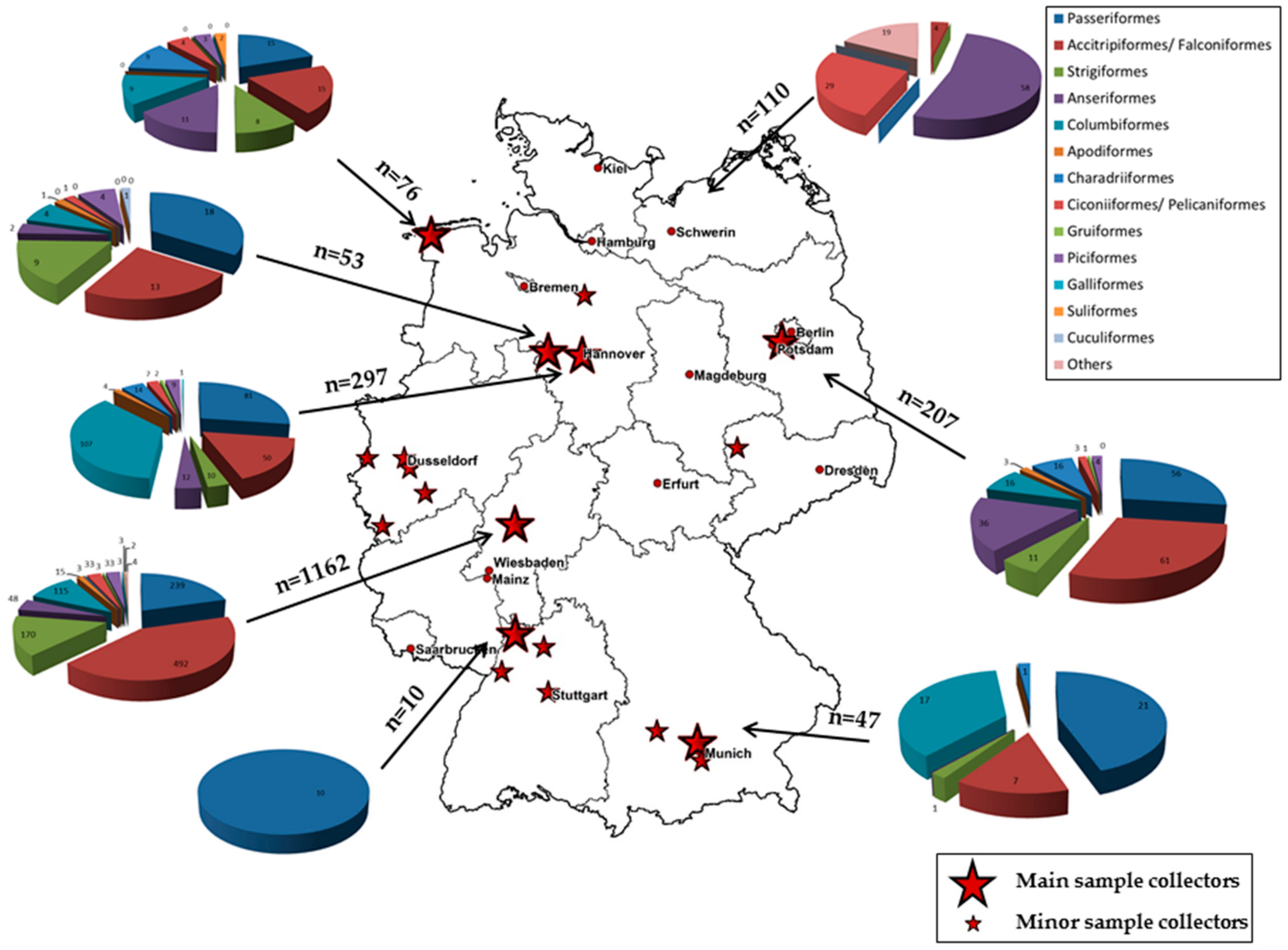
1. Introduction
Wild birds play an important role as reservoir hosts and as a transport shuttle for zoonotic arboviruses and their arthropod hosts to Central Europe. In particular, migratory birds play an important role in the spread of novel viruses to new areas along the major wild bird flyways across Asia, Africa, and Europe. Therefore, monitoring studies in wild birds can be used as an early warning system for the incursion of a number of zoonotic pathogens. We have monitored the infection and seroprevalence status of migratory and resident birds for many years, and have set up a German nationwide wild bird surveillance network for zoonotic arthropod-borne virus infections, with special emphasis on zoonotic flaviviruses, such as West Nile virus (WNV) and Usutu virus (USUV).
