Household Air Pollution Is Associated with Chronic Cough but Not Hemoptysis after Completion of Pulmonary Tuberculosis Treatment in Adults, Rural Eastern Democratic Republic of Congo
Abstract
1. Introduction
2. Materials and Methods
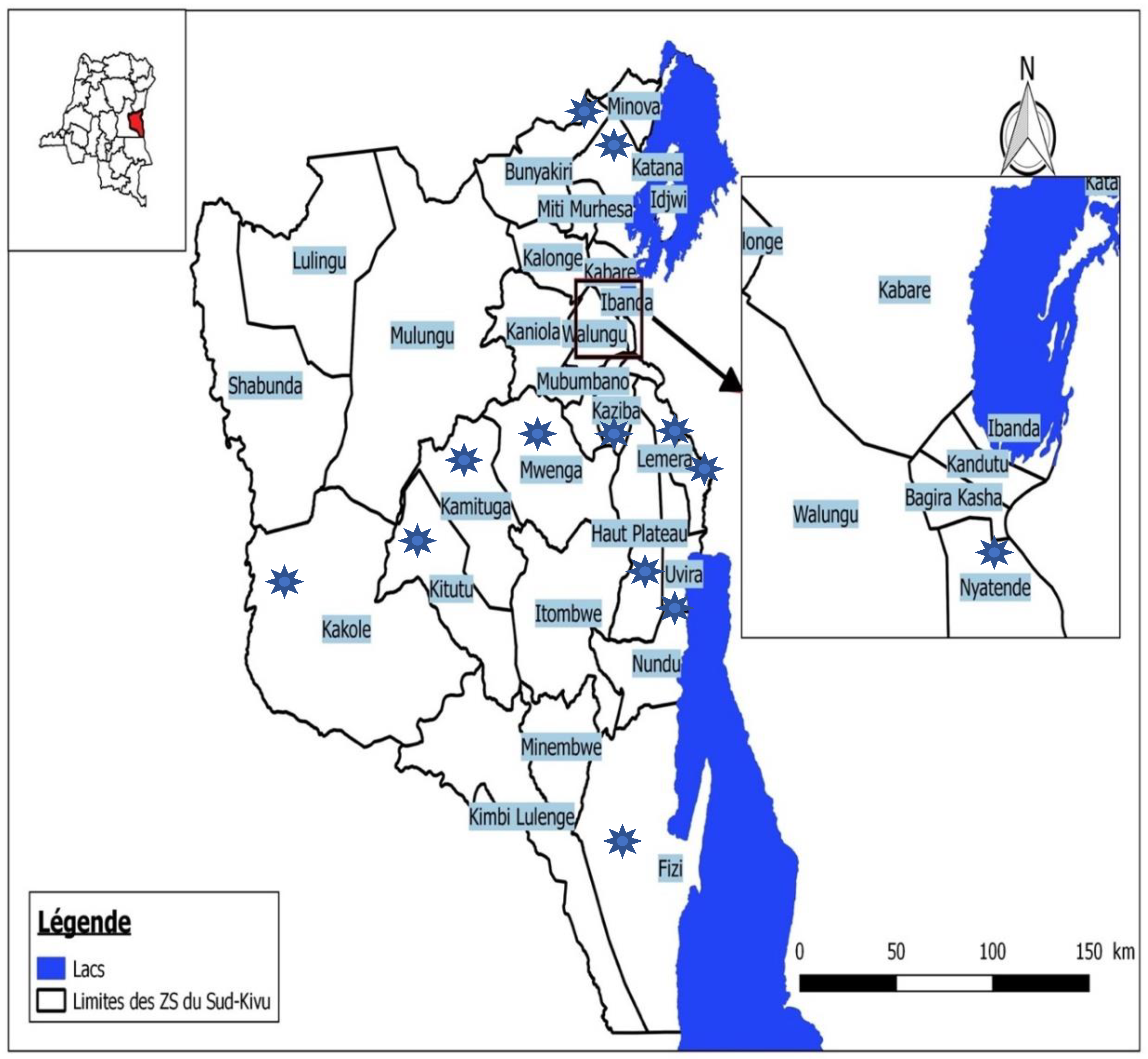
2.1. Design and Settings
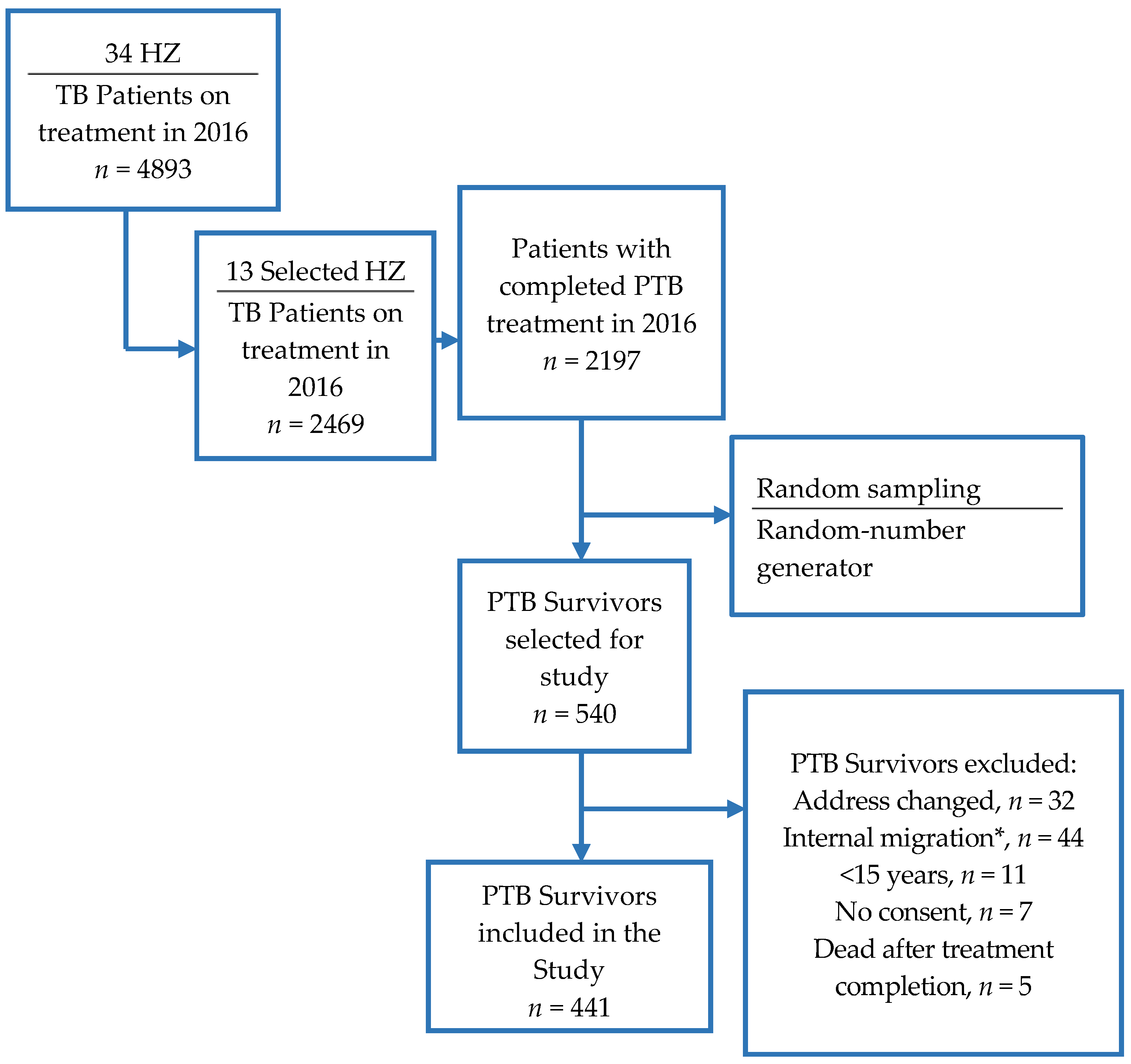
2.2. Participants
2.3. Instrument
2.4. Predictor Variables
2.5. Operational Definition of Variables
2.6. Outcome Variables
2.7. Statistical Analysis
2.8. Ethics Clearance
3. Results
3.1. Covariate Distribution Among PTB survivors by Gender
3.2. Reported Prevalence of Chronic Cough and Hemoptysis in PTB Survivors by Gender on Selected Covariates
3.3. Effects of HAP (Smoke from Biomass Fuel and Kerosene) and other Predictors on Chronic Cough or Hemoptysis among Former PTB Patients, by Gender
4. Discussion
5. Conclusions
Author Contributions
Acknowledgment
Conflicts of Interest
References
- Pai, M. Time for high-burden countries to lead the tuberculosis research agenda. PLoS Med. 2018, 15, e1002544. [Google Scholar] [CrossRef] [PubMed]
- Prediction of Under-Detection of Paediatric Tuberculosis in the Democratic Republic of Congo: Experience of Six Years in the South-Kivu Province. Available online: https://www.ncbi.nlm.nih.gov/pmc/ articles/PMC5217857/ (accessed on 13 June 2018).
