Infections in Healthcare Workers in Germany—22-Year Time Trends †
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Seidler, A.; Nienhaus, A.; Diel, R. Review of epidemiological studies on the occupational risk of tuberculosis in low-incidence areas. Respiration 2005, 72, 431–446. [Google Scholar] [CrossRef]
- Baussano, I.; Nunn, P.; Williams, B.; Pivetta, E.; Bugiani, M.; Scano, F. Tuberculosis among health care workers. Emerg. Infect. Dis. 2011, 17, 488–494. [Google Scholar] [CrossRef]
- Diel, R.; Niemann, S.; Nienhaus, A. Risk of tuberculosis transmission among healthcare workers. ERJ Open Res. 2018, 4, 00161-2017. [Google Scholar] [CrossRef]
- Westermann, C.; Peters, C.; Lisiak, B.; Lamberti, M.; Nienhaus, A. The prevalence of hepatitis C among healthcare workers: A systematic review and meta-analysis. Occup. Environ. Med. 2015, 72, 880–888. [Google Scholar] [CrossRef]
- Kuster, S.P.; Shah, P.S.; Coleman, B.L.; Lam, P.P.; Tong, A.; Wormsbecker, A.; McGeer, A. Incidence of influenza in healthy adults and healthcare workers: A systematic review and meta-analysis. PLoS ONE 2011, 6, e26239. [Google Scholar] [CrossRef]
- Ho, P.L.; Becker, M.; Chan-Yeung, M.M. Emerging occupational lung infections. Int. J. Tuber. Lung Dis. 2005, 11, 710–721. [Google Scholar]
- Poon, L.L.; Guan, Y.; Nicholls, J.M.; Yuen, K.Y.; Peiris, J.S. The aetiology, origins and diagnosis of severe acute respiratory syndrome. Lancet Infect. Dis. 2004, 4, 663–671. [Google Scholar] [CrossRef]
- Lee, N.; Hui, D.; Wu, A.; Chan, P.; Cameron, P.; Joynt, G.M.A.; Ahuja, A.; Yung, M.Y.; Leung, C.B.; To, K.F.; et al. Major outbreak of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 2003, 348, 1986–1994. [Google Scholar] [CrossRef]
- Booth, C.M.; Matukas, L.M.; Tomlinson, G.A.; Rachlis, A.R.; Rose, D.B.; Dwosh, H.A.; Walmsley, S.L.; Mazzulli, T.; Avendano, M.; Derkach, P.; et al. Clinical features and short term outcomes of 144 patients with SARS in the Greater Toronto Area. JAMA 2003, 289, 2801–2809. [Google Scholar] [CrossRef]
- Kuster, S.P.; Coleman, B.L.; Raboud, J.; McNeil, S.; De Serres, G.; Gubbay, J.; Hatchette, T.; Katz, K.C.; Loeb, M.; Low, D.; et al. Working Adult Influenza Cohort Study Group. Risk factors for influenza among health care workers during 2009 pandemic, Toronto, Ontario, Canada. Emerg. Infect. Dis. 2013, 19, 606–615. [Google Scholar] [CrossRef]
- Lietz, J.; Westermann, C.; Nienhaus, A.; Schablon, A. The Occupational Risk of Influenza A (H1N1) Infection among Healthcare Personnel during the 2009 Pandemic: A Systematic Review and Meta-Analysis of Observational Studies. PLoS ONE 2016, 11, e0162061. [Google Scholar] [CrossRef]
- Dulon, M.; Peters, C.; Schablon, A.; Nienhaus, A. MRSA carriage among healthcare workers in non-outbreak settings in Europe and the United States: A systematic review. BMC Infect. Dis. 2014, 14, 363. [Google Scholar] [CrossRef]
- Sepkowitz, K.A.; Eisenberg, L. Occupational deaths among healthcare workers. Emerg. Infect. Dis. 2005, 11, 1003–1008. [Google Scholar] [CrossRef]
- Hofmann, F.; Kralj, N.; Beie, M. Needle stick injuries in health care: Frequency, causes and preventive strategies. Gesundheitswesen 2002, 64, 259–266. [Google Scholar] [CrossRef]
- Green-McKenzie, J.; McCarthy, R.B.; Shofer, F.S. Characterisation of occupational blood and body fluid exposures beyond the Needlestick Safety and Prevention Act. J. Infect. Prev. 2016, 17, 226–232. [Google Scholar] [CrossRef]
- Council of the European Union. Council Directive 2010/32/EU of May 2010 implementing the Framework Agreement on prevention from sharp injuries in the hospital and healthcare sector concluded by HOSPEEM and EPSU. Off. J. Eur. Union 2010, L134, 66–72. [Google Scholar]
- TRBA 250—Technical Rules for Bilological Agents. Biological Agents in Health Care and Welfare Facilities (Technische Regel Biologische Arbeitsstoffe 250) Last Amendment 2015. Available online: https://www.baua.de/EN/Service/Legislative-texts-and-technical-rules/Rules/TRBA/pdf/TRBA-250.pdf?__blob=publicationFile&v=2 (accessed on 4 October 2018).
