Sources and Characteristics of Particulate Matter in Subway Tunnels in Seoul, Korea
Abstract
1. Introduction
2. Materials and Methods
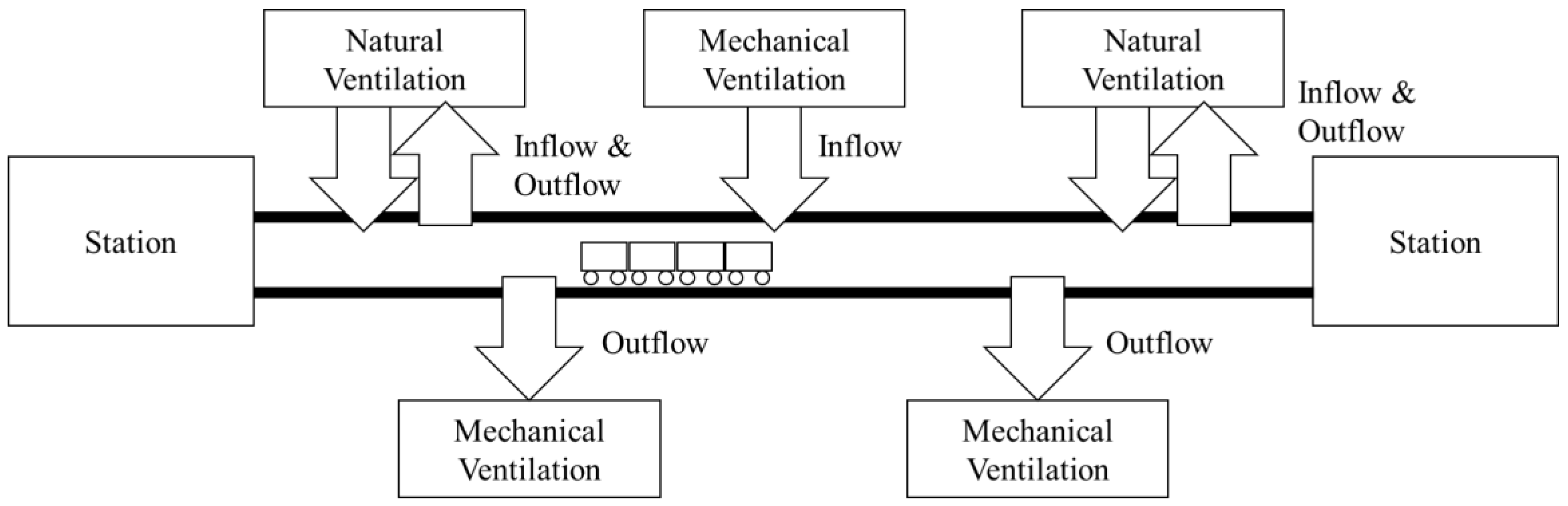
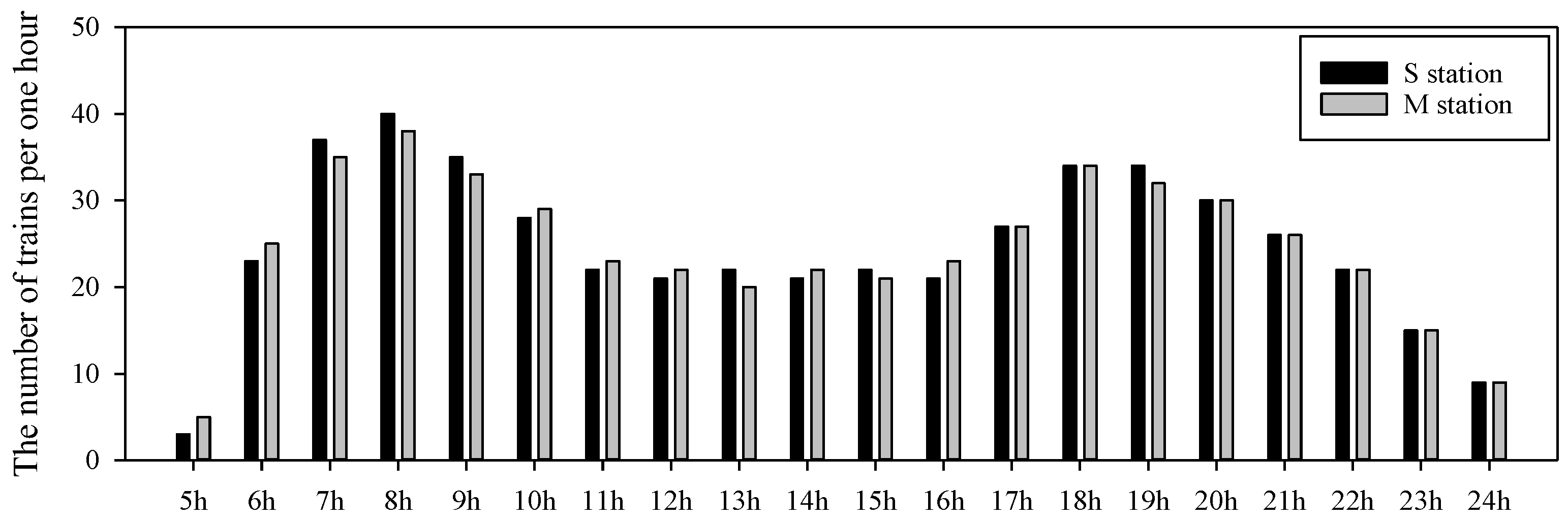
2.1. Study Area
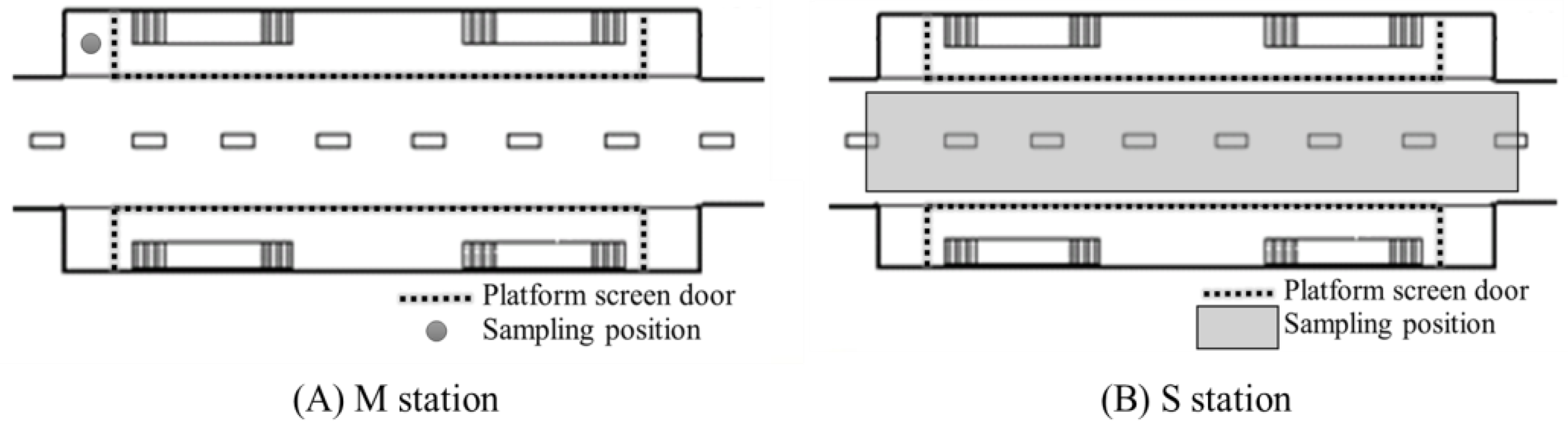
2.2. Collection and Analysis of Samples
2.3. Estimated Carbonate by Ion Balance
3. Results and Discussion
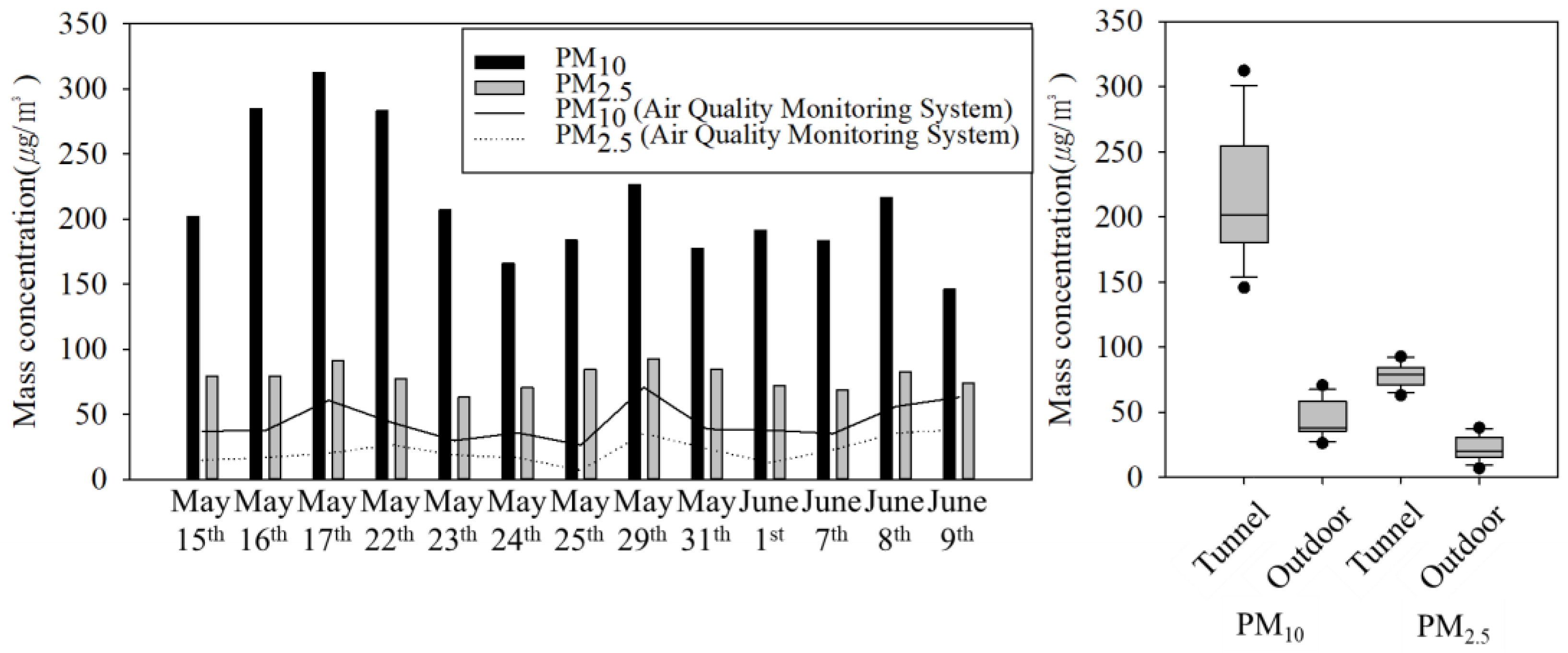
3.1. PM Mass
3.2. Chemical Composition of PM
3.2.1. Carbonaceous Compounds
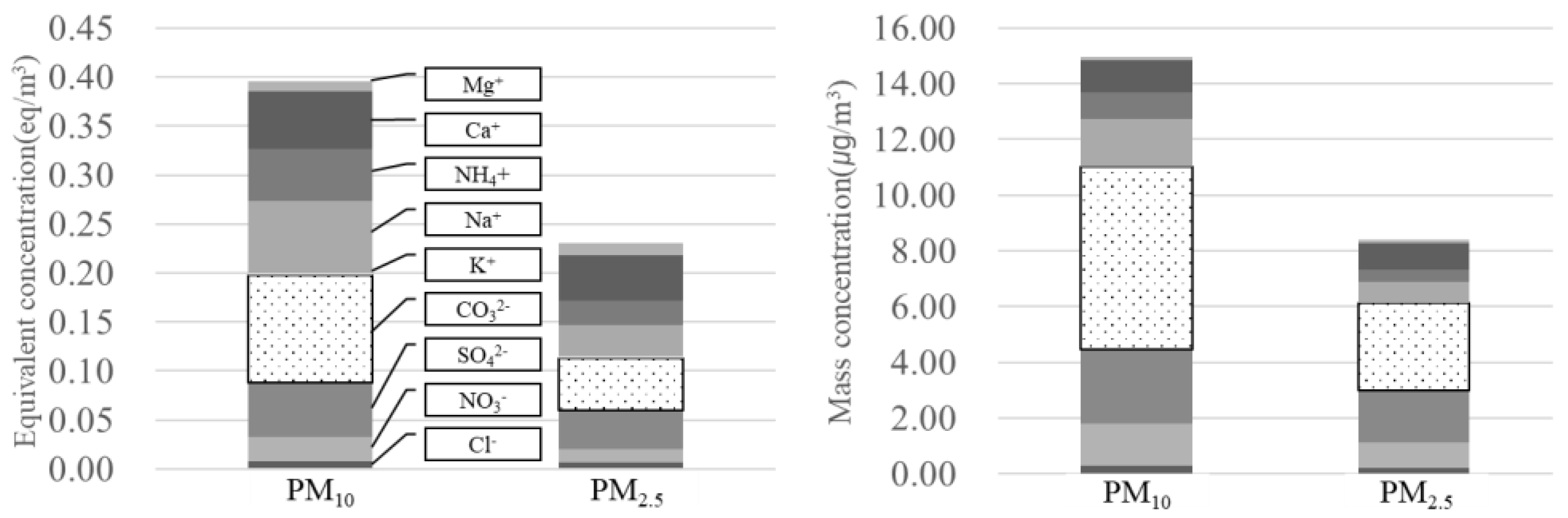
3.2.2. Ionic Compounds
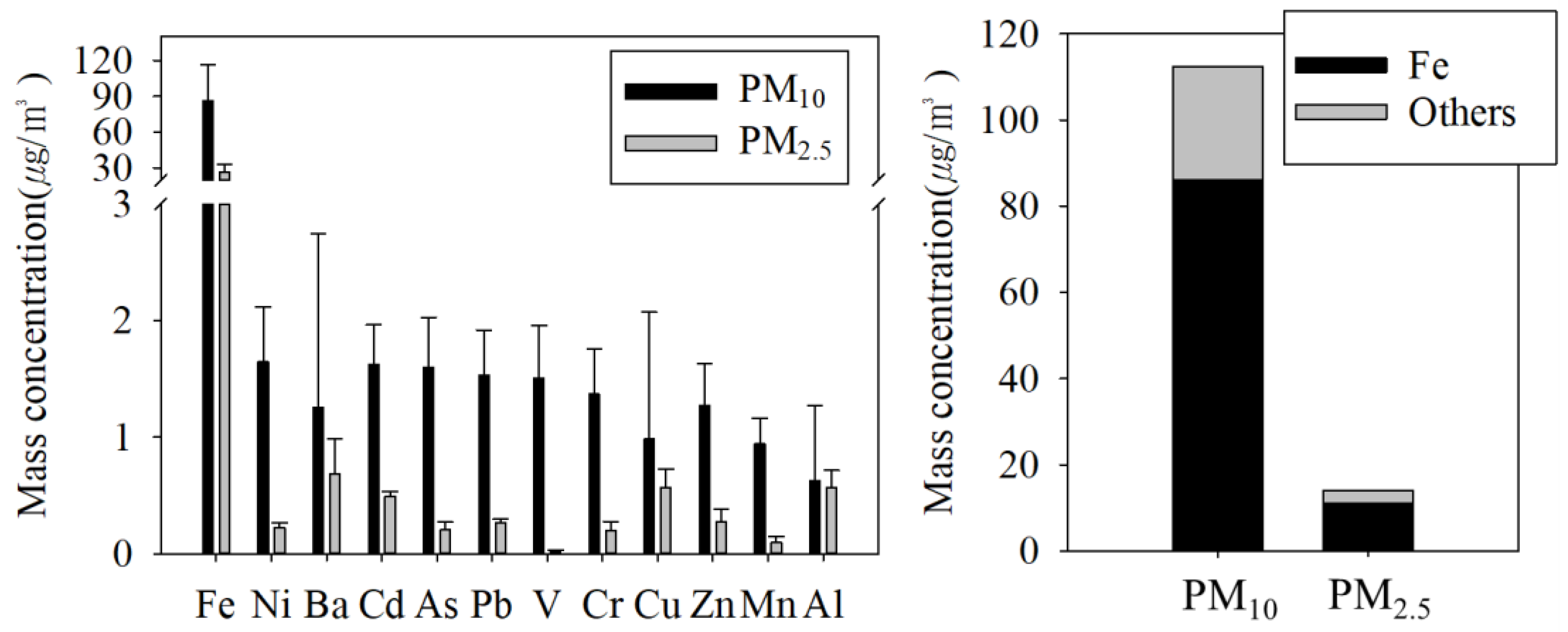
3.2.3. Inorganic Compounds
3.3. Morphology and Energy-Dispersive Spectroscopy (EDX)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pan, S.; Fan, L.; Liu, J.P.; Xie, J.C.; Sun, Y.Y.; Cui, N.; Zhang, L.L.; Zheng, B.Y. A review of the piston effect in subway stations. Adv. Mech. Eng. 2013, 5. [Google Scholar] [CrossRef]
- Lee, Y.; Choi, K.; Jung, W.; Versoza, M.E.; Barabad, M.L.M.; Kim, T.; Park, D. Generation characteristics of nanoparticles emitted from subways in operation. Aerosol Air Qual. Res. 2018, 18, 2230–2239. [Google Scholar] [CrossRef]
- Abbasi, S.; Wahlstrom, J.; Olander, L.; Larsson, C.; Olofsson, U.; Sellgren, U. A study of airborne wear particles generated from organic railway brake pads and brake discs. Wear 2011, 273, 93–99. [Google Scholar] [CrossRef]
- Arias-Cuevas, O.; Li, Z.; Lewis, R.; Gallardo-Hernandez, E.A. Rolling-sliding laboratory tests of friction modifiers in dry and wet wheel-rail contacts. Wear 2010, 268, 543–551. [Google Scholar] [CrossRef]
- Olofsson, U. A study of airborne wear particles generated from the train traffic-block braking simulation in a pin-on-disc machine. Wear 2011, 271, 86–91. [Google Scholar] [CrossRef]
- Xu, B.; Hao, J. Air quality inside subway metro indoor environment worldwide: A review. Environ. Int. 2017, 107, 33–46. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Review of Evidence on Health Aspects of Air Pollution—REVIHAAP Project: Technical Report; WHO: Copenhagen, Denmark, 2013. [Google Scholar]
- He, G.; Fan, M.; Zhou, M. The effect of air pollution on mortality in China: Evidence from the 2008 Beijing Olympic Games. J. Environ. Econ. Manag. 2016, 79, 18–39. [Google Scholar] [CrossRef]
