom poisoning in Northern Italy: A 21-year retrospective analysis. Hum. Exp. Toxicol. 2018, 37, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Helfer, A.G.; Meyer, M.R.; Michely, J.A.; Maurer, H.H. Direct analysis of the mushroom poisons α- and β-amanitin in human urine using a novel on-line turbulent flow chromatography mode coupled to liquid chromatography-high resolution-mass spectrometry/mass spectrometry. J. Chromatogr. A 2014, 1325, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Index Fungorum. Available online: http://www.indexfungorum.org/names/names.asp (accessed on 1 July 2018).
- The Portal of the Swiss Government. Federal Law: Verordnung des EDI über Lebensmittel Pflanzlicher Herkunft, Pilze und Speisesalz. Available online: https://www.admin.ch/opc/de/classified-compilation/20143412/201805010000/817.022.17.pdf (accessed on 1 July 2018).
- Swiss Mushroom Control Association. Recommendation List (Password-Protected Document). Schweizerische Vereinigung Amtlicher Pilzkontrollorgane, VAPKO. Available online: www.vapko.ch (accessed on 19 July 2018).
- Swiss Mushroom Control Association. List of Poisonous Mushrooms. Schweizerische Vereinigung Amtlicher Pilzkontrollorgane, VAPKO. Available online: http://vapko.ch/phocadownload/public/TOUS/Giftpilzliste-Listedeschampignonstoxiques-plusmortels.pdf (accessed on 1 July 2018).
- Bon, M. Pareys Buch der Pilze, 2nd ed.; Kosmos: Stuttgart, Germany, 2005; ISBN 3-440-09970-9. [Google Scholar]
- Breitenbach, J.; Kränzlin, F. Fungi of Switzerland, 2nd ed.; Mycologia Luzern: Luzern, Switzerland, 1984; Volume 1, ISBN 3856042105. [Google Scholar]
- Breitenbach, J.; Kränzlin, F. Fungi of Switzerland, 1st ed.; Mycologia Luzern: Luzern, Switzerland, 1986; Volume 2, ISBN 3856042202. [Google Scholar]
- Breitenbach, J.; Kränzlin, F. Fungi of Switzerland, 1st ed.; Mycologia Luzern: Luzern, Switzerland, 1991; Volume 3, ISBN 385604230X. [Google Scholar]
- Breitenbach, J.; Kränzlin, F. Fungi of Switzerland, 1st ed.; Mycologia Luzern: Luzern, Switzerland, 1994; Volume 4, ISBN 3856042407. [Google Scholar]
- Breitenbach, J.; Kränzlin, F. Fungi of Switzerland, 1st ed.; Mycologia Luzern: Luzern, Switzerland, 2000; Volume 5, ISBN 3856042504. [Google Scholar]
- Breitenbach, J.; Kränzlin, F. Fungi of Switzerland, 1st ed.; Mycologia Luzern: Luzern, Switzerland, 2005; Volume 6, ISBN 3856042601. [Google Scholar]
- Gawlikowski, T.; Romek, M.; Satora, L. Edible mushroom-related poisoning: A study on circumstances of mushroom col-lection, transport, and storage. Hum. Exp. Toxicol. 2015, 34, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Kupferschmidt, H.; Rauber-Lüthy, C. Akute Vergiftungen. In Internistiche Notfälle, 8th ed.; Schoenenberger, R.A., Haefeli, W.E., Schifferli, J., Eds.; Thieme: Stuttgart, Germany, 2008; pp. 490–492. ISBN 9783135106083. [Google Scholar]
- ©2018 UpToDate. Clinical Manifestations and Evaluation of Mushroom Poisoning. Available online: https://www.uptodate.com/contents/clinical-manifestations-and-evaluation-of-mushroom-poisoning?search=mushroom&source=search_result&selectedTitle=1~37&usage_type=default&display_rank=1 (accessed on 1 May 2018).
- Baier, J. Mushrooms and Toadstools: An Illustrated Guide; JG Press: North Dighton, MA, USA, 1995; ISBN 1572151331. [Google Scholar]
- ©2018 UpToDate. Management of Mushroom Poisoning. Available online: https://www.uptodate.com/contents/management-of-mushroom-poisoning?topicRef=13893&source=see_link#H20098225 (accessed on 1 May 2018).