- Federal Ministry of Labour and Social Affairs. Ordinanz for Preventive Occupational Medical Care (Verordnung zur Arbeitsmedizinischen Vorsorge—ArbMedVV). Available online: https://www.bmas.de/SharedDocs/Downloads/DE/PDF-Publikationen/a453-arbeitsmedizinischen-vorsorge.pdf?__blob=publicationFile (accessed on 4 October 2018).
- Nienhaus, A.; Kesavachandran, C.; Wendeler, D.; Haamann, F.; Dulon, M. Infectious diseases in healthcare workers—An analysis of the standardised data set of a German compensation board. J. Occup. Med. Toxicol. 2012, 7, 8. [Google Scholar] [CrossRef]
- Westermann, C.; Dulon, M.; Wendeler, D.; Nienhaus, A. Hepatitis C among healthcare personnel: Secondary data analyses of costs and trends for hepatitis C infections with occupational causes. J. Occup. Med. Toxicol. 2016, 11, 52. [Google Scholar] [CrossRef]
- Hofmann, F.; Kralj, N. Criteria for successful hepatitis B vaccination in adults: Results of a case study. Infection 2009, 37, 266–269. [Google Scholar] [CrossRef]
- Voigt, K.; Rühle, F.; Bergmann, A.; Schübel, J.; Hirsch, K.; Riemenschneider, H. Vaccination status among nurses in hospitals. Results of a cross sectional study at Harzklinikum Dorothea Christiane Erxleben Quedlinburg. Pflege 2016, 29, 205–212. (In German) [Google Scholar] [CrossRef]
- Ramich, T.; Eickholz, P.; Wicker, S. Work-related infections in dentistry: Risk perception and preventive measures. Clin. Oral. Investig. 2017, 21, 2473–2479. [Google Scholar] [CrossRef]
- von Laer, A.; Harder, T.; Zimmermann, R.; Dudareva, S. Virushepatitis B und D im Jahr 2017. Epidemiol. Bull. 2018, 30, 285–294. [Google Scholar]
- Zimmermann, R.; Meurs, L.; Schmidt, D.; Kollan, C.; Dudareva, S.; Bremer, V. Zur Situation bei wichtigen Infektionskrankheiten in Deutschland Hepatitis C im Jahr 2017. Epidemiol. Bull. 2018, 29, 271–281. [Google Scholar]
- Wittmann, A.; Kralj, N.; Köver, J.; Gasthaus, K.; Hofmann, F. Study of blood contact in simulated surgical needlestick injuries with single or double latex gloving. Infect. Control Hosp. Epidemiol. 2009, 30, 53–56. [Google Scholar] [CrossRef]
