Gene Drive for Mosquito Control: Where Did It Come from and Where Are We Headed?
Abstract
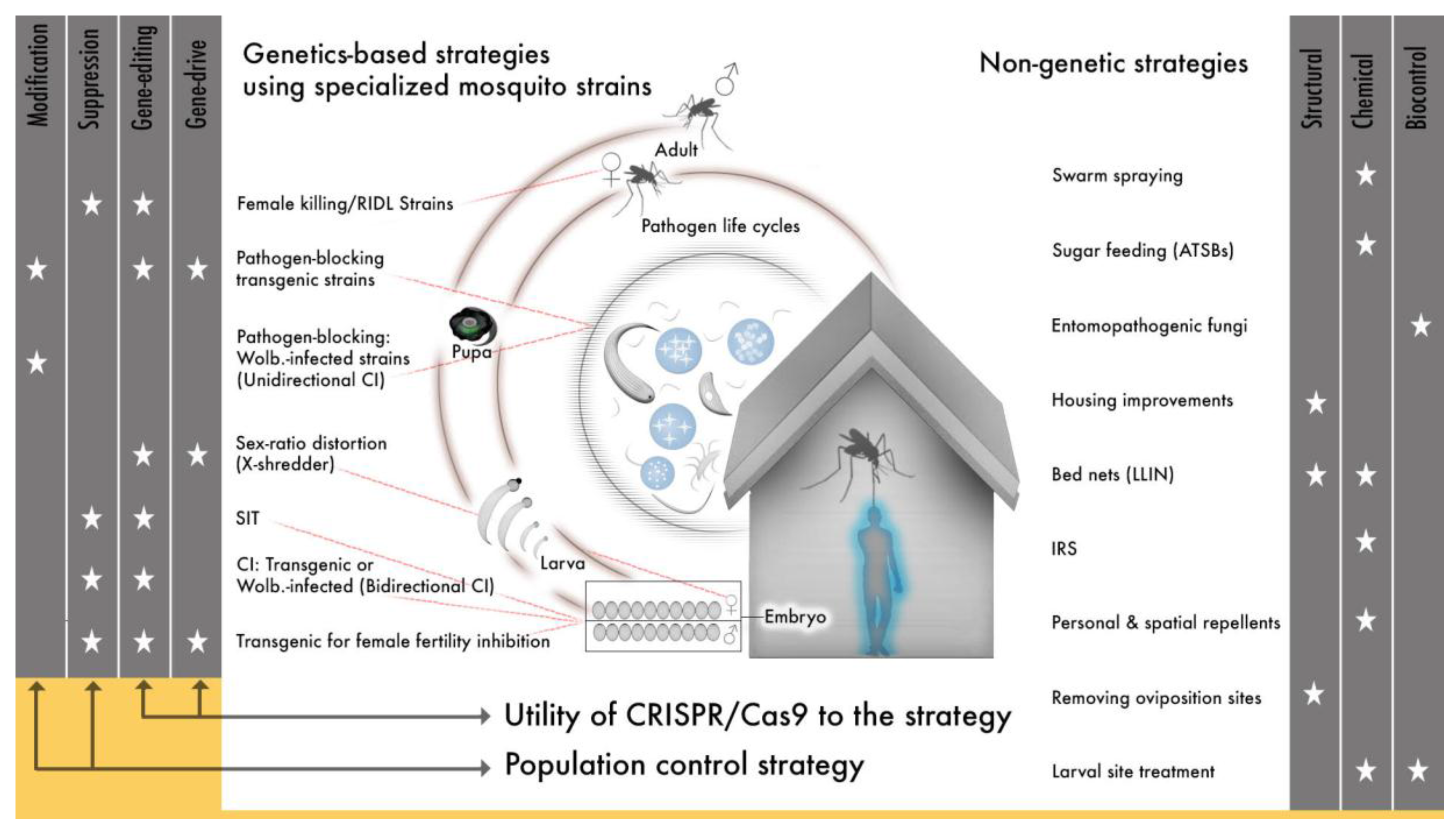
1. Toward Gene drive for Vector-Borne Disease Control: A History
1.1. Initial Proposals for Genetic Control of Insect Vectors of Disease
1.2. Early Definitions and Perspectives on Genetics for Vector Control
1.3. The Birth (or Coming-of-Age) of a New Field: Vector Biology Leverages Natural Genetic Phenomena to Develop Important Tools in Mosquito Genetics
1.3.1. Transposons as a Basis for Genetic Transformation
1.3.2. The Skewing of a Population toward Desirable Genotypes Using Underdominance and Cytoplasmic Incompatibility
1.3.3. The Application of Naturally Occurring Selfish Elements to Synthetic Gene drives in Mosquitoes
1.3.4. The Application of the Endosymbiont Wolbachia to Gene drive in Mosquitoes
1.4. Synthetic Approaches to Genetic Strategies for Mosquito Control
1.4.1. The “Flightless Female”: An Illustration of a Fully Realized Population Suppression Strategy Using Synthetic Gene Modules
1.4.2. Synthetic Engineering for Complete Pathogen Blocking
1.5. A Technology-Driving Need for Site-Specific Gene Editing
2. What Cas9 Has Done for Synthetic Genetic Engineering and Gene drive
3. A Dramatic Shift in the Application of Genetic Approaches to Mosquito and Mosquito-Transmitted Disease Control: Perspectives on the Future
“Clearly, the technology described here is not to be used lightly. Given the suffering caused by some species, neither is it obviously one to be ignored.”—Austin Burt [75].
3.1. Long-Standing Considerations
3.1.1. Early Successes Provide Templates for Predicting Success
3.1.2. Identify and Measure Appropriate Proxies for Assessing the Fitness of a Treatment Strain
3.1.3. The Importance of Modeling
3.1.4. Which Tools, Where
3.2. New Considerations
3.2.1. Gene drive Resistance
3.2.2. Off-Target Effects of the Cas9/sgRNA Complex
3.3.3. Containing an Efficient Gene drive
3.3.4. Globalization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- MacDonald, G. The Epidemiology and Control of Malaria; Oxford University Press: London, UK, 1957. [Google Scholar]
- Bhatt, S.; Weiss, D.; Cameron, E.; Bisanzio, D.; Mappin, B. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature 2015. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. On some peculiar pigmented cells found in two mosquitos fed on malarial blood. Br. Med. J. 1897, 2, 1786–1788. [Google Scholar] [CrossRef] [PubMed]
- Grassi, G.B. Rapporti tra la Malaria e Peculiari Insetti (Zanzaroni e Zanzare Palustri). Available online: http://www.lincei.it/pubblicazioni/rendicontiFMN/rol/visabs.php?lang=en&type=mat&fileId=1046 (accessed on 3 August 2017).
- Reed, W.; Carroll, J.; Agramonte, A. The Etiology of the Yellow Fever. An Additional Note. J. Am. Med. Assoc. 1901, XXXVI, 431–440. [Google Scholar] [CrossRef]
- Manson, P. On the development of Filaria sanguinis hominis, and on the mosquito considered as a nurse*. J. Linn. Soc. Lond. Zool. 1878, 14, 304–311. [Google Scholar] [CrossRef]
- Kourí, G.; Guzmán, M.G.; Bravo, J. Hemorrhagic dengue in Cuba: History of an epidemic. Bull. Pan Am. Health Organ. 1986, 20, 24–30. [Google Scholar] [PubMed]
- Soper, F.; Wilson, D.B. Anopheles gambiae in Brazil, 1930 to 1940; The Rockafellar Foundation: New York, NY, USA, 1943. [Google Scholar]
- Soper, F.L. The elimination of urban yellow fever in the Americas through the eradication of Aedes aegypti. Am. J. Public Health Nations Health 1963, 53, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Carson, R. Silent Spring; Houghton Mifflin: Boston, MA, USA, 1962; ISBN 0618249060. [Google Scholar]
- NIOSH. NIOSH Special Occupation Hazard Reviw: DDT. DHHS (NIOSH) Publication Number 78-200. Available online: https://www.cdc.gov/niosh/docs/78-200/default.html (accessed on 3 August 2017).
- Muller, H.J. Artificial transmutation of the gene. Science 1927, 66, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Serebrovskii, A.S. On the possibility of a new method for the control of insect pests. Zool. Zhurnal 1940, 19, 618–630. [Google Scholar]
- Knipling, E.F. Possibilities of insect control or eradication through the use of sexually sterile males. J. Econ. Entomol. 1955, 48, 902–904. [Google Scholar] [CrossRef]
- Curtis, C.F. Genetic control of insect pests: Growth industry or lead balloon? Biol. J. Linn. Soc. 1985, 26, 359–374. [Google Scholar] [CrossRef]
- Vanderplank, F.L. Hybridization between Glossina Species and Suggested New Method for Control of Certain Species of Tsetse. Nature 1944, 154, 607–608. [Google Scholar] [CrossRef]
- Vanderplank, F.L. Experiments in the hybridisation of Tsetse-flies (Glossina, Diptera) and the possibility of a new control method. Trans. R. Entomol. Soc. Lond. 1947, 98, 1–18. [Google Scholar] [CrossRef]
- Vanderplank, F.L. Experiments in Cross-Breeding Tsetse-Flies (Glossina Species). Ann. Trop. Med. Parasitol. 1948, 42, 131–152. [Google Scholar] [CrossRef] [PubMed]
- WHO Scientific Group on the Genetics of Vectors and Insecticide Resistance. WHO Technical Report Series No. 268; Genetics of Vectors and Insecticide Resistance. Available online: http://apps.who.int/iris/bitstream/10665/40573/1/WHO_TRS_268.pdf (accessed on 3 August 2017).