- Beelen, R.; Hoek, G.; van den Brandt, P.A.; Goldbohm, R.A.; Fischer, P.; Schouten, L.J.; Jerrett, M.; Hughes, E.; Armstrong, B.; Brunekreef, B. Long-term effects of traffic-related air pollution on mortality in a Dutch cohort (NLCS-AIR study). Environ. Health Perspect. 2008, 116, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Nieuwenhuijsen, M.J. Exposure Assessment in Occupational and Environmental Epidemiology; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Karlsson, H.L.; Nilsson, L.; Moller, L. Subway particles are more genotoxic than street particles and induce oxidative stress in cultured human lung cells. Chem. Res. Toxicol. 2005, 18, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.L.; Ljungman, A.G.; Lindbom, J.; Moller, L. Comparison of genotoxic and inflammatory effects of particles generated by wood combustion, a road simulator and collected from street and subway. Toxicol. Lett. 2006, 165, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Maher, B.A.; Ahmed, I.A.M.; Karloukovski, V.; MacLaren, D.A.; Foulds, P.G.; Allsop, D.; Mann, D.M.A.; Torres-Jardon, R.; Calderon-Garciduenas, L. Magnetite pollution nanoparticles in the human brain. Proc. Natl. Acad. Sci. USA 2016, 113, 10797–10801. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.W.; Kang, S.J.; Matsubayashi, T.; Sawada, Y.; Ueda, M. The effectiveness of platform screen doors for the prevention of subway suicides in South Korea. J. Affect. Disord. 2016, 194, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Ho, D.X.; Jeon, J.S.; Kim, J.C. A noticeable shift in particulate matter levels after platform screen door installation in a Korean subway station. Atmos. Environ. 2012, 49, 219–223. [Google Scholar] [CrossRef]
- Park, D.; Lee, T.; Hwang, D.; Jung, W.; Lee, Y.; Cho, K.; Kim, D.; Lee, K. Identification of the sources of PM10 in a subway tunnel using positive matrix factorization. Air Waste Manag. Assoc. 2014, 64, 1361–1368. [Google Scholar] [CrossRef]
- Querol, X.; Moreno, T.; Karanasiou, A.; Reche, C.; Alastuey, A.; Viana, M.; Font, O.; Gil, J.; de Miguel, E.; Capdevila, M. Variability of levels and composition of PM10 and PM2.5 in the Barcelona metro system. Atmos. Chem. Phys. 2012, 12, 5055–5076. [Google Scholar] [CrossRef]
- Namgung, H.G.; Kim, J.B.; Woo, S.H.; Park, S.; Kim, M.; Kim, M.S.; Bae, G.N.; Park, D.; Kwon, S.B. Generation of nanoparticles from friction between railway brake disks and pads. Environ. Sci. Technol. 2016, 50, 3453–3461. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.H.; Kim, J.B.; Bae, G.N.; Hwang, M.S.; Tahk, G.H.; Yoon, H.H.; Yook, S.J. Investigation of diurnal pattern of generation and resuspension of particles induced by moving subway trains in an underground tunnel. Aerosol Air Qual. Res. 2018, 18, 2240–2252. [Google Scholar] [CrossRef]
