In Vitro Evaluation of the Inhibitory Activity of Thymoquinone in Combatting Candida albicans in Denture Stomatitis Prevention
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microbiology Test
2.1.1. Exposing Acrylic Specimens to Candida albicans
2.1.2. Evaluation
Slide Count
Serial Dilution Test
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kossioni, A.E. The prevalence of denture stomatitis and its predisposing conditions in an older Greek population. Gerodontology 2011, 28, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Arendorf, T.M. Denture stomatitis: A review. J. Oral Rehabil. 1987, 14, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Gümrü, B.; Kadir, T.; Uygun-Can, B.; Ozbayrak, S. Distribution and phospholipase activity of Candida species in different denture stomatitis types. Mycopathologia 2006, 162, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Newton, A. Denture sore mouth: A possible aetiology. Br. Dent. J. 1962, 112, 357–360. [Google Scholar]
- Webb, B.C.; Thomas, C.J.; Willcox, M.D.; Harty, D.W.; Knox, K.W. Candida-associated denture stomatitis. Aetiology and management: A review. Part I. Factors influencing distribution of Candida species in the oral cavity. Aust. Dent. J. 1998, 43, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Budtz-Jorgensen, E. Clinical aspects of Candida infection in denture wearers. J. Am. Dent. Assoc. 1978, 96, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Gendreau, L.; Loewy, Z.G. Epidemiology and etiology of denture stomatitis. J. Prosthodont. 2011, 20, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Karbach, J.; Walter, C.; Al-Nawas, B. Evaluation of saliva flow rates, Candida colonization and susceptibility of Candida strains after head and neck radiation. Clin. Oral Investig. 2012, 16, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Jeganathan, S.; Lin, C.C. Denture stomatitis—A review of the aetiology, diagnosis and management. Aust. Dent. J. 1992, 37, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.A.; Toledo, B.C.; Santos, C.T.; Pereira Costa, A.C.; Back-Brito, G.N.; Kaminagakura, E.; Jorge, A.O. Opportunistic microorganisms in individuals with lesions of denture stomatitis. Diagn. Microbiol. Infect. Dis. 2013, 76, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Vanden Abbeele, A.; de Meel, H.; Ahariz, M.; Perraudin, J.P.; Beyer, I.; Courtois, P. Denture contamination by yeasts in the elderly. Gerodontology 2008, 25, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Bergendal, T.; Holmberg, K.; Nord, C.E. Yeast colonization in the oral cavity and feces in patients with denture stomatitis. Acta Odontol. Scand. 1979, 37, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Bergendal, T.; Holmberg, K. Studies of Candida serology in denture stomatitis patients. Scand. J. Dent. Res. 1982, 90, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Radford, D.R.; Challacombe, S.J.; Walter, J.D. Denture plaque and adherence of Candida albicans to denture-base materials in vivo and in vitro. Crit. Rev. Oral Biol. Med. 1999, 10, 99–116. [Google Scholar] [CrossRef] [PubMed]
- Redding, S.; Bhatt, B.; Rawls, H.R.; Siegel, G.; Scott, K.; Lopez-Ribot, J. Inhibition of Candida albicans biofilm formation on denture material. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2009, 107, 669–672. [Google Scholar] [CrossRef] [PubMed]
- Nett, J.E.; Marchillo, K.; Spiegel, C.A.; Andes, D.R. Development and validation of an in vivo Candida albicans biofilm denture model. Infect. Immun. 2010, 78, 3650–3659. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.S.; Marchini, L.; Bernardes, L.A.; Paulino, L.C.; Nobrega, F.G. Biofilm microbial communities of denture stomatitis. Oral Microbiol. Immunol. 2008, 23, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, P.; Thompson, A.; Xie, Z.; Kashleva, H.; Ganguly, S.; Mitchell, A.P.; Dongari-Bagtzoglou, A. Role of Bcr1-activated genes Hwp1 and Hyr1 in Candida albicans oral mucosal biofilms and neutrophil evasion. PLoS ONE 2011, 6, e16218. [Google Scholar] [CrossRef] [PubMed]
- Katragkou, A.; Kruhlak, M.J.; Simitsopoulou, M.; Chatzimoschou, A.; Taparkou, A.; Cotton, C.J.; Paliogianni, F.; Diza-Mataftsi, E.; Tsantali, C.; Walsh, T.J.; et al. Interactions between human phagocytes and Candida albicans biofilms alone and in combination with antifungal agents. J. Infect. Dis. 2010, 201, 1941–1949. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; McCormick, T.S.; Imamura, Y.; Mukherjee, P.K.; Ghannoum, M.A. Interaction of Candida albicans with adherent human peripheral blood mononuclear cells increases C. albicans biofilm formation and results in differential expression of pro- and anti-inflammatory cytokines. Infect. Immun. 2007, 75, 2612–2620. [Google Scholar] [CrossRef] [PubMed]
- Gornitsky, M.; Paradisl, I.; Landaverde