- Berger, K.J.; Guss, D.A. Mycotoxins revisited: Part II. J. Emerg. Med. 2005, 28, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Parant, F.; Peltier, L.; Lardet, G.; Pulce, C.; Descotes, J.; Moulsma, M. Phalloidin syndrome: Role of Elisa-based assay for the detection of alpha- and gamma-amanitins in urine. Preliminary results. Acta Clin. Belg. 2006, 61 (Suppl. 1), 11–17. [Google Scholar] [CrossRef] [PubMed]
- Bühlmann Laboratories. Amanitin ELISA Product Information. Available online: https://www.buhlmannlabs.ch/products-solutions/special-products/amanitin/ (accessed on 1 July 2018).
- Schenk-Jaeger, K.; Rauber-Lüthy, C.; Reichert, C.; Kupferschmidt, H. Vergiftung mit Knollenblätterpilzen (Amanita phalloides) und Anderen Amatoxin-haltigen Pilzen (Lepiota-, Galerina- und Amanita-Arten). Toxinfo Suisse. 2017. Available online: http://toxinfo.ch/customer/files/32/MB-Amanita-D-2017.pdf (accessed on 1 July 2018).
- Faulstich, H.; Jahn, W.; Wieland, T. Silybin inhibition of amatoxin uptake in the perfused rat liver. Arzneimittelforschung 1980, 30, 452–454. [Google Scholar] [PubMed]
- Mengs, U.; Pohl, R.T.; Mitchell, T. Legalon® SIL: The antidote of choice in patients with acute hepatotoxicity from amatoxin poisoning. Curr. Pharm. Biotechnol. 2012, 13, 1964–1970. [Google Scholar] [CrossRef]
- Poucheret, P.; Fons, F.; Doré, J.C.; Michelot, D.; Rapior, S. Amatoxin poisoning treatment decision-making: Pharmaco-therapeutic clinical strategy assessment using multi-dimensional multivariate statistic analysis. Toxicon 2010, 55, 1338–1345. [Google Scholar] [CrossRef]
- Ganzert, M.; Felgenhauer, N.; Schuster, T.; Eyer, T.; Gourdin, C.; Zilker, T. Knollenblätterpilzvergiftung. Silibinin und Kombination von Silibinin und Penicillin im Vergleich. Dtsch. Med. Wochenchr. 2008, 133, 2261–2267. [Google Scholar] [CrossRef]
- Giannini, L.; Vannacci, A.; Missanelli, A.; Mastroianni, R.; Mannaioni, P.F.; Moroni, F.; Masini, E. Amatoxin poisoning: A 15-year retrospective analysis and follow-up evaluation of 105 patients. Clin. Toxicol. 2007, 45, 539–542. [Google Scholar] [CrossRef] [PubMed]
| Patient ID | Age (Years) | Sex | Latency to Onset of Symptoms | Mushroom Species | Type of Acquisition | Consumed Cooked or Raw | Consultation with a Mycologist or Tox Info Suisse | Symptoms/Signs, and Complications | Therapy | Abnormal Laboratory Findings (on Presentation, except Mentioned Otherwise) | Length of Hospital Stay and Transfer to Other Unit |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 62 | M | >12 h | Boletus edulis, Macrolepiota procera, Cantharellus cibarius (not definitely identified) | self-harvested | not known | both | diarrhoea, abdominal pain, vertigo, sweating | activated charcoal | normal findings, alpha-amanitin negative | <24 h, discharged from ED |
| 2 | 15 | W | 6 h | unknown | self-harvested | not known | both | nausea, vomiting | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin, penicillin G | sodium 128 mmol/L, WBC 17.1 G/L, alpha-amanitin negative | <24 h, discharged from ED |
| 3 | 22 | M | 6 h | unknown | self-harvested | not known | both | nausea, vomiting, abdominal pain | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin, penicillin G | WBC 18.1 G/L, alpha-amanitin negative | <24 h, discharged from ED |
| 4 | 48 | M | 5 h | unknown | self-harvested | not known | both | nausea, vomiting, diarrhoea, abdominal pain | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin, penicillin G | creatinine 106 µmol/L, CRP 8 mg/L, bilirubin total 27 µmol/L, alpha-amanitin negative | <24 h, discharged from ED |
