Uranium and Associated Heavy Metals in Ovis aries in a Mining Impacted Area in Northwestern New Mexico
Abstract
:1. Introduction
Study Purpose and Aims
2. Materials and Methods
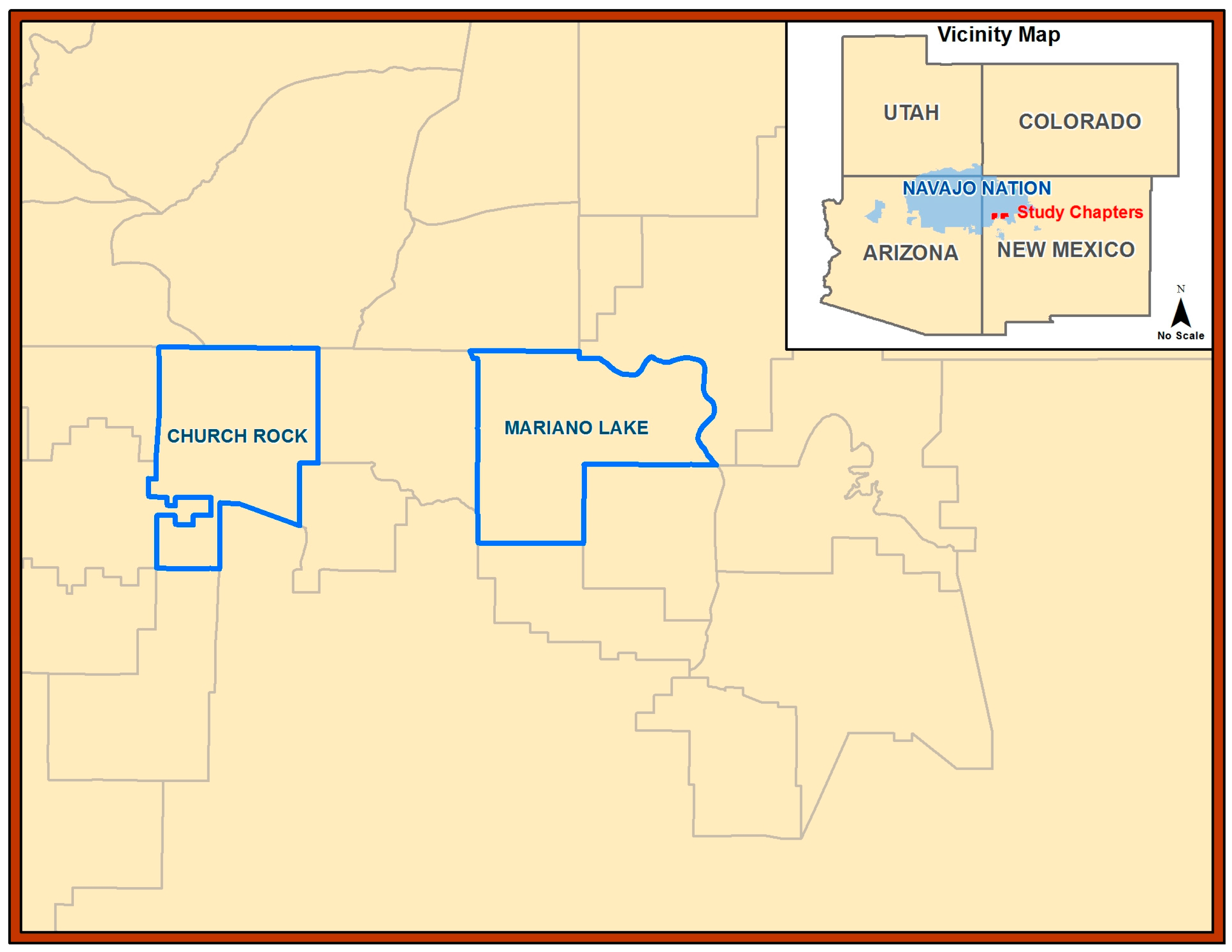
2.1. Study Setting
2.2. O. aries Tissue Samples
2.3. Soil Samples
2.4. Biota Samples
2.5. Water Samples
2.6. Statistical Analysis
2.7. Sample Preparation and Analysis
3. Results
3.1. Human Harvester Questionnaire
3.2. Sheep 1
3.3. Sheep 2
3.4. Sheep 3
3.5. Sheep Forage
4. Discussion
4.1. Implications for Human Intake
4.2. Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Santiago-Rivera, A.L.; Morse, G.S.; Haase, R.F.; McCaffrey, R.J.; Tarbell, A. Exposure to an environmental toxin, quality of life and psychological distress. J. Environ. Psychol. 2007, 27, 33–43. [Google Scholar] [CrossRef]
- McLemore, V.T. Uranium Industry in New Mexico—History, Production and Present Status; New Mexico Bureau of Geology & Mineral Resources: Socorro, NM, USA, 1983; Volume 5, pp. 45–51. [Google Scholar]
- Brugge, D.; Panikkar, B. The ethical issues in uranium mining research in the Navajo Nation. Acc. Res. 2007, 12, 121–153. [Google Scholar]
- Hund, L.; Bedrick, E.J.; Miller, C.; Huerta, G.; Nez, T.; Ramone, S.; Shuey, C.; Cajero, M.; Lewis, J. A Bayesian framework for estimating disease risk due to exposure to uranium mine and mill waste on the Navajo Nation. J. R. Stat. Soc. A 2015, 178, 1069–1091. [Google Scholar] [CrossRef]
- Taylor, D.M.; Taylor, S.K. Environmental uranium and human health. Rev. Environ. Health 1997, 12, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Gilman, A.P.; Villenueve, D.C.; Secours, V.E.; Yagminas, A.P.; Tracy, B.L.; Quinn, J.M.; Valli, V.E.; Willes, R.J.; Moss, M.A. Uranyl nitrate: 28-day and 91-day toxicity studies in the Sprague-Dawley rat. Toxicol. Sci. 1998, 41, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Tracy, B.L.; Quinn, J.M.; Lahey, J.; Gilman, A.P.; Mancuso, K.; Yagminas, A.P.; Villenueve, D.C. Absorption and retention of uranium from drinking water by rats and rabbits. Health Phys. 1992, 62, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Dang, H.S.; Pullat, V.R.; Sharma, R.C. Distribution of uranium in human organs of an urban Indian population and its relationship with clearance half-lives. Health Phys. 1995, 68, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Eisler, R. Arsenic hazards to fish, wildlife, and invertebrates: A synoptic view. U. S. Fish Wildl. Serv. Biol. Rep. 1988, 85, 12. [Google Scholar]
- Kirkam, M.B. Cadmium in plants on polluted soils: Effects of soil factors, hyperaccumulation, and amendments. Geoderma 2006, 137, 19–32. [Google Scholar] [CrossRef]