- Midander, K.; Elihn, K.; Wallen, A.; Belova, L.; Karlsson, A.K.B.; Wallinder, I.O. Characterisation of nano- and micron-sized airborne and collected subway particles, a multi-analytical approach. Sci. Total Environ. 2012, 427, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Kim, B.; Ryu, J.; Maskey, S.; Kim, J.C.; Sohn, J.; Ro, C.U. Source identification of particulate matter collected at underground subway stations in Seoul, Korea using quantitative single-particle analysis. Atmos. Environ. 2010, 44, 2287–2293. [Google Scholar] [CrossRef]
- Guo, L.; Hu, Y.J.; Hu, Q.Q.; Lin, J.; Li, C.L.; Chen, J.M.; Li, L.N.; Fu, H.B. Characteristics and chemical compositions of particulate matter collected at the selected metro stations of Shanghai, China. Sci. Total Environ. 2014, 496, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Moreno, T.; Martins, V.; Querol, X.; Jones, T.; BeruBe, K.; Minguillon, M.C.; Amato, F.; Capdevila, M.; de Miguel, E.; Centelles, S.; et al. A new look at inhalable metalliferous airborne particles on rail subway platforms. Sci. Total Environ. 2015, 505, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Kim, B.; Malek, M.A.; Koo, Y.S.; Jung, J.N.; Son, Y.S.; Kim, J.C.; Kim, H.; Ro, C.U. Chemical speciation of size-segregated floor dusts and airborne magnetic particles collected at underground subway stations in Seoul, Korea. J. Hazard. Mater. 2012, 213, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, A.C. Heterogeneous reactions of sulfur dioxide with carbonaceous particles. Int. J. Chem. Kinet. 1982, 14, 269–277. [Google Scholar] [CrossRef]
- Ibusuki, T.; Barnes, H.M. Manganese(II) catalyzed sulfur dioxide oxidation in aqueous solution at environmental concentrations. Atmos. Environ. (1967) 1984, 18, 145–151. [Google Scholar] [CrossRef]
- Sinha, A.; Bhattacharya, J. Environmental Kuznets curve estimation for NO2 emission: A case of Indian cities. Ecol. Indic. 2016, 67, 1–11. [Google Scholar] [CrossRef]
- Krachler, M.; Mohl, C.; Emons, H.; Shotyk, W. Atmospheric deposition of V, Cr, and Ni since the late glacial: Effects of climatic cycles, human impacts, and comparison with crustal abundances. Environ. Sci. Technol. 2003, 37, 2658–2667. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lee, K.B.; Kim, J.S.; Park, D.; Kim, S.D. Study on the estimation of configuration ratio of ion compound with carbonate in subway. J. Odor. Indoor Environ. 2016, 15, 154–163. [Google Scholar] [CrossRef]