| 5 | 46 | W | 6 h | unknown | self-harvested | not known | both | nausea, vomiting, abdominal pain | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin, penicillin G | WBC 17.1 G/L, alpha-amanitin negative | <24 h, discharged from ED |
| 6 | 64 | M | 4 h | Boletaceae edible | commercial sources | not known | mycologist | nausea, vomiting | intravenous fluids, metoclopramide | ASAT 43 U/L, LDH 527 U/L, WBC 12.7 G/L | <24 h, discharged from ED |
| 7 | 60 | W | 4 h | Boletaceae edible | commercial sources | not known | mycologist | nausea, vomiting | intravenous fluids, meclizine/pyridoxine, esomeprazole, domperidone | unknown, alpha-amanitin negative | <24 h, discharged from ED |
| 8 | 27 | W | 0.2 h | unknown | self-harvested | cooked | no | nausea, feeling of faintness, paraesthesia | no therapy | unknown | <24 h, discharged from ED |
| 9 | 36 | W | 4.5 h | unknown | commercial sources | cooked | Tox Info Suisse | nausea, vomiting, diarrhoea, abdominal pain | intravenous fluids, metoclopramide, butyl scopolamine | normal findings | <24 h, discharged from ED |
| 10 | 34 | W | 2 h | Russula xerampelina (not definitely identified) | self-harvested | not known | Tox Info Suisse | nausea, vomiting, diarrhoea, abdominal pain | intravenous fluids, paracetamol | ASAT 58 U/L, ALAT 85 U/L, AP 174 U/L, GGT 160 U/L, LDH 530 U/L, CRP 48 mg/L ¶ | <24 h, discharged from ED |
| 11 | 30 | W | >6 h | Agaricus edible (not definitely identified) | commercial sources | not known | no | nausea, vomiting, abdominal pain, hypaesthesia | intravenous fluids, metoclopramide, ondansetrone | normal findings | <24 h, discharged from ED |
| 12 | 60 | W | 0.5 h | unknown | self-harvested | not known | mycologist | nausea, abdominal pain, dry mouth | intravenous fluids, activated charcoal, silibinin | normal findings, alpha-amanitin negative | <24 h, discharged from ED |
| 13 | 60 | M | 0.5 h | unknown | self-harvested | not known | mycologist | nausea, abdominal pain, dry mouth | intravenous fluids, activated charcoal, silibinin | normal findings, alpha-amanitin negative | <24 h, discharged from ED |
| 14 ∇ | 67 | W | 12 h | Amanita phalloides | self-harvested | not known | both | prolonged nausea and vomiting: on the 4th day acute renal failure, macrohaematuria, retinal haemorrhages, hemiplegia, progressive somnolence, suspected thrombotic thrombocytopenic purpura | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin, metoclopramide | initially normal; on the 4th day increase in liver enzymes (peak on the 6th day; ASAT 108 U/L, ALAT 74 U/L) and creatinine (peak on the 11th day; 398 μmol/L), thrombocytopenia (nadir of 23 G/L on the 9th day); alpha-amanitin positive | 792 h, internal medicine, abdominal surgery, intensive unit care, nephrology |
| 15 | 42 | M | >12 h | unknown | self-harvested | not known | no | nausea, diarrhoea | no therapy | unknown | <24 h, discharged from ED |
| 16 ∇ | 48 | W | >12 h | unknown | self-harvested | cooked | both | nausea, vomiting, diarrhoea, vertigo, fatigue | intravenous fluids, silibinin | ASAT 43 U/L, ALAT 61 U/L, AP 106U/L, GGT 118 U/L, bilirubin total 22 µmol/L ¶,# alpha-amanitin urinalysis negative | 84 h, internal medicine |
| 17 | 23 | M | 6 h | Amanita muscaria (not definitely identified) | commercial sources | not known | Tox Info Suisse | asymptomatic | no therapy | sodium 148 mmol/L | <24 h, discharged from ED |