- Caldas, E.D.; Machado, L.L. Cadmium, mercury and lead in medicinal herbs in Brazil. Food Chem. Toxicol. 2004, 42, 599–603. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, M.J.; Parker, D.R.; Clark, J.M. Metals and micronutrients-food safety issues. Field Crops Res. 1999, 60, 143–163. [Google Scholar] [CrossRef]
- Navarro-Alarcon, M.; Cabrera-Vique, C. Selenium in food and the human body: A review. Sci. Total Environ. 2008, 400, 115–141. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Cann, C.I.; Calzolari, E.; Vivoli, R.; Garavelli, V.; Bergomi, M. Reproductive outcomes in a population exposed long-term to inorganic selenium via drinking water. Sci. Total Environ. 2000, 250, 1–7. [Google Scholar] [CrossRef]
- Meeker, J.D.; Rossano, M.G.; Protas, B.; Diamond, M.P.; Puscheck, E.; Daly, D.; Paneth, N.; Wirth, J.J. Cadmium, lead, and other metals in relation to semen quality: Human evidence for molybdenum as a male reproductive toxicant. Environ. Health Perspect. 2008, 116, 1473–1479. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Rossano, M.G.; Protas, B.; Padmanahban, V.; Diamond, M.P.; Puscheck, E.; Daly, D.; Paneth, N.; Wirth, J.J. Environmental exposure to metals and male reproductive hormones: Circulating testosterone is inversely associated with blood molybdenum. Fertil. Steril. 2010, 93, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.; Singh, S.P. Effects of molybdenum on fertility of male rats. BioMetals 2002, 15, 65–72. [Google Scholar] [CrossRef] [PubMed]
- De lemos, J.L.; Brugge, D.; Cajero, M.; Downs, M.; Durant, J.L.; George, C.M.; Henio-Adeky, S.; Nez, T.; Manning, T.; Rock, T.; et al. Development of risk maps to minimize uranium exposures in the Navajo Churchrock mining district. Environ. Health 2009, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, W.S.; Weber, C.W.; Dahozy-Arviso, K. Use and nutrient content of traditional Navajo foods. Ecol. Food Nutr. 1985, 17, 323–344. [Google Scholar] [CrossRef]
- Lapham, S.C.; Millard, J.B.; Samet, J.M. Health implications of radionuclide levels in cattle raised near U mining and milling facilities in Ambrosia Lake, New Mexico. Health Phys. 1989, 36, 327–340. [Google Scholar] [CrossRef]
- Millard, J.B.; Lapham, S.C.; Hahn, P.; Jere, B. Radionuclide Levels in Sheep and Cattle Grazing Near Uranium Mining and milling at Churchrock, NM; New Mexico Environmental Improvement Division: Santa Fe, NM, USA, 1986.
- Ruttenber, A.J.; Kreiss, K.; Douglas, R.L.; Buhl, T.E.; Millard, J. The assessment of human exposures to radionuclides from a uranium mill tailings release and mine dewatering effluent. Health Phys. 1984, 47, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Samuel-Nakamura, C.; Hodge, F.S.; Valentine, J.L.; Robbins, W.A. Heavy metal contamination in Thelesperma megapotamicum. J. Toxicol. Environ. Health Sci. 2017, 9, 14–22. [Google Scholar] [CrossRef]