WNV is an arthropod-borne, single-stranded RNA virus, belonging to the family
Flaviviridae [
1], and is considered to be the most widespread flavivirus in the world [
2,
3]. The virus circulates between mosquitoes, which act as vectors, and wild birds, which act as reservoir hosts [
4]. The composition of bird and mosquito species differs between the geographical regions with WNV circulation [
5]. Wild birds serve as amplifying hosts; they develop a strong and long-term viremia, and are capable of infecting bird-biting mosquitoes [
6]. As a rule, most birds undergo subclinical infection and do not develop clinical symptoms [
7]. However, there are some highly-susceptible bird species, such as birds of prey, jays, and crows, which have been shown to develop severe and usually fatal encephalitis [
6,
7,
8]. As shown by Nehmeth et al. [
9], persistently WNV-infected birds can act as carriers between WNV endemic areas, and distribute WNV to new, previously disease-free areas. As every year millions of wild birds migrate between Europe and Africa, they can act as an entry portal, by overwintering in, or passing through, WNV-endemic areas [
10,
11].
Infection of susceptible non-avian vertebrates is usually asymptomatic, but, humans and horses in particular, can develop disease as a consequence of WNV infections, which may range from mild febrile illness (West Nile fever) to encephalitis with fatal outcome [
12,
13,
14]. In Europe, several countries, such as Ukraine, Romania, Russia, France, Italy, and Hungary, have reported WNV infections in humans and horses during the last decades [
2,
15,
16,
17]. In the last few years, WNV cases have been observed, particularly in Southern Europe, often associated with the major flyways of migratory birds [
18]. In previous wild bird studies in Germany, from 2007 to 2013, neutralizing antibodies against WNV could be detected, primarily in migratory birds, but WNV-specific RNA has not been found yet [
19,
20,
21,
22].
