Abstract
Background: Chest ultrasound (CUS) has been shown to be a sensitive and specific imaging modality for pneumothorax, pneumonia, and pleural effusions. However, the role of chest ultrasound in the diagnosis of thoracic tuberculosis (TB) is uncertain. We performed a systematic search in the medical literature to better define the potential role and value of chest ultrasound in diagnosing thoracic tuberculosis. Aim: To describe existing literature with regard to the diagnostic value of chest ultrasound in thoracic tuberculosis. Methods: MEDLINE, EMBASE, and Scopus databases were searched for relevant articles. We included studies that used chest ultrasound for the diagnosis or management of any form of thoracic tuberculosis, including pulmonary, pleural, mediastinal, and military forms. Results: We identified five main fields of chest ultrasound application: (1) Detection, characterization, and quantification of TB; (2) detection of residual pleural thickening after evacuation; (3) chest ultrasound-guided needle biopsy; (4) identification of pathologic mediastinal lymph nodes in children; and (5) identification of parenchymal ultrasound patterns. Effusion was also detected, in early stages, with signs of organization in 24–100% of patients. A low to moderate (10–23%), false negative rate was reported for chest ultrasound-guided needle biopsy. CUS was able to identify mediastinal lymph nodes in as many as 67% of patients with negative chest radiography. Conclusions: Very few studies with important methodological limitations analyze the role of chest ultrasound in the diagnosis of TB. The scarce available data suggests potential targets of future diagnostic or feasibility trials, such as the detection of tuberculosis–related pleural effusion, residual pleural thickening, lymphadenopathy, TB parenchymal patterns, or the use of CUS in biopsy guidance.
1. Introduction
In 2015, an estimated 10.4 million patients were newly diagnosed with tuberculosis (TB) worldwide, with 1.4 million cases resulting in death [1]. To achieve the End TB 2035 target [1] of a 90% reduction in the TB incidence rate by 2035, an acceleration of the present decline rate is urgently needed. Early and accurate diagnosis in high- and low-resource settings is the first pillar of the End TB strategy and is pivotal in enabling early treatment [2].
One of the main reasons for the under-detection of TB is the limited availability of diagnostic methods [3]. Moreover, when a diagnostic apparatus is available, many patients present with non-specific symptoms and negative laboratory exams. This delays diagnosis and aggravates prognosis.
The diagnosis of active TB has always used a composite approach, uniting radiology with three other technologies: Microscopy (sputum smears), culture-based methods, and molecular tests [4]. Unfortunately, facilities using these technologies are not always available in resource-limited settings, especially in peripheral health centers.
Point-of-care chest ultrasonography (CUS), intended for lung, pleural, and mediastinal ultrasonography, is becoming an attractive, non-invasive medical imaging modality in both affluent and resource-limited settings [5]. A recent consensus standardized terminology and indications, regarding critical care lung ultrasound [6]—a method which evolved as a highly sensitive and specific imaging tool for diagnosing chest conditions, such as pneumothorax, pneumonia, and pulmonary edema. Resource-limited settings are of special interest, as radiological equipment and expertise are scarce, or even absent, due to their high costs or poor maintenance. A focused assessment of extra-pulmonary TB has been also proposed [7]. However, the role of lung, and more broadly, chest ultrasound in defining TB has been scarcely investigated. It is currently unclear if CUS could play a clinically-relevant role in diagnosing or excluding active tubercular chest lesions.
The aim of this review is to describe existing studies that have used CUS for the diagnosis of thoracic TB, to ascertain whether it represents a useful tool for the diagnosis and follow up of TB.
2. Methods
This systematic review was conducted, following the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) [8] statement. The methodological quality of each study was critically evaluated, using the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS 2) [9].