- Global tuberculosis report 2018. Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 16 October 2018).
- Long Term Complications after Completion of Pulmonary Tuberculosis Treatment: A Quest for a Public Health Approach. Available online: https://www.clinicaltuberculosisjournal.com/article/S2405-5794(16)30005-5/abstract (accessed on 18 June 2018).
- Complications of Tuberculosis. Available online: https://insights.ovid.com/pubmed?pmid=25028786 (accessed on 11 June 2018).
- ERS/ATS Workshop Report on Respiratory Health Effects of Household Air Pollution. Available online: http://erj.ersjournals.com/content/51/1/1700698 (accessed on 11 June 2018).
- International Energy Agency. Available online: https://www.iea.org/ (accessed on 10 November 2018).
- Gordon, S.B.; Bruce, N.G.; Grigg, J.; Hibberd, P.L.; Kurmi, O.P.; Lam, K.H.; Mortimer, K.; Asante, K.P.; Balakrishnan, K.; et al. Respiratory risks from household air pollution in low and middle income countries. Lancet Respir. Med. 2014, 2, 823–860. [Google Scholar] [CrossRef]
- Akkara, S.A.; Shah, A.D.; Adalja, M.; Akkara, A.G.; Rathi, A.; Shah, D.N. Pulmonary tuberculosis: The day after. Int. J. Tuberc. Lung Dis. 2013, 17, 810–813. [Google Scholar] [CrossRef] [PubMed]
- Household Air Pollution and Health. Available online: http://www.who.int/mediacentre/factsheets /fs292/en/ (accessed on 9 November 2015).
- Pulmonary Tuberculosis Treated with Directly Observed Therapy: Serial Changes in Lung Structure and Function. Chest. Available online: http://www.sciencedirect.com/science/article/pii/S0012369215474951 (accessed on 13 June 2018).
- Chronic Pulmonary Function Impairment Caused by Initial and Recurrent Pulmonary Tuberculosis Following Treatment. Available online: http://thorax.bmj.com/content/55/1/32 (accessed on 12 June 2018).
- André, E.; Rusumba, O.; Evans, C.A.; Ngongo, P.; Sanduku, P.; Elvis, M.M.; Celestin, H.N.; Alain, I.R.; Musafiri, E.M.; Kabuayi, J.P.; et al. Patient-led active tuberculosis case-finding in the Democratic Republic of the Congo. Bull. World Health Organ. 2018, 96, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Saleh, S.; van Zyl-Smit, R.; Allwood, B.; Lawin, H.; Mbatchou Ngahane, B.H.; Ayakaka, I.; Moyo, E.; El-Sony, A.; Mortimer, K.; Rylance, J. Questionnaires for Lung Health in Africa across the Life Course. Int. J. Environ. Res. Public Health 2018, 15, 1615. [Google Scholar] [CrossRef] [PubMed]
- Indoor Air Pollution from Biomass Fuel Smoke Is a Major Health Concern in the Developing World. Available online: http://www.sciencedirect.com/science/article/pii/S003592030800271X (accessed on 12 November 2015).