USUV is a close relative of WNV, which was probably introduced to Europe (Italy) in 1996 [
23,
24]. The first large outbreak of USUV occurred in 2001, in Austria, with a significant die-off of Eurasian Blackbirds (
Turdus merula) and Great Grey Owls (
Strix nebulosa) [
25]. Since then, the virus has spread to different European countries, such as Hungary, Switzerland, Spain, Belgium, Czech Republic, France, and Croatia [
26,
27,
28]. In 2010, USUV was isolated from a pool of
Culex pipiens pipiens mosquitoes in Southwestern Germany [
29]. The following two years (2011/12), USUV caused a massive die-off in Eurasian Blackbirds in the Upper Rhine valley [
20,
30], resulting in a continuous decline of the species population in USUV-suitable areas [
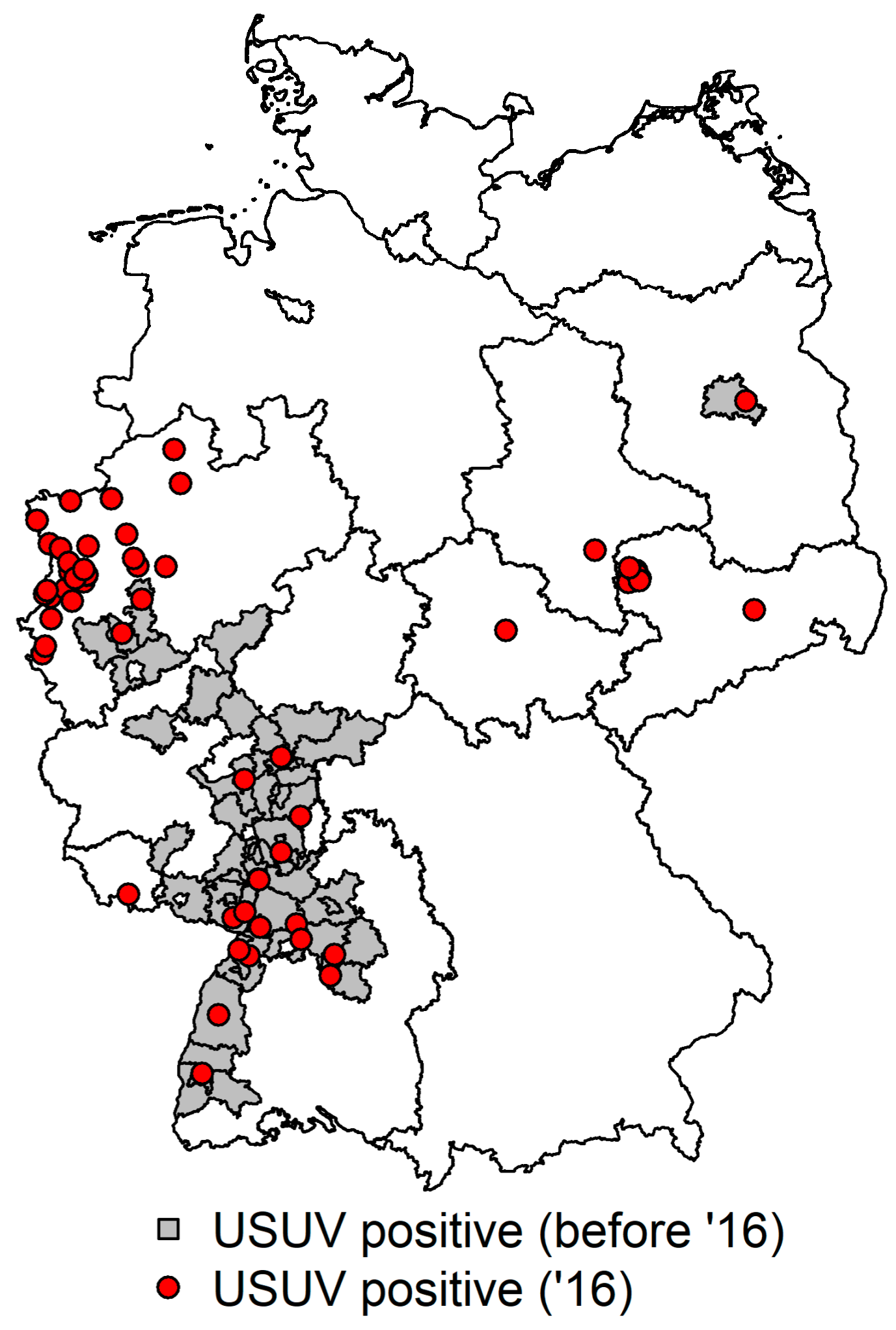
31]. The German Federal States Rhineland-Palatinate, Baden-Wuerttemberg, and Hesse were the major distribution areas for USUV, and sporadic cases were also observed in Cologne and Bonn. Between 2013 and 2015, the number of USUV positive birds decreased. In 2016, a dramatic increase in the number of USUV positive birds was detected. Besides the known USUV epidemic areas spanning the Upper Rhine valley, a cumulative emergence of USUV in the Northern parts of North-Rhine Westphalia up to the border to The Netherlands has been found as well as a higher occurrence of USUV positive birds in the region of Leipzig. Surprisingly, the causative USUV strains found in 2016 represented four lineages, of which two putative novel lineages most likely have been introduced into Germany just recently [
32,
33], from where they probably have spread to other western European countries causing mass-die-offs, preferentially in Eurasian Blackbirds in The Netherlands and Belgium [
33,
34,
35,
36]. While USUV originally was considered as an arbovirus with low zoonotic potential, recent data from various European countries indicate that there also might be a much higher number of clinical neuroinvasive USUV infections in humans than assumed to date [
37,
38].