- Curtis, C.F. Possible use of translocations to fix desirable genes in insect pest populations. Nature 1968, 218, 368–369. [Google Scholar] [CrossRef] [PubMed]
- Curtis, C.F.; Graves, P.M. Methods for replacement of malaria vector populations. J. Trop. Med. Hyg. 1988, 91, 43–48. [Google Scholar] [PubMed]
- Hamilton, W.D. Extraordinary Sex Ratios. Science 1967, 156, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Knipling, E.F.; Laven, H.; Craig, G.B.; Pal, R.; Kitzmiller, J.B.; Smith, C.N.; Brown, A.W. Genetic control of insects of public health importance. Bull. World Health Organ. 1968, 38, 421–438. [Google Scholar] [PubMed]
- Curtis, C.F.; Adak, T. Population replacement in Culex fatigans by means of cytoplasmic incompatibility: 1. Laboratory experiments with non-overlapping generations. Bull. World Health Organ. 1974, 51, 249–255. [Google Scholar] [PubMed]
- Curtis, C.F. Population replacement in Culex fatigans by means of cytoplasmic incompatibility: 2. Field cage experiments with overlapping generations. Bull. World Health Organ. 1976, 53, 107–119. [Google Scholar] [PubMed]
- Wood, R.J.; Cook, L.M.; Hamilton, A.; Whitelaw, A. Transporting the Marker Gene re (Red Eye) into a Laboratory Cage Population of Aedes Aegypti (Diptera: Culicidae), Using Meiotic Drive at the MD Locus. J. Med. Entomol. 1977, 14, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.H.; Sakai, R.K.; Vernick, K.D.; Paskewitz, S.; Seeley, D.C.; Miller, L.H.; Collins, W.E.; Campbell, C.C.; Gwadz, R.W. Genetic selection of a Plasmodium-refractory strain of the malaria vector Anopheles gambiae. Science 1986, 234, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Graves, P.M.; Curtis, C.F. A cage replacement experiment involving introduction of genes for refractoriness to Plasmodium yoelii nigeriensis into a population of Anopheles gambiae (Diptera: Culicidae). J. Med. Entomol. 1982, 19, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Beaty, B.J.; Prager, D.J.; James, A.A.; Jacobs-Lorena, M.; Miller, L.H.; Law, J.H.; Collins, F.H.; Kafatos, F.C. From Tucson to genomics and transgenics: The vector biology network and the emergence of modern vector biology. PLoS Negl. Trop. Dis. 2009, 3. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.H.; James, A.A. Genetic modification of mosquitoes. Sci. Med. 1996, 3, 52–61. [Google Scholar]
- James, A.A.; Blackmer, K.; Racioppi, J.V. A salivary gland-specific, maltase-like gene of the vector Aedes aegypti. Gene 1989, 75, 73–83. [Google Scholar] [CrossRef]
- Meredith, S.E.O.; James, A.A. Biotechnology as applied to vectors and vector control. Ann. Parasito. Hum. Comp. 1990, 65, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.H.; Sakai, R.K.; Romans, P.; Gwadz, R.W.; Kantoff, P.; Coon, H.G. Stable integration and expression of a bacterial gene in the mosquito Anopheles gambiae. Science 1987, 237, 779–781. [Google Scholar] [CrossRef] [PubMed]
- Burt, A.; Trivers, R. Genes in Conflict: The Biology of Selfish Genetic Elements; Belknap Press of Harvard University Press: Cambridge, UK, 2006; ISBN 9780674027220. [Google Scholar]
- McClintock, B. The Discovery and Characterization of Tranposable Elements: The Collected Papers of Barbara McClintock; Garland Publishing: New York, NY, USA, 1987. [Google Scholar]
- Engels, W.R. Invasions of P elements. Genetics 1997, 145, 11–15. [Google Scholar] [PubMed]
- Charlesworth, B.; Sniegowski, P.; Stephan, W. The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 1994, 371, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Mackay, T. Transposable element-induced fitness mutations in Drosophila melanogaster. Genet. Res. 1986. [Google Scholar] [CrossRef]
- Fitzpatrick, B.; Sved, J. High levels of fitness modifiers induced by hybrid dysgenesis in Drosophila melanogaster. Genet. Res. 1986. [Google Scholar] [CrossRef]
- Spradling, A.C.; Rubin, G.M. Transposition of cloned P elements into Drosophila germ line chromosomes. Science 1982, 218, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Warren, A.; Crampton, J. Transposable genetic elements in the genome of the mosquito, Aedes aegypti. In Molecular Insect Science; Hagedorn, H., Hildebrand, H., Kidwell, M., Law, J., Eds.; Plenum Press: New York, NY, USA, 1990; p. 339. [Google Scholar]
- Allen, M.L.; O’Brochta, D.A.; Atkinson, P.W.; Levesque, C.S. Stable, Germ-line Transformation of Culex quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 2001, 38, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Grossman, G.L.; Rafferty, C.S.; Clayton, J.R.; Stevens, T.K.; Mukabayire, O.; Benedict, M.Q. Germline transformation of the malaria vector, Anopheles gambiae, with the piggyBac transposable element. Insect Mol. Biol. 2001, 10, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, F.; Oliveira, S.; Rocha, B. Germline transformation of Aedes fluviatilis (Diptera: Culicidae) with the piggyBac transposable element. Mem. Inst. Oswaldo Cruz 2006. [Google Scholar] [CrossRef]
- Nolan, T.; Bower, T.; Brown, A.; Crisanti, A. piggyBac-mediated germline transformation of the malaria mosquito Anopheles stephensi using the red fluorescent protein DsRed as a selectable marker. J. Biol. 2002. [Google Scholar] [CrossRef]
- Perera, O.; Harrell, I.; Handler, A. Germ-line transformation of the South American malaria vector, Anopheles albimanus, with a piggyBac/EGFP transposon vector is routine and highly efficient. Insect Mol. Biol. 2002, 4, 291–297. [Google Scholar] [CrossRef]
- Catteruccia, F.; Nolan, T.; Loukeris, T.G.; Blass, C.; Savakis, C.; Kafatos, F.C.; Crisanti, A. Stable germline transformation of the malaria mosquito Anopheles stephensi. Nature 2000, 405, 959–962. [Google Scholar] [CrossRef] [PubMed]
- Jasinskiene, N.; Coates, C.J.; Benedict, M.Q.; Cornel, A.J.; Rafferty, C.S.; James, A.A.; Collins, F.H. Stable transformation of the yellow fever mosquito, Aedes aegypti, with the Hermes element from the housefly. Proc. Natl. Acad. Sci. USA 1998, 95, 3743–3747. [Google Scholar] [CrossRef] [PubMed]
- Coates, C.J.; Jasinskiene, N.; Miyashiro, L.; James, A.A. Mariner transposition and transformation of the yellow fever mosquito, Aedes aegypti. Proc. Natl. Acad. Sci. USA 1998, 95, 3748–3751. [Google Scholar] [CrossRef] [PubMed]
- Labbé, G.M.C.; Nimmo, D.D.; Alphey, L. piggybac- and PhiC31-mediated genetic transformation of the Asian tiger mosquito, Aedes albopictus (Skuse). PLoS Negl. Trop. Dis. 2010, 4, e788. [Google Scholar] [CrossRef] [PubMed]
- Adelman, Z.N.; Basu, S.; Myles, K.M. Gene insertions and deletion in mosquitoes. In Genetic Control of Malaria and Dengue; Adelman, Z.N., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 139–168. [Google Scholar]
- Kidwell, M.; Ribeiro, J. Can transposable elements be used to drive disease refractoriness genes into vector populations? Parasitol. Today 1992. [Google Scholar] [CrossRef]
- James, A.A. Gene drive systems in mosquitoes: Rules of the road. Trends Parasitol. 2005, 21, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Gould, F.; Schliekelman, P. Population genetics of autocidal control and strain replacement. Annu. Rev. Entomol. 2004, 49, 193–217. [Google Scholar] [CrossRef] [PubMed]
- Rasgon, J.L.; Gould, F. Transposable element insertion location bias and the dynamics of gene drive in mosquito populations. Insect Mol. Biol. 2005, 14, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Scali, C.; Nolan, T.; Sharakhov, I.; Sharakhova, M.; Crisanti, A.; Catteruccia, F. Post-integration behavior of a Minos transposon in the malaria mosquito Anopheles stephensi. Mol. Genet. Genom. 2007, 278, 575–584. [Google Scholar] [CrossRef] [PubMed]
- O’Brochta, D.A.; Sethuraman, N.; Wilson, R.; Hice, R.H.; Pinkerton, A.C.; Levesque, C.S.; Bideshi, D.K.; Jasinskiene, N.; Coates, C.J.; James, A.A.; et al. Gene vector and transposable element behavior in mosquitoes. J. Exp. Biol. 2003, 206, 3823–3834. [Google Scholar] [CrossRef] [PubMed]
- Macias, V.M.; Jimenez, A.J.; Burini-Kojin, B.; Pledger, D.; Jasinskiene, N.; Phong, C.H.; Chu, K.; Fazekas, A.; Martin, K.; Marinotti, O.; et al. nanos-Driven expression of piggyBac transposase induces mobilization of a synthetic autonomic transposon in Anopheles stephensi. Insect Biochem. Mol. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- O’Brochta, D.A.; Alford, R.T.; Pilitt, K.L.; Aluvihare, C.U.; Harrell, R.A., II. piggyBac transposon remobilization and enhancer detection in Anopheles mosquitoes. Proc. Natl. Acad. Sci. USA 2011, 108, 16339–16344. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.; Bax, N.; Grewe, P. Engineered underdominance allows efficient and economical introgression of traits into pest populations. J. Theor. Biol. 2001, 212, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Akbari, O.S.; Matzen, K.D.; Marshall, J.M.; Huang, H.; Ward, C.M.; Hay, B.A. A Synthetic Gene Drive System for Local, Reversible Modification and Suppression of Insect Populations. Curr. Biol. 2013, 23, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Yen, J.H.; Barr, A.R. The etiological agent of cytoplasmic incompatibility in Culex pipiens. J. Invertebr. Pathol. 1973, 22, 242–250. [Google Scholar] [CrossRef]
- Yen, J.H.; Barr, A.R. New hypothesis of the cause of cytoplasmic incompatibility in Culex pipiens L. Nature 1971, 232, 657–658. [Google Scholar] [CrossRef] [PubMed]
- Stouthamer, R.; Breeuwer, J.A.J.; Hurst, G.D.D. Wolbachia pipientis: Microbial Manipulator of Arthropod Reproduction. Annu. Rev. Microbiol. 1999, 53, 71–102. [Google Scholar] [CrossRef] [PubMed]
- LePage, D.P.; Metcalf, J.A.; Bordenstein, S.R.; On, J.; Perlmutter, J.I.; Shropshire, J.D.; Layton, E.M.; Funkhouser-Jones, L.J.; Beckmann, J.F.; Bordenstein, S.R. Prophage WO genes recapitulate and enhance Wolbachia-induced cytoplasmic incompatibility. Nature 2017, 543, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Sinkins, S.; Curtis, C.; O’Neill, S. The potential application of inherited symbiont systems to pest control. Explore our Research. In Influential Passangers: Inherited Microorganisms and Arthropod Reproduction; O’Neill, S.L., Hoffman, A.A., Werren, J.H., Eds.; Oxford University Press: New York, NY, USA, 1997; pp. 155–175. ISBN 0198577869. [Google Scholar]