| 18 | 70 | W | 0.2 h | Lepista nuda, Hypholoma fasciculaea or laterium | commercial sources (Lepista), unknown (Hypholoma) | not known | both | nausea, abdominal pain | intravenous fluids, metoclopramide | bilirubin total 17 µmol/L | <24 h, discharged from ED |
| 19 ∇ | 74 | M | 5.5 h | Amanita phalloides | self-harvested | cooked | both | nausea, vomiting, diarrhoea, vertigo, cramps | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin | ASAT 52 U/L, ALAT 42 U/L, GGT 68 U/L (peak on the 6th day; 79 U/L), creatinine 119 µmol/L, glomerular filtration rate (GFR) 52 mL/min, CRP 7 mg/L, INR 3.8, bilirubin total 17 µmol/L (peak on the 6th day; 38 µmol/L),, alpha-amanitin positive | 120 h, intensive care |
| 20 | 59 | M | 5 h | unknown | self-harvested | cooked | Tox Info Suisse | nausea, diarrhoea, abdominal pain, hallucinations | intravenous fluids | potassium 3.3 mmol/L, CRP 7 mg/L, alpha-amanitin negative | <24 h, discharged from ED |
| 21 | 47 | W | 2 h | Boletus satanas | self-harvested | cooked | Tox Info Suisse | nausea, vomiting, diarrhoea, abdominal pain, headache, chills | intravenous fluids, metoclopramide, butylscopolamine, esomeprazole | potassium 3.3 mmol/L | <24 h, discharged from ED |
| 22 | 36 | W | 1.5 h | Boletus satanas | self-harvested | raw | both | nausea, vomiting, diarrhoea | intravenous fluids, activated charcoal, metoclopramide | potassium 3.3 mmol/L | <24 h, discharged from ED |
| 23 | 73 | W | n.a. | Clitocybe nebularis, Xerocomus chrysenteron, X. badius, Lepista nuda, Lactarius semisangui fluus, Russula variata, Amanita rubescens, Laccaria amethystina, Craterellus cornuco pioides, Pseudohyd num gelatinosum | self-harvested | cooked | both | asymptomatic | intravenous fluids, activated charcoal | unknown | <24 h, discharged from ED |
| 24 ∇ | 51 | W | 0.5 h | same as patient ID 23 | self-harvested | cooked | both | nausea, diarrhoea, thoracic pain | intravenous fluids, activated charcoal | normal laboratory findings | 55 h, cardiology |
| 25 | 71 | M | n.a. | same as patient ID 23 | self-harvested | cooked | both | asymptomatic | intravenous fluids, activated charcoal | unknown | <24 h, discharged from ED |
| 26 | 58 | M | n.a. | same as patient ID 23 | self-harvested | cooked | both | asymptomatic | intravenous fluids, activated charcoal | unknown | <24 h, discharged from ED |
| 27 | 71 | M | 0.4 h | Armillaria mellea, Xerocomus chrysenteron, Chlorophyl lum rachodes | self-harvested | cooked | both | nausea, vertigo, fatigue, confusion, sweating | intravenous fluids, activated charcoal, metoclopramide | ASAT 39 U/L, GGT 44 U/L, LDH 621 U/L, INR 2.15 | <24 h, discharged from ED |
| 28 | 69 | W | 0.4 h | Armillaria mellea, Xerocomus chrysenteron, Chlorophyl lum rachodes | self-harvested | cooked | both | nausea, vomiting, vertigo | intravenous fluids, activated charcoal, metoclopramide | potassium 3.3 mmol/L, | <24 h, discharged from ED |
| 29 | 33 | W | >12 h | Clitocybe nebularis, Boletaceae | self-harvested | raw | both | nausea, diarrhoea, headache, dry mouth | intravenous fluids, activated charcoal, silibinin | normal laboratory findings | <24 h, discharged from ED |
| 30 | 49 | W | 6 h | unknown | commercial sources | cooked | Tox Info Suisse | nausea, vomiting, diarrhoea, abdominal pain | intravenous fluids, metoclopramide | normal laboratory findings | <24 h, discharged from ED |