- Wrenn, M.E.; Durbin, P.W.; Howard, B.; Lipsztein, J.; Rundo, J.; Still, E.T.; Willis, D.L. Metabolism of ingested U and Ra. Health Phys. 1985, 48, 601–633. [Google Scholar] [CrossRef] [PubMed]
- Carriere, M.; Avoscan, L.; Collins, R.; Carrot, F.; Khodja, H.; Ansoborlo, E.; Gouget, G. Influence of uranium speciation on normal rat kidney (NRK-52E) proximal cell cytotoxicity. Chem. Res. Toxicol. 2004, 17, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Smyth, J.B.A.; Wang, J.H.; Barlow, R.M.; Humphreys, D.J.; Robins, M.; Stodulski, J.B.J. Experimental acute selenium intoxication in lambs. J. Comp. Pathol. 1990, 102, 197–209. [Google Scholar] [CrossRef]
- Zachara, B.A.; Gromadzinska, J.; Wasowicz, W.; Zbrog, A. Red blood cell and plasma glutathione peroxidase activities and selenium concentration in patients with chronic kidney disease: A review. Acta Biochim. Pol. 2006, 53, 663–677. [Google Scholar] [PubMed]
- Navarro-Alarcon, M.; Lopez-Martinez, M.C. Essentiality of selenium in the human body: Relationship with different diseases. Sci. Total Environ. 2000, 249, 347–371. [Google Scholar] [CrossRef]
- Cristaldi, L.A.; McDowell, L.R.; Buergelt, C.D.; Davis, P.A.; Wilkinson, N.S.; Martin, F.G. Tolerance of inorganic selenium in wether sheep. Small Rumin. Res. 2006, 56, 205–213. [Google Scholar] [CrossRef]
- Pugh, D.G. Sheep Goat Medicine, 1st ed.; Saunders: Philadelphia, PA, USA, 2002. [Google Scholar]
- Zachara, B.A.; Mikolajczak, J.; Trafikowska, U. Effect of various dietary selenium (Se) intakes on tissue Se levels and Glutathione Peroxidase activities in lambs. J. Vet. Med. A 1993, 40, 310–318. [Google Scholar] [CrossRef]
- Dreesen, D.R.; Cokal, E.J. Plant uptake assay to determine bioavailability of inorganic contaminants. Water Air Soil Pollut. 1984, 22, 85–93. [Google Scholar] [CrossRef]
- Soudek, P.; Petrova, S.; Benesova, D.; Dvorakova, M.; Vanek, T. Uranium uptake by hydroponically cultivated crop plants. J. Environ. Radioact. 2011, 102, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Anke, M.; Seeber, O.; Muller, R.; Schafer, U.; Zerull, J. Uranium transfer in the food chain from soil to plants, animals and man. Chem. Erde 2009, 69, 75–90. [Google Scholar] [CrossRef]
- Stojanovic, M.; Stevanovic, D.; Illes, D.; Grubisic, M.; Milojkovic, J. The effect of uranium content in the tailings on some cultivated plants. Water Air Soil Pollut. 2009, 200, 101–108. [Google Scholar] [CrossRef]
- Shahandeh, H.; Hossner, L.R. Role of soil properties in phytoaccumulation of uranium. Water Air Soil Pollut. 2002, 141, 165–180. [Google Scholar] [CrossRef]