- Turelli, M.; Hoffmann, A.A. Microbe-induced cytoplasmic incompatibility as a mechanism for introducing transgenes into arthropod populations. Insect Mol. Biol. 1999, 8, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Wade, M.J.; Beeman, R.W. The population dynamics of maternal-effect selfish genes. Genetics 1994, 138, 1309–1314. [Google Scholar] [PubMed]
- Beeman, R.; Friesen, K.; Denell, R. Maternal-effect selfish genes in flour beetles. Science 1992, 256. [Google Scholar] [CrossRef]
- Chen, C.; Huang, H.; Ward, C.M.; Su, J.T.; Schaeffer, L.V.; Guo, M.; Hay, B.A. A Synthetic Maternal-Effect Selfish Genetic Element Drives Population Replacement in Drosophila. Science 2007, 316, 597–600. [Google Scholar]
- Belfort, M.; Derbyshire, V.; Parker, M.M.; Cousineau, B.; Lambowitz, A.M. Mobile Introns: Pathways and Proteins. In Mobile DNA II; American Society of Microbiology: Washington, DC, USA, 2002; pp. 761–783. [Google Scholar]
- Chevalier, B.S.; Stoddard, B.L. Homing endonucleases: Structural and functional insight into the catalysts of intron/intein mobility. Nucleic Acids Res. 2001, 29, 3757–3774. [Google Scholar] [CrossRef] [PubMed]
- Coen, D.; Deutsch, J.; Netter, P.; Petrochilo, E. Mitochondrial genetics. I. Methodology and phenomenology. Symp. Soc. Exp. Biol. 1970, 24, 449–496. [Google Scholar] [PubMed]
- Stoddard, B.L. Homing endonuclease structure and function. Q. Rev. Biophys. 2005, 38, 49–95. [Google Scholar] [CrossRef] [PubMed]
- Burt, A. Site-specific selfish genes as tools for the control and genetic engineering of natural populations. Proc. Biol. Sci. 2003, 270, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, J.; Taylor, G.; Havranek, J. Computational reprogramming of homing endonuclease specificity at multiple adjacent base pairs. Nucleic Acids Res. 2010. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, J.; Havranek, J.; Duarte, C.; Sussman, D. Computational redesign of endonuclease DNA binding and cleavage specificity. Nature 2006. [Google Scholar] [CrossRef] [PubMed]
- Jarjour, J.; West-Foyle, H.; Certo, M. High-resolution profiling of homing endonuclease binding and catalytic specificity using yeast surface display. Nucleic Acids Res. 2009. [Google Scholar] [CrossRef] [PubMed]
- Windbichler, N.; Miriam, M.; Papathanos, P.A.; Thyme, S.B.; Li, H.; Ulge, U.Y.; Hovde, B.T.; Baker, D.; Monnat, R.J.; College, B.; et al. A synthetic homing endonuclease-based gene drive system in the human malaria mosquito. Nature 2011, 473, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Laven, H. Eradication of Culex pipiens fatigans through Cytoplasmic Incompatibility. Nature 1967, 216, 383–384. [Google Scholar] [CrossRef] [PubMed]
- Kriesner, P.; Hoffmann, A.A.; Lee, S.F.; Turelli, M.; Weeks, A.R. Rapid Sequential Spread of Two Wolbachia Variants in Drosophila simulans. PLoS Pathog. 2013, 9, e1003607. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.H.; Paskewitz, S.M. Malaria: Current and Future Prospects for Control. Annu. Rev. Entomol. 1995, 40, 195–219. [Google Scholar] [CrossRef] [PubMed]
- Sinkins, S.P.; O’Neill, S.L. Wolbachia as a vehicle to modify insect populations. In Insect Transgenesis: Methods and Applications; CRC Press: Boca Raton, FL, USA, 2000; pp. 271–288. [Google Scholar]
- Calvitti, M.; Moretti, R.; Lampazzi, E.; Bellini, R.; Dobson, S.L. Characterization of a new Aedes albopictus (Diptera: Culicidae)-Wolbachia pipientis (Rickettsiales: Rickettsiaceae) symbiotic association generated by artificial transfer of the wPip strain from Culex pipiens (Diptera: Culicidae). J. Med. Entomol. 2010, 47, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Calvitti, M.; Moretti, R.; Skidmore, A.R.; Dobson, S.L. Wolbachia strain wPip yields a pattern of cytoplasmic incompatibility enhancing a Wolbachia-based suppression strategy against the disease vector Aedes albopictus. Parasit. Vectors 2012, 5, 254. [Google Scholar] [CrossRef] [PubMed]
- Moretti, R.; Calvitti, M. Male mating performance and cytoplasmic incompatibility in a wPip Wolbachia trans-infected line of Aedes albopictus (Stegomyia albopicta). Med. Vet. Entomol. 2013, 27, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Puggioli, A.; Calvitti, M.; Moretti, R.; Bellini, R. wPip Wolbachia contribution to Aedes albopictus SIT performance: Advantages under intensive rearing. Acta Trop. 2016, 164, 473–481. [Google Scholar] [CrossRef] [PubMed]
- MosquitoMate. Available online: http://mosquitomate.com/?v=3.0 (accessed on 1 January 2017).
- Walker, T.; Johnson, P.H.; Moreira, L.A.; Iturbe-Ormaetxe, I.; Frentiu, F.D.; McMeniman, C.J.; Leong, Y.S.; Dong, Y.; Axford, J.; Kriesner, P.; et al. The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 2011, 476, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Montgomery, B.L.; Popovici, J.; Iturbe-Ormaetxe, I.; Johnson, P.H.; Muzzi, F.; Greenfield, M.; Durkan, M.; Leong, Y.S.; Dong, Y.; et al. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 2011, 476, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Frentiu, F.D.; Zakir, T.; Walker, T.; Popovici, J.; Pyke, A.T.; van den Hurk, A.; McGraw, E.A.; O’Neill, S.L. Limited Dengue Virus Replication in Field-Collected Aedes aegypti Mosquitoes Infected with Wolbachia. PLoS Negl. Trop. Dis. 2014, 8, e2688. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.H.; Carrasco, A.M.; Frentiu, F.D.; Chenoweth, S.F.; Beebe, N.W.; van den Hurk, A.F.; Simmons, C.P.; O’Neill, S.L.; McGraw, E.A. Wolbachia Reduces the Transmission Potential of Dengue-Infected Aedes aegypti. PLoS Negl. Trop. Dis. 2015, 9, e0003894. [Google Scholar] [CrossRef] [PubMed]
- Weinert, L.A.; Araujo-Jnr, E.V.; Ahmed, M.Z.; Welch, J.J. The incidence of bacterial endosymbionts in terrestrial arthropods. Proc. R. Soc. B Biol. Sci. 2015, 282, 20150249. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, D. The Importance of Long-Term Social Research in Enabling Participation and Developing Engagement Strategies for New Dengue Control Technologies. PLoS Negl. Trop. Dis. 2012, 6, e1785. [Google Scholar] [CrossRef] [PubMed]
- Nikoh, N.; Tanaka, K.; Shibata, F.; Kondo, N.; Hizume, M.; Shimada, M.; Fukatsu, T. Wolbachia genome integrated in an insect chromosome: Evolution and fate of laterally transferred endosymbiont genes. Genome Res. 2008, 18, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Sun, L.V.; Vamathevan, J.; Riegler, M.; Deboy, R.; Brownlie, J.C.; McGraw, E.A.; Martin, W.; Esser, C.; Ahmadinejad, N.; et al. Phylogenomics of the reproductive parasite Wolbachia pipientis wMel: A streamlined genome overrun by mobile genetic elements. PLoS Biol. 2004, 2, E69. [Google Scholar] [CrossRef] [PubMed]
- Kondo, N.; Nikoh, N.; Ijichi, N. Genome fragment of Wolbachia endosymbiont transferred to X chromosome of host insect. Proc. Natl. Acad. Sci. USA 2002. [Google Scholar] [CrossRef] [PubMed]
- Klasson, L.; Kambris, Z.; Cook, P. Horizontal gene transfer between Wolbachia and the mosquito Aedes aegypti. BMC. Genom. 2009. [Google Scholar] [CrossRef] [PubMed]
- Fenn, K.; Conlon, C.; Jones, M.; Quail, M. Phylogenetic relationships of the Wolbachia of nematodes and arthropods. PLoS Pathog. 2006. [Google Scholar] [CrossRef] [PubMed]
- Hotopp, J.C.D. Horizontal gene transfer between bacteria and animals. Trends Genet. 2011. [Google Scholar] [CrossRef]
- Hotopp, J.C.D.; Clark, M.E.; Oliveira, D.C.S.G.; Foster, J.M.; Fischer, P.; Torres, M.C.M.; Giebel, J.D.; Kumar, N.; Ishmael, N.; Wang, S.; et al. Widespread Lateral Gene Transfer from Intracellular Bacteria to Multicellular Eukaryotes. Science 2007, 317, 1753–1756. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.; Ganatra, M.; Kamal, I.; Ware, J.; Makarova, K. The Wolbachia genome of Brugia malayi: Endosymbiont evolution within a human pathogenic nematode. PLoS Biol. 2005. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Lees, R.S.; Nimmo, D.; Aw, D.; Jin, L.; Gray, P.; Berendonk, T.U.; White-Cooper, H.; Scaife, S.; Kim Phuc, H.; et al. Female-specific flightless phenotype for mosquito control. Proc. Natl. Acad. Sci. USA 2010, 107, 4550–4554. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, M.P.; Su, Z.; Alphey, N.; Alphey, L.S.; Coleman, P.G.; Wein, L.M. Analyzing the control of mosquito-borne diseases by a dominant lethal genetic system. Proc. Natl. Acad. Sci. USA 2007, 104, 9540–9545. [Google Scholar] [CrossRef] [PubMed]
- Phuc, H.; Andreasen, M.H.; Burton, R.S.; Vass, C.; Epton, M.J.; Pape, G.; Fu, G.; Condon, K.C.; Scaife, S.; Donnelly, C.A.; et al. Late-acting dominant lethal genetic systems and mosquito control. BMC Biol. 2007, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.D.; Donnelly, C.A.; Wood, R.J.; Alphey, L.S. Insect population control using a dominant, repressible, lethal genetic system. Science 2000, 287, 2474–2476. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, D.O.; McKemey, A.R.; Garziera, L.; Lacroix, R.; Donnelly, C.A.; Alphey, L.; Malavasi, A.; Capurro, M.L. Suppression of a Field Population of Aedes aegypti in Brazil by Sustained Release of Transgenic Male Mosquitoes. PLoS Negl. Trop. Dis. 2015, 9, e0003864. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.F.; McKemey, A.R.; Nimmo, D.; Curtis, Z.; Black, I.; Morgan, S.A.; Oviedo, M.N.; Lacroix, R.; Naish, N.; Morrison, N.I.; et al. Successful suppression of a field mosquito population by sustained release of engineered male mosquitoes. Nat. Biotechnol. 2012, 30, 828–830. [Google Scholar] [CrossRef] [PubMed]
- Labbé, G.M.C.; Scaife, S.; Morgan, S.A.; Curtis, Z.H.; Alphey, L. Female-Specific Flightless (fsRIDL) Phenotype for Control of Aedes albopictus. PLoS Negl. Trop. Dis. 2012, 6, e1724. [Google Scholar] [CrossRef]
- Marinotti, O.; Jasinskiene, N.; Fazekas, A.; Scaife, S.; Fu, G.; Mattingly, S.T.; Chow, K.; Brown, D.M.; Alphey, L.; James, A.A. Development of a population suppression strain of the human malaria vector mosquito, Anopheles stephensi. Malar. J. 2013, 12, 142. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; McGrath, J.; Wang, P.; Lee, T. Cellular Toxicity Induced by SRF-Mediated Transcriptional Squelching. Toxicol. Sci. 2006, 96, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Environmental Assessment for Investigational Use of Aedes aegypti OX513A. Available online: https://www.fda.gov/downloads/AnimalVeterinary/DevelopmentApprovalProcess/GeneticEngineering/GeneticallyEngineeredAnimals/UCM514698.pdf (accessed on 5 August 2016).
