Relationship between Telomere Length, Genetic Traits and Environmental/Occupational Exposures in Bladder Cancer Risk by Structural Equation Modelling
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.1.1. Data Collection
2.1.2. Analysis on DNA from Peripheral Blood Leucocytes
2.1.3. 32P-Post-Labeling Analysis of DNA Adducts
2.1.4. Genotyping
2.1.5. Leucocytes Telomere Length Analysis
2.2. Statistical Analysis
3. Results
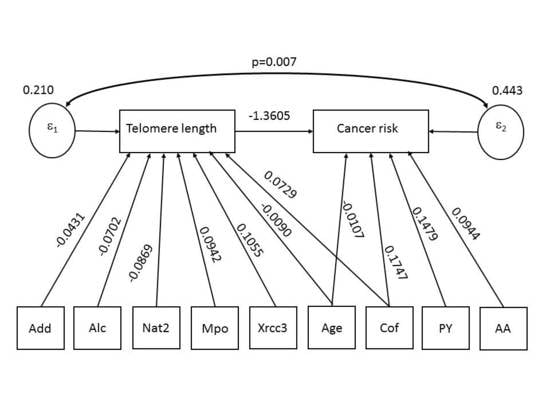
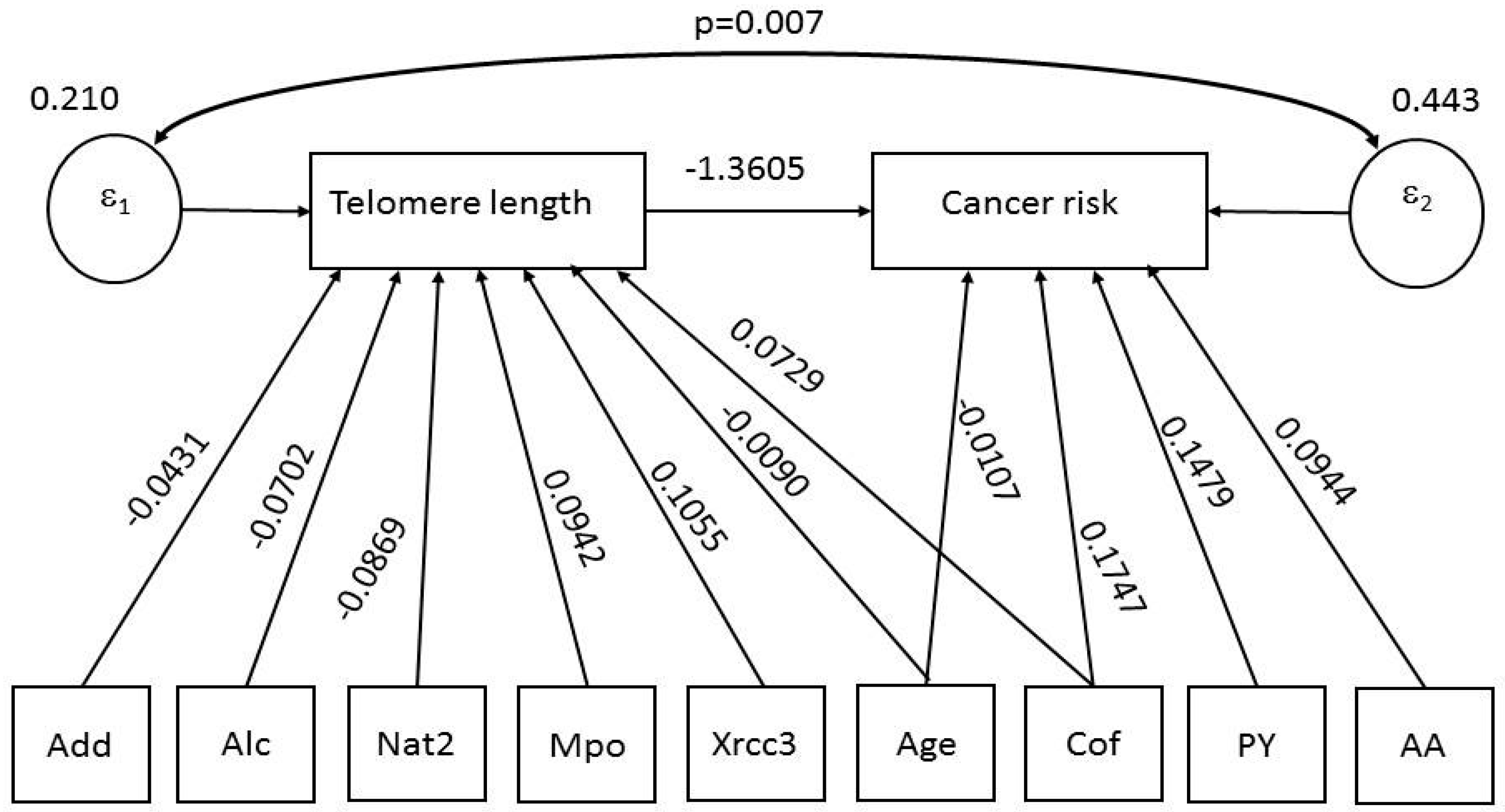
- Direct effects. The first model shows that LTL is negatively associated with age (p = 0.000), DNA adducts (p = 0.017), alcohol intake (p = 0.017) and NAT2 (p = 0.018), and positively association with coffee (p = 0.016), MPO (p = 0.009) and XRCC3 (p = 0.004). The second model shows that BC risk significantly increased with consumption of cigarettes (p = 0.000), cumulative exposure to AAs (p = 0.003) and coffee (p = 0.006), while it decreased with LTL (p = 001) and age (p = 0.019).
- Indirect effects. The first model shows no indirect effects. The second model shows that, via LTL reduction, BC risk increased with age (p = 0.007) and NAT2 (p = 0.011), while it decreased with MPO (p = 0.029) and XRCC3 (p = 0.003).
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Blackburn, E.H.; Greider, C.W.; Szostak, J.W. Telomeres and telomerase: The path from maize, Tetrahymena and yeast to human cancer and aging. Nat. Med. 2006, 12, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Calado, R.T.; Young, N.S. Telomere diseases. N. Engl. J. Med. 2009, 361, 2353–2365. [Google Scholar] [CrossRef] [PubMed]
- Shay, J.W.; Wright, W.E. Telomerase activity in human cancer. Curr. Opin. Oncol. 1996, 8, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Frenck, R.W., Jr.; Blackburn, E.H.; Shannon, K.M. The rate of telomere sequence loss in human leukocytes varies with age. Proc. Natl. Acad. Sci. USA 1998, 95, 5607–5610. [Google Scholar] [CrossRef] [PubMed]
- Von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem. Sci. 2002, 27, 339–344. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, S.; Funk, W.E.; Hou, L. Environmental and occupational exposure to chemicals and telomere length in human studies. Occup. Environ. Med. 2013, 70, 743–749. [Google Scholar] [CrossRef] [PubMed]
- McGrath, M.; Wong, J.Y.; Michaud, D.; Hunter, D.J.; De Vivo, I. Telomere length, cigarette smoking, and bladder cancer risk in men and women. Cancer Epidemiol. Biomark. Prev. 2007, 16, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Jönsson, B.A.G.; Lindh, C.H.; Albin, M.; Broberg, K. N-nitrosamines are associated with shorter telomere length. Scand. J. Work Environ. Health 2011, 37, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Pavanello, S.; Pesatori, A.C.; Dioni, L.; Hoxha, M.; Bollati, V.; Siwinska, E.; Mielzyńska, D.; Bolognesi, C.; Bertazzi, P.A.; Baccarelli, A. Shorter