- Pope, D.; Diaz, E.; Smith-Sivertsen, T.; Lie, R.T.; Bakke, P.; Balmes, J.R.; Smith, K.R.; Bruce, N.G. Exposure to household air pollution from wood combustion and association with respiratory symptoms and lung function in nonsmoking women: Results from the RESPIRE trial, Guatemala. Environ. Health Perspect. 2015, 123, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Household Air Pollution, Chronic Respiratory Disease and Pneumonia in Malawian Adults: A Case-Control Study. Available online: https://wellcomeopenresearch.org/articles/2-103/v1 (accessed on 11 June 2018).
- Bonjour, S.; Adair-Rohani, H.; Wolf, J.; Bruce, N.G.; Mehta, S.; Prüss-Ustün, A.; Lahiff, M.; Rehfuess, E.A.; Mishra, V.; Smith, K.R. Solid fuel use for household cooking: country and regional estimates for 1980–2010. Environ. Health Perspect. 2013, 121, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Assad, N.A.; Balmes, J.; Mehta, S.; Cheema, U.; Sood, A. Chronic obstructive pulmonary disease secondary to household air pollution. Semin. Respir. Crit. Care Med. 2015, 36, 408–421. [Google Scholar] [CrossRef] [PubMed]
- Post-Tuberculous Lung Function Impairment in a Tuberculosis Reference Clinic in Cameroon. Available online: http://search.proquest.com/docview/1787977597?rfr_id=info%3Axri%2Fsid%3Aprimo (accessed on 12 June 2018).
- GlobalData Report Store—Cigarettes in DR Congo. Available online: https://www.globaldata.com/store /report/cg0465mr--cigarettes-in-dr-congo-2018/ (accessed on 12 June 2018).
- Siddharthan, T.; Grigsby, M.R.; Goodman, D.; Chowdhury, M.; Rubinstein, A.; Irazola, V.; Gutierrez, L.; Miranda, J.J.; Bernabe-Ortiz, A.; Alam, D.; et al. Association between Household Air Pollution Exposure and Chronic Obstructive Pulmonary Disease Outcomes in 13 Low- and Middle-Income Country Settings. Am. J. Respir. Crit. Care Med. 2018, 197, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Effect of Indoor Air Pollution from Biomass and Solid Fuel Combustion on Prevalence of Self-Reported Asthma among Adult Men and Women in India: Findings from a Nationwide Large-Scale Cross-Sectional Survey. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5560475/ (accessed on 9 November 2018).
- Tuberculosis Country Profiles. Available online: http://www.who.int/tb/country/data/profiles/en/ (accessed on 12 June 2018).
- Prevalence and Outcomes of Xpert MTB/RIF-Detected Drug Susceptible and Resistant Tuberculosis in a Post-Conflict Region of the Democratic Republic of the Congo, 2012–2015. Available online: https://www.atsjournals.org/doi/abs/10.1164/ajrccm-conference.2017.195.1_MeetingAbstracts.A2112 (accessed on 10 November 2018).
- Risk Factors for Tuberculosis Treatment Failure, Default, or Relapse and Outcomes of Retreatment in Morocco. Available online: https://doi.org/10.1186/1471-2458-11-140 (accessed on 15 June 2018).