- Clyde, K.; Kyle, J.L.; Harris, E. Recent advances in deciphering viral and host determinants of dengue virus replication and pathogenesis. J. Virol. 2006, 80, 11418–11431. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Das, S.; Cirimotich, C.; Souza-Neto, J.A.; McLean, K.J.; Dimopoulos, G. Engineered anopheles immunity to Plasmodium infection. PLoS Pathog. 2011, 7, e1002458. [Google Scholar] [CrossRef] [PubMed]
- Erickson, S.M.; Xi, Z.; Mayhew, G.F.; Ramirez, J.L.; Aliota, M.T.; Christensen, B.M.; Dimopoulos, G. Mosquito infection responses to developing filarial worms. PLoS Negl. Trop. Dis. 2009, 3, e529. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Coppens, I.; Gårdsvoll, H.; Ploug, M.; Jacobs-Lorena, M. Plasmodium ookinetes coopt mammalian plasminogen to invade the mosquito midgut. Proc. Natl. Acad. Sci. USA 2011, 108, 17153–17158. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Shields, A.R.; Jupatanakul, N.; Dimopoulos, G.; Mongin, E. Suppressing Dengue-2 Infection by Chemical Inhibition of Aedes aegypti Host Factors. PLoS Negl. Trop. Dis. 2014, 8, e3084. [Google Scholar] [CrossRef] [PubMed]
- Kuadkitkan, A.; Wikan, N.; Fongsaran, C.; Smith, D.R. Identification and characterization of prohibitin as a receptor protein mediating DENV-2 entry into insect cells. Virology 2010, 406, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Olson, K.E.; Adelman, Z.N.; Travanty, E.A.; Sanchez-Vargas, I.; Beaty, B.J.; Blair, C.D. Developing arbovirus resistance in mosquitoes. Insect Biochem. Mol. Biol. 2002, 32, 1333–1343. [Google Scholar] [CrossRef]
- Osta, M.A.; Christophides, G.K.; Vlachou, D.; Kafatos, F.C. Innate immunity in the malaria vector Anopheles gambiae: Comparative and functional genomics. J. Exp. Biol. 2004, 207, 2551–2563. [Google Scholar] [CrossRef] [PubMed]
- Pike, A.; Vadlamani, A.; Sandiford, S.L.; Gacita, A.; Dimopoulos, G. Characterization of the Rel2-regulated transcriptome and proteome of Anopheles stephensi identifies new anti-Plasmodium factors. Insect Biochem. Mol. Biol. 2014, 52, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Niare, O.; Markianos, K.; Volz, J.; Oduol, F.; Touré, A.; Bagayoko, M.; Sangaré, D.; Traoré, S.F.; Wang, R.; Blass, C.; et al. Genetic Loci Affecting Resistance to Human Malaria Parasites in a West African Mosquito Vector Population. Science 2002, 298, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Riehle, M.M.; Markianos, K.; Lambrechts, L.; Xia, A.; Sharakhov, I.; Koella, J.C.; Vernick, K.D. A major genetic locus controlling natural Plasmodium falciparum infection is shared by East and West African Anopheles gambiae. Malar. J. 2007, 6, 87. [Google Scholar] [CrossRef] [PubMed]
- Riehle, M.A.; Moreira, C.K.; Lampe, D.; Lauzon, C.; Jacobs-Lorena, M. Using bacteria to express and display anti-Plasmodium molecules in the mosquito midgut. Int. J. Parasitol. 2007, 37, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.L.; Dimopoulos, G. The Toll immune signaling pathway control conserved anti-dengue defenses across diverse Ae. aegypti strains and against multiple dengue virus serotypes. Dev. Comp. Immunol. 2010, 34, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.C.; Barillas-Mury, C.; Jacobs-Lorena, M. Hemocyte differentiation mediates the mosquito late-phase immune response against Plasmodium in Anopheles gambiae. Proc. Natl. Acad. Sci. USA 2015, 112, E3412–E3420. [Google Scholar] [CrossRef] [PubMed]
- Corby-Harris, V.; Drexler, A.; Watkins de Jong, L.; Antonova, Y.; Pakpour, N.; Ziegler, R.; Ramberg, F.; Lewis, E.E.; Brown, J.M.; Luckhart, S.; et al. Activation of Akt signaling reduces the prevalence and intensity of malaria parasite infection and lifespan in Anopheles stephensi mosquitoes. PLoS Pathog. 2010, 6, e1001003. [Google Scholar] [CrossRef] [PubMed]
- Hauck, E.S.; Antonova-Koch, Y.; Drexler, A.; Pietri, J.; Pakpour, N.; Liu, D.; Blacutt, J.; Riehle, M.A.; Luckhart, S. Overexpression of phosphatase and tensin homolog improves fitness and decreases Plasmodium falciparum development in Anopheles stephensi. Microbes Infect. 2013, 15, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Luckhart, S.; Giulivi, C.; Drexler, A.L.; Antonova-Koch, Y.; Sakaguchi, D.; Napoli, E.; Wong, S.; Price, M.S.; Eigenheer, R.; Phinney, B.S.; et al. Sustained activation of Akt elicits mitochondrial dysfunction to block Plasmodium falciparum infection in the mosquito host. PLoS Pathog. 2013, 9, e1003180. [Google Scholar] [CrossRef] [PubMed]
- Ito, J.; Ghosh, A.; Moreira, L.A.; Wimmer, E.A.; Jacobs-Lorena, M. Transgenic anopheline mosquitoes impaired in transmission of a malaria parasite. Nature 2002, 417, 452–455. [Google Scholar] [CrossRef] [PubMed]
- Adelman, Z.N.; Basu, S.; Myles, K.M. Engineering pathogen resistance in mosquitoes. In Genetic Control of Malaria and Dengue; Adelman, Z.N., Ed.; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Isaacs, A.T.; Jasinskiene, N.; Tretiakov, M.; Thiery, I.; Zettor, A.; Bourgouin, C.; James, A.A. Transgenic Anopheles stephensi coexpressing single-chain antibodies resist Plasmodium falciparum development. Proc. Natl. Acad. Sci. USA 2012, 109, E1922–E1930. [Google Scholar] [CrossRef] [PubMed]
- Mathur, G.; Sanchez-Vargas, I.; Alvarez, D.; Olson, K.E.; Marinotti, O.; James, A.A. Transgene-mediated suppression of dengue viruses in the salivary glands of the yellow fever mosquito, Aedes aegypti. Insect Mol. Biol. 2010, 19, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Fraser, M.J.; Ciszczon, T.; Elick, T.; Bauser, C. Precise excision of TTAA-specific lepidopteran transposons piggyBac (IFP2) and tagalong (TFP3) from the baculovirus genome in cell lines from two species of Lepidoptera. Insect Mol. Biol. 1996, 5, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Arcà, B.; Zabalou, S.; Loukeris, T.; Savakis, C. Mobilization of a Minos transposon in Drosophila melanogaster chromosomes and chromatid repair by heteroduplex formation. Genetics 1997, 145, 267–279. [Google Scholar] [PubMed]
- Spradling, A.; Stern, D.; Beaton, A.; Rhem, E. The Berkeley Drosophila Genome Project gene disruption project: Single P-element insertions mutating 25% of vital Drosophila genes. Genetics 1999, 153, 135–177. [Google Scholar]
- Spradling, A.; Stern, D.; Kiss, I. Gene disruptions using P transposable elements: An integral component of the Drosophila genome project. Proc. Natl. Acad. Sci. USA 1995, 92, 10824–10830. [Google Scholar] [CrossRef] [PubMed]
- Lyman, R.; Lawrence, F.; Nuzhdin, S.; Mackay, T. Effects of single P-element insertions on bristle number and viability in Drosophila melanogaster. Genetics 1996, 143, 277–292. [Google Scholar] [PubMed]
- Marrelli, M.T.; Moreira, C.K.; Kelly, D.; Alphey, L.; Jacobs-Lorena, M. Mosquito transgenesis: What is the fitness cost? Trends Parasitol. 2006, 22, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, R.; Choi, M.; Stoddard, B.L. Redesign of extensive protein-DNA interfaces of meganucleases using iterative cycles of in vitro compartmentalization. Proc. Natl. Acad. Sci. USA 2014, 111, 4061–4066. [Google Scholar] [CrossRef] [PubMed]
- Windbichler, N.; Papathanos, P.A.; Catteruccia, F.; Ranson, H.; Burt, A.; Crisanti, A. Homing endonuclease mediated gene targeting in Anopheles gambiae cells and embryos. Nucleic Acids Res. 2007, 35, 5922–5933. [Google Scholar] [CrossRef] [PubMed]
- Nimmo, D.D.; Alphey, L.; Meredith, J.M.; Eggleston, P. High efficiency site-specific genetic engineering of the mosquito genome. Insect Mol. Biol. 2006, 15, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Meredith, J.M.; Basu, S.; Nimmo, D.D.; Larget-Thiery, I.; Warr, E.L.; Underhill, A.; McArthur, C.C.; Carter, V.; Hurd, H.; Bourgouin, C.; et al. Site-specific integration and expression of an anti-malarial gene in transgenic Anopheles gambiae significantly reduces Plasmodium infections. PLoS ONE 2011, 6, e14587. [Google Scholar] [CrossRef] [PubMed]