- Dulon, M.; Lisiak, B.; Wendeler, D.; Nienhaus, A. Workers’ Compensation Claims for Needlestick Injuries Among Healthcare Personnel in Hospitals, Doctors’ Surgeries and Nursing Institutions. Gesundheitswesen 2018, 80, 176–182. (In German) [Google Scholar]
- Dulon, M.; Lisiak, B.; Wendeler, D.; Nienhaus, A. Causes of needlestick injuries in three healthcare settings: Analysis of accident notifications registered six months after the implementation of EU Directive 2010/32/EU in Germany. J. Hosp. Infect. 2017, 95, 306–311. [Google Scholar] [CrossRef]
- Dulon, M.; Wendeler, D.; Nienhaus, A. Seroconversion after needlestick injuries—Analyses of statutory accident insurance claims in Germany. GMS Hyg. Infect. Control 2018, 6, 13. [Google Scholar]
- Himmelreich, H.; Rabenau, H.F.; Rindermann, M.; Stephan, C.; Bickel, M.; Marzi, I.; Wicker, S. The management of needlestick injuries. Dtsch. Arztebl. Int. 2013, 110, 61–67. [Google Scholar] [CrossRef]
- Nienhaus, A.; Schablon, A.; Preisser, A.M.; Ringshausen, F.C.; Diel, R. Tuberculosis in healthcare workers—A narrative review from a German perspective. J. Occup. Med. Toxicol. 2014, 9, 9. [Google Scholar] [CrossRef]
- Diel, R.; Loddenkemper, R.; Nienhaus, A. Consequences of tuberculosis among asylum seekers for health care workers in Germany. J. Occup. Med. Toxicol. 2016, 11, 4. [Google Scholar] [CrossRef][Green Version]
- Atti, M.L.C.D.; Gattinara, G.C.; Ciliento, G.; Lancella, L.; Russo, C.; Coltella, L.; Raponi, M. Prolonged in-hospital exposure to an infant with active pulmonary tuberculosis. Epidemiol. Infect. 2011, 139, 139–142. [Google Scholar] [CrossRef]
- Schablon, A.; Peters, C.; Diel, R.; Diner, G.; Anske, U.; Pankow, W.; Ringshausen, F.C.; Nienhaus, A. Serial IGRA testing of trainees in the healthcare sector in a country with low incidence for tuberculosis—A prospective cohort study. GMS Hyg. Infect. Control 2013, 8. [Google Scholar] [CrossRef]
- Nienhaus, A.; Ringshausen, F.C.; Costa, J.T.; Schablon, A.; Tripodi, D. IFN-γ release assay versus tuberculin skin test for monitoring TB infection in healthcare workers. Expert Rev. Anti-Infect. Ther. 2013, 11, 37–48. [Google Scholar] [CrossRef]