- Loxham, M.; Cooper, M.J.; Gerlofs-Nijland, M.E.; Cassee, F.R.; Davies, D.E.; Palmer, M.R.; Teagle, D.A.H. Physicochemical characterization of airborne particulate matter at a mainline underground railway station. Environ. Sci. Technol. 2013, 47, 3614–3622. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Oh, M.; Yoon, Y.; Park, E.; Lee, K. Source identification of PM10 pollution in subway passenger cabins using positive matrix factorization. Atmos. Environ. 2012, 49, 180–185. [Google Scholar] [CrossRef]
- Martins, V.; Moreno, T.; Mendes, L.; Eleftheriadis, K.; Diapouli, E.; Alves, C.A.; Duarte, M.; de Miguel, E.; Capdevila, M.; Querol, X.; et al. Factors controlling air quality in different European subway systems. Environ. Res. 2016, 146, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Birch, M.E.; Cary, R.A. Elemental carbon-based method for monitoring occupational exposures to particulate diesel exhaust. Aerosol Sci. Technol. 1996, 25, 221–241. [Google Scholar] [CrossRef]
- Jankowski, N.; Schmidl, C.; Marr, I.L.; Bauer, H.; Puxbaum, H. Comparison of methods for the quantification of carbonate carbon in atmospheric PM10 aerosol samples. Atmos. Environ. 2008, 42, 8055–8064. [Google Scholar] [CrossRef]
- Krzyzanowski, M.; Cohen, A. Update of WHO air quality guidelines. Air Qual. Atmos. Health 2008, 1, 7–13. [Google Scholar] [CrossRef]
- Yang, H.J.; Chen, G.X.; Gao, G.Q.; Wu, G.N.; Zhang, W.H. Experimental research on the friction and wear properties of a contact strip of a pantograph-catenary system at the sliding speed of 350 km/h with electric current. Wear 2015, 332, 949–955. [Google Scholar] [CrossRef]
- Moreno, T.; Reche, C.; Minguillon, M.C.; Capdevila, M.; de Miguel, E.; Querol, X. The effect of ventilation protocols on airborne particulate matter in subway systems. Sci. Total Environ. 2017, 584–585, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.H.; Lin, Y.L.; Liu, C.C. Levels of PM10 and PM2.5 in Taipei rapid transit system. Atmos. Environ. 2008, 42, 7242–7249. [Google Scholar] [CrossRef]
- Lee, Y.; Jeong, W.; Choi, K.; Kim, T.; Park, D. Estimation of particulate matters in subway tunnels using a kriging method. J. Odor. Indoor Environ. 2016, 15, 126–133. [Google Scholar] [CrossRef]
- Johansson, C.; Johansson, P.A. Particulate matter in the underground of Stockholm. Atmos. Environ. 2003, 37, 3–9. [Google Scholar] [CrossRef]
- Mugica-Álvarez, V.; Figueroa-Lara, J.; Romero-Romo, M.; Sepúlveda-Sánchez, J.; López-Moreno, T. Concentrations and properties of airborne particles in the Mexico City subway system. Atmos. Environ. 2012, 49, 284–293. [Google Scholar] [CrossRef]