telomere length in peripheral blood lymphocytes of workers exposed to polycyclic aromatic hydrocarbons. Carcinogenesis 2010, 31, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Broberg, K.; Björk, J.; Paulsson, K.; Höglund, M.; Albin, M. Constitutional short telomeres are strong genetic susceptibility markers for bladder cancer. Carcinogenesis 2005, 26, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Modica, F.; Guarrera, S.; Fiorito, G.; Pardini, B.; Viberti, C.; Allione, A.; Critelli, R.; Bosio, A.; Casetta, G.; et al. Shorter leukocyte telomere length is independently associated with poor survival in patients with bladder cancer. Cancer Epidemiol. Biomark. Prev. 2014, 23, 2439–2446. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, Y.; Kota, K.K.; Kallakury, B.; Mikhail, N.N.; Sayed, D.; Mokhtar, A.; Maximous, D.; Yassin, E.H.; Gouda, I.; et al. Strong association between long and heterogeneous telomere length in blood lymphocytes and bladder cancer risk in Egyptian. Carcinogenesis 2015, 36, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Hung, R.J.; Boffetta, P.; Brennan, P.; Malaveille, C.; Gelatti, U.; Placidi, D.; Carta, A.; Hautefeuille, A.; Porru, S. Genetic polymorphisms of MPO, COMT, MnSOD, NQO1, interactions with environmental exposures and bladder cancer risk. Carcinogenesis 2004, 25, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Hung, R.J.; Boffetta, P.; Brennan, P.; Malaveille, C.; Hautefeuille, A.; Donato, F.; Gelatti, U.; Spaliviero, M.; Placidi, D.; Carta, A.; et al. GST, NAT, SULT1A1, CYP1B1 genetic polymorphisms, interactions with environmental exposures and bladder cancer risk in a high-risk population. Int. J. Cancer 2004, 110, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Hung, R.J.; Brennan, P.; Malaveille, C.; Donato, F.; Placidi, D.; Carta, A.; Hautefeuille, A.; Boffetta, P.; Porru, S. Polymorphisms of the DNA repair genes XRCC1, XRCC3, XPD, interaction with environmental exposures, and bladder cancer risk in a case-control study in northern Italy. Cancer Epidemiol. Biomark. Prev. 2003, 12, 1234–1240. [Google Scholar]
- Pavanello, S.; Mastrangelo, G.; Placidi, D.; Campagna, M.; Pulliero, A.; Carta, A.; Arici, C.; Porru, S. CYP1A2 polymorphisms, occupational and environmental exposures and risk of bladder cancer. Eur. J. Epidemiol. 2010, 25, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Burger, M.; Catto, J.W.; Dalbagni, G.; Grossman, H.B.; Herr, H.; Karakiewicz, P.; Kassouf, W.; Kiemeney, L.A.; La Vecchia, C.; Shariat, S.; et al. Epidemiology and risk factors of urothelial bladder cancer. Eur. Urol. 2013, 63, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Grotenhuis, A.J.; Dudek, A.M.; Verhaegh, G.W.; Witjes, J.A.; Aben, K.K.; van der Marel, S.L.; Vermeulen, S.H.; Kiemeney, L.A. Prognostic relevance of urinary bladder cancer susceptibility loci. PLoS ONE 2014, 9, e89164. [Google Scholar] [CrossRef] [PubMed]