2.1. Search Strategy
Two investigators (FG and LP), independently and unaware of the other investigator’s decisions, searched PubMed, EMBASE, and Scopus for studies assessing the role of lung ultrasound in thoracic TB. Searches were carried out without language restrictions and from the database’s inception until April 2017. Any inconsistencies that remained after discussion were resolved by consensus with a third author (NV), available for mediation. In PubMed, we used the following search strategy: (tuberculosis) AND (ultrasound OR ultrasonography) OR (miliary OR lung OR chest) AND/OR (recovery OR pattern OR diagnosis OR effusion). A similar search strategy was run in EMBASE and Scopus. Reference lists of included articles and those relevant to the topic were hand-searched for identification of additional, potentially-relevant articles.
2.2. Study Selection
We included all studies reporting patient data (including original studies, case-reports, and case series) using chest ultrasound for the diagnosis or management of any form of thoracic tuberculosis, namely its pulmonary, pleural, mediastinal, and miliary forms. Studies were excluded if they (1) used ultrasound for non-thoracic organs or (2) used endoscopic ultrasound. Studies were included, irrespective of the methodology employed. This was to ensure that a comprehensive understanding of the available evidence was achieved.
2.3. Data Extraction and Analysis
The two authors (FG, LP) independently extracted data, using a standardized spreadsheet. Any disagreement was resolved by consensus with a third author (DP). The following information was extracted: (i) Study population characteristics (i.e., author, year, number of participants, mean age, and percentage of females); (ii) study design and inclusion criteria; (iii) details of the ultrasound procedure; (iv) diagnostic criteria used for detecting TB and the final diagnosis made (pulmonary, pleural, or mediastinal TB); and (v) the anatomical site of investigation (pleural cavity, mediastinum, or lung).
The extracted information was organized in tables, describing each study’s characteristics, methods, and its main results. Patient characteristics—such as age, number of participants, and number of female cases—were summarized as the mean or proportions, where appropriate. Due to the different design of the studies included, the purely descriptive aim of 8 of the 12 studies, and the high heterogeneity of the outcomes reported, we decided not to meta-analyze the selected studies, instead reporting the main findings as descriptive results. Where some degree of diagnostic accuracy was reported, we calculated it as the percentage of patients with a positive ultrasound (US) examination, over the total of confirmed or suspected TB patients.
3. Results
The search identified 4442 non-duplicate records. After title and abstract screening, as well as a full text review, 12 studies were included, as shown in Figure 1 [10,11,12,13,14,15,16,17,18,19,20], with one study included from the reference screening of a previous article [21].
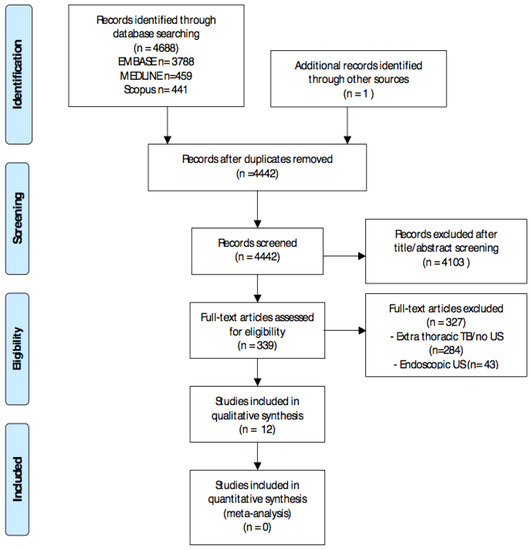
Figure 1.
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram. Abbreviations: Tuberculosis (TB) and ultrasound (US).
3.1. Study and Patient Characteristics
Study and patient characteristics are summarized in Table 1. The 12 analyzed studies included 300 participants, with a mean age of 32, 46% of which were females. We found that four studies were conducted in Asia, five in Europe, and three in Africa. Hemi-thoraxes (costophrenic recesses) were the most common site analyzed (n = 5), followed by the mediastinum (n = 4) and lung parenchyma (n = 3). Reference methods for the confirmation of tuberculosis strongly differed across studies.

Table 1.
Descriptive characteristics of the study participants, study design, and chest ultrasound (CUS) techniques used.