- Ringshausen, F.C.; Schablon, A.; Nienhaus, A. Interferon-gamma release assays for the tuberculosis serial testing of health care workers: A systematic review. J. Occup. Med. Toxicol. 2012, 7, 6. [Google Scholar] [CrossRef]
- Torres, C.J.; Silva, R.; Sá, R.; Cardoso, M.J.; Nienhaus, A. Results of five-year systematic screening for latent tuberculosis infection in healthcare workers in Portugal. J. Occup. Med. Toxicol. 2010, 5, 22. [Google Scholar] [CrossRef]
- Nienhaus, A.; Schablon, A.; Bâcle, C.L.; Siano, B.; Diel, R. Evaluation of the interferon-gamma release assay in healthcare workers. Int. Arch. Occup. Environ. Health 2008, 81, 295–300. [Google Scholar] [CrossRef]
- Gilardi, F.; Castelli Gattinara, G.; Vinci, M.R.; Ciofi Degli Atti, M.; Santilli, V.; Brugaletta, R.; Santoro, A.; Montanaro, R.; Lavorato, L.; Raponi, M.; et al. Seasonal Influenza Vaccination in Health Care Workers. A Pre-Post Intervention Study in an Italian Paediatric Hospital. Int. J. Environ. Res. Public Health 2018, 15, 841. [Google Scholar] [CrossRef]
- Imai, C.; Toizumi, M.; Hall, L.; Lambert, S.; Halton, K.; Merollini, K. A systematic review and meta-analysis of the direct epidemiological and economic effects of seasonal influenza vaccination on healthcare workers. PLoS ONE 2018, 13, e0198685. [Google Scholar] [CrossRef]
- Thomas, R.E.; Jefferson, T.; Lasserson, T.J. Influenza vaccination for healthcare workers who care for people aged 60 or older living in long-term care institutions. Cochrane Database Syst. Rev. 2016, 6, CD005187. [Google Scholar] [CrossRef]
- De Serres, G.; Skowronski, D.M.; Ward, B.J.; Gardam, M.; Lemieux, C.; Yassi, A.; Patrick, D.M.; Krajden, M.; Loeb, M.; Collignon, P.; et al. Influenza Vaccination of Healthcare Workers: Critical Analysis of the Evidence for Patient Benefit Underpinning Policies of Enforcement. PLoS ONE 2017, 12, e0163586. [Google Scholar] [CrossRef]
- Stranzinger, J.; Kindel, J.; Henning, M.; Wendeler, D.; Nienhaus, A. Prevalence of CMV infection among staff in a metropolitan children’s hospital—Occupational health screening findings. GMS Hyg. Infect. Control 2016, 11. [Google Scholar] [CrossRef]