- Cumberbatch, M.G.; Cox, A.; Teare, D.; Catto, J.W. Contemporary occupational carcinogen exposure and bladder cancer: A systematic review and meta-analysis. JAMA Oncol. 2015, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Schwender, H.; Selinski, S.; Blaszkewicz, M.; Marchan, R.; Ickstadt, K.; Golka, K.; Hengstler, J.G. Distinct SNP combinations confer susceptibility to urinary bladder cancer in smokers and non-smokers. PLoS ONE 2012, 7, e51880. [Google Scholar] [CrossRef] [PubMed]
- Porru, S.; Pavanello, S.; Carta, A.; Arici, C.; Simeone, C.; Izzotti, A.; Mastrangelo, G. Complex relationships between occupation, environment, DNA adducts, genetic polymorphisms and bladder cancer in a case-control study using a structural equation modeling. PLoS ONE 2014, 9, e94566. [Google Scholar] [CrossRef] [PubMed]
- Cawthon, R.M. Telomere measurement by quantitative PCR. Nucleic Acids Res. 2002, 30, e47. [Google Scholar] [CrossRef] [PubMed]
- Kline, R. Principles and Practice of Structural Equation Modeling, 2nd ed.; The Guilford Press: New York, NY, USA, 2005. [Google Scholar]
- Bentler, P.; Chou, C. Practical issues in structural modeling. Sociol. Methods Res. 1987, 16, 78–117. [Google Scholar] [CrossRef]
- Worthington, R.; Whittaker, T. Scale Development Research. A Content Analysis and Recommendations for Best Practices. Couns. Psychol. 2006, 34, 806–838. [Google Scholar] [CrossRef]
- Hair, J.; Black, W.; Babin, B.; Anderson, R.; Tatham, R. Multivariate Data Analysis, 6th ed.; Pearson Educational, Inc.: Upper Saddle River, NJ, USA, 2006. [Google Scholar]
- Talaska, G.; al-Juburi, A.Z.; Kadlubar, F.F. Smoking related carcinogen-DNA adducts in biopsy samples of human urinary bladder: Identification of N-(deoxyguanosin-8-yl)-4-aminobiphenyl as a major adduct. Proc. Natl. Acad. Sci. USA 1991, 88, 5350–5354. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, S.; Tada-Oikawa, S.; Kawanishi, S. Site-specific DNA damage at the GGG sequence by UVA involves acceleration of telomere shortening. Biochemistry 2001, 40, 4763–4768. [Google Scholar] [CrossRef] [PubMed]
- Matullo, G.; Palli, D.; Peluso, M.; Guarrera, S.; Carturan, S.; Celentano, E.; Krogh, V.; Munnia, A.; Tumino, R.; Polidoro, S.; et al. XRCC1, XRCC3, XPD gene polymorphisms, smoking and 32P-DNA adducts in a sample of healthy subjects. Carcinogenesis 2001, 22, 1437–1445. [Google Scholar] [CrossRef] [PubMed]
- Piedrafita, F.J.; Molander, R.B.; Vansant, G.; Orlova, E.A.; Pfahl, M.; Reynolds, W.F. An Alu element in the myeloperoxidase promoter contains a composite SP1-thyroid hormone-retinoic acid response element. J. Biol. Chem. 1996, 271, 14412–14420. [Google Scholar] [CrossRef] [PubMed]