- Martins, V.; Moreno, T.; Minguillon, M.C.; van Drooge, B.L.; Reche, C.; Amato, F.; de Miguel, E.; Capdevila, M.; Centelles, S.; Querol, X. Origin of inorganic and organic components of PM2.5 in subway stations of Barcelona, Spain. Environ. Pollut. 2016, 208, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.C.; Watson, J.G.; Crow, D.; Lowenthal, D.H.; Merrifield, T. Comparison of IMPROVE and NIOSH carbon measurements. Aerosol Sci. Technol. 2001, 34, 23–34. [Google Scholar] [CrossRef]
- Zhang, C.M.; Song, W.; Zhang, X.C.; Li, R.; Zhao, S.J.; Fan, C.M. Synthesis, characterization and evaluation of resin-based carbon spheres modified by oxygen functional groups for gaseous elemental mercury capture. J. Mater. Sci. 2018, 53, 9429–9448. [Google Scholar] [CrossRef]
- Hering, S.; Cass, G. The Magnitude of bias in the measurement of PM2.5 arising from volatilization of particulate nitrate from teflon filters. J. Air Waste Manag. Assoc. 1999, 49, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Namgung, H.-G.; Kim, J.B.; Kim, M.-S.; Kim, M.; Park, S.; Woo, S.-H.; Bae, G.-N.; Park, D.; Kwon, S.-B. Size distribution analysis of airborne wear particles released by subway brake system. Wear 2017, 372–373, 169–176. [Google Scholar] [CrossRef]
- Minguillon, M.C.; Reche, C.; Martins, V.; Amato, F.; de Miguel, E.; Capdevila, M.; Centelles, S.; Querol, X.; Moreno, T. Aerosol sources in subway environments. Environ. Res. 2018, 167, 314–328. [Google Scholar] [CrossRef] [PubMed]
- Chillrud, S.N.; Epstein, D.; Ross, J.M.; Sax, S.N.; Pederson, D.; Spengler, J.D.; Kinney, P.L. Elevated airborne exposures of teenagers to manganese, chromium, and iron from steel dust and New York City’s subway system. Environ. Sci. Technol. 2004, 38, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.G.; Jiang, H.M.; Dong, C.Y.; Yan, Q.; Yu, L.Z.; Yu, Y. Magnetic and geochemical characterization of iron pollution in subway dusts in Shanghai, China. Geochem. Geophys. Geosyst. 2011, 12. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, L.; Zhu, D.; Gao, H.O.; Xie, Y.; Li, H.; Xu, X.; Wang, H. Characteristics of particulate matter (PM) concentrations influenced by piston wind and train door opening in the Shanghai subway system. Transp. Res. D 2016, 47, 77–88. [Google Scholar] [CrossRef]
- Salma, I.; Weidinger, T.; Maenhaut, W. Time-resolved mass concentration, composition and sources of aerosol particles in a metropolitan underground railway station. Atmos. Environ. 2007, 41, 8391–8405. [Google Scholar] [CrossRef]