- Stranzinger, J.; Kozak, A.; Schilgen, B.; Paris, D.; Nießen, T.; Schmidt, L.; Wille, A.; Wagner, N.L.; Nienhaus, A. Are female daycare workers at greater risk of cytomegalovirus infection? A secondary data analysis of CMV seroprevalence between 2010 and 2013 in Hamburg, Germany. GMS Hyg. Infect. Control 2016, 11. [Google Scholar] [CrossRef]
- Meyer-Rüsenberg, B.; Loderstädt, U.; Richard, G.; Kaulfers, P.M.; Gesse, C. Epidemic keratoconjunctivitis: The current situation and recommendations for prevention and treatment. Dtsch. Arztebl. Int. 2011, 108, 475–480. [Google Scholar]
- Pleyer, U.; Birnbaum, F. Adenoviral keratoconjunctivitis. Ophthalmologe 2015, 112, 459–469. (In German) [Google Scholar] [CrossRef]
- Peters, C.; Dulon, M.; Kleinmüller, O.; Nienhaus, A.; Schablon, A. MRSA Prevalence and Risk Factors among Health Personnel and Residents in Nursing Homes in Hamburg, Germany—A Cross-Sectional Study. PLoS ONE 2017, 12, e0169425. [Google Scholar] [CrossRef]
- Peters, C.; Kleinmüller, O.; Nienhaus, A.; Schablon, A. Prevalence and risk factors of MRSA colonisations: A cross-sectional study among personnel in outpatient care settings in Hamburg, Germany. BMJ Open 2018, 8, e021204. [Google Scholar] [CrossRef]
- Schablon, A.; Kleinmüller, O.; Nienhaus, A.; Peters, C. MRSA prevalence among patient transport staff in Hamburg. GMS Hyg. Infect. Control 2018, 13. [Google Scholar] [CrossRef]
- Peters, C.; Dulon, M.; Lietz, J.; Nienhaus, A. Infection Control Professionals’ Views on the Handling of MRSA-Colonised Healthcare Workers. Gesundheitswesen 2017, 79, 648–654. (In German) [Google Scholar]
- Dulon, M.; Haamann, F.; Nienhaus, A. Involvement of occupational physicians in the management of MRSA-colonised healthcare workers in Germany—A survey. J. Occup. Med. Toxicol. 2013, 8, 16. [Google Scholar] [CrossRef]
- Haamann, F.; Dulon, M.; Nienhaus, A. MRSA as an occupational disease: A case series. Int. Arch. Occup. Environ. Health 2011, 84, 259–266. [Google Scholar] [CrossRef][Green Version]
- Dulon, M.; Wendeler, D.; Haamann, F.; Nienhaus, A. Infections as occupational diseases—Analysis of the data of the Accident Insurance Institution for the Health and Welfare Services for 2007 to 2011. Zbl. Arbeitsmed. 2013, 63, 34–43. [Google Scholar]