- Ji, G.; Gu, A.; Zhou, Y.; Shi, X.; Xia, Y.; Long, Y.; Song, L.; Wang, S.; Wang, X. Interactions between exposure to environmental polycyclic aromatic hydrocarbons and DNA repair gene polymorphisms on bulky DNA adducts in human sperm. PLoS ONE 2010, 5, e13145. [Google Scholar] [CrossRef] [PubMed]
- Lunn, R.M.; Langlois, R.G.; Hsieh, L.L.; Thompson, C.L.; Bell, D.A. XRCC1 polymorphisms: Effects on aflatoxin B1-DNA adducts and glycophorin a variant frequency. Cancer Res. 1999, 59, 2557–2561. [Google Scholar] [PubMed]
- Pavanello, S.; Hoxha, M.; Dioni, L.; Bertazzi, P.A.; Snenghi, R.; Nalesso, A.; Ferrara, S.D.; Montisci, M.; Baccarelli, A. Shortened telomeres in individuals with abuse in alcohol consumption. Int. J. Cancer 2011, 129, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Crous-Bou, M.; Giovannucci, E.; De Vivo, I. Coffee Consumption Is Positively Associated with Longer Leukocyte Telomere Length in the Nurses’ Health Study. J. Nutr. 2016, 146, 1373–1378. [Google Scholar] [CrossRef] [PubMed]
- Cardin, R.; Piciocchi, M.; Martines, D.; Scribano, L.; Petracco, M.; Farinati, F. Effects of coffee consumption in chronic hepatitis C: A randomized controlled trial. Dig. Liver Dis. 2013, 45, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Nettleton, J.A.; Diez-Roux, A.; Jenny, N.S.; Fitzpatrick, A.L.; Jacobs, D.R., Jr. Dietary patterns, food groups, and telomere length in the Multi-Ethnic Study of Atherosclerosis (MESA). Am. J. Clin. Nutr. 2008, 88, 1405–1412. [Google Scholar] [PubMed]
- Bakuradze, T.; Lang, R.; Hofmann, T.; Eisenbrand, G.; Schipp, D.; Galan, J.; Richling, E. Consumption of a dark roast coffee decreases the level of spontaneous DNA strand breaks: A randomized controlled trial. Eur. J. Nutr. 2015, 54, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Bichler, J.; Cavin, C.; Simic, T.; Chakraborty, A.; Ferk, F.; Hoelzl, C.; Schulte-Hermann, R.; Kundi, M.; Haidinger, G.; Angelis, K.; et al. Coffee consumption protects human lymphocytes against oxidative and 3-amino-1-methyl-5H-pyrido[4,3-b]indole acetate (Trp-P-2) induced DNA-damage: Results of an experimental study with human volunteers. Food Chem. Toxicol. 2007, 45, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Steinkellner, H.; Hoelzl, C.; UhI, M.; Cavin, C.; Haidinger, G.; Gsur, A.; Schmid, R.; Kundi, M.; Bichler, J.; Knausmüller, S. Coffee consumption induces GSTP in plasma and protects lymphocytes against (+/2)-anti-benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide induced DNA-damage: Results of controlled human intervention trials. Mutat. Res. 2005, 591, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Hoelzl, C.; Knasmüller, S.; Wagner, K.H.; Elbling, L.; Huber, W.; Kager, N.; Ferk, F.; Ehrlich, V.; Nersesyan, A.; Neubauer, O.; et al. Instant coffee with high chlorogenic acid levels protects humans against oxidative damage of macromolecules. Mol. Nutr. Food Res. 2010, 54, 1722–1733. [Google Scholar] [CrossRef] [PubMed]