3.2. Main Findings
The main findings of the included studies are reported in Table 2. We found that five main fields of application of chest ultrasound in the field of thoracic TB emerged. The first concerned the diagnosis of pleural effusions, initially investigated in 1989, in 21 young subjects [10]. Effusions seemed to facilitate the determination of underlying lung lesions [12], while CUS could detect pleural effusions in early stages [16]. Secondly, CUS seemed to be accurate in detecting residual pleural thickening after evacuation [20].

Table 2.
Main findings of the studies included.
A third use of chest ultrasound focused on biopsies of TB lesions, as reported in three studies. In two studies, some degree of accuracy could be extrapolated, which resulted in moderate to high (76–90%) rates of success [13,21].
The fourth application aimed at identifying pathological mediastinal lymph nodes in children with suspected mediastinal disease, as reported by three studies, all in the pediatric setting [14,15,21] with variable diagnostic performance (40–100%). Only one study monitored the response after treatment, using the reduction of lymph node volume as an indicator for treatment success [14].
Finally, CUS was used to describe parenchymal patterns in one study, which only enrolled patients with miliary TB [18] and reported on only one case [19].
The qualitative assessment of the included studies showed a predominance of high or unclear risk of bias and concern over applicability, across all features analyzed, especially with regards patient selection and the reference standard used.
4. Discussion
The main finding of our systematic review is that the few available studies on chest ultrasound have focused on five fields of interest: detection of pleural effusion, assessment of residual pleural thickening, the helpfulness of trans-thoracic needle biopsy, assessment of mediastinal lymphadenopathies, and detection of pulmonary involvement in miliary TB. To our knowledge, this is the first review summarizing evidence for the use of chest ultrasound in the diagnosis of thoracic tuberculosis.
Surprisingly, apart from a single reported case, we found no study analyzing parenchymal ultrasound patterns, typical of pulmonary TB. Considering the pulmonary involvement of TB has been extensively studied by other radiological means [22], we expected to find evidence to include or exclude CUS as an alternative imaging technique in active TB. In the wider field of chest ultrasound, lung ultrasound (LUS) has gained acceptance for the diagnosis of consolidations and interstitial syndromes [23], hence its potential role in defining TB-related, pneumonic infiltrates. However, there appears to be scant available data at the moment, analyzing the use of LUS in the definition of TB parenchymal infiltrates.
The only study that attempted to report lung patterns describes the presence of B-lines and subpleural granularity, but only enrolled ten patients with miliary TB, which is distinct from pulmonary TB. Additionally, B-lines are, by definition, a highly non-specific sign of interstitial involvement, hence their role must be confirmed before LUS can be used to differentiate TB from other interstitial and alveolar lung diseases. Further, the presence, severity, and distribution of subpleural consolidation in patients with sole pulmonary involvement is still unknown.
There is an increasing interest in employing chest ultrasound in low- and middle-income countries [24]. Chest ultrasound has a relatively steep learning curve: It is ionization-free and is increasingly available [25] at reasonable costs. Moreover, it can be portable and operated with rechargeable batteries. Ultrasound gel, the only routine supply item needed, can easily be produced locally [26], thus making it an attractive option in resource-limited settings. On the other hand, inter-observer variability and diagnostic errors represent important pitfalls, and should be investigated specifically for TB. While the conditions for a wider implementation are favorable, none of the studies were performed in low-income countries. This observation, added to the paucity of available evidence, indicates that the use of CUS for the diagnosis of thoracic TB is still a clinical niche.
No conclusions can be drawn on diagnostic accuracy, as we did not find any clinical trial that compared CUS versus other imaging modalities. In the search for early diagnosis techniques, high negative predictive values become an important endpoint to target. If the benefits of point of care ultrasonography can be transposed to the diagnosis and management of TB—a disease that may yield highly unspecific signs [12]—it remains yet to be challenged by an adequately-powered, diagnostic trial.
The strength of our review stems from the strict search performed and pragmatic clinical questioning. Limitations of the review are the paucity of available data and extreme heterogeneity of the studies included, in terms of aim, methodology, and outcomes. The low or unclear quality of many of the included studies also greatly limits generalizations. Additionally, only five studies [12,13,14,15,21] compared the result of the CUS technique to a reference standard for the diagnosis of TB. Most importantly, no prospective studies comparing CUS to other imaging modalities in diagnosing TB were identified.