- Aarnio, P.; Yli-Tuomi, T.; Kousa, A.; Makela, T.; Hirsikko, A.; Hameri, K.; Raisanen, M.; Hillamo, R.; Koskentalo, T.; Jantunen, M. The concentrations and composition of and exposure to fine particles (PM2.5) in the Helsinki subway system. Atmos. Environ. 2005, 39, 5059–5066. [Google Scholar] [CrossRef]
- Bukowiecki, N.; Gehrig, R.; Hill, M.; Lienemann, P.; Zwicky, C.N.; Buchmann, B.; Weingartner, E.; Baltensperger, U. Iron, manganese and copper emitted by cargo and passenger trains in Zurich (Switzerland): Size-segregated mass concentrations in ambient air. Atmos. Environ. 2007, 41, 878–889. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, G.X.; Zhang, S.D.; Gao, G.Q.; Wu, G.N.; Zhang, W.H.; Zhou, Z.R. Comparative investigation into the friction and wear behaviours of a Cu-Ag contact wire/carbon strip and a pure copper contact wire/carbon strip at high speeds. Wear 2017, 376, 1552–1557. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Zhang, Y.Z.; Du, S.M.; Song, C.F.; Yang, Z.H.; Shangguan, B. Tribological properties of pure carbon strip affected by dynamic contact force during current-carrying sliding. Tribol. Int. 2018, 123, 256–265. [Google Scholar] [CrossRef]
- Ma, C.-J.; Lee, K.-B.; Kim, S.-D.; Sera, K. Chemical properties and source profiles of particulate matter collected on an underground subway platform. Asian J. Atmos. Environ. 2015, 9, 165–172. [Google Scholar] [CrossRef]
- Karlsson, H.L.; Gustafsson, J.; Cronholm, P.; Moller, L. Size-dependent toxicity of metal oxide particles—A comparison between nano- and micrometer size. Toxicol. Lett. 2009, 188, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Isozaki, H.; Oosawa, J.; Kawano, Y.; Hirasawa, R.; Kubota, S.; Konishi, S. Measures against electrolytic rail corrosion in Tokyo Metro Subway Tunnels. Procedia Eng. 2016, 165, 583–592. [Google Scholar] [CrossRef]
- Guo, H.B.; Barnard, A.S. Naturally occurring iron oxide nanoparticles: Morphology, surface chemistry and environmental stability. J. Mater. Chem. A 2013, 1, 27–42. [Google Scholar] [CrossRef]
- Choi, S.I.; Feng, J.; Kim, S.B.; Jo, Y.M. Magnetization of metal mesh for fine dust capture. Aerosol Air Qual. Res. 2018, 18, 1932–1943. [Google Scholar] [CrossRef]
- Salma, I.; Posfai, M.; Kovacs, K.; Kuzmann, E.; Homonnay, Z.; Posta, J. Properties and sources of individual particles and some chemical species in the aerosol of a metropolitan underground railway station. Atmos. Environ. 2009, 43, 3460–3466. [Google Scholar] [CrossRef]
- Kang, S.; Hwang, H.; Park, Y.; Kim, H.; Ro, C.U. Chemical compositions of subway particles in Seoul, Korea determined by a quantitative single particle analysis. Environ. Sci. Technol. 2008, 42, 9051–9057. [Google Scholar] [CrossRef] [PubMed]