- Rathod, M.A.; Patel, D.; Das, A.; Tipparaju, S.R.; Shinde, S.S.; Anderson, R.F. Inhibition of radical-induced DNA strand breaks by water-soluble constituents of coffee: Phenolics and caffeine metabolites. Free Radic. Res. 2013, 47, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; Jeong, H.G. Protective effects of kahweol and cafestol against hydrogen peroxide-induced oxidative stress and DNA damage. Toxicol. Lett. 2007, 173, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Cavin, C.; Bezencon, C.; Guignard, G.; Schilter, B. Coffee diterpenes prevent benzo[a]pyrene genotoxicity in rat and human culture systems. Biochem. Biophys. Res. Commun. 2003, 306, 488–495. [Google Scholar] [CrossRef]
- Peluso, M.; Airoldi, L.; Armelle, M.; Martone, T.; Coda, R.; Malaveille, C.; Giacomelli, G.; Terrone, C.; Casetta, G.; Vineis, P. White blood cell DNA adducts, smoking, and NAT2 and GSTM1 genotypes in bladder cancer: A case-control study. Cancer Epidemiol. Biomark. Prev. 1998, 7, 341–346. [Google Scholar]
- Kadlubar, F.F. Carcinogenic aromatic amine metabolism and DNA adduct detection in human. In Xenobiotics and Cancer; Emster, L., Ed.; Taylor & Francis, Ltd.: London, UK, 1991; pp. 329–338. [Google Scholar]
- Frederickson, S.M.; Messing, E.M.; Renikoff, C.A.; Swaminathan, S. Relationship between in vivo acetylator phenotypes and cytosolic N-acetyltransferase and O-acetyltransferase activities in human uroepithelial cells. Cancer Epidemiol. Biomark. Prev. 1994, 3, 25–32. [Google Scholar]
- Jongeneelen, F.J. Biological exposure limit for occupational exposure to coal tar pitch volatiles at cokeovens. Int. Arch. Occup. Environ. Health 1992, 63, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Kao, J.; Patterson, F.K.; Hall, J. Skin penetration and metabolism of topically applied chemicals in six mammalian species, including man: An in vitro study with benzo[a]pyrene and testosterone. Toxicol. Appl. Pharmacol. 1985, 81, 502–516. [Google Scholar] [CrossRef]
- Ng, K.M.; Chu, I.; Bronaugh, R.L.; Franklin, C.A.; Somers, D.A. Percutaneous absorption and metabolism of pyrene, benzo[a]pyrene, and di(2-ethylhexyl) phthalate: Comparison of in vitro and in vivo results in the hairless guinea pig. Toxicol. Appl. Pharmacol. 1992, 115, 216–223. [Google Scholar] [CrossRef]
- VanRooij, J.G.; De Roos, J.H.; Bodelier-Bade, M.M.; Jongeneelen, F.J. Absorption of polycyclic aromatic hydrocarbons through human skin: Differences between anatomical sites and individuals. J. Toxicol. Environ. Health 1993, 38, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Albert, R.E.; Miller, M.L.; Cody, T.; Andringa, A.; Shukla, R.; Shukla, R.; Baxter, C.S. Benzo[a]pyrene-induced skin damage and tumor promotion in the mouse. Carcinogenesis 1991, 12, 1273–1280. [Google Scholar] [CrossRef] [PubMed]