- Fries, G.F.; Paustenbach, D.J. Evaluation of potential transmission of 2,3,7,8-tetracholorodibenzo-p-dioxin-contaminated incinerator emissions to humans via food. J. Toxicol. Environ. Health 1990, 29, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Beyer, W.N.; Conner, E.E.; Gerould, S. Estimates of soil ingestion by wildlife. J. Wildl. Manag. 1994, 58, 375–382. [Google Scholar] [CrossRef]
- Cordova, F.J.; Wallace, J.D.; Pieper, R.D. Forage intake by grazing livestock: A review. J. Range Manag. 1978, 31, 430–438. [Google Scholar] [CrossRef]
- Gong, Y.; Hodgson, J.; Lambert, M.G.; Gordon, I.L. Short-term ingestive behavior of sheep and goats grazing grasses and legumes. 1. Comparison of bite weight, bite rate, and bite dimensions for forages at two stages of maturity. N. Z. J. Agric. Res. 1996, 39, 63–73. [Google Scholar] [CrossRef]
- De Lemos, J.L.; Bostick, B.C.; Quicksall, A.N.; Landis, J.D.; George, C.C.; Slagowski, N.L.; Rock, T.; Brugge, D.; Lewis, J.; Durant, J.L. Rapid dissolution of soluble uranyl phases in arid, mine-impacted catchments near Church Rock, NM. Environ. Sci. Technol. 2008, 42, 3951–3957. [Google Scholar] [CrossRef]
- National Research Council (NRC). Nutrient Requirements of Small Ruminants: Sheep, Goats, Cervids, and New World Camelids; The National Academy Press: Washington, DC, USA, 2007. [Google Scholar]
- National Research Council (NRC). Mineral Tolerance of Animals, 2nd ed.; The National Academy Press: Washington, DC, USA, 2005. [Google Scholar]
- World Health Organization (WHO). Environmental Health, Criteria 58-Selenium; World Health Organization: Geneva, Switzerland, 1987; pp. 91–223. [Google Scholar]
- Koenig, K.M.; Rode, L.M.; Cohen, H.; Buckley, W.T. Effects of diet and chemical form of selenium on selenium metabolism in sheep. J. Anim. Sci. 1997, 75, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Saha, U.; Fayiga, A.; Hancock, D.; Sonon, L. Selenium in animal nutrition: Deficiencies in soils and forages, requirements, supplementation and toxicity. IJAAS 2016, 2, 112–125. [Google Scholar] [CrossRef]
- Davis, T.Z.; Stegelmeier, B.L.; Welch, K.D.; Pfister, J.A.; Panter, K.E.; Hall, J.O. Comparative oral dose toxicokinetics of selenium compounds commonly found in selenium accumulator plants. J. Anim. Sci. 2014, 91, 4501–4509. [Google Scholar] [CrossRef] [PubMed]
- Beresford, N.A.; Howard, B.J. The importance of soil adhered to vegetation as a source of radionuclides ingested by grazing animals. Sci. Total Environ. 1991, 107, 237–254. [Google Scholar] [CrossRef]
- Jacobs, L.W. Selenium in Agriculture and the Environment; American Society of Agronomy Inc.: Madison, WI, USA, 1989. [Google Scholar]
- Lewis, J. DiNEH Study Water Quality Data. Unpublished work. 2017. [Google Scholar]
- Environmental Protection Agency (EPA). Drinking Water Standards and Health Advisories Table 2009. Available online: https://www3.epa.gov/region9/water/drinking/files/dwshat-v09.pdf (accessed on 15 May 2016).