- Beerli, R.R.; Barbas, C.F. Engineering polydactyl zinc-finger transcription factors. Nat. Biotechnol. 2002, 20, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Segal, D.J.; Ghiara, J.B.; Barbas, C.F. Design of polydactyl zinc-finger proteins for unique addressing within complex genomes. Proc. Natl. Acad. Sci. USA 1997, 94, 5525–5530. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.C.; Holmes, M.C.; Wang, J.; Guschin, D.Y.; Lee, Y.-L.; Rupniewski, I.; Beausejour, C.M.; Waite, A.J.; Wang, N.S.; Kim, K.A.; et al. An improved zinc-finger nuclease architecture for highly specific genome editing. Nat. Biotechnol. 2007, 25, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-G.; Cha, J.; Chandrasegaran, S. Hybrid restriction enzymes: Zinc finger fusions to Fok I cleavage domain (Flavobacterium okeanokoites/chimeric restriction endonuclease/protein engineering/recognition and cleavage domains). Proc. Natl. Acad. Sci. USA 1996, 93, 1156–1160. [Google Scholar] [CrossRef] [PubMed]
- Christian, M.; Cermak, T.; Doyle, E.L.; Schmidt, C.; Zhang, F.; Hummel, A.; Bogdanove, A.J.; Voytas, D.F. Targeting DNA Double-Strand Breaks with TAL Effector Nucleases. Genetics 2010, 186, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Boch, J.; Scholze, H.; Schornack, S.; Landgraf, A.; Hahn, S.; Kay, S.; Lahaye, T.; Nickstadt, A.; Bonas, U. Breaking the code of DNA binding specificity of TAL-type III effectors. Science 2009, 326, 1509–1512. [Google Scholar] [CrossRef] [PubMed]
- Moscou, M.J.; Bogdanove, A.J. A simple cipher governs DNA recognition by TAL effectors. Science 2009, 326, 1501. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kweon, J.; Kim, A.; Chon, J.K.; Yoo, J.Y.; Kim, H.J.; Kim, S.; Lee, C.; Jeong, E.; Chung, E.; et al. A library of TAL effector nucleases spanning the human genome. Nat. Biotechnol. 2013, 31, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Aryan, A.; Anderson, M.A.E.; Myles, K.M.; Adelman, Z.N. Germline excision of transgenes in Aedes aegypti by homing endonucleases. Sci. Rep. 2013, 3, 1603. [Google Scholar] [CrossRef] [PubMed]
- Smidler, A.L.; Terenzi, O.; Soichot, J.; Levashina, E.A.; Marois, E. Targeted mutagenesis in the malaria mosquito using TALE nucleases. PLoS ONE 2013, 8, e74511. [Google Scholar] [CrossRef] [PubMed]
- DeGennaro, M.; McBride, C.S.; Seeholzer, L.; Nakagawa, T.; Dennis, E.J.; Goldman, C.; Jasinskiene, N.; James, A.A.; Vosshall, L.B. orco mutant mosquitoes lose strong preference for humans and are not repelled by volatile DEET. Nature 2013, 498, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Karberg, M.; Long, M.; Jones, J.P.; Sullenger, B.; Lambowitz, A.M. Group II Introns Designed to Insert into Therapeutically Relevant DNA Target Sites in Human Cells. Science 2000, 289, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Mali, P.; Yang, L.; Esvelt, K.M.; Aach, J.; Guell, M.; DiCarlo, J.E.; Norville, J.E.; Church, G.M. RNA-guided human genome engineering via Cas9. Science 2013, 339, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A.; et al. Multiplex Genome Engineering Using CRISPR/Cas Systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Aryan, A.; Overcash, J.M.; Samuel, G.H.; Anderson, M.A.E.; Dahlem, T.J.; Myles, K.M.; Adelman, Z.N. Silencing of end-joining repair for efficient site-specific gene insertion after TALEN/CRISPR mutagenesis in Aedes aegypti. Proc. Natl. Acad. Sci. USA 2015, 112, 4038–4043. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Lin, J.; Held, N.L.; Clem, R.J.; Passarelli, A.L.; Franz, A.W.E. Heritable CRISPR/Cas9-Mediated Genome Editing in the Yellow Fever Mosquito, Aedes aegypti. PLoS ONE 2015, 10, e0122353. [Google Scholar] [CrossRef] [PubMed]
- Kistler, K.E.; Vosshall, L.B.; Matthews, B.J. Genome Engineering with CRISPR-Cas9 in the Mosquito Aedes aegypti. Cell Rep. 2015, 11, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.B.; Basu, S.; Jiang, X.; Qi, Y.; Timoshevskiy, V.A.; Biedler, J.K.; Sharakhova, M.V.; Elahi, R.; Anderson, M.A.E.; Chen, X.-G.; et al. A male-determining factor in the mosquito Aedes aegypti. Science 2015, 348, 1268–1270. [Google Scholar] [CrossRef] [PubMed]
- Hammond, A.; Galizi, R.; Kyrou, K.; Simoni, A.; Siniscalchi, C.; Katsanos, D.; Gribble, M.; Baker, D.; Marois, E.; Russell, S.; et al. A CRISPR-Cas9 gene drive system targeting female reproduction in the malaria mosquito vector Anopheles gambiae. Nat. Biotechnol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gantz, V.M.; Jasinskiene, N.; Tatarenkova, O.; Fazekas, A.; Macias, V.M.; Bier, E.; James, A.A. Highly efficient Cas9-mediated gene drive for population modification of the malaria vector mosquito Anopheles stephensi. Proc. Natl. Acad. Sci. USA 2015, 112, E6736–E6743. [Google Scholar] [CrossRef] [PubMed]
- Gantz, V.M.; Bier, E. The mutagenic chain reaction: A method for converting heterozygous to homozygous mutations. Science. 2015, 348, 442–444. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Akbari, O.S.; White, B.J. Highly efficient site-specific mutagenesis in malaria mosquitoes using CRISPR. bioRxiv 2017. [Google Scholar] [CrossRef]
- Deveau, H.; Garneau, J.E.; Moineau, S. CRISPR/Cas System and Its Role in Phage-Bacteria Interactions. Annu. Rev. Microbiol. 2010, 64, 475–493. [Google Scholar] [CrossRef] [PubMed]
- Wiedenheft, B.; Sternberg, S.H.; Doudna, J.A. RNA-guided genetic silencing systems in bacteria and archaea. Nature 2012, 482, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Ishino, Y.; Shinagawa, H.; Makino, K.; Amemura, M.; Nakata, A. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J. Bacteriol. 1987, 169, 5429–5433. [Google Scholar] [CrossRef] [PubMed]
- Gasiunas, G.; Barrangou, R.; Horvath, P.; Siksnys, V. Cas9-crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc. Natl. Acad. Sci. USA 2012, 109, E2579–E2586. [Google Scholar] [CrossRef] [PubMed]
- Brouns, S.J.J.; Jore, M.M.; Lundgren, M.; Westra, E.R.; Slijkhuis, R.J.H.; Snijders, A.P.L.; Dickman, M.J.; Makarova, K.S.; Koonin, E.V.; van der Oost, J. Small CRISPR RNAs Guide Antiviral Defense in Prokaryotes. Science 2008, 321, 960–964. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Bui, M.; Yang, T.; White, B.J.; Akbari, O.S. Germline Cas9 Expression Yields Highly Efficient Genome Engineering in a Major Worldwide Disease Vector. Aedes Aegypti. bioRxiv 2017. [Google Scholar] [CrossRef]
- Smith, R.C.; Atkinson, P.W. Mobility properties of the Hermes transposable element in transgenic lines of Aedes aegypti. Genetica 2011, 139, 7–22. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.; Orsetti, J.; Klocko, A.D.; Aluvihare, C.; Peckham, E.; Atkinson, P.W.; Lehane, M.J.; O’Brochta, D.A. Post-integration behavior of a Mos1 mariner gene vector in Aedes aegypti. Insect Biochem. Mol. Biol. 2003, 33, 853–863. [Google Scholar] [CrossRef]
- Arensburger, P.; Kim, Y.-J.; Orsetti, J.; Aluvihare, C.; O’Brochta, D.A.; Atkinson, P.W. An active transposable element, Herves, from the African malaria mosquito Anopheles gambiae. Genetics 2005, 169, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Sethuraman, N.; Fraser, M.J.; Eggleston, P.; O’Brochta, D.A. Post-integration stability of piggyBac in Aedes aegypti. Insect Biochem. Mol. Biol. 2007, 37, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Rodicio, M.R.; Chater, K.F. Small DNA-free liposomes stimulate transfection of streptomyces protoplasts. J. Bacteriol. 1982, 151, 1078–1085. [Google Scholar] [PubMed]
- Sellem, C.H.; Belcour, L. Intron open reading frames as mobile elements and evolution of a group I intron. Mol. Biol. Evol. 1997, 14, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Galizi, R.; Doyle, L.A.; Menichelli, M.; Bernardini, F.; Deredec, A.; Burt, A.; Stoddard, B.L.; Windbichler, N.; Crisanti, A. A synthetic sex ratio distortion system for the control of the human malaria mosquito. Nat. Commun. 2014, 5, 3977. [Google Scholar] [CrossRef] [PubMed]
- Roman, J.; Woodson, S.A. Integration of the Tetrahymena group I intron into bacterial rRNA by reverse splicing in vivo. Proc. Natl. Acad. Sci. USA 1998, 95, 2134–2139. [Google Scholar] [CrossRef] [PubMed]
- Bernardini, F.; Galizi, R.; Menichelli, M.; Papathanos, P.-A.; Dritsou, V.; Marois, E.; Crisanti, A.; Windbichler, N. Site-specific genetic engineering of the Anopheles gambiae Y chromosome. Proc. Natl. Acad. Sci. USA 2014, 111, 7600–7605. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.; Corfas, R.; Matthews, B.; Ritchie, S. Multimodal integration of carbon dioxide and other sensory cues drives mosquito attraction to humans. Cell 2014, 156, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Liesch, J.; Bellani, L.L.; Vosshall, L.B.; Riehle, M.; Brown, M. Functional and Genetic Characterization of Neuropeptide Y-Like Receptors in Aedes aegypti. PLoS Negl. Trop. Dis. 2013, 7, e2486. [Google Scholar] [CrossRef] [PubMed]