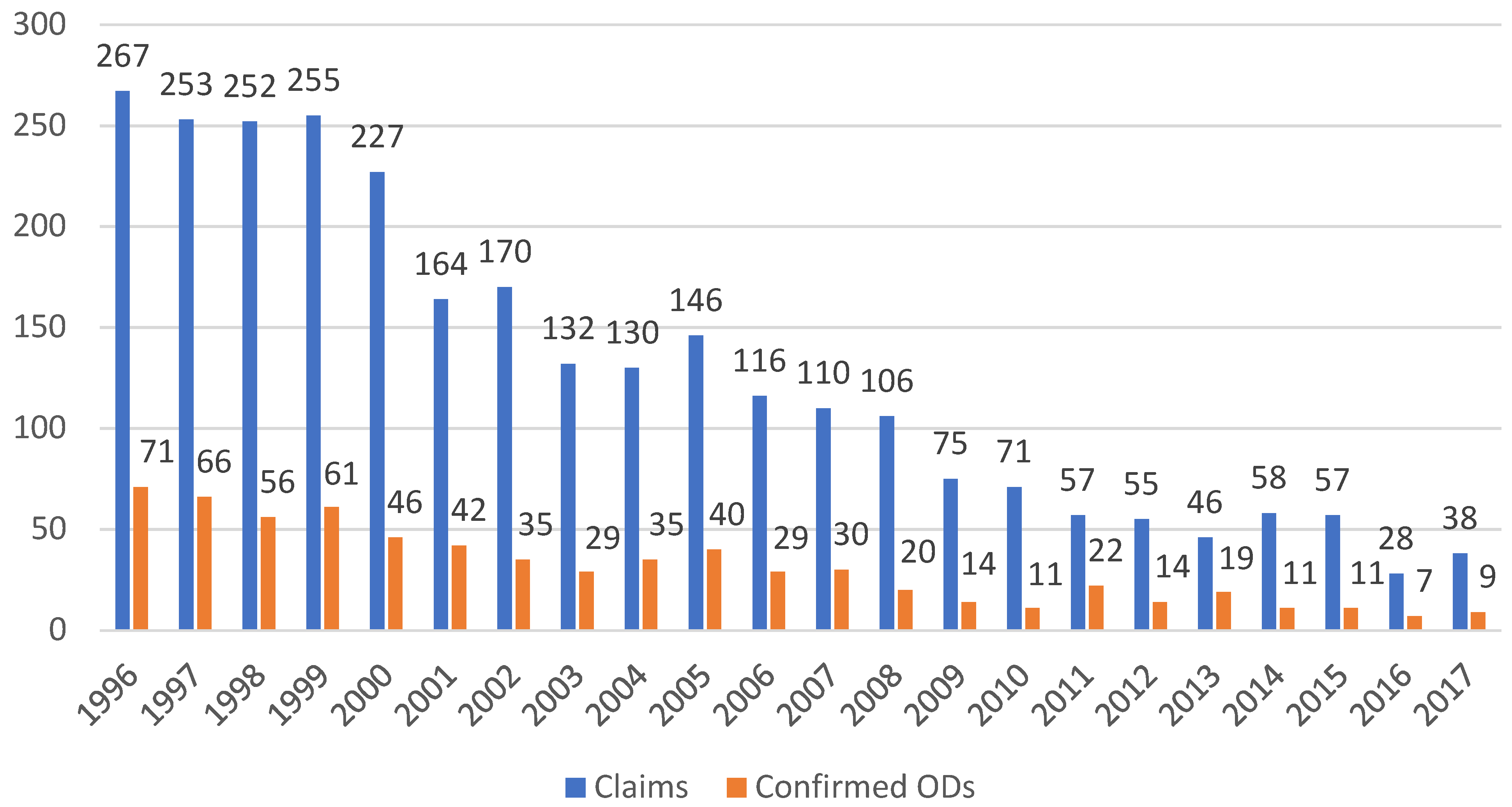
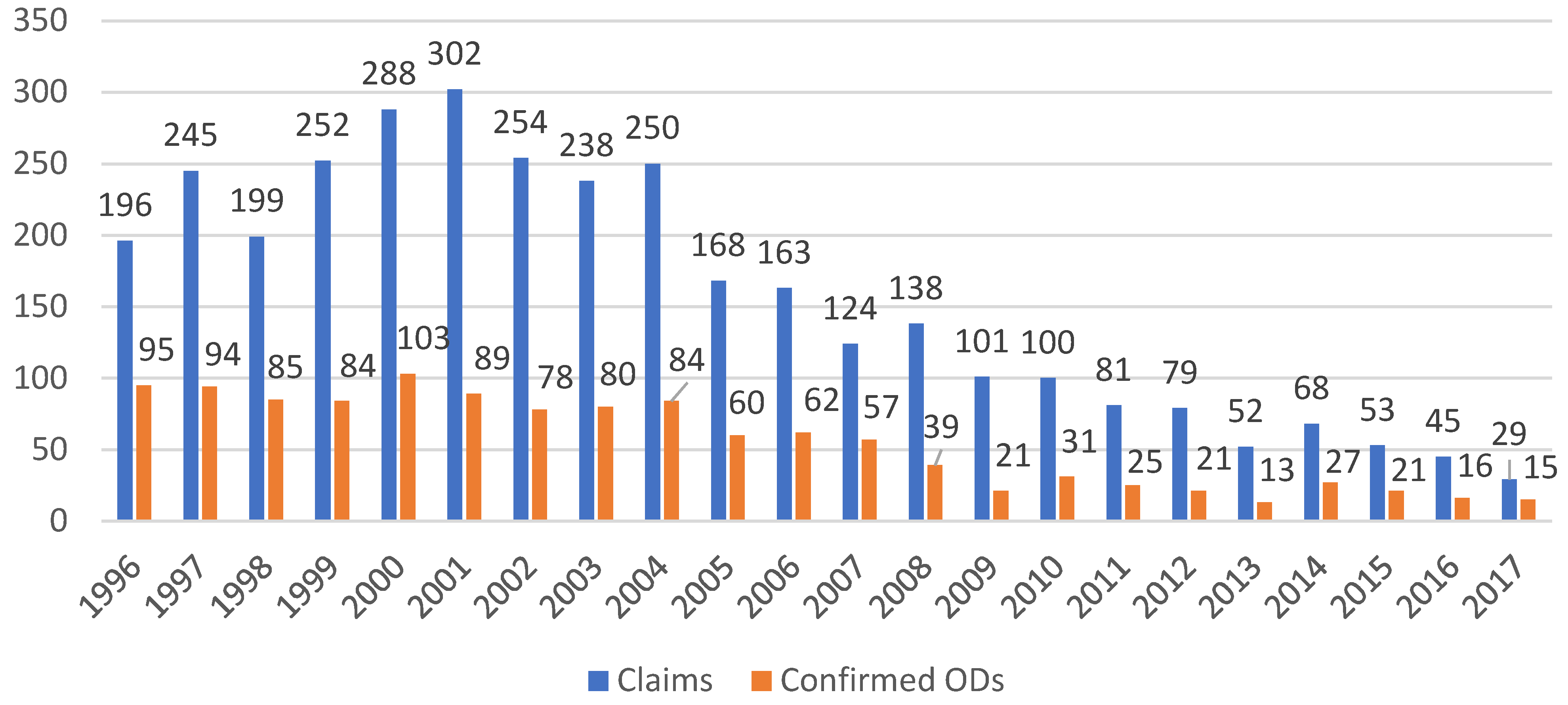
| Year | Full-Time Workers in Thousands (N) | All Claims (N) | Claims per 100,000 Full-Time Workers | ODs Confirmed | ODs per 100,000 Full-Time Workers |
|---|---|---|---|---|---|
| 1996 | 3238 | 816 | 25.2 | 226 | 7.0 |
| 1997 | 3157 | 884 | 28.0 | 238 | 7.5 |
| 1998 | 2888 | 849 | 29.4 | 246 | 8.5 |
| 1999 | 2831 | 889 | 31.4 | 252 | 8.9 |
| 2000 | 2990 | 882 | 29.5 | 216 | 7.2 |
| 2001 | 3019 | 791 | 26.2 | 186 | 6.2 |
| 2002 | 3189 | 810 | 25.4 | 232 | 7.3 |
| 2003 | 3627 | 809 | 22.3 | 200 | 5.5 |
| 2004 | 4213 | 1,066 | 25.3 | 199 | 4.7 |
| 2005 | 3431 | 988 | 28.8 | 296 | 8.6 |
| 2006 | 3595 | 798 | 22.2 | 239 | 6.7 |
| 2007 | 3589 | 786 | 21.9 | 211 | 5.9 |
| 2008 | 3773 | 717 | 19.0 | 200 | 5.3 |
| 2009 | 4010 | 818 | 20.4 | 255 | 6.4 |
| 2010 | 4089 | 646 | 15.8 | 304 | 7.4 |
| 2011 | 4299 | 761 | 17.7 | 324 | 7.5 |
| 2012 | 4406 | 727 | 16.5 | 368 | 8.4 |
| 2013 | 4531 | 879 | 19.4 | 422 | 9.3 |
| 2014 | 4585 | 729 | 15.9 | 360 | 7.9 |
| 2015 | 4587 | 711 | 15.5 | 301 | 6.6 |
| 2016 | 4753 | 789 | 16.6 | 290 | 6.1 |
| 2017 | 4935 | 760 | 15.4 | 340 | 6.9 |
| Total | 17,905 | 5905 |
| Year | Hepatitis B | Hepatitis C | ||
|---|---|---|---|---|
| Claims per 100,000 full-time workers | ODs per 100,000 full-time workers | Claims per 100,000 full-time workers | ODs per 100,000 full-time workers | |
| 1996 | 8.3 | 2.2 | 6.1 | 2.9 |
| 1997 | 8.0 | 2.1 | 7.8 | 3.0 |
| 1998 | 8.7 | 1.9 | 6.9 | 2.9 |
| 1999 | 9.0 | 2.2 | 8.9 | 3.0 |
| 2000 | 7.6 | 1.5 | 9.6 | 3.4 |
| 2001 | 5.4 | 1.4 | 10.0 | 3.0 |
| 2002 | 5.3 | 1.1 | 8.0 | 2.5 |
| 2003 | 3.6 | 0.8 | 6.6 | 2.2 |
| 2004 | 3.1 | 0.8 | 5.9 | 2.0 |
| 2005 | 4.3 | 1.2 | 4.9 | 1.8 |
| 2006 | 3.2 | 0.8 | 4.5 | 1.7 |
| 2007 | 3.1 | 0.8 | 3.5 | 1.6 |
| 2008 | 2.8 | 0.5 | 3.7 | 1.0 |
| 2009 | 1.9 | 0.4 | 2.5 | 0.5 |
| 2010 | 1.7 | 0.3 | 2.5 | 0.8 |
| 2011 | 1.3 | 0.5 | 1.9 | 0.6 |
| 2012 | 1.3 | 0.3 | 1.8 | 0.5 |
| 2013 | 1.0 | 0.4 | 1.2 | 0.3 |
| 2014 | 1.3 | 0.2 | 1.5 | 0.6 |
| 2015 | 1.2 | 0.2 | 1.2 | 0.5 |
| 2016 | 0.6 | 0.2 | 1.0 | 0.3 |