- Navajo Nation Environmental Protection Agency (NNEPA). Navajo Nation Surface Water Quality Standards. Available online: https://www.epa.gov/sites/production/files/2014-12/documents/navajo-tribe.pdf (accessed on 6 January 2017).
- Tsuji, L.J.S.; Manson, H.; Wainman, B.C.; Vanspronsen, E.P.; Shecapio-Blacksmith, J.; Rabbitskin, T. Identifying potential receptors and routes of contaminant exposure in the traditional territory of the Ouje-Bougoumou Cree: Land use and a geographical information system. Environ. Monit. Assess. 2007, 127, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Ballew, C.; White, L.L.; Strauss, K.F.; Benson, L.J.; Mendlein, J.M.; Mokdad, A.H. Intake of nutrients and food sources among the Navajo: Findings from the Navajo health and nutrition survey. J. Nutr. 1997, 127, 2085S–2093S. [Google Scholar] [PubMed]
- Keys, A. Diet and the epidemiology of coronary heart disease. J. Am. Med. Assoc. 1957, 164, 1912–1919. [Google Scholar] [CrossRef] [PubMed]
- Joint Expert Committee on Food Additives (JECFA). Evaluation of Certain Food Additives and Contaminants. In Thirty-Third Report of the Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Expert Committee on Food Additives; WHO Technical Report Series No. 776; World Health Organization: Geneva, Switzerland, 1989. [Google Scholar]
- Joint Expert Committee on Food Additives (JECFA). Joint Expert Committee on Food Additives (JECFA). Joint FAO/WHO Expert Committee on food additives. In Sixty-First Meeting: Summary and Conclusions; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Food and Nutrition Board. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; The National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Food and Nutrition Board. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium and Carotenoids; The National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Chambers, J.C.; Sidle, R.C. Fate of heavy metals in an abandoned lead-zinc tailings pond: I. Vegetation. J. Environ. Qual. 1991, 20, 745–751. [Google Scholar] [CrossRef]
| Heavy Metal | Upper Limits ppm (μg/L) | Reference | EPA MCLs 1 μg/L |
|---|---|---|---|
| As | 0.2 ppm (200 μg/L) | Pugh, 2002 | 10 μg/L 1 |
| Cd | 0.10–0.05 ppm (10–50 μg/L) | Pugh, 2002 | 5 μg/L 1 |
| Pb | 0.05–1 ppm (50–1000 μg/L) | Pugh, 2002; NRC 2, 2005 | 15 μg/L 1,3 |
| Se | 0.05 ppm (50 μg/L) | Pugh, 2002 | 50 μg/L 1 |
| U | -- | -- | 30 μg/L 1,4 |
| Heavy Metal | Requirement 1 or Maximum Tolerable Concentrations 2 | Reference |
|---|---|---|
| As | MTC 2: 30 mg/kg of diet | NRC 3, 2005 |