| 31 | 73 | W | 1 h | unknown | self-harvested | raw | no | nausea, vomiting, diarrhoea | intravenous fluids, metoclopramide | LDH 513 U/L, creatinine 95 µmol/L, GFR 50 mL/min | <24 h, discharged from ED |
| 32 | 36 | W | >12 h | unknown | restaurant meal | cooked | no | nausea, abdominal pain | intravenous fluids, metoclopramide | ALAT 102 U/L, GGT 90 U/L, bilirubin total 49 µmol/L | <24 h, discharged from ED |
| 33 ∇ | 58 | W | 0.5 h | unknown | self-harvested | raw | Tox Info Suisse | salivation | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin | GGT 95 U/L, alpha-amanitin negative | 72 h, intensive care |
| 34 ∇ | 79 | M | 4.5 h | unknown | self-harvested | cooked | both | nausea, vomiting, vertigo, unconsciousness | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin | potassium 5.7 mmol/L, creatinine 88 µmol/L, GFR 87 mL/min, WBC 11.9 G/L, alpha-amanitin negative | 120 h, intensive care |
| 35 ∇ | 67 | M | >12 h | Amanita phalloides | self-harvested | not known | mycologist | nausea, vomiting, diarrhoea | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin, gastric and jejunal tube | CRP 30 mg/L, bilirubin total 37 µmol/L, WBC 13.1 G/L, INR 1.3, alpha-amanitin positive | 216 h, intensive care |
| 36 | 53 | W | >6 h | unknown | self-harvested | cooked | Tox Info Suisse | nausea, vomiting, diarrhoea, vertigo, unconsciousness | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin | normal findings, alpha-amanitin negative | <24 h, ward not documented |
| 37 ∇ | 65 | W | 0.3 h | Mushrooms with gills, Lactarius semisanguifluus, Boletaceae | self-harvested | not known | both | nausea, vomiting, diarrhoea, tachycardia, sweating | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin | ASAT 41 U/L, ALAT 40 U/L, GGT 95 U/L, LDH 508 U/L, creatinine 79 mmol/L, GFR 68 mL/min, CRP 9 mg/L, WBC 18.6 G/L, haemoglobin 117 g/L, alpha-amanitin negative | 72 h, gastroenterology ward |
| 38 ∇ | 41 | W | 7 h | unknown | self-harvested | not known | Tox Info Suisse | nausea, vomiting, paraesthesia | intravenous fluids, activated charcoal, N-acetylcysteine, silibinin | normal findings | 30 h, intensive care unit |
| 39 | 30 | W | 4 h | Macrolepiota procera | self-harvested | not known | both | nausea, vomiting, diarrhoea, abdominal pain | intravenous fluids, activated charcoal | normal findings, alpha-amanitin negative | <24 h, discharged from ED |
| 40 | 54 | M | 0.8 h | Amanita pantherina | self-harvested | cooked | mycologist | nausea, vomiting, abdominal pain, fatigue, mild visual disturbances, agitation, psychological excitation, tachycardia | intravenous fluids, activated charcoal | ASAT 40 U/L, creatinine 104 µmol/L | <24 h, discharged from ED |
| 41 | 54 | W | 0.8 h | Amanita pantherina | self-harvested | cooked | mycologist | abdominal pain, fatigue, agitation, mild visual disturbances | intravenous fluids, activated charcoal | normal findings | <24 h, discharged from ED |
| 42 | 19 | M | >12 h | Psilocybe | purchased on the street | raw | no | vertigo, feeling of faintness, unconsciousness, dyspnoea | intravenous fluids | normal findings | <24 h, discharged from ED |
| 43 | 46 | M | 0.3 h | Boletus edulis (not definitely identified) | restaurant meal | cooked | no | nausea, abdominal pain | intravenous fluids, metoclopramide | normal findings | <24 h, discharged from ED |
| 44 | 53 | W | 7 h | Boletus edulis (not definitely identified) | commercial sources | cooked | no | nausea, diarrhoea, headache, fever | intravenous fluids | normal findings | <24 h, discharged from ED |