- Oshi, S.N.; Alobu, I.; Ukwaja, K.N.; Oshi, D.C. Investigating gender disparities in the profile and treatment outcomes of tuberculosis in Ebonyi state, Nigeria. Epidemiol. Infect. 2015, 143, 932–942. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Wang, X.; Ma, A.; Wang, Q.; Han, X.; Li, Y. Factors associated with patient and provider delays for tuberculosis diagnosis and treatment in Asia: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0120088. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, K.; Gordon, S.B.; Jindal, S.K.; Accinelli, R.A.; Balmes, J.; Martin, W.J. Household air pollution is a major avoidable risk factor for cardiorespiratory disease. Chest 2012, 142, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, A.L.; Miyashita, L.; Liu, N.M.; Lesosky, M.; Flitz, G.; Ndamala, C.; Balmes, J.R.; Gordon, R.B.; Mortimer, K.; Grigg, J. Use of cleaner-burning biomass stoves and airway macrophage black carbon in Malawian women. Sci. Total Environ. 2018, 635, 405–411. [Google Scholar] [CrossRef] [PubMed]
| All N = 441 | Males N = 298 (67.6) | Females N = 143 (32.4) | p-Value (Males vs. Females) | |
|---|---|---|---|---|
| Age (years), mean (±SD) | 44.6 (±14.9) | 44.9 (±14.6) | 43.9 (±15.6) | 0.24 |
| Age group 15–35 years 36–55 years >55 years | 121 (27.4) 214 (48.5) 106 (24.0) | 71 (23.8) 153 (51.3) 74 (24.8) | 50 (35.0) 61 (42.7) 32 (22.4) | 0.047 |
| Unmarried | 101 (22.9) | 57 (19.1) | 44 (30.7) | 0.006 |
| Kitchen outdoor Indoor + outdoor Indoor | 218 (49.4) 52 (11.8) 171 (38.8) | 145 (48.7) 34 (11.4) 119 (39.9) | 73 (51.1) 18 (12.6) 52 (36.4) | 0.76 |
| No kitchen ventilation | 82 (18.6) | 56 (18.8) | 26 (18.2) | 0.88 |
| Cooking > 3 h/day | 53 (34.7) | 92 (30.9) | 61 (42.7) | 0.015 |
| Unclean energy | 368 (83.5) | 248 (83.2) | 120 (83.9) | 0.85 |
| Tobacco smoke exposure | 81 (18.4) | 54 (18.1) | 27 (18.9) | 0.85 |
| Poverty | 433 (98.2) | 292 (98.0) | 141 (98.6) | 0.65 |
| Vermin/pests in home | 406 (92.1) | 271 (90.9) | 135 (94.4) | 0.21 |
| No sunlight in bedroom | 193 (43.8) | 140 (47.0) | 53 (37.1) | 0.049 |
| Any comorbidity | 113 (25.6) | 80 (26.9) | 33 (23.1) | 0.40 |
| Hospitalization for TB | 82 (18.6) | 45 (15.1) | 37 (25.9) | 0.006 |
| TB retreatment | 29 (6.6) | 21 (7.1) | 8 (5.6) | 0.57 |
| Ignore treatment outcome | 168 (38.1) | 108 (36.2) | 60 (42.0) | 0.25 |
| No DOT | 68 (15.4) | 47 (15.8) | 21 (14.7) | 0.77 |
| TB treatment >6 months | 49 (11.1) | 37 (12.4) | 12 (8.4) | 0.21 |
| Chronic Cough (N = 247) | Hemoptysis (N = 35) | |||||
|---|---|---|---|---|---|---|
| All N = 247/441 (56.0) | Males N = 171/298 (57.0) | Females N = 76/143 (53.1) | All N = 35/441 (7.9) | Males N = 23/298 (7.7) | Females N = 12/143 (8.4) | |