- Bibikova, M.; Beumer, K.; Trautman, J.K.; Carroll, D. Enhancing gene targeting with designed zinc finger nucleases. Science 2003, 300, 764. [Google Scholar] [CrossRef] [PubMed]
- Aryan, A.; Anderson, M.A.E.; Myles, K.M.; Adelman, Z.N. TALEN-based gene disruption in the dengue vector Aedes aegypti. PLoS ONE 2013, 8, e60082. [Google Scholar] [CrossRef] [PubMed]
- Kohno, H.; Suenami, S.; Takeuchi, H.; Sasaki, T.; Kubo, T. Production of Knockout Mutants by CRISPR/Cas9 in the European Honeybee, Apis mellifera L. Zool. Sci. 2016, 33, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, G.; Zhu, Y.-N.; Xiang, H.; Wang, W. Advances and perspectives in the application of CRISPR/Cas9 in insects. Zool. Res. 2016, 37, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Liu, Y.; Ai, D.; Jiang, X.; Dong, S.; Wang, G. A Pheromone Antagonist Regulates Optimal Mating Time in the Moth Helicoverpa armigera. Curr. Biol. 2017, 27, 1610–1615. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Ren, M.; Wang, Z.; Zhang, B.; Rong, Y.S.; Jiao, R.; Gao, G. Highly Efficient Genome Modifications Mediated by CRISPR/Cas9 in Drosophila. Genetics 2013, 195, 289–291. [Google Scholar] [CrossRef] [PubMed]
- Ishizu, H.; Iwasaki, Y.W.; Hirakata, S.; Ozaki, H.; Iwasaki, W.; Siomi, H.; Siomi, M.C. Somatic Primary piRNA Biogenesis Driven by cis-Acting RNA Elements and trans-Acting Yb. Cell Rep. 2015, 12, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Gratz, S.J.; Ukken, F.P.; Rubinstein, C.D.; Thiede, G.; Donohue, L.K.; Cummings, A.M.; O’Connor-Giles, K.M. Highly specific and efficient CRISPR/Cas9-catalyzed homology-directed repair in Drosophila. Genetics 2014, 196, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Bassett, A.R.; Tibbit, C.; Ponting, C.P.; Liu, J.-L. Highly efficient targeted mutagenesis of Drosophila with the CRISPR/Cas9 system. Cell Rep. 2013, 4, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Sebo, Z.L.; Lee, H.B.; Peng, Y.; Guo, Y. A simplified and efficient germline-specific CRISPR/Cas9 system for Drosophila genomic engineering. Fly 2014, 8, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Awata, H.; Watanabe, T.; Hamanaka, Y.; Mito, T.; Noji, S.; Mizunami, M. Knockout crickets for the study of learning and memory: Dopamine receptor Dop1 mediates aversive but not appetitive reinforcement in crickets. Sci. Rep. 2015, 5, 15885. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, J.; Chen, D.; Yang, P.; Jiang, F.; Wang, X.; Kang, L. CRISPR/Cas9 in locusts: Successful establishment of an olfactory deficiency line by targeting the mutagenesis of an odorant receptor co-receptor (Orco). Insect Biochem. Mol. Biol. 2016, 79, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Heinze, S.D.; Kohlbrenner, T.; Ippolito, D.; Meccariello, A.; Burger, A.; Mosimann, C.; Saccone, G.; Bopp, D. CRISPR-Cas9 targeted disruption of the yellow ortholog in the housefly identifies the brown body locus. Sci. Rep. 2017, 7, 4582. [Google Scholar] [CrossRef] [PubMed]
- Gilles, A.F.; Schinko, J.B.; Averof, M. Efficient CRISPR-mediated gene targeting and transgene replacement in the beetle Tribolium castaneum. Development 2015, 142, 2832–2839. [Google Scholar] [CrossRef] [PubMed]
- Afrane, Y.A.; Bonizzoni, M.; Yan, G. Secondary Malaria Vectors of Sub-Saharan Africa: Threat to Malaria Elimination on the Continent? Curr. Top. Malar. 2016. [Google Scholar] [CrossRef]
- Alphey, L.; Beard, C.B.; Billingsley, P.; Coetzee, M.; Crisanti, A.; Curtis, C.; Eggleston, P.; Godfray, C.; Hemingway, J.; Jacobs-Lorena, M.; et al. Malaria control with genetically manipulated insect vectors. Science 2002, 298, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Harbach, R.E. The Phylogeny and Classification of Anopheles. In Anopheles Mosquitoes—New Insights into Malaria Vectors; InTech: London, UK, 2013. [Google Scholar]
- Service, M.W.; Townson, H. The Anopheles vector. Essent. Malariol. 2002, 4, 59–84. [Google Scholar]
- Patterson, R.S.; Weidhaas, D.E.; Ford, H.R.; Lofgren, C.S. Suppression and Elimination of an Island Population of Culex pipiens quinquefasciatus with Sterile Males. Science 1970, 168, 1368–1369. [Google Scholar] [CrossRef] [PubMed]
- Asman, S.M.; McDonald, P.T.; Prout, T. Field Studies of Genetic Control Systems for Mosquitoes. Annu. Rev. Entomol. 1981, 26, 289–318. [Google Scholar] [CrossRef] [PubMed]
- Weidhaas, D.; Schmidt, C.; Seabrook, E. Field studies on the release of sterile males for the control of Anopheles quadrimaculatus. Mosq. News 1962, 22, 283–291. [Google Scholar]
- Morlan, H.B.; McCray, E.M., Jr.; Kilpatrick, J.W. Field Tests with Sexually Sterile Males for control of Aedes aegypti. Mosq. News 1962, 22, 295–300. [Google Scholar]
- Krishnamurthy, B.S.; Ray, S.N.; Joshi, G. A note on preliminary field studies of the use of irradiated males for reduction of C. fatigans Wied populations. Indian J. Malariol. 1962, 4, 365–373. [Google Scholar]
- Spielman, A. Why entomological antimalaria research should not focus on transgenic mosquitoes. Parasitol. Today 1994, 10, 374–376. [Google Scholar] [CrossRef]
- Benedict, M.Q.; Robinson, A.S. The first releases of transgenic mosquitoes: An argument for the sterile insect technique. Trends Parasitol. 2003, 19, 349–355. [Google Scholar] [CrossRef]
- Lambrechts, L.; Koella, J.C.; Boëte, C. Can transgenic mosquitoes afford the fitness cost? Trends Parasitol. 2008, 24, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L.; Benedict, M.; Bellini, R.; Clark, G.G.; Dame, D.A.; Service, M.W.; Dobson, S.L. Sterile-insect methods for control of mosquito-borne diseases: An analysis. Vector Borne Zoonotic Dis. 2010, 10, 295–311. [Google Scholar] [CrossRef] [PubMed]
- Fitz-Earle, M.; Holm, D.G.; Suzuki, D.T. Genetic control of insect populations: I. Cage studies of chromsome replacement by compound autosomes in Drosophila melanogaster. Genetics 1973, 74, 461–475. [Google Scholar] [PubMed]
- James, A. Control of Disease Transmission through Genetic Modification of Mosquitoes. In Insect Transgenesis: Methods and Applications; Handler, A.M., James, A.A., Eds.; CRC Press: Boca Raton, FL, USA, 2000; pp. 319–332. [Google Scholar]
- Rasgon, J.L.; Scott, T.W. Crimson: A Novel Sex-Linked Eye Color Mutant of Culex pipiens L. (Diptera: Culicidae). J. Med. Entomol. 2004, 41, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Dame, D.A.; Curtis, C.F.; Benedict, M.Q.; Robinson, A.S.; Knols, B.G.J. Historical applications of induced sterilisation in field populations of mosquitoes. Malar. J. 2009, 8, S2. [Google Scholar] [CrossRef] [PubMed]
- Fried, M. Determination of Sterile-Insect Competitiveness. J. Econ. Entomol. 1971, 64, 869–872. [Google Scholar] [CrossRef]
- Spielman, A.; Kitron, U.; Pollack, R.J. Time limitation and the role of research in the worldwide attempt to eradicate malaria. J. Med. Entomol. 1993, 30, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Eckhoff, P.A.; Wenger, E.A.; Godfray, H.C.J.; Burt, A. Impact of mosquito gene drive on malaria elimination in a computational model with explicit spatial and temporal dynamics. Proc. Natl. Acad. Sci. USA 2017, 114, E255–E264. [Google Scholar] [CrossRef] [PubMed]
- Unckless, R.L.; Messer, P.W.; Connallon, T.; Clark, A.G. Modeling the manipulation of natural populations by the mutagenic Chain reaction. Genetics 2015, 201, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Unckless, R.L.; Clark, A.G.; Messer, P.W. Evolution of resistance against CRISPR/Cas9 gene drive. Genetics 2017, 205, 827–841. [Google Scholar] [CrossRef] [PubMed]
- Robert, M.A.; Okamoto, K.W.; Gould, F.; Lloyd, A.L. Antipathogen genes and the replacement of disease-vectoring mosquito populations: A model-based evaluation. Evol. Appl. 2014, 7, 1238–1251. [Google Scholar] [CrossRef] [PubMed]
- WHO Global Programme to Eliminate Lymphatic Filariasis: Progress Report, 2014. Available online: http://www.who.int/lymphatic_filariasis/resources/who_wer9038/en/ (accessed on 3 August 2017).
- Thomas, M.B.; Godfray, H.C.J.; Read, A.F.; van den Berg, H.; Tabashnik, B.E.; van Lenteren, J.C.; Waage, J.K.; Takken, W. Lessons from Agriculture for the Sustainable Management of Malaria Vectors. PLoS Med. 2012, 9, e1001262. [Google Scholar] [CrossRef] [PubMed]
- Bill and Melinda Gates Foundation. From Aspiration to Action. Available online: https://www.mmv.org/sites/default/files/uploads/docs/publications/Aspiration-to-Action.pdf (accessed on 3 August 2017).