5. Conclusions
There is a striking scarcity of studies analyzing the use of chest ultrasound as an imaging technique to aid TB diagnosis. CUS is a promising imaging technique for the detection of TB-related effusion, residual pleural thickening, mediastinal lymphadenopathy, and trans-thoracic biopsy guidance. CUS may prove beneficial, especially in low-income countries, where incidence is high and radiological techniques are scarce. Further research is urgently needed to define pulmonary TB patterns, the feasibility of CUS, and diagnostic accuracy.
Author Contributions
All individuals listed as authors have contributed substantially to the design, performance, or reporting of the study. Each contribution is indicated as follows: F.D.G. and L.P. designed the study; F.D.G., L.P., N.V., and D.P. performed the systematic search; L.P., F.D.G., D.P., and N.V. drafted the manuscript; V.L., L.M., A.S., M.A.M.H., R.C., G.P., and M.J.S. critically reviewed the manuscript. All authors read and approved the final version of this manuscript.
Funding
This research received no external funding.
Acknowledgments
Thank you to Micheal Afewerki for the English review.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| CUS | chest ultrasound |
| TB | tuberculosis |
References
- World Health Organization. Global Tuberculosis Report 2016; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization. Global Strategy and Targets for Tuberculosis Prevention, Care and Control after 2015; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Sulis, G.; Centis, R.; Sotgiu, G.; D’Ambrosio, L.; Pontali, E.; Spanevello, A.; Matteelli, A.; Zumla, A.; Migliori, G.B. Recent developments in the diagnosis and management of tuberculosis. NPJ Prim. Care Respir. Med. 2016, 26, 16078. [Google Scholar] [CrossRef] [PubMed]
- Pai, M.; Behr, M.A.; Dowdy, D.; Dheda, K.; Divangahi, M.; Boehme, C.C.; Ginsberg, A.; Swaminathan, S.; Spigelman, M.; Getahun, H.; et al. Tuberculosis. Nat. Rev. Dis. Prim. 2016, 2, 16076. [Google Scholar] [CrossRef] [PubMed]
- Becker, D.M.; Tafoya, C.A.; Becker, S.L.; Kruger, G.H.; Tafoya, M.J.; Becker, T.K. The use of portable ultrasound devices in low- and middle-income countries: A systematic review of the literature. Trop. Med. Int. Health 2016, 21, 294–311. [Google Scholar] [CrossRef] [PubMed]
- Volpicelli, G.; Elbarbary, M.; Blaivas, M.; Lichtenstein, D.A.; Mathis, G.; Kirkpatrick, A.W.; Melniker, L.; Gargani, L.; Noble, V.E.; Via, G.; et al. International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med. 2012, 38, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Heller, T.; Wallrauch, C.; Goblirsch, S.; Brunetti, E. Focused assessment with sonography for HIV-associated tuberculosis (FASH): A short protocol and a pictorial review. Crit. Ultrasound J. 2012, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed]
- Whiting, P.F.; Weswood, M.E.; Rutjes, A.W.S.; Reitsma, J.B.; Bossuyt, P.N.; Kleijnen, J. Evaluation of QUADAS, a tool for the quality assessment of diagnostic accuracy studies. BMC Med. Res. Methodol. 2006, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Carazo Martínez, O.; Vargas Serrano, B.; Rodríguez Romero, R. Real-time ultrasound evaluation of tuberculous pleural effusions. J. Clin. Ultrasound 1989, 17, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Akhan, O.; Demirkazik, F.B.; Ozmen, M.N.; Balkanci, F.; Ozkara, S.; Cöplü, L.; Emri, A.; Besim, A. Tuberculous pleural effusions: Ultrasonic diagnosis. J. Clin. Ultrasound 1992, 20, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Yang, P.C.; Chang, D.B.; Yu, C.J.; Lee, L.N.; Wu, H.D.; Kuo, S.H.; Luh, K.T. Ultrasound guided aspiration biopsy for pulmonary tuberculosis with unusual radiographic appearances. Thorax 1993, 48, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Gulati, M.; Venkataramu, N.; Gupta, S.; Sood, B.P.; Sheena, D.M.; Gupta, S.K.; Suri, S. Ultrasound guided fine needle aspiration biopsy in mediastinal tuberculosis. Int. J. Tuberc. Lung Dis. 2000, 4, 1164–1168. [Google Scholar] [PubMed]