| Age group 15–35 years 36–55 years >55 years | 66 (26.7) 122 (49.4) 59 (23.9) | 37 (21.6) 89 (52.1) 45 (26.3) | 29 (38.2) 33 (43.4) 14 (18.4) | 7 (20.0) 18 (51.4) 10 (28.6) | 5 (21.7) 10 (43.5) 8 (34.8) | 2 (16.7) 8 (66.7) 2 (16.7) |
| Unmarried | 55 (22.3) | 33 (19.3) | 22 (29.0) | 6 (17.1) | 3 (13.0) | 3 (25.0) |
| Kitchen outdoor Indoor + outdoor Indoor | 127 (51.4) 25 (10.1) 95 (38.5) | 88 (51.5) 16 (9.7) 67 (39.2) | 39 (51.3) 9 (11.8) 28 (36.8) | 17 (48.6) 1 (2.9) 17 (48.6) | 12 (52.2) 1 (4.4) 10 (43.5) | 5 (41.7) 0 (0.0) 7 (58.3) |
| No kitchen ventilation | 45 (18.2) | 32 (18.7) | 13 (17.1) | 6 (17.1) | 4 (17.4) | 2 (16.7) |
| Cooking > 3 h/day | 102 (41.3) | 59 (34.5) | 43 (56.6) | 9 (25.7) | 3 (13.0) | 6 (50.0) |
| Unclean energy | 210 (85.0) | 142 (83.0) | 68 (89.5) | 31 (88.6) | 19 (82.6) | 12 (100.0) |
| Tobacco smoke exposure | 42 (17.0) | 33 (19.3) | 9 (11.8) | 6 (17.1) | 6 (26.1) | 0 (0.0) |
| Poverty | 244 (98.8) | 169 (98.8) | 75 (98.7) | 34 (97.1) | 22 (95.7) | 12 (100.0) |
| Vermin/pests in home | 225 (91.1) | 156 (91.2) | 69 (90.8) | 32 (91.4) | 21 (91.3) | 11 (91.7) |
| No sunlight in bedroom | 106 (42.9) | 79 (46.2) | 27 (35.5) | 15 (42.9) | 10 (43.5) | 5 (41.7) |
| Any comorbidity | 74 (30.0) | 53 (31.0) | 21 (27.6) | 10 (28.6) | 7 (30.4) | 3 (25.0) |
| Hospitalization for TB | 51(20.7) | 29 (17.0) | 22 (29.0) | 6 (17.1) | 4 (17.4) | 2 (16.7) |
| TB retreatment | 16 (6.5) | 13 (7.6) | 3 (4.0) | 5 (14.3) | 3 (13.0) | 2 (16.7) |
| Ignore treatment outcome | 89 (36.3) | 60 (35.9) | 29 (38.2) | 19 (54.3) | 13 (56.5) | 6 (50.0) |
| No DOT | 35 (14.7) | 28 (16.4) | 7 (9.2) | 6 (17.1) | 6 (26.1) | 0 (0.0) |
| TB treatment > 6 months | 41 (16.6) | 31 (18.3) | 10 (13.2) | 3 (8.6) | 2 (8.7) | 1 (8.3) |
| Chronic Cough | ||||||
|---|---|---|---|---|---|---|
| Crude OR [95% CI] | Adjusted* OR [95% CI] | |||||
| All | Males | Females | All | Males | Females | |
| Sex | 0.84 [0.56–1.26] | 0.84 [0.54–1.28] | - | - | ||
| Age group 15–35 years 36–55 years >55 years | Ref 1.11 [0.71–1.73] 1.05 [0.62–1.77] | Ref 1.28 [0.73–2.25] 1.43 [0.74–2.76] | Ref 0.85 [0.40–1.81] 0.56 [0.23–1.38] | Ref 1.21 [0.75–1.94] 1.11 [0.64–1.93] | Ref 1.55 [0.85–2.81] 1.71 [0.86–3.40] | Ref 0.76 [0.33–1.73] 0.42 [0.15–1.15 |
| Unmarried | 0.92 [0.59–1.44] | 1.03 [0.57–1.84] | 0.83 [0.41–1.70] | - | - | - |
| Kitchen Outdoor Indoor + outdoor Indoor | Ref 0.66 [0.36–1.22] 0.90 [0.60–1.34] | Ref 0.58 [0.27–1.22] 0.84 [0.51–1.36] | Ref 0.87 [0.31–2.45] 1.02 [0.50–2.08] | - | - | - |
| No kitchen ventilation | 0.95 [0.58–1.53] | 0.99 [0.55–1.78] | 0.86 [0.37–2.01] | - | - | - |