| 45 | 43 | W | 3 h | unknown | self-harvested | cooked | both | nausea, vomiting, diarrhoea | intravenous fluids, activated charcoal, metoclopramide | normal findings, alpha-amanitin negative | <24 h, discharged from ED |
| 46 | 48 | M | 1 h | unknown | self-harvested | cooked | both | nausea, vomiting, diarrhoea | intravenous fluids, activated charcoal, metoclopramide | GGT 52 U/L | <24 h, discharged from ED |
| 47 ∇ | 40 | M | 1.5 h | Boletus satanas | self-harvested | raw | both | nausea, vomiting, diarrhoea, tachycardia, hypotension, fever, suspected disseminated intravascular coagulation (DIC) | intravenous fluids, activated charcoal, metoclopramide | ASAT 36 U/L, INR 1.32, persistent thrombocytopenia: nadir on the 3th day 102 G/L | 105 h, internal medicine ward |
| 48 | 24 | M | >12 h | unknown | self-harvested | cooked | Tox Info Suisse | headache | no therapy | ASAT 38 U/L, ALAT 126 U/L | <24 h, discharged from ED |
| 49 | 23 | M | 1 h | Amanita muscaria (not definitely identified) | purchased on the street | raw | no | nausea, vomiting, fatigue, aggression | no therapy | WBC 14.4 G/L | <24 h, discharged from ED |
| 50 | 28 | W | 4 h | Lycoperdales, Xerocomus chrysenteron | self-harvested | raw | both | nausea, vomiting, diarrhoea, chills | no therapy | unknown | <24 h, discharged from ED |
| 51 | 26 | M | 1 h | unknown psycho-active mushrooms | purchased on the street | raw | Tox Info Suisse | nausea, vomiting, extrapyramidal symptoms, panic, sweating | no therapy | unknown | <24 h, discharged from ED |
| Family | Species | n | Comment | Symptoms and Signs | Interval from Ingestion to Symptoms |
|---|---|---|---|---|---|
| Agaricaceae (n = 5) | Agaricus bisporus | 1 | Edible (in Switzerland sold only from cultivated mycelium) | nausea, vomiting, abdominal pain, hypesthesia | >6 h |
| Chlorophyllum rachodes | 2 | Edible (in Switzerland) | nausea, vomiting, vertigo, confusion, sweating, fatigue | 0.4 h | |
| Macrolepiota procera | 2 | Edible | nausea, vomiting, diarrhea, abdominal pain, vertigo, sweating | 4–>12 h | |
| Amanitaceae (n = 11) | Amanita muscaria | 2 | Poisonous | nausea, vomiting, aggression, fatigue | 1–6 h |
| Amanita pantherina | 2 | Poisonous | nausea, vomiting, abdominal pain, agitation, psychological excitation, visual disturbances, tachycardia, fatigue | 0.75 h | |
| Amanita phalloides | 3 | Poisonous | nausea, vomiting, diarrhea, vertigo, cramps, hemiplegia, somnolence, acute renal failure, macroheamaturia, retinal hemorrhages, thrombotic thrombocytopenic purpura | 5.5–>12 h | |
| Amanita rubescens | 4 | Edible | nausea, diarrhea, thoracic pain, asymptomatic | 0.5 h | |
| Boletaceae (n = 21) | n.a. | 4 | Not further identified Edible, non-edible, poisonous depending on the species | nausea, vomiting, diarrhea, headache, dry mouth, sweating, tachycardia | 0.3–>12 h |
| Boletus edulis | 3 | Edible | nausea, diarrhea, abdominal pain, vertigo, sweating, headache, fever | 0.3–>12 h | |
| Boletus satanas | 3 | Poisonous Current name: Rubroboletus satanas | nausea, vomiting, diarrhea, abdominal pain, headache, tachycardia, hypotension, chills, fever, suspected DIC | 1.5–2 h | |
| Xerocomus chrysenteron | 7 | Edible Current name: Xerocomellus chrysenteron | nausea, vomiting, diarrhea, vertigo, confusion, sweating, thoracic pain, fatigue, chills, asymptomatic | 0.4–4 h | |
| Xerocomus badius | 4 | Edible Current name: Imleria badia | nausea, diarrhea, thoracic pain, asymptomatic | 0.5 h | |