- Barreaux, P.; Barreaux, A.; Sternberg, E.; Suh, E. Priorities for Broadening the Malaria Vector Control Tool Kit. Trends Parasitol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Jeffries, C.; Walker, T. Biological Control of Mosquito Vectors: Past, Present, and Future. Insects 2016, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Verily. Available online: https://verily.com/projects/interventions/debug/ (accessed on 1 January 2017).
- Eliminate Dengue Program. Available online: http://www.eliminatedengue.com/project (accessed on 1 January 2017).
- Shenoy, R.K.; Suma, T.K.; Kumaraswami, V.; Rahmah, N.; Dhananjayan, G.; Padma, S. Antifilarial drugs, in the doses employed in mass drug administrations by the Global Programme to Eliminate Lymphatic Filariasis, reverse lymphatic pathology in children with Brugia malayi infection. Ann. Trop. Med. Parasitol. 2009, 103, 235–247. [Google Scholar] [CrossRef] [PubMed]
- White, M.T.; Griffin, J.T.; Churcher, T.S.; Ferguson, N.M.; Basáñez, M.-G.; Ghani, A.C. Modelling the impact of vector control interventions on Anopheles gambiae population dynamics. Parasit. Vectors 2011, 4, 153. [Google Scholar] [CrossRef] [PubMed]
- Fillinger, U.; Ndenga, B.; Githeko, A.; Lindsay, S.W. Integrated malaria vector control with microbial larvicides and insecticide-treated nets in western Kenya: A controlled trial. Bull. World Health Organ. 2009, 87, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Deredec, A.; Burt, A.; Godfray, H.C.J. The Population Genetics of Using Homing Endonuclease Genes in Vector and Pest Management. Genetics 2008, 179, 2013–2026. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.M.; Buchman, A.; Sanchez, C.H.M.; Akbari, O.S. Overcoming evolved resistance to population-suppressing homing-based gene drives. Sci. Rep. 2017, 7, 3776. [Google Scholar] [CrossRef] [PubMed]
- Noble, C.; Olejarz, J.; Esvelt, K.M.; Church, G.M.; Nowak, M.A. Evolutionary dynamics of CRISPR gene drives. Sci. Adv. 2017, 3, e1601964. [Google Scholar] [CrossRef] [PubMed]
- Champer, J.; Liu, J.; Oh, S.Y.; Reeves, R.; Luthra, A.; Oakes, N.; Clark, A.G.; Messer, P.W. Reducing resistance allele formation in CRISPR gene drives. bioRxiv 2017. [Google Scholar] [CrossRef]
- Esvelt, K.M.; Smidler, A.L.; Catteruccia, F.; Church, G.M. Concerning RNA-guided gene drives for the alteration of wild populations. Elife 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhao, Y. Self-processing of ribozyme-flanked RNAs into guide RNAs in vitro and in vivo for CRISPR-mediated genome editing. J. Integr. Plant Biol. 2014, 56, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Port, F.; Bullock, S.L. Augmenting CRISPR applications in Drosophila with tRNA-flanked sgRNAs. Nat. Methods 2016, 13, 852–854. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, S.; Fujii, W.; Ogawa, T.; Sugiura, K.; Naito, K. Development of a mono-promoter-driven CRISPR/Cas9 system in mammalian cells. Sci. Rep. 2016, 5, 18341. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhou, G.; Wu, J.; Bian, G.; Lu, P.; Raikhel, A.S.; Xi, Z. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar] [CrossRef] [PubMed]
- Rancès, E.; Ye, Y.H.; Woolfit, M.; McGraw, E.A.; O’Neill, S.L. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLoS Pathog. 2012, 8, e1002548. [Google Scholar] [CrossRef] [PubMed]
- Calvitti, M.; Marini, F.; Desiderio, A.; Puggioli, A.; Moretti, R. Wolbachia density and cytoplasmic incompatibility in Aedes albopictus: Concerns with using artificial Wolbachia infection as a vector suppression tool. PLoS ONE 2015, 10, e0121813. [Google Scholar] [CrossRef] [PubMed]
- Akbari, O.S.; Antoshechkin, I.; Amrhein, H.; Williams, B.; Diloreto, R.; Sandler, J.; Hay, B.A. The Developmental Transcriptome of the Mosquito Aedes aegypti, an Invasive Species and Major Arbovirus Vector. G3 (Bethesda) 2013, 3, 1493–1509. [Google Scholar] [CrossRef] [PubMed]
- Macias, V.; Coleman, J.; Bonizzoni, M.; James, A.A. piRNA pathway gene expression in the malaria vector mosquito Anopheles stephensi. Insect Mol. Biol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Black, W.C.; Hess, A.M.; Foy, B.D. Comparative genomics of small RNA regulatory pathway components in vector mosquitoes. BMC Genom. 2008, 9, 425. [Google Scholar] [CrossRef] [PubMed]
- Ronsseray, S.; Lehmann, M.; Anxolabéhère, D. The Maternally Inherited Regulation of P Elements in Drosophila melanogaster Can Be Elicited by Two P Copies at Cytological Site 1A on the X Chromosome. Genet. Soc. Am. 1991, 129, 501–512. [Google Scholar]
- Kawaoka, S.; Mitsutake, H.; Kiuchi, T.; Kobayashi, M.; Yoshikawa, M.; Suzuki, Y.; Sugano, S.; Shimada, T.; Kobayashi, J.; Tomari, Y.; et al. A role for transcription from a piRNA cluster in de novo piRNA production. RNA 2012, 18, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Olovnikov, I.; Ryazansky, S.; Shpiz, S.; Lavrov, S.; Abramov, Y.; Vaury, C.; Jensen, S.; Kalmykova, A. De novo piRNA cluster formation in the Drosophila germ line triggered by transgenes containing a transcribed transposon fragment. Nucleic Acids Res. 2013, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Shpiz, S.; Ryazansky, S.; Olovnikov, I.; Abramov, Y.; Kalmykova, A. Euchromatic transposon insertions trigger production of novel Pi- and endo-siRNAs at the target sites in the Drosophila germline. PLoS Genet. 2014, 10, e1004138. [Google Scholar] [CrossRef] [PubMed]
- Le Thomas, A.; Stuwe, E.; Li, S.; Du, J.; Marinov, G.; Rozhkov, N.; Chen, Y.C.; Luo, Y.; Sachidanandam, R.; Toth, K.F.; et al. Transgenerationally inherited piRNAs trigger piRNA biogenesis by changing the chromatin of piRNA clusters and inducing precursor processing. Genes 2014, 28, 1167–1180. [Google Scholar] [CrossRef] [PubMed]
- Boivin, A.; Gally, C.; Netter, S.; Anxolabéhère, D.; Ronsseray, S. Telomeric associated sequences of Drosophila recruit polycomb-group proteins in vivo and can induce pairing-sensitive repression. Genetics 2003, 164, 195–208. [Google Scholar] [PubMed]
- Cho, S.W.; Kim, S.; Kim, Y.; Kweon, J.; Kim, H.S.; Bae, S.; Kim, J.-S. Analysis of off-target effects of CRISPR/Cas-derived RNA-guided endonucleases and nickases. Genome Res. 2014, 24, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.; Kazuno, A.-A.; Kelsoe, J.; Nakanishi, M.; Takumi, T.; Kato, T. Exome sequencing in the knockin mice generated using the CRISPR/Cas system. Sci. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
- IGSR: The International Genome Sample Resource. Available online: http://www.internationalgenome.org/ (accessed on 1 January 2017).