- Wolterbeek, A.P.; Schoevers, E.J.; Rutten, A.A.; Feron, V.J. A critical appraisal of intratracheal instillation of benzo[a]pyrene to Syrian golden hamsters as a model in respiratory tract carcinogenesis. Cancer Lett. 1995, 89, 107–116. [Google Scholar] [CrossRef]
- IARC. Monographs on the evaluation of carcinogenic risks to humans. In Some Non-Heterocyclic Polycyclic Aromatic Hydrocarbons and Some Related Exposures; International Agency for Research on Cancer: Lyon, France, 2010; Volume 92, pp. 765–771. [Google Scholar]
- Cawthon, R.M.; Smith, K.R.; O’Brien, E.; Sivatchenko, A.; Kerber, R.A. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet 2003, 361, 393–395. [Google Scholar] [CrossRef]
- Weischer, M.; Nordestgaard, B.G.; Cawthon, R.M.; Freiberg, J.J.; Tybjaerg-Hansen, A.; Bojesen, S.E. Short telomere length, cancer survival, and cancer risk in 47,102 individuals. J. Natl. Cancer Inst. 2013, 105, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Willeit, P.; Willeit, J.; Mayr, A.; Weger, S.; Oberhollenzer, F.; Brandstätter, A.; Kronenberg, F.; Kiechl, S. Telomere length and risk of incident cancer and cancer mortality. JAMA 2010, 304, 69–75. [Google Scholar] [CrossRef] [PubMed]
| Variables | Cases (N = 96) | Controls (N = 94) | Mann-Withney | ||
|---|---|---|---|---|---|
| Average ± SD | N (%) | Average ± SD | N (%) | p | |
| Age (years) | 61.5 ± 10.9 | 96 (100) | 60.3 ± 11.7 | 94 (100) | 0.6224 |
| Years of school | 7.41 ± 3.35 | 95 (99) | 8.38 ± 3.67 | 93 (99) | 0.0530 |
| Tobacco smoke (pack-years) | 32.8 ± 20.6 | 96 (100) | 23.2 ± 20.6 | 94 (100) | 0.0008 |
| coffee consumption (coffee-years) | 2.76 ± 2.67 | 96 (100) | 2.05 ± 1.49 | 94 (100) | 0.1158 |
| Alcohol consumption (alcohol-years) | 93.4 ± 104 | 96 (100) | 107 ± 117 | 94 (100) | 0.4154 |
| BMI (Kg/m2) | 26.0 ± 3.61 | 85 (86) | 25.9 ± 3.20 | 88 (94) | 0.8329 |
| Vegetables consumption (vegetables/week) | 2.43 ± 0.80 | 96 (100) | 2.52 ± 0.60 | 94 (100) | 0.8142 |
| Cumulative exposure to AA | 79.7 ± 78.0 | 11 (11) | 18.6 ± 17.5 | 6 (6) | 0.1880 |
| Cumulative exposure to PAHs | 46.5 ± 39.1 | 37 (38) | 31.5 ± 31.6 | 35 (37) | 0.4922 |
| DNA adducts (ln) | 1.11 ± 1.31 | 96 (100) | 0.84 ± 1.11 | 94 (100) | 0.1831 |
| LTL (T/S) | 1.55 ± 1.14 | 96 (100) | 2.03 ± 1.42 | 94 (100) | 0.0123 |
| Endogenous Variables | Exogenous Variables | Direct Effects | Indirect Effects | ||||
|---|---|---|---|---|---|---|---|
| Beta Coefficient | 95% Confidence Interval | p-Value | Beta Coefficient | 95% Confidence Interval | p-Value | ||
| LTL (RQ) | Age | −0.0090 | −0.0132; −0.0049 | 0.000 | 0 | ||
| DNA Adducts (ln) | −0.0431 | −0.0787; −0.0075 | 0.017 | 0 | |||
| Alc | −0.0702 | −0.1278; −0.0127 | 0.017 | 0 | |||