| 2017 | 0.8 | 0.2 | 0.6 | 0.3 |
| HIV/AIDS | Epidemic Kerato-Conjunctivitis | Cytomegaly | Influenza | |||||
|---|---|---|---|---|---|---|---|---|
| Year | Claims (N) | Confirmed ODs (N) | Claims (N) | Confirmed ODs (N) | Claims (N) | Confirmed ODs (N) | Claims (N) | Confirmed ODs (N) |
| 1996 | 8 | 4 | 8 | 20 | 5 | 0 | 2 | 0 |
| 1997 | 3 | 1 | 13 | 8 | 4 | 1 | 1 | 0 |
| 1998 | 5 | 5 | 44 | 20 | 4 | 1 | 0 | 1 |
| 1999 | 6 | 3 | 50 | 27 | 9 | 2 | 2 | 0 |
| 2000 | 12 | 1 | 44 | 23 | 2 | 0 | 1 | 1 |
| 2001 | 8 | 0 | 9 | 16 | 7 | 0 | 0 | 0 |
| 2002 | 9 | 1 | 19 | 14 | 5 | 1 | 6 | 0 |
| 2003 | 3 | 0 | 18 | 4 | 2 | 3 | 1 | 0 |
| 2004 | 11 | 2 | 7 | 5 | 2 | 0 | 0 | 0 |
| 2005 | 9 | 2 | 16 | 6 | 1 | 0 | 0 | 0 |
| 2006 | 12 | 0 | 23 | 12 | 3 | 0 | 0 | 0 |
| 2007 | 4 | 4 | 24 | 8 | 4 | 0 | 0 | 0 |
| 2008 | 11 | 2 | 10 | 5 | 2 | 1 | 1 | 0 |
| 2009 | 5 | 0 | 23 | 4 | 5 | 2 | 53 | 32 |
| 2010 | 5 | 1 | 16 | 18 | 4 | 1 | 5 | 4 |
| 2011 | 10 | 10 | 21 | 5 | 4 | 0 | 12 | 3 |
| 2012 | 2 | 3 | 42 | 21 | 1 | 0 | 2 | 0 |
| 2013 | 3 | 6 | 35 | 22 | 3 | 1 | 11 | 1 |
| 2014 | 5 | 0 | 15 | 7 | 1 | 0 | 9 | 1 |
| 2015 | 3 | 0 | 13 | 4 | 5 | 1 | 6 | 0 |
| 2016 | 3 | 5 | 5 | 3 | 3 | 2 | 8 | 4 |
| 2017 | 4 | 2 | 4 | 1 | 5 | 1 | 4 | 1 |
| Total | 141 | 34 | 456 | 255 | 81 | 17 | 124 | 48 |
| TB | LTBI | All | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year | Claims (N) | A | Confirmed ODs (N) | B | Claims (N) | A | Confirmed ODs (N) | B | Claims (N) | A | Confirmed ODs (N) | B |
| 1996 | 173 | 5.3 | 43 | 1.3 | - | 173 | 5.3 | 43 | 1.3 | |||
| 1997 | 201 | 6.4 | 51 | 1.6 | - | 201 | 6.4 | 51 | 1.6 | |||
| 1998 | 155 | 5.4 | 48 | 1.7 | - | 155 | 5.4 | 48 | 1.7 | |||
| 1999 | 124 | 4.4 | 47 | 1.7 | - | 124 | 4.4 | 47 | 1.7 | |||
| 2000 | 136 | 4.6 | 24 | 0.8 | - | 136 | 4.6 | 24 | 0.8 | |||
| 2001 | 125 | 4.1 | 29 | 1.0 | - | 125 | 4.1 | 29 | 1.0 | |||
| 2002 | 125 | 3.9 | 33 | 1.0 | - | 125 | 3.9 | 33 | 1.0 | |||
| 2003 | 134 | 3.7 | 40 | 1.1 | - | 134 | 3.7 | 40 | 1.1 | |||
| 2004 | 191 | 4.5 | 50 | 1.2 | - | 191 | 4.5 | 50 | 1.2 | |||
| 2005 | 234 | 6.8 | 99 | 2.9 | - | 234 | 6.8 | 99 | 2.9 | |||