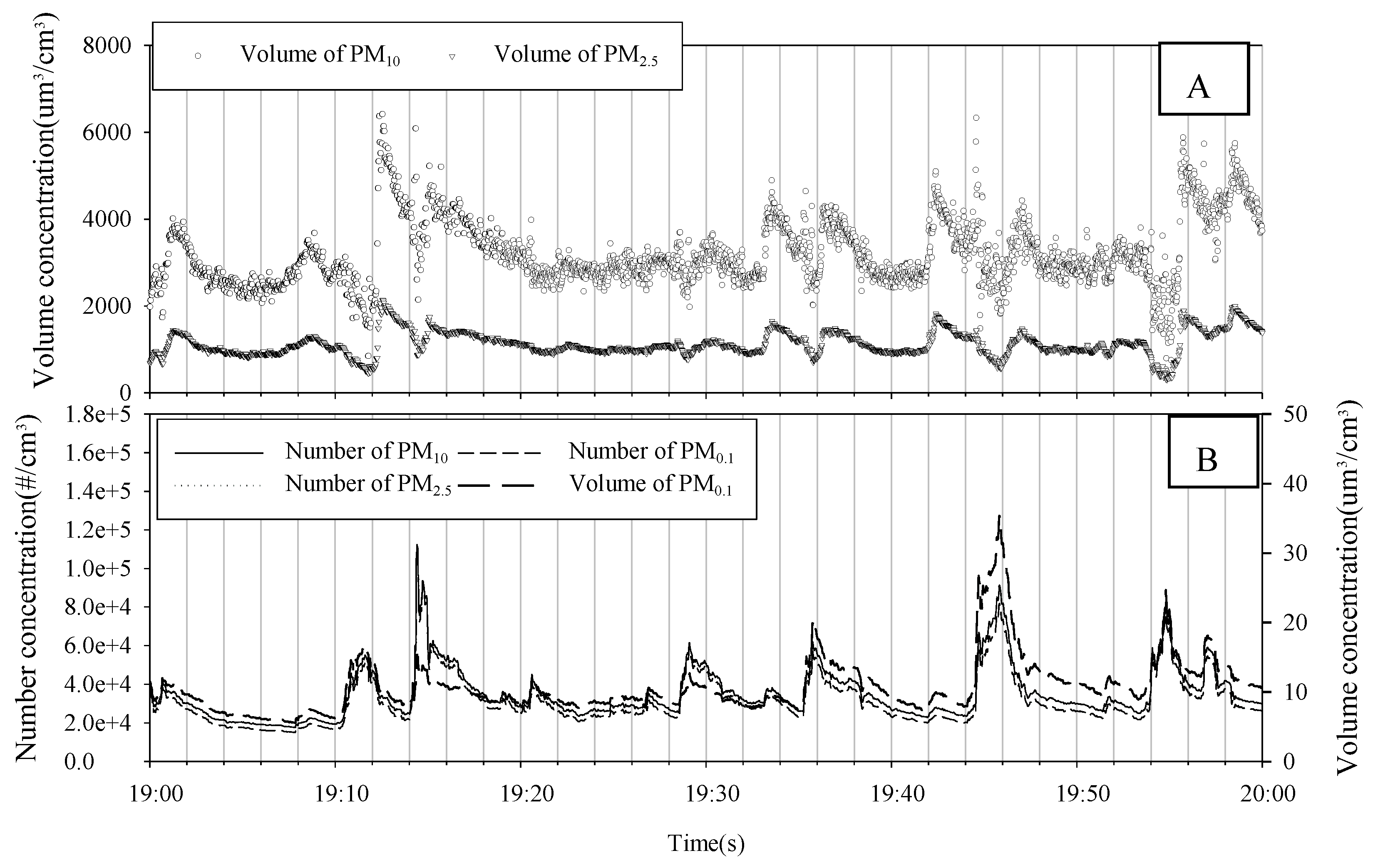
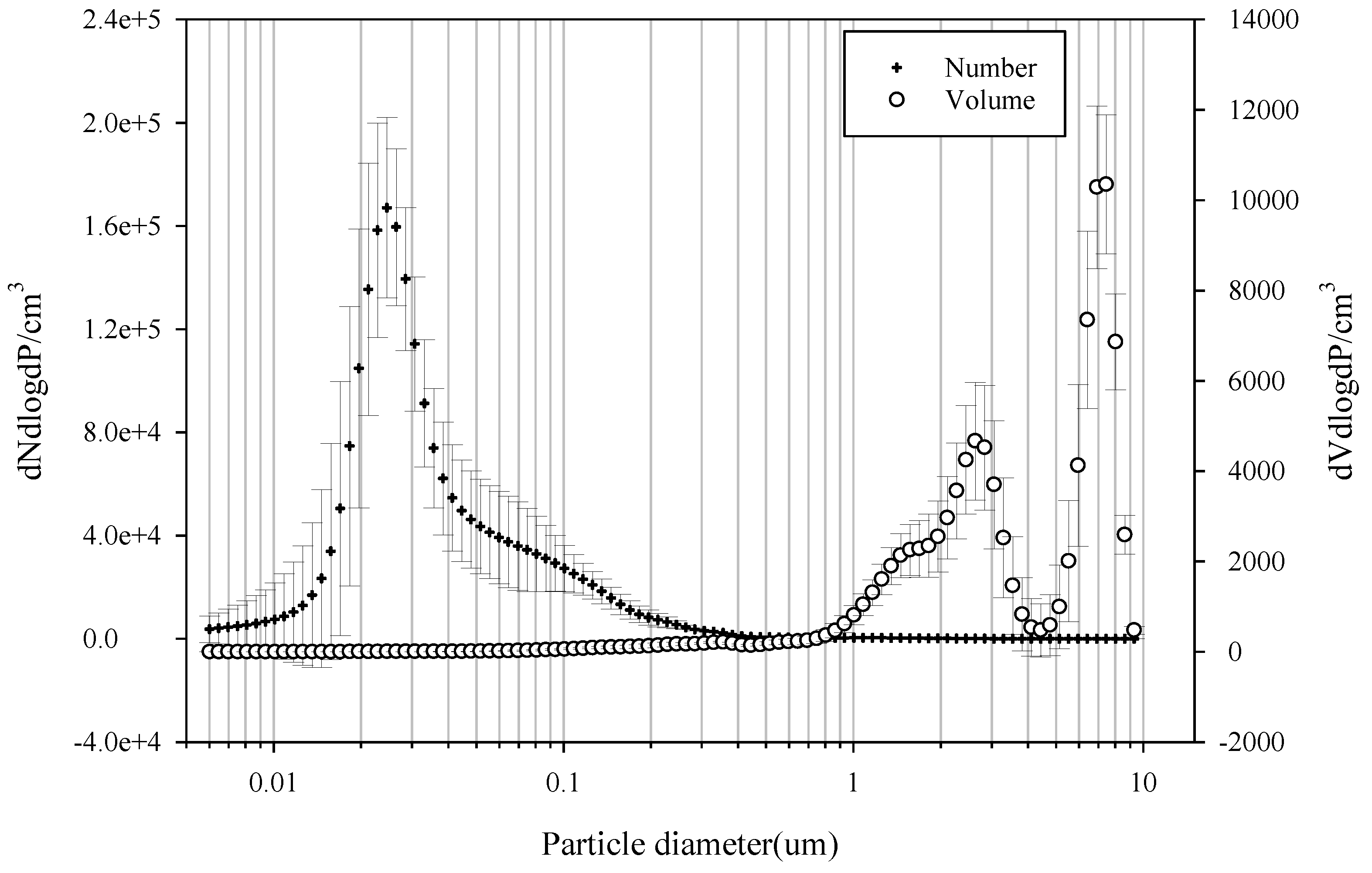
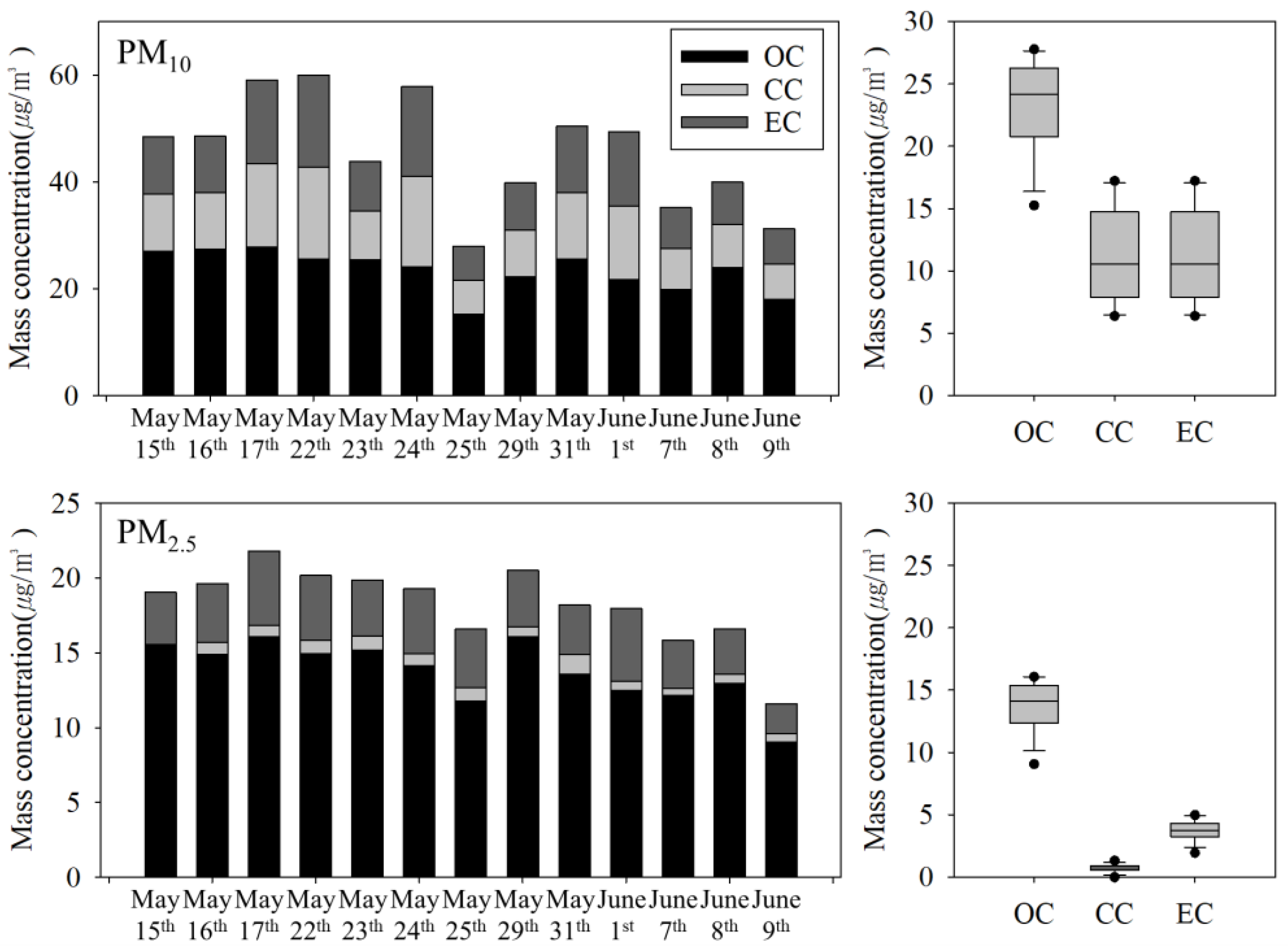
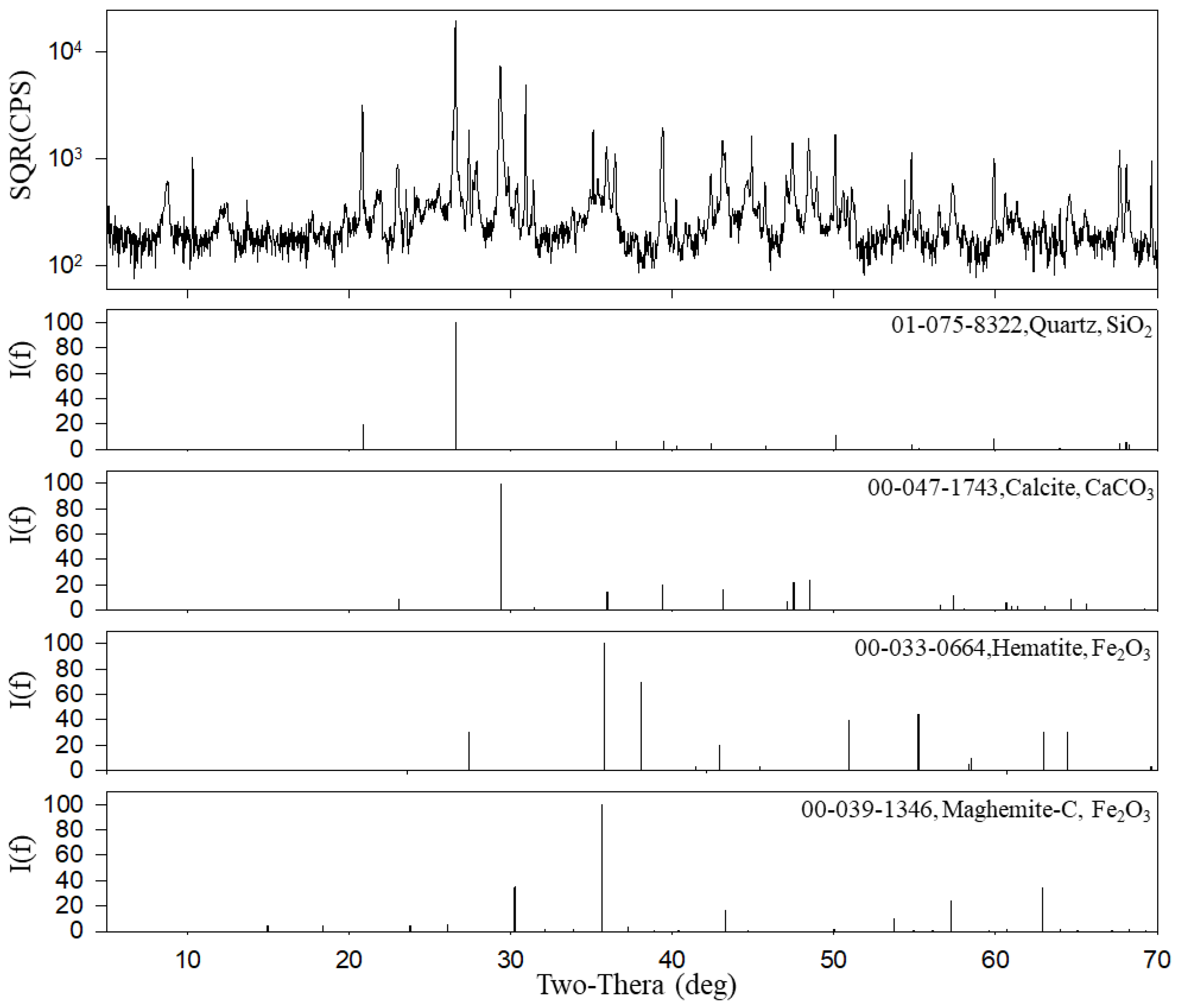



| Item | Filter | Particle Diameter | Flow Rate (L/min) | Analysis Instruments | Sampling Period and Site |
|---|---|---|---|---|---|
| Low-volume air sampler (PMS-104) | Zefluor | PM10 and PM2.5 | 16.7 | Ion compound (IC), Metal compound (ICP-AES) | 15 May to 9 June 2017 (n = 13) M station, Seoul |
| Mini-volume air sampler (MiniVol TAS) | Quartz | PM10 and PM2.5 | 5.0 | Organic and Elemental Carbon (Carbon analyzer) | |
| Electrical low-pressure impactor (ELPI) | Aluminum foil | 14 stages (D50 1: 10, 5.3, 3.6, 2.5, 1.6, 0.94, 0.60, 0.38, 0.25, 0.15, 0.094, 0.054, 0.030, and 0.016 µm) | 10.0 | Morphology and Chemical by size distribution (SEM and TEM/EDX) | 15 May to 17 May 2017 M station, Seoul |
| Low-volume air sampler PMS-104 | Zefluor | PM10 | 16.7 | 18 April 2018 S station, Seoul | |
| Sieve (mesh 18: Φ = 1.00 mm) | Zipper bag | Floor dust under 1 mm | - | Chemical form (XRD) | Three times within 15 May to 9 June 2017 S station, Seoul |
| Program Activity | Carbon | Carrier Gas | Ramp Time (s) | Program Temperature (°C) |
|---|---|---|---|---|
| Oven purge | - | He | 10 | Ambient |
| 1st ramp | OC1 | 60 | 315 | |
| 2nd ramp | OC2 | 60 | 475 | |
| 3rd ramp | OC3 | 60 | 615 | |
| 4th ramp | OC4 and CC | 90 | 870 | |
| Cooling for EC | CC | 30 | 0 | |
| Stabilize temp | PC and EC | He/O2 | 45 | 550 |
| He/O2 1st ramp | PC and EC | 45 | 625 | |
| 2nd ramp | EC | 45 | 700 | |
| 3rd ramp | 45 | 775 | ||
| 4th ramp | 45 | 850 | ||
| 5th ramp | 120 | 910 | ||
| External standard, calibration and cool down | - | Calibration gas and He/O2 | 120 | 0 |
| PM10 | PM2.5 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| This Study | In Seoul [16] | In Mexico City [43] | This Study | In Seoul [29] | In Barcelona [44] | In Mexico City [43] | |||
| Mass (g/m3) | Ratio (%) | Mass (g/m3) | Mass (g/m3) | Ratio (%) | Mass (g/m3) | ||||
| Total | 213.7 ± 50.4 | 100 | 200.75 | 89.55 | 78.4 ± 8.8 | 100 | 55.1 | 20.7–93.2 | 48.34 |
| TC * | 34.5 ± 6.8 | 16.1 | - | - | 17.5 ± 2.6 | 22.3 | - | 3.2–17.1 | - |
| Anion ** | 11.0 ± 5.1 | 5.2 | 18.16 + | - | 6.7 ± 3.4 | 8.6 | 6.4 ** | - | - |
| Cation | 4.0 ± 1.8 | 1.9 | 9.82 ++ | - | 2.5 ± 1.3 | 3.2 | 3 | - | - |
| Inorganic *** | 11.1 ± 1.5 | 5.2 | 8.68 +++ | - | 2.9 ± 0.6 | 3.7 | - | - | - |
| Fe | 86.1 ± 30.5 | 40.3 | 72.51 | 5.57 | 26.3 ± 6.5 | 33.6 | - | - | 3.1 |
| Fe2O3 **** | 123.1 ± 37.7 | 57.6 | - | - | 37.7 ± 9.3 | 48 | - | 6.9–52.4 | - |