| PY | 0.0385 | −0.0107; 0.0879 | 0.125 | 0 | |||
| Cof | 0.0729 | 0.0138; 0.1320 | 0.016 | 0 | |||
| AA | −0.0240 | −0.0865; 0.0383 | 0.450 | 0 | |||
| Nat2 | −0.0869 | −0.1591; −0.0147 | 0.018 | 0 | |||
| MPO | 0.0942 | 0.0238; 0.1647 | 0.009 | 0 | |||
| XRCC3 | 0.1055 | 0.0333; 0.1777 | 0.004 | 0 | |||
| XRCC1 | 0.0335 | −0.0360; 0.1031 | 0.345 | 0 | |||
| MnSOD | −0.0014 | −0.1202; 0.1174 | 0.981 | 0 | |||
| CYP1B1 | 0.0098 | −0.0673; 0.0870 | 0.803 | 0 | |||
| BC risk | LTL (RQ) | −1.3605 | −2.1939; −0.5271 | 0.001 | 0 | ||
| Age | −0.0107 | −0.0198; −0.0017 | 0.019 | 0.0123 | 0.0034; 0.0213 | 0.007 | |
| DNA Adducts (ln) | −0.0439 | −0.1127; 0.0249 | 0.212 | 0.0587 | −0.0033; 0.1207 | 0.064 | |
| Alc | −0.0663 | −0.1761; 0.0434 | 0.236 | 0.0956 | −0.0081; 0.1992 | 0.071 | |
| PY | 0.1478 | 0.0726; 0.2231 | 0.000 | −0.0525 | −0.1283; 0.0233 | 0.174 | |
| Cof | 0.1747 | 0.0505; 0.2989 | 0.006 | −0.0992 | −0.2058; 0.0073 | 0.068 | |
| AA | 0.0944 | 0.0314; 0.1575 | 0.003 | 0.0328 | −0.0517; 0.1172 | 0.447 | |
| Nat2 | 0 | 0.1183 | 0.0275; 0.2090 | 0.011 | |||
| MPO | 0 | −0.1283 | −0.2438; −0.0128 | 0.029 | |||
| XRCC3 | 0 | −0.1436 | −0.2385; −0.0487 | 0.003 | |||
| XRCC1 | 0 | −0.0456 | −0.1379; 0.0466 | 0.332 | |||
| MnSOD | 0.0904 | −0.0757; 0.2566 | 0.286 | 0.0019 | −0.1600; 0.1638 | 0.982 | |
| CYP1B1 | 0 | −0.0134 | −0.1186; 0.0918 | 0.803 | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavanello, S.; Carta, A.; Mastrangelo, G.; Campisi, M.; Arici, C.; Porru, S. Relationship between Telomere Length, Genetic Traits and Environmental/Occupational Exposures in Bladder Cancer Risk by Structural Equation Modelling. Int. J. Environ. Res. Public Health 2018, 15, 5. https://doi.org/10.3390/ijerph15010005
Pavanello S, Carta A, Mastrangelo G, Campisi M, Arici C, Porru S. Relationship between Telomere Length, Genetic Traits and Environmental/Occupational Exposures in Bladder Cancer Risk by Structural Equation Modelling. International Journal of Environmental Research and Public Health. 2018; 15(1):5. https://doi.org/10.3390/ijerph15010005
Chicago/Turabian StylePavanello, Sofia, Angela Carta, Giuseppe Mastrangelo, Manuela Campisi, Cecilia Arici, and Stefano Porru. 2018. "Relationship between Telomere Length, Genetic Traits and Environmental/Occupational Exposures in Bladder Cancer Risk by Structural Equation Modelling" International Journal of Environmental Research and Public Health 15, no. 1: 5. https://doi.org/10.3390/ijerph15010005
APA StylePavanello, S., Carta, A., Mastrangelo, G., Campisi, M., Arici, C., & Porru, S. (2018). Relationship between Telomere Length, Genetic Traits and Environmental/Occupational Exposures in Bladder Cancer Risk by Structural Equation Modelling. International Journal of Environmental Research and Public Health, 15(1), 5. https://doi.org/10.3390/ijerph15010005