| Cooking > 3 h/day | 1.97 [1.31–2.97] ** | 1.50 [0.90–2.49] | 3.55 [1.75–7.18] *** | 1.45 [0.93–2.27] | 1.06 [0.61–1.85] | 2.74 [1.25–6.07] * |
| Unclean energy | 1.29 [0.78–2.14] | 0.97 [0.52–1.79] | 2.45 [0.97–6.22] | 2.10 [1.10–4.00] * | 1.61 [0.74–3.50] | 3.93 [1.15–13.4] * |
| Tobacco smoke exposure | 0.81 [0.50–1.32] | 1.21 [0.66–2.21] | 0.37 [0.15–0.88] * | - | - | - |
| Poverty | 2.15 [0.51–9.12] | 2.75 [0.50–15.24] | 1.14 [0.07–18.53] | - | - | - |
| Vermin/pests in home | 0.73 [0.36–1.50] | 1.09 [0.50–2.41] | 0.15 [0.02–1.25] | - | - | - |
| No sunlight in bedroom | 0.92 [0.63–1.35] | 0.93 [0.59–1.47] | 0.87 [0.44–1.71] | - | - | - |
| Any comorbidity | 1.70 [1.09–2.65] * | 1.66 [0.98–2.84] | 1.75 [0.79–3.90] | 1.63 [0.97–2.74] | 1.56 [0.83–2.93] | 1.74 [0.67–4.53] |
| Hospitalization for TB | 1.37 [0.84–2.24] | 1.42 [0.73–2.74 | 1.41 [0.66–3.02] | 1.34 [0.75–2.41] | 1.14 [0.53–2.43] | 1.98 [0.75–5.24] |
| TB retreatment | 0.96 [0.45–2.06] | 1.22 [0.49–3.05] | 0.51 [0.12–2.22] | - | - | - |
| Ignore treatment outcome | 0.82 [0.56–1.21] | 0.89 [0.56–1.43] | 0.72 [0.34–1.40] | - | - | - |
| No DOT | 0.81 [0.48–1.35] | 1.11 [0.59–2.10] | 0.38 [0.15–1.02] | - | - | - |
| TB treatment > 6 months | 4.63 [2.11–10.1] *** | 4.47 [1.80–11.1] ** | 4.92 [1.04–23.4] * | 3.80 [1.62–8.96] ** | 4.45 [1.64–12.1] ** | 2.94 [0.50–17.43] |
| Hemoptysis | ||||||
|---|---|---|---|---|---|---|
| Crude OR [95% CI] | Adjusted § OR [95% CI] | |||||
| All | Male | Female | All | Male | Female | |
| Sex | 1.10 [0.53–2.27] | - | - | 1.20 [0.57–2.55] | - | - |
| Age group 15–35 years 36–55 years >55 years | Ref 1.50 [0.61–3.69] 1.70 [0.62–4.63] | Ref 0.92 [0.30–2.81] 1.6 [0.50–5.15] | Ref 3.62 [0.73–17.91] 1.6 [0.21–11.97] | Ref 1.66 [0.66–4.20] 1.70 [0.61–4.74] | Ref 0.99 [0.32–3.09] 1.60 [0.48–5.31] | Ref 5.25 [0.81–34.0] 2.18 [0.22–21.8] |
| Unmarried | 0.68 [0.27–1.68] | 0.61 [0.18–2.14] | 0.73 [0.19–2.84] | - | - | - |
| Kitchen Outdoor Indoor + Outdoor Indoor | Ref 0.23 [0.03–1.08] 1.31 [0.65–2.64] | Ref 0.34 [0.04–2.68] 1.02 [0.42–2.44] | Ref NE 2.12 [0.63–7.08] | - | - | - |
| No kitchen ventilation | 0.90 [0.36–2.24] | 0.90 [0.30–2.77] | 0.89 [0.18–4.34] | - | - | - |
| Cooking > 3 h/day | 0.63 [0.29–1.38] | 0.31 [0.09–1.08] | 1.38 [0.42–4.51] | 0.63 [0.28–1.40] | 0.34 [0.10–1.18] | 1.28 [0.36–4.59] |
| Unclean energy | 1.59 [0.54–4.64] | 0.95 [0.31–2.94] | NE | 1.86 [0.61–5.62] | 1.07 [0.33–3.41] | NE |
| Tobacco smoke exposure | 0.91 [0.37–2.28] | 1.67 [0.63–4.45] | NE | - | - | - |
| Poverty | 0.60 [0.07–4.99] | 0.41 [0.05–3.64] | NE | - | - | - |
| Vermin/pests in home | 0.91 [0.27–3.15] | 1.05 [0.23–4.74] | 0.62 [0.07–5.52] | - | - | - |
| No sunlight in bedroom | 0.97 [0.48–1.93] | 0.86 [0.36–2.02] | 1.24 [0.37–4.11] | - | - | - |