| 2006 | 175 | 4.9 | 83 | 2.3 | 20 | 0.6 | 5 | 0.1 | 195 | 5.4 | 88 | 2.5 |
| 2007 | 253 | 7.1 | 56 | 1.6 | 33 | 0.9 | 6 | 0.2 | 286 | 8.0 | 62 | 1.7 |
| 2008 | 195 | 5.2 | 73 | 1.9 | 40 | 1.1 | 17 | 0.5 | 235 | 6.2 | 90 | 2.4 |
| 2009 | 124 | 3.1 | 61 | 1.5 | 187 | 4.7 | 65 | 1.6 | 311 | 7.8 | 126 | 3.1 |
| 2010 | 102 | 2.5 | 72 | 1.8 | 199 | 4.9 | 125 | 3.1 | 301 | 7.4 | 197 | 4.8 |
| 2011 | 115 | 2.7 | 68 | 1.6 | 285 | 6.6 | 166 | 3.9 | 400 | 9.3 | 234 | 5.4 |
| 2012 | 146 | 3.3 | 66 | 1.5 | 227 | 5.2 | 179 | 4.1 | 373 | 8.5 | 245 | 5.6 |
| 2013 | 160 | 3.5 | 77 | 1.7 | 383 | 8.5 | 244 | 5.4 | 543 | 12.0 | 321 | 7.1 |
| 2014 | 94 | 2.1 | 81 | 1.8 | 297 | 6.5 | 205 | 4.5 | 391 | 8.5 | 286 | 6.2 |
| 2015 | 155 | 3.4 | 57 | 1.2 | 235 | 5.1 | 178 | 3.9 | 390 | 8.5 | 235 | 5.1 |
| 2016 | 235 | 4.9 | 80 | 1.7 | 268 | 5.6 | 161 | 3.4 | 503 | 10.6 | 241 | 5.1 |
| 2017 | 222 | 4.5 | 98 | 2.0 | 251 | 5.1 | 201 | 4.1 | 473 | 9.6 | 299 | 6.1 |
| Total | 3574 | 1336 | 2425 | 1,553 | 5999 | 3889 | ||||||
| MRSA | ||
|---|---|---|
| Year | Claims (N) | Confirmed ODs (N) |
| 2006 | 114 | 1 |
| 2007 | 88 | 5 |
| 2008 | 98 | 11 |
| 2009 | 102 | 8 |
| 2010 | 49 | 9 |
| 2011 | 52 | 9 |
| 2012 | 60 | 9 |
| 2013 | 44 | 7 |
| 2014 | 58 | 5 |
| 2015 | 54 | 7 |
| 2016 | 47 | 2 |
| 2017 | 39 | 1 |
| Total | 805 | 74 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nienhaus, A. Infections in Healthcare Workers in Germany—22-Year Time Trends. Int. J. Environ. Res. Public Health 2018, 15, 2656. https://doi.org/10.3390/ijerph15122656
Nienhaus A. Infections in Healthcare Workers in Germany—22-Year Time Trends. International Journal of Environmental Research and Public Health. 2018; 15(12):2656. https://doi.org/10.3390/ijerph15122656
Chicago/Turabian StyleNienhaus, Albert. 2018. "Infections in Healthcare Workers in Germany—22-Year Time Trends" International Journal of Environmental Research and Public Health 15, no. 12: 2656. https://doi.org/10.3390/ijerph15122656
APA StyleNienhaus, A. (2018). Infections in Healthcare Workers in Germany—22-Year Time Trends. International Journal of Environmental Research and Public Health, 15(12), 2656. https://doi.org/10.3390/ijerph15122656