- Bosch-Marcet, J.; Serres-Créixams, X.; Borrás-Pérez, V.; Coll-Sibina, M.T.; Guitet-Juliá, M.; Coll-Rosell, E. Value of sonography for follow-up of mediastinal lymphadenopathy in children with tuberculosis. J. Clin. Ultrasound 2007, 35, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Moseme, T.; Andronikou, S. Through the eye of the suprasternal notch: Point-of-care sonography for tuberculous mediastinal lymphadenopathy in children. Pediatr. Radiol. 2014, 44, 681–684. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, V.; Gombar, S.; Kumar, N.; Goyal, N.; Gupta, K. Pregnancy with bilateral tubercular pleural effusion: Challenges. Trop. Doct. 2014, 44, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Bahr, H.M.; El-Shafey, M.; Hantera, M.S.; Abo-El magd, G.H.; El-Batsh, A.H. Ultrasound guided needle pleural biopsy in patients with undiagnosed pleural effusion. Egypt J. Chest Dis. Tuberc. 2014, 63, 113–118. [Google Scholar] [CrossRef]
- Hunter, L.; Bélard, S.; Janssen, S.; van Hoving, D.J.; Heller, T. Miliary tuberculosis: Sonographic pattern in chest ultrasound. Infection 2016, 44, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Heuvelings, C.C.; Bélard, S.; Janssen, S.; Wallrauch, C.; Grobusch, M.P.; Brunetti, E.; Giordani, M.T.; Heller, T. Chest ultrasonography in patients with HIV: A case series and review of the literature. Infection 2016, 44, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.F.; Su, M.C.; Weng, H.H.; Wu, J.T.; Chiu, C.T. Sonographic septation: A predictor of sequelae of tuberculous pleurisy after treatment. Thorax 2009, 64, 806–809. [Google Scholar] [CrossRef] [PubMed]
- Bosch-Marcet, J.; Serres-Créixams, X.; Zuasnabar-Cotro, A.; Codina-Puig, X.; Català-Puigbó, M.; Simon-Riazuelo, J.L. Comparison of ultrasound with plain radiography and CT for the detection of mediastinal lymphadenopathy in children with tuberculosis. Pediatr. Radiol. 2004, 34, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, L.; Parlatano, D.; Boccuzzi, F.; Onoscuri, M.; Volpicelli, G.; Veltri, A. The imaging spectrum of pulmonary tuberculosis. Acta Radiol. 2015, 56, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Reissig, A.; Copetti, R.; Mathis, G.; Mempel, C.; Schuler, A.; Zechner, P.; Aliberti, S.; Neumann, R.; Kroegel, C.; Hoyer, H. Lung ultrasound in the diagnosis and follow-up of community-acquired pneumonia: A prospective, multicenter, diagnostic accuracy study. Chest 2012, 142, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Berlet, T. Thoracic ultrasound for the diagnosis of pneumonia in adults: A meta-analysis. Respir. Res. 2015, 16, 89. [Google Scholar] [CrossRef] [PubMed]
- Sippel, S.; Muruganandan, K.; Levine, A.; Shah, S. Review article: Use of ultrasound in the developing world. Int J. Emerg. Med. 2011, 4, 72. [Google Scholar] [CrossRef] [PubMed]
- Binkowski, A.; Riguzzi, C.; Price, D.; Fahimi, J. Evaluation of a cornstarch-based ultrasound gel alternative for low-resource settings. J. Emerg. Med. 2014, 47, e5–e9. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).