| Cantharellaceae (n = 5) | Cantharellus cibarius | 1 | Edible | diarrhea, abdominal pain, vertigo, sweating | >12 h |
| Craterellus cornucopioides | 4 | Edible | nausea, diarrhea, thoracic pain, asymptomatic | 0.5 h | |
| Hydnangiaceae (n = 4) | Laccaria amethystina | 4 | Edible | nausea, diarrhea, thoracic pain, asymptomatic | 0.5 h |
| Incertae sedis (n = 4) | Pseudohydnum gelatinosum | 4 | Edible | nausea, diarrhea, thoracic pain, asymptomatic | 0.5 h |
| Physalacriaceae (n = 2) | Armillaria mellea | 2 | Edible but has to be pre-cooked in boiling water | nausea, vomiting, vertigo, confusion, sweating, fatigue | 0.4 h |
| Russulaceae (n = 10) | Russula variata | 4 | Edible Current name: Russula cyanoxantha | nausea, diarrhea, thoracic pain, asymptomatic | 0.5 h |
| Russula xerampelina | 1 | Edible | nausea, vomiting, diarrhea, abdominal pain | 2 h | |
| Lactarius semisanguifluus | 5 | Edible | nausea, vomiting, diarrhea, sweating, thoracic pain, tachycardia, asymptomatic | 0.3–0.5 h | |
| Strophariaceae (n = 1) | Hypholoma fasciculaea or laterium | 1 | Poisonous | nausea, abdominal pain | 0.2 h |
| Tricholomataceae (n = 10) | Clitocybe nebularis | 5 | Edible in Switzerland but has to be prepared in boiling water Considered non-edible in Germany | nausea, diarrhea, headache, dry mouth, thoracic pain, asymptomatic | 0.5–>12 h |
| Lepista nuda | 5 | Edible | nausea, abdominal pain, diarrhea, thoracic pain, asymptomatic | 0.2–0.5 h | |
| n.a. | Lycoperdales | 1 | Outdated order Edible and non-edible | nausea, vomiting, diarrhea, chills | 4 h |
| n (%) | |
|---|---|
| Gastrointestinal | 44 (86.3) |
| nausea | 42 (82.4) |
| vomiting | 30 (58.8) |
| diarrhoea | 23 (45.1) |
| abdominal pain | 18 (35.3) |
| Neurological/Psychiatric | 23 (45.1) |
| vertigo | 8 (15.7) |
| agitation/aggression/panic/psychological excitation | 5 (9.8) |
| headache | 4 (7.8) |
| paraesthesia/hypaesthesia | 3 (5.9) |
| unconsciousness | 3 (5.9) |
| feeling of faintness | 2 (3.9) |
| mild visual disturbances | 2 (3.9) |
| confusion | 1 (2) |
| extrapyramidal symptoms | 1 (2) |
| hemiplegia | 1 (2) |
| spasms | 1 (2) |
| hallucinations | 1 (2) |
| cholinergic symptoms (e.g., sweating, salivation) | 5 (9.8) |
| anticholinergic symptoms (e.g., dry mouth) | 3 (5.9) |
| Cardiovascular | 5 (9.8) |
| tachycardia | 3 (5.9) |
| thoracic pain | 1 (2) |
| hypotension | 1 (2) |
| dyspnoea | 1 (2) |
| Miscellaneous/other | 10 (19.6) |
| fatigue | 5 (9.8) |
| chills/fever | 4 (7.8) |
| renal failure | 1 (2) |
| (suspected) thrombotic thrombocytopenic purpura | 1 (2) |
| (suspected) disseminated intravascular coagulation (DIC) | 1 (2) |
| Cluster Size (Number of Persons Involved) | Patient ID | Mushroom Species | Symptoms and Signs | Interval from Ingestion to Symptoms | Comment |
|---|---|---|---|---|---|
| 4 | 2–5 | unknown | nausea (n = 4), vomiting (n = 4), abdominal pain (n = 3), diarrhoea (n = 1) | 5–6 h | |
| 2 | 6–7 | Boletaceae | nausea (n = 2), vomiting (n = 2) | 4 h | |
| 2 | 12–13 | unknown | nausea (n = 2), abdominal pain (n = 2), dry mouth (n = 2) | 0.5 h | |
| 2 | 14 | Amanita phalloides | nausea (n = 1), vomiting (n = 1), acute renal failure (n = 1), macrohaematuria (n = 1), retinal haemorrhages (n = 1), hemiplegia (n = 1), progressive somnolence, (n = 1), suspected thrombotic thrombocytopenic purpura (n = 1) | 12 h | Only one patient of this cluster consulted our ED |
| 2 | 16, 48 | unknown | nausea (n = 1), vomiting (n = 1), diarrhoea (n = 1), fatigue (n = 1), vertigo (n = 1), headache (n = 1) | >12 h | |
| 2 | 21 | Boletus satanas | nausea (n = 1), vomiting (n = 1), abdominal pain (n = 1), diarrhoea (n = 1), chills (n = 1), headache (n = 1) | 2 h | Only one patient of this cluster consulted our ED |
| 3 | 22, 47 | Boletus satanas | nausea (n = 2), vomiting (n = 2), diarrhoea (n = 2), fever (n = 1), tachycardia (n = 1), hypotension (n = 1), suspected DIC (n = 1) | 1.5 h | Only two patients of this cluster consulted our ED |
| 4 | 23–26 | Xerocomus chrysenteron, X. badius, Lepista nuda, Clitocybe nebularis, Lactarius semisanguifluus, Russula variata, Amanita rubescens, Laccaria amethystina, Craterellus cornucopioides, Pseudohydnum gelatinosum | nausea (n = 1), diarrhoea (n = 1), thoracic pain (n = 1) | 0.5 h | Only one patient had symptoms, the other three were asymptomatic |
| 2 | 27, 28 | Xerocomus chrysenteron, Chlorophyllum rachodes, Armillaria mellea | nausea (n = 2), vertigo (n = 2), vomiting (n = 1), sweating (n = 1), fatigue (n = 1), confusion (n = 1) | 0.4 h | |
| 2 | 35 | Amanita phalloides | nausea (n = 1), vomiting (n = 1), diarrhoea (n = 1) | >12 h | Only one patient of this cluster consulted our ED |
| 2 | 37 | Mushrooms with gills, Lactarius semisanguifluus, Boletaceae | nausea (n = 1), vomiting (n = 1), diarrhoea (n = 1), sweating (n = 1), tachycardia (n = 1) | 0.3 h | Only one patient of this cluster consulted our ED |
| 4 | 40, 41 | Amanita pantherina | abdominal pain (n = 2), fatigue (n = 2), agitation (=2), mild visual disturbances (n = 2), nausea (n = 1), vomiting (n = 1), psychological excitation (n = 1), tachycardia (n = 1) | 0.8 h | Two patients of this cluster visited another ED |
| 3 | 45, 46 | unknown | nausea (n = 2), vomiting (n = 2), diarrhoea (n = 2) | 1–3 h | Only two patients of this cluster consulted our ED |
| 3 | 50 | Lycoperdales, Xerocomus chrysenteron | nausea (n = 2), vomiting (n = 2), diarrhoea (n = 2), chills (n = 1), asymptomatic (n = 1) | 4–12 h | Only one patient of this cluster consulted our ED |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keller, S.A.; Klukowska-Rötzler, J.; Schenk-Jaeger, K.M.; Kupferschmidt, H.; Exadaktylos, A.K.; Lehmann, B.; Liakoni, E. Mushroom Poisoning—A 17 Year Retrospective Study at a Level I University Emergency Department in Switzerland. Int. J. Environ. Res. Public Health 2018, 15, 2855. https://doi.org/10.3390/ijerph15122855
Keller SA, Klukowska-Rötzler J, Schenk-Jaeger KM, Kupferschmidt H, Exadaktylos AK, Lehmann B, Liakoni E. Mushroom Poisoning—A 17 Year Retrospective Study at a Level I University Emergency Department in Switzerland. International Journal of Environmental Research and Public Health. 2018; 15(12):2855. https://doi.org/10.3390/ijerph15122855
Chicago/Turabian StyleKeller, Sarah A., Jolanta Klukowska-Rötzler, Katharina M. Schenk-Jaeger, Hugo Kupferschmidt, Aristomenis K. Exadaktylos, Beat Lehmann, and Evangelia Liakoni. 2018. "Mushroom Poisoning—A 17 Year Retrospective Study at a Level I University Emergency Department in Switzerland" International Journal of Environmental Research and Public Health 15, no. 12: 2855. https://doi.org/10.3390/ijerph15122855
APA StyleKeller, S. A., Klukowska-Rötzler, J., Schenk-Jaeger, K. M., Kupferschmidt, H., Exadaktylos, A. K., Lehmann, B., & Liakoni, E. (2018). Mushroom Poisoning—A 17 Year Retrospective Study at a Level I University Emergency Department in Switzerland. International Journal of Environmental Research and Public Health, 15(12), 2855. https://doi.org/10.3390/ijerph15122855