Whereas USUV has been endemic in Germany since 2010, WNV has not been detected so far, but an incursion is possible. A spreading tendency of the virus in the Northern direction is already apparent, and vectors and hosts are already present. This study continued the molecular and serological surveillance for WNV and USUV in wild birds in Germany, which has been going since 2014. This is the first time that such an extensive monitoring study for WNV and USUV in wild birds with different collection sites distributed all over Germany has been carried out. As WNV and USUV have similar transmission cycles between birds as main amplifying hosts and mosquitoes as vectors, co-circulation as well as overlapping transmission cycles cannot be excluded.
3. Results
We investigated 1902 blood samples for WNV- and USUV-specific RNA. WNV-specific RNA was not detected in any of the samples, whereas four live wild birds were positive for USUV-specific genome sequences by qRT-PCR. In detail, in 2014, one Eurasian Blackbird from Giessen, in the federal state Hesse, in 2015, one Eurasian Blackbird and one City Pigeon (
Columba livia f. domestica) from Dusseldorf (North-Rhine Westphalia), and in 2016, another Eurasian Blackbird from Giessen (Hesse) tested positive. The molecular results of the wild bird blood samples for each year are highlighted in
Table 2.
The investigated organ samples (brain, liver, spleen or heart as available) from diseased, euthanized or dead-found wild birds, from 2016, from all over Germany, were also analyzed for RNA of WNV and USUV. WNV-specific RNA was not found. In total, together with the results obtained from our cooperation partners (for details see sample collection), we can summarize that for 2016, USUV RNA was detected in organ samples from 73 birds of various species belonging to the orders Passeriformes and Strigiformes (for details see
Table 3).
During the period from 2014 to 2016, 58 out of 1825 wild bird sera showed specific neutralizing antibodies against WNV. In 2014, WNV-specific antibodies could be detected in 22 wild birds (
Table 4), in 2015, in 20 wild birds (
Table 5), and in 2016, in 16 wild birds (
Table 6). The titers ranged from 1/10 to 1/240, but were mostly found to be 1/40 or lower. The WNV antibody positive birds were mainly long-distance, partial or short-distance migrants, but also some resident species were affected. The resident and/or partial migrant birds included one Eurasian Magpie (ND
50 1/10), three Eurasian Blackbirds (ND
50 1/10), two Hooded Crows (ND
50 1/10 and 1/20), six Northern Goshawks (ND
50 1/10 to 1/15), four City Pigeons (ND
50 1/10), one Eurasian Green Woodpecker (ND
50 1/20), and one Eurasian Tawny Owl (ND
50 1/10). Only low or no USUV-specific antibodies could be detected in these wild birds, thus unspecific cross-reactivity was excluded (for details see
Table 4,
Table 5 and
Table 6).
In the same period, 56 birds with USUV-neutralizing antibodies were detected among the 1825 sera—three of them in 2014, 32 in 2015, and 21 in 2016. The neutralizing titers varied between 1/10 and 1/1920. Most birds belonged to resident species, but also short-distance and particularly, partial migrants were affected. The most frequently affected resident species were Eurasian Blackbirds (11 specimens), but antibodies were also detected in some bird species from the zoological orders, Accipitriformes and Strigiformes (for details see
Table 4,
Table 5 and
Table 6). Furthermore, also two Eurasian Magpies, one House Sparrow, one Great Tit, three Carrion Crows and one Hooded Crow, from the order Passeriformes, which are resident species or partial migrants, were found to have low USUV antibody titers, ranging from 1/10 to 1/15.
In seven wild birds from this study, it was not possible to discriminate between WNV or USUV titers by VNT, because the antibody titers for both viruses were the same or differed only slightly (1–1.5 fold). In detail, these were one Eurasian Blackbird (WNV ND50 1/20; USUV ND50 1/15), one Typical Warbler (WNV ND50 1/10; USUV ND50 1/10), one City Pigeon (WNV ND50 1/15; USUV ND50 1/15) and one White-tailed Eagle (WNV ND50 1/240; USUV ND50 1/120) in 2014, and two Common Buzzards (WNV ND50 1/15; USUV ND50 1/10) and one Eurasian Woodcock (WNV ND50 1/10; USUV ND50 1/10) in 2015.