| Cd | MTC 2: 10 mg/kg DM 4 diet | NRC 3, 2005; 2007 |
| Pb | MTC 2: 100 mg/kg diet DM 4 | NRC 3, 2005; 2007 |
| Mo | R 1: 0.10–0.5 3 mg/kg DM 4 | Pugh, 2002; NRC 3, 2007 |
| MTC 2: 5 mg/kg diet DM 4 | NRC 3, 2005; 2007 | |
| Se | R 1: 0.10–0.20 ppm (0.10–0.20 mg/kg) diet | Pugh, 2002 |
| Chronic ingestion: 0.25 mg/kg BW 5 | Pugh, 2002 | |
| MTC 2: 5 mg/kg DM 4 | NRC 3, 2005; 2007 |
| HM | Soil (Range) n = 8 | Shoot/Root (Range) n = 8/8 | BF 1 | Sheep Tissue n = 1 |
|---|---|---|---|---|
| Se | 0.91–1.85 | 0.87–1.37/1.38–1.45 | 0.85 | 3.93 Liver |
| 2.04/0.53 KM 2/KC 3 | ||||
| 1.30 Wool | ||||
| Cd | 0.05–0.07 | 0.05–0.08/0.06–0.11 | 1.16 | 0.06 Liver |
| 0.01 Intestine | ||||
| ng 4/0.01 KM 2/KC 3 | ||||
| As | 2.33–4.53 | 0.84–1.41/1.40–1.54 | 0.34 | 0.66 Wool |
| 0.09/0.08 KM 2/KC 3 | ||||
| 0.08 Bone | ||||
| Pb | 3.91–9.07 | 1.15–1.94/2.03–2.72 | 0.26 | 1.12 Wool |
| 0.21/0.15 KM 2/KC 3 | ||||
| 0.20 Bone | ||||
| Mo | 0.10–0.13 | 0.79–1.16/0.30–0.37 | 8.53 | 1.20 Liver |
| 0.43/0.05 KM 2/KC 3 | ||||
| 0.08 Lung | ||||
| U | 0.81–0.93 | 0.18–0.34/0.20–0.52 | 0.28 | 0.02/ng 4 KM/KC |
| 0.06 Wool | ||||
| 0.001 Liver |
| HM | Soil (Range) n = 8 | Shoot/Root (Range) n = 8/8 | BF 1 | Sheep Tissue n = 1 |
|---|---|---|---|---|
| Se | 1.17–1.88 | 1.13–1.61/1.81–3.50 | 0.80 | 5.93 Liver |
| 2.83/0.68 KM 2/KC 3 | ||||
| 1.88 Wool | ||||
| Cd | 0.06–0.17 | 0.08–0.16/0.20–0.29 | 1.53 | 1.02/0.01 KM 2/KC 3 |
| 0.22 Liver | ||||
| 0.04 Wool | ||||
| As | 1.58–3.79 | 0.78–1.40/1.23–1.41 | 0.5 | 0.56 Wool |
| 0.10/0.06 KM 2/KC 3 | ||||
| 0.10 Muscle | ||||
| Pb | 5.28–8.35 | 0.93–2.14/2.99–3.18 | 0.29 | 1.09 Bone |
| 1.07 Wool | ||||
| 0.21/0.25 KM 2/KC 3 | ||||
| Mo | 0.06–0.18 | 0.53–1.00/1.03–1.34 | 7.5 | 1.39 Liver |
| 0.66/ng 4 KM 2/KC 3 | ||||
| 0.25 Lung | ||||
| U | 0.48–1.15 | 0.08–0.31/0.28–0.42 | 0.23 | 0.001/ng 4 KM 2/KC 3 |
| 0.08 Wool | ||||
| 0.01 Lung |
| HM | Soil (Range) n = 8 | Shoot/Root (Range) n = 8/8 | BF 1 | Sheep Tissue n = 1 |
|---|---|---|---|---|
| Se | 2.29–2.80 | 1.19–2.62/2.58–3.36 | 0.78 | 3.28 Liver |
| 2.62/0.60 KM 2/KC 3 | ||||
| 3.85 Wool | ||||
| Cd | 0.07–0.08 | 0.09–0.17/0.14–0.22 | 1.71 | 0.63/0.02 KM 2/KC 3 |
| 0.23 Liver | ||||
| 0.05 Wool | ||||
| As | 1.20–1.75 | 0.55–0.63/0.82–1.06 | 0.42 | 0.71 Wool |
| 0.09/0.07 KM 2/KC 3 | ||||
| 0.14 Liver | ||||
| Pb | 5.23–6.37 | 1.36–1.58/2.42–2.62 | 0.25 | 1.90 Wool |
| 0.70 Bone | ||||
| 0.55 Liver | ||||
| Mo | 0.07–1.03 | 1.13–1.18/0.61–1.08 | 8.9 | 1.47 Liver |
| 0.47/0.11 KM 2/KC 3 | ||||
| 0.23 Wool | ||||