| Any comorbidity | 1.18 [0.55–2.53] | 1.21 [0.48–3.06] | 1.12 [0.29–4.41] | - | - | - |
| Hospitalization for TB | 0.90 [0.36–2.24] | 1.20 [0.39–3.71] | 0.56 [0.11–2.63] | - | - | - |
| TB retreatment | 2.56 [0.95–7.45] | 2.14 [0.58–7.86] | 4.17 [0.74–23.4] | 3.04 [1.04–5.09] * | 1.93 [0.50–7.54] | 8.71 [1.00–77.2] |
| Ignore treatment outcome | 2.05 [1.02–4.10] * | 2.46 [1.04–5.83] * | 1.43 [0.44–4.66] | 2.24 [1.09–4.58] * | 2.44 [1.01–5.88] * | 2.06 [0.57–7.54] |
| No DOT | 1.15 [0.46–2.88] | 2.01 [0.75–5.41] | NE | - | - | - |
| TB treatment > 6 months | 0.73 [0.22–2.50] | 0.65 [0.15–2.91] | 0.99 [0.12–8.41] | - | - | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katoto, P.D.M.C.; Murhula, A.; Kayembe-Kitenge, T.; Lawin, H.; Bisimwa, B.C.; Cirhambiza, J.P.; Musafiri, E.; Birembano, F.; Kashongwe, Z.; Kirenga, B.; et al. Household Air Pollution Is Associated with Chronic Cough but Not Hemoptysis after Completion of Pulmonary Tuberculosis Treatment in Adults, Rural Eastern Democratic Republic of Congo. Int. J. Environ. Res. Public Health 2018, 15, 2563. https://doi.org/10.3390/ijerph15112563
Katoto PDMC, Murhula A, Kayembe-Kitenge T, Lawin H, Bisimwa BC, Cirhambiza JP, Musafiri E, Birembano F, Kashongwe Z, Kirenga B, et al. Household Air Pollution Is Associated with Chronic Cough but Not Hemoptysis after Completion of Pulmonary Tuberculosis Treatment in Adults, Rural Eastern Democratic Republic of Congo. International Journal of Environmental Research and Public Health. 2018; 15(11):2563. https://doi.org/10.3390/ijerph15112563
Chicago/Turabian StyleKatoto, Patrick D.M.C., Aime Murhula, Tony Kayembe-Kitenge, Herve Lawin, Bertin C. Bisimwa, Jean Paul Cirhambiza, Eric Musafiri, Freddy Birembano, Zacharie Kashongwe, Bruce Kirenga, and et al. 2018. "Household Air Pollution Is Associated with Chronic Cough but Not Hemoptysis after Completion of Pulmonary Tuberculosis Treatment in Adults, Rural Eastern Democratic Republic of Congo" International Journal of Environmental Research and Public Health 15, no. 11: 2563. https://doi.org/10.3390/ijerph15112563
APA StyleKatoto, P. D. M. C., Murhula, A., Kayembe-Kitenge, T., Lawin, H., Bisimwa, B. C., Cirhambiza, J. P., Musafiri, E., Birembano, F., Kashongwe, Z., Kirenga, B., Mfinanga, S., Mortimer, K., De Boever, P., Nawrot, T. S., Nachega, J. B., & Nemery, B. (2018). Household Air Pollution Is Associated with Chronic Cough but Not Hemoptysis after Completion of Pulmonary Tuberculosis Treatment in Adults, Rural Eastern Democratic Republic of Congo. International Journal of Environmental Research and Public Health, 15(11), 2563. https://doi.org/10.3390/ijerph15112563