The neutralization assay results of all wild bird species between 2014 and 2016 are presented in the
Supplemental Table S1.
4. Discussion
Wild birds play an important role in virus transmission and spread to new and previously unaffected areas. Every year, migratory birds travel over long distances and successively pass from WNV endemic regions onto new areas [
11]. However, WNV viremia in birds usually does not last longer than 6 days, which is too short for long-distance projections, e.g., from the endemic areas in Southern Europe to Northwestern Europe [
6,
10,
40]. Therefore, local bird–mosquito–bird transmission cycles along the routes are required, to allow dispersal. Moreover, other factors (e.g., migratory or climate stress) may prolong viremia in the birds. Finally, persistent subclinical WNV infections lasting over several weeks have also been described for several bird species [
9,
41]. In the last few years, WNV has been found in horses and birds in different European countries, such as Hungary, Italy and Austria [
42,
43,
44,
45]. Due to the geographic proximity, an introduction of WNV to Germany will occur in just a matter of time—the virus is “
ante portas”.
However, WNV-specific nucleic acids could not be detected in any of the 1902 avian blood samples in the present study. Therefore, there is currently no indication for an autochthonous WNV cycle in resident and migratory birds in Germany. These data are in accordance with earlier WNV studies in birds [
19,
20,
21]. Furthermore, no WNV-specific RNA has been detected in mosquitoes in the different German mosquito surveillance studies to date [
46,
47].
In contrast, USUV, a closely related flavivirus, was introduced to Europe about twenty years ago, and phylogenetic analyses revealed the occurrence of different USUV lineages: USUV Europe 1–5 and USUV Africa 1–3 [
27,
33,
48]. The virus has been endemic in Germany since 2011/2012, when it caused a massive die-off in Eurasian Blackbirds and Great Grey Owls in the Upper Rhine valley [
30]. During the following years (2013–2015), the number of yearly USUV positive cases was low. But in 2016, besides the known USUV epidemic regions in the Upper Rhine valley, a higher occurrence of USUV was found in the Northern parts of North-Rhine Westphalia, up to the border to The Netherlands as well as in the region of Leipzig (Eastern Germany). Phylogenetic analysis of the causative German USUV strains demonstrated the circulation of four lineages, two of which probably have recently spread from Germany to the further affected Western European countries (Belgium, The Netherlands) [
32,
33].
In the here-presented live bird survey, spanning 2014–2016, only four USUV genome positive animals were detected during blood investigations, which was not surprising as mostly healthy-looking or orphan wild birds, or wild birds found with injuries caused by trauma, were sampled and not primarily birds with neurological symptoms. The positive birds originated from the known USUV epidemic regions (Dusseldorf and Giessen) in Germany. In contrast, the many USUV-diseased or dead-found wild birds identified during dead bird sampling from all over Germany in 2016 illustrate an onward spread and new virus incursions. Several cases in 2016 have been described in publications by Cadar et al. 2017 and Sieg et al. 2017, to date [
32,
33]. A complete detailed map, showing the origin of all USUV positive dead birds, in 2016, in Germany, is given in
Figure 2.
The serological results showed that 58 out of 1825 wild birds, belonging to 10 bird orders, had WNV-neutralizing antibodies, corresponding to 3.18%. Affected birds were mainly long-distance (L), short-distance (S), and partial migrants (P), which probably came in contact with the virus in their overwintering regions in Southern Europe and/or Africa. The number of WNV positive birds as well as the large number of different wild bird species are in line with previous studies [
19,
20,
21]. WNV antibodies were also detected in 18 resident and/or partial migrant bird species (R, P); however, their WNV-neutralizing antibody titers were quite low (ND
50 1/10 and 1/20) and the only real resident birds (R) among them were one Eurasian Magpie and one Eurasian Tawny Owl (
Table 7). These results are in accordance with previous studies in Germany, but it is still unclear why low WNV-neutralizing antibodies occur in partial migrant and/or resident bird species. An explanation for the WNV seropositivity in raptors, such as the Northern Goshawk, could be that these birds became infected by predating infected migratory birds or scavenging carcasses [
8,
49]. These wild birds have also been classified as facultative/partial migrants so that the infections may have occurred outside Germany. We were also able to detect very low WNV antibody titers in City Pigeons, which usually also are a resident species, but some specimens also migrate over short distances and may come into contact with infected birds in the neighboring countries. In Greece, Domestic Pigeons have been shown to be suitable sentinels for WNV [
50]. Thereby, seroconverted pigeons indicated regions with enzootic virus transmission to warn health authorities at an early stage. Therefore, the number of investigated pigeons should be increased in further studies, to increase sensitivity of the surveillance network.