| U | 0.36–0.38 | 0.09–0.10/0.14–0.23 | 0.51 | 0.002/0.001 KM 2/KC 3 |
| 0.09 Wool | ||||
| 0.01 Lung |
| Sample Type | Heavy Metal Concentration mg/kg | Reference |
|---|---|---|
| Soil | U: 3–8 | deLemos et al., 2009 [18] |
| U: 5.1 ± 2.0 | deLemos et al., 2008 [41] | |
| U: 4.2 ± 2.0 (low impact area) | deLemos et al., 2008 [41] | |
| Se: 2.3/0.7 (control) | Dreesen & Cokal, 1984 [32] | |
| Vegetation | U: 0.5–7.7 | deLemos et al., 2009 [18] |
| Roots | U: 5.0 | deLemos et al., 2009 [18] |
| Shoots | U: 2.4 | deLemos et al., 2009 [18] |
| Sheep muscle | U: 0.88–0.148 | Millard et al., 1986 [21]; Ruttenber et al., 1984 [22] |
| Pb: 6.06–38.48 | ||
| Sheep liver | U: 1.776–2.96 | Millard et al., 1986 [21]; Ruttenber et al., 1984 [22] |
| Pb: 38.48–429.2 | ||
| Sheep kidney | U: 4.44–6.512 | Millard et al., 1986 [21]; Ruttenber et al., 1984 [22] |
| Pb: 41.44–1050.8 | ||
| Sheep bone | U: 35.52–65.12 | Ruttenber et al., 1984 [22] |
| Pb: 340.4 |
| Heavy Metal | Weekly Intake (μg/kg BW 1) | PTWI 2 (μg/kg BW 1) | % of PTWI | Once a Week Intake (μg) | RDI 3 or RDA 4 or UL 5 (μg/day) | % of RDI or RDA or UL |
|---|---|---|---|---|---|---|
| As | 1.64 | 7 | 10.9 | -- | -- | -- |
| Cd | 2.98 | 25 | 42.5 | -- | -- | -- |
| Pb | 6.2 | 15 | 24.8 | -- | -- | -- |
| Mo | -- | -- | -- | 92.34 | RDA: 45 UL: 2000 | 205 4.6 |
| Se | -- | -- | -- | 392.26 | RDI: 55 UL: 400 | 713 98 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samuel-Nakamura, C.; Robbins, W.A.; Hodge, F.S. Uranium and Associated Heavy Metals in Ovis aries in a Mining Impacted Area in Northwestern New Mexico. Int. J. Environ. Res. Public Health 2017, 14, 848. https://doi.org/10.3390/ijerph14080848
Samuel-Nakamura C, Robbins WA, Hodge FS. Uranium and Associated Heavy Metals in Ovis aries in a Mining Impacted Area in Northwestern New Mexico. International Journal of Environmental Research and Public Health. 2017; 14(8):848. https://doi.org/10.3390/ijerph14080848
Chicago/Turabian StyleSamuel-Nakamura, Christine, Wendie A. Robbins, and Felicia S. Hodge. 2017. "Uranium and Associated Heavy Metals in Ovis aries in a Mining Impacted Area in Northwestern New Mexico" International Journal of Environmental Research and Public Health 14, no. 8: 848. https://doi.org/10.3390/ijerph14080848
APA StyleSamuel-Nakamura, C., Robbins, W. A., & Hodge, F. S. (2017). Uranium and Associated Heavy Metals in Ovis aries in a Mining Impacted Area in Northwestern New Mexico. International Journal of Environmental Research and Public Health, 14(8), 848. https://doi.org/10.3390/ijerph14080848