- Hwang, W.Y.; Fu, Y.; Reyon, D.; Maeder, M.L.; Tsai, S.Q.; Sander, J.D.; Peterson, R.T.; Yeh, J.-R.J.; Joung, J.K. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Yang, Z.; Xu, J.; Sun, J.; Mao, D.; Hu, Y.; Yang, S.-J.; Qiao, H.-H.; Wang, X.; Hu, Q.; et al. Enhanced Specificity and Efficiency of the CRISPR/Cas9 System with Optimized sgRNA Parameters in Drosophila. Cell Rep. 2014, 9, 1151–1162. [Google Scholar] [CrossRef] [PubMed]
- Crosetto, N.; Mitra, A.; Silva, M.J.; Bienko, M.; Dojer, N.; Wang, Q.; Karaca, E.; Chiarle, R.; Skrzypczak, M.; Ginalski, K.; et al. Nucleotide-resolution DNA double-strand break mapping by next-generation sequencing. Nat. Methods 2013, 10, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Mali, P.; Aach, J.; Stranges, P.B.; Esvelt, K.M.; Moosburner, M.; Kosuri, S.; Yang, L.; Church, G.M. Cas9 transcriptional activators for target specificity screening and paired nickases for cooperative genome engineering. Nat. Biotechnol. 2013, 31, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.X.; Mirzazadeh, R.; Garnerone, S.; Scott, D.; Schneider, M.W.; Kallas, T.; Custodio, J.; Wernersson, E.; Li, Y.; Gao, L.; et al. BLISS is a versatile and quantitative method for genome-wide profiling of DNA double-strand breaks. Nat. Commun. 2017, 8, 15058. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.; Scott, D.; Weinstein, J.; Ran, F. DNA targeting specificity of RNA-guided Cas9 nucleases. Nature 2013. [Google Scholar] [CrossRef] [PubMed]
- Iyer, V.; Shen, B.; Zhang, W.; Hodgkins, A.; Keane, T.; Huang, X.; Skarnes, W.C. Off-target mutations are rare in Cas9-modified mice. Nat. Methods 2015. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, K.A.; Wu, W.-H.; Colgan, D.F.; Tsang, S.H.; Bassuk, A.G.; Mahajan, V.B. Unexpected mutations after CRISPR-Cas9 editing in vivo. Nat. Methods 2016. [Google Scholar] [CrossRef] [PubMed]
- Zetsche, B.; Gootenberg, J.S.; Abudayyeh, O.O.; Slaymaker, I.M.; Makarova, K.S.; Essletzbichler, P.; Volz, S.E.; Joung, J.; van der Oost, J.; Regev, A.; et al. Cpf1 Is a Single RNA-Guided Endonuclease of a Class 2 CRISPR-Cas System. Cell 2015, 163, 759–771. [Google Scholar] [CrossRef] [PubMed]
- Kleinstiver, B.P.; Pattanayak, V.; Prew, M.S.; Tsai, S.Q.; Nguyen, N.T.; Zheng, Z.; Joung, J.K. High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 2016, 529, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Slaymaker, I.M.; Gao, L.; Zetsche, B.; Scott, D.A.; Yan, W.X.; Zhang, F. Rationally engineered Cas9 nucleases with improved specificity. Science 2016, 351, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Ran, F.A.; Hsu, P.D.; Lin, C.-Y.; Gootenberg, J.S.; Konermann, S.; Trevino, A.E.; Scott, D.A.; Inoue, A.; Matoba, S.; Zhang, Y.; et al. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 2013, 154, 1380–1389. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Sandler, J.D.; Reyon, D.; Cascio, V.M.; Joung, J.K. Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat. Biotechnol. 2014, 32, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Pattanayak, V.; Lin, S.; Guilinger, J.P.; Ma, E.; Doudna, J.A.; Liu, D.R. High-throughput profiling of off-target DNA cleavage reveals RNA-programmed Cas9 nuclease specificity. Nat. Biotechnol. 2013, 31, 839–842. [Google Scholar] [CrossRef] [PubMed]
- Haeussler, M.; Schönig, K.; Eckert, H.; Eschstruth, A.; Mianné, J.; Renaud, J.-B.; Schneider-Maunoury, S.; Shkumatava, A.; Teboul, L.; Kent, J.; et al. Evaluation of off-target and on-target scoring algorithms and integration into the guide RNA selection tool CRISPOR. Genome Biol. 2016, 17, 148. [Google Scholar] [CrossRef] [PubMed]
- Oliveros, J.C.; Franch, M.; Tabas-Madrid, D.; San-León, D.; Montoliu, L.; Cubas, P.; Pazos, F. Breaking-Cas—interactive design of guide RNAs for CRISPR-Cas experiments for ENSEMBL genomes. Nucleic Acids Res. 2016, 44, W267–W271. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.M. The effect of gene drive on containment of transgenic mosquitoes. J. Theor. Biol. 2009, 258, 250–265. [Google Scholar] [CrossRef] [PubMed]
- Akbari, O.S.; Bellen, H.J.; Bier, E.; Bullock, S.L.; Burt, A.; Church, G.M.; Cook, K.R.; Duchek, P.; Edwards, O.R.; Esvelt, K.M.; et al. Safeguarding gene drive experiments in the laboratory. Science 2015, 349, 927–929. [Google Scholar] [CrossRef] [PubMed]
- DiCarlo, J.E.; Chavez, A.; Dietz, S.L.; Esvelt, K.M.; Church, G.M. Safeguarding CRISPR-Cas9 gene drives in yeast. Nat. Biotechnol. 2015, 33, 1250–1255. [Google Scholar] [CrossRef] [PubMed]
- Oye, K.A.; Esvelt, K.; Appleton, E.; Catteruccia, F.; Church, G.; Kuiken, T.; Lightfoot, S.B.-Y.; McNamara, J.; Smidler, A.; Collins, J.P. Regulating gene drives. Science 2014, 345, 626–628. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Luo, L.; Gao, X.J. Cas9-triggered chain ablation of cas9 as a gene drive brake. Nat. Biotechnol. 2016, 34, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Noble, C.; Min, J.; Olejarz, J.; Buchthal, J.; Chavez, A.; Smidler, A.L.; DeBenedictis, E.A.; Church, G.M.; Nowak, M.A.; Esvelt, K.M. Daisy-chain gene drives for the alteration of local populations. bioRxiv 2016. [Google Scholar] [CrossRef]
- Mukundarajan, H.; Hol, F.J.H.; Castillo, E.A.; Newby, C.; Prakash, M. Using Mobile Phones as Acoustic Sensors for High-Throughput Surveillance of Mosquito Ecology. bioRxiv 2017. [Google Scholar] [CrossRef]
- Grubaugh, N.D.; Sharma, S.; Krajacich, B.J.; Fakoli, L.S., III; Bolay, F.K.; Diclaro, J.W., II; Johnson, W.E.; Ebel, G.D.; Foy, B.D.; Brackney, D.E. Xenosurveillance: A Novel Mosquito-Based Approach for Examining the Human-Pathogen Landscape. PLoS Negl. Trop. Dis. 2015, 9, e0003628. [Google Scholar] [CrossRef] [PubMed]
- Oxitec. Available online: http://www.oxitec.com/ (accessed on 1 January 2017).
- Munhenga, G.; Brooke, B.D.; Spillings, B.; Essop, L.; Hunt, R.H.; Midzi, S.; Govender, D.; Braack, L.; Koekemoer, L.L. Field study site selection, species abundance and monthly distribution of anopheline mosquitoes in northern Kruger National Park, South Africa. Malar. J. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Ageep, T.B.; Damiens, D.; Alsharif, B.; Ahmed, A.; Salih, E.H.; Ahmed, F.T.; Diabaté, A.; Lees, R.S.; Gilles, J.R.; El Sayed, B.B. Participation of irradiated Anopheles arabiensis males in swarms following field release in Sudan. Malar. J. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Lees, R.S.; Gilles, J.R.; Hendrichs, J.; Vreysen, M.J.; Bourtzis, K.; KBourtzis, K. Back to the future: The sterile insect technique against mosquito disease vectors. Curr. Opin. Insect Sci. 2015, 10, 156–162. [Google Scholar] [CrossRef]
- VectorBase: Population Biology. Available online: https://www.vectorbase.org/popbio (accessed on 1 January 2017).
- Pigott, D.M.; Kraemer, M.U. Enhancing Infectious Disease Mapping with Open Access Resources. Available online: http://www.eurosurveillance.org/images/dynamic/EE/V19N49/art20989.pdf (accessed on 11 December 2014).
- Malaria Atlas Project. Available online: http://www.map.ox.ac.uk/explorer/ (accessed on 1 January 2017).
| Genetic Techniques | ||||||
|---|---|---|---|---|---|---|
| Transposons | ϕC31 | HEGs | ZFNs | TALENs | CRISPR/Cas9 | |
| Origin and biological basis | Transposable elements discovered in the 1950's in maize, specifically DNA (type II) transposons. | ϕC31 transposase-mediated integration of bacteriophage DNA into bacterial genomes | Group I introns discovered prior to 1970 in yeast that show higher than Mendelian inheritance proportions | Zinc-binding domain from transcription factors for DNA recognition, FokI endonuclease for DNA cleavage, first fused for site-specific DNA cleavage in 1996 | TALE proteins discovered in Xanthomonas species were decoded in 2009 and conjugated to FokI endonuclease | S. pyogenes CRISPR/Cas9 biology for destruction and memory of parasitic nucleic acid |
| Initial use for genome modification | 1982 (D. melanogaster) | 1982 (S. pyogenes) | 1998 (E.coli) | 2001 (Xenopus laevis) | 2010 (S. cerevisiae) | 2012 (Human cell lines) |
| Year used in mosquitoes | 1998 | 2006 | 2011 | 2013 | 2013 | 2015 |
| Mechanism | Transposase mediated transfer of DNA between ITRs either from a plasmid into the mosquito genome (transformation) or from one place in a genome to another | ϕC31 integrase mediates integration of a plasmid bearing an attB site into a complementary attP site on the mosquito genome | HEG encoded endonuclease recognizes and cleaves genomic DNA such that a gene cassette can be integrated by cell HDR machinery | Pairs of zinc-finger domains recognize and bind a sequence of nucleotide triplets, each endonuclease cuts the DNA backbone, together creating a double-stranded break | Pairs of TALE domains recognize and bind a sequence of nucleotides, each endonuclease cuts DNA backbone, together creating a double-stranded break | Cas9 protein bound to a sgRNA through scaffold sequence on the sgRNA recognizes a genomic sequence that is complementary to ~20 nucleotides on the sgRNA |
| Mutation efficiency * | NA | NA | 1–9% | <7% | <16% | >90% |
| Transformation efficiency * | ~2% | 10–18% | <1% | <6% | ~2% | ~2% |
| Gene drive * | Potentially | No | Yes | No | No | Yes |
| Drive efficiency | <<0.05% | NA | 56% | NA | NA | 90–100% |
| Benefits | First available transformation technique in mosquitoes, some natural transposons are extremely efficient gene drives. | Insertion into a known site | Insertion into a known site, high efficiency, recognition of sites on X-chromosome only in An. gambiae | Site-specific editing. Double stranded cuts allowed targeted mutagenesis-first gene editing technique for reverse genetics | Site-specific editing, efficient mutagenesis, TALEN expression plasmids could be cloned in-house. | Site-specific, easily re-engineered, adaptable to different species. Highly efficient for mutagenesis and as a drive. |
| Drawbacks | Random insertion, low transformation efficiency, very low mobility of synthetic transposon once integrated into the genome | Requires pre-insertion of the attP site using transposition | Requires pre-existing target-sites, re-engineering of the HEG, or transgenesis for insertion of target sites. Drive mechanism generates drive-resistant alleles. | Expensive, requires modular protein engineering, some codons were not recognized by any zinc-fingers, requires in vitro optimization | Requires protein engineering, timing: introduced just before Cas9 was demonstrated for gene-editing | Drive mechanism generates drive-resistant alleles |
| References | [40,42,43,44,45,46,47,48,49,50,57,173,174,175,176] | [50,132,142,143,177] | [73,75,79,152,178,179,180,181] | [144,145,146,147,154,182,183] | [148,150,153,159,184,185] | [156,159,160,161,162,163,164,166,167,169,170,171,172] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macias, V.M.; Ohm, J.R.; Rasgon, J.L. Gene Drive for Mosquito Control: Where Did It Come from and Where Are We Headed? Int. J. Environ. Res. Public Health 2017, 14, 1006. https://doi.org/10.3390/ijerph14091006
Macias VM, Ohm JR, Rasgon JL. Gene Drive for Mosquito Control: Where Did It Come from and Where Are We Headed? International Journal of Environmental Research and Public Health. 2017; 14(9):1006. https://doi.org/10.3390/ijerph14091006
Chicago/Turabian StyleMacias, Vanessa M., Johanna R. Ohm, and Jason L. Rasgon. 2017. "Gene Drive for Mosquito Control: Where Did It Come from and Where Are We Headed?" International Journal of Environmental Research and Public Health 14, no. 9: 1006. https://doi.org/10.3390/ijerph14091006
APA StyleMacias, V. M., Ohm, J. R., & Rasgon, J. L. (2017). Gene Drive for Mosquito Control: Where Did It Come from and Where Are We Headed? International Journal of Environmental Research and Public Health, 14(9), 1006. https://doi.org/10.3390/ijerph14091006