| Unknown | 30.0 ± 22.1 | 14 | - | - | 11.1 ± 9.7 | 14.1 | - | - | - |
| Particle Size (µm) | C K | O K | S K | Fe K | Cu L | Ca K | Si K | Ba L | Br L | K K | Mo L | Mg K |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.016 | 91.58 | 7.21 | 1.21 | |||||||||
| 0.030 | 84.81 | 12.25 | 2.94 | |||||||||
| 0.054 | 95.65 | 4.35 | ||||||||||
| 0.094 | 80.95 | 11.49 | 3.39 | 1.73 | 1.89 | 0.56 | ||||||
| 0.150 | 83.17 | 8.62 | 1.48 | 1.45 | 1.87 | 3.42 | ||||||
| 0.250 | 70.67 | 8.26 | 8.86 | 12.21 | ||||||||
| 0.380 | 64.03 | 17.95 | 1.55 | 14.80 | 0.7 | 0.96 | ||||||
| 0.600 | 60.89 | 14.31 | 0.87 | 20.54 | 1.32 | 1.27 | 0.80 | |||||
| 0.940 | 40.37 | 18.65 | 0.78 | 34.19 | 2.06 | 1.83 | 2.11 | |||||
| 1.600 | 21.69 | 22.97 | 0.79 | 47.20 | 2.38 | 3.09 | 1.87 | |||||
| 2.500 | 28.07 | 20.43 | 0.60 | 44.14 | 1.95 | 2.73 | 1.64 | 0.44 | ||||
| 3.600 | 38.62 | 18.28 | 0.76 | 37.96 | 1.63 | 2.74 |
| Element | A | B | C |
|---|---|---|---|
| Iron (Fe) | 43.98 | 43.94 | 71.32 |
| Oxygen (O) | 40.40 | 40.58 | 16.33 |
| Silicon (Si) | 6.59 | 6.59 | 3.71 |
| Magnesium (Mg) | 0.34 | 0.34 | 1.42 |
| Aluminum (Al) | 1.18 | 1.17 | 1.76 |
| Calcium (Ca) | 1.77 | 1.76 | |
| Sodium (Na) | 2.80 | 2.79 | |
| Phosphorus (P) | 0.68 | 0.68 | |
| Sulfur (S) | 0.47 | 0.47 | |
| Chlorine (Cl) | 0.54 | 0.54 | |
| Potassium (K) | 1.14 | 1.14 | |
| Titanium (Ti) | 0.12 | ||
| Zirconium (Zr) | |||
| Sum | 100.00 | 100.00 | 100.00 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.; Lee, Y.-C.; Kim, T.; Choi, J.S.; Park, D. Sources and Characteristics of Particulate Matter in Subway Tunnels in Seoul, Korea. Int. J. Environ. Res. Public Health 2018, 15, 2534. https://doi.org/10.3390/ijerph15112534
Lee Y, Lee Y-C, Kim T, Choi JS, Park D. Sources and Characteristics of Particulate Matter in Subway Tunnels in Seoul, Korea. International Journal of Environmental Research and Public Health. 2018; 15(11):2534. https://doi.org/10.3390/ijerph15112534
Chicago/Turabian StyleLee, Yongil, Young-Chul Lee, Taesung Kim, Jin Seok Choi, and Duckshin Park. 2018. "Sources and Characteristics of Particulate Matter in Subway Tunnels in Seoul, Korea" International Journal of Environmental Research and Public Health 15, no. 11: 2534. https://doi.org/10.3390/ijerph15112534
APA StyleLee, Y., Lee, Y.-C., Kim, T., Choi, J. S., & Park, D. (2018). Sources and Characteristics of Particulate Matter in Subway Tunnels in Seoul, Korea. International Journal of Environmental Research and Public Health, 15(11), 2534. https://doi.org/10.3390/ijerph15112534