WNV seropositive birds were also detected among resident birds in The Netherlands, which according to Lim et al. [
49] might indicate that the virus already is circulating in this country. Among the WNV seropositive birds from The Netherlands, there was a large number of Eurasian Coots. Similar findings also were made in different other countries, such as Iran, Spain and the Czech Republic, where a high percentage of the investigated Eurasian Coots demonstrated WNV-neutralizing antibodies [
51,
52,
53]. The species probably plays a special role because they develop a significant antibody response to WNV, thus they may be suitable sentinel animals for WNV. Unfortunately, our sample panel on Eurasian Coots is too small (only four birds in our panel over three years) to draw reliable conclusions. In contrast to Lim et al. [
49], who preferentially investigated waterfowls, our focus was on Passerines and birds of prey, which are known to be highly-susceptible to WNV infections; therefore, more than 1000 avian blood samples from these bird groups occurred in our sample panel.
Due to the fact that some partial migrant and resident birds showed neutralizing antibodies against WNV, local circulation of the virus in Germany cannot be excluded. In principle, the potential vector,
Culex pipiens, is present in Germany, and the susceptibility of indigenous mosquitoes to WNV infection could be demonstrated [
54]. Therefore, mosquito sampling in areas where seropositive resident birds have been found seems to be an important surveillance tool to detect local virus circulation. However, despite large scale mosquito screening projects, with over 143 trapping sites, all over Germany, and additional mass-collection of mosquitoes at predisposed places (such as flood areas or big watercourses or rivers), during the main mosquito season, since 2009, WNV-specific RNA has not been detected in mosquitoes so far [
47]. The same applies to the detection of WNV-specific RNA in wild birds in this, and in former, studies [
20,
21]. Therefore, local circulation of the virus so far undetected in mosquito populations, as assumed by Lim et al. 2017 [
49] for The Netherlands, is questionable for Germany. In the case of local WNV circulation, an increase of antibody response in resident and/or partial migrant birds should occur over the years, but this was not observed in this study. Instead, we observed a regressive number of birds with specific WNV-neutralizing antibodies from 2014 to 2016, although the number of investigated samples in 2016 was three times as high as in 2014 (see
Table 1 and
Table 7).
In contrast to the WNV situation, most of the USUV antibody positive birds in our study were resident birds or facultative/partial migrants. The high neutralizing titers varied between 1/60 and 1/1920. The serological investigation showed a relatively small number of USUV antibody positive birds in 2014—neutralizing antibodies could be detected in just three of 248 birds (1.21%). In the following year, 32 of 821 birds (3.9%) and in 2016, 21 of 756 (2.78%) birds, demonstrated neutralizing antibodies against USUV. In 2015 and 2016, we detected significantly more birds with USUV antibodies, with partially high neutralization titers. However, the numbers of USUV antibody positive birds remained low and a comprehensive diffusion of endemic areas was not detectable. In contrast to the situation in Austria, where four to five years after the USUV outbreak, the percentage of seroreactors increased to over 50% and an establishment of herd immunity was seen [
55], this has not occurred in Germany yet. Our results show sporadic USUV antibody positive birds in the known USUV epidemic regions. These results are in accordance with the previous study from 2011–2013 by Ziegler et al. 2015 [
20]. In other USUV endemic countries, such as Italy, where USUV has been circulating for many years, there is also no evidence for the establishment of herd immunity, as seen in the studies in Austria [
56]. Further wild bird investigations will show whether the percentage of USUV positive seroreactors will increase in the future, and if an entry of WNV into the wild bird population takes place in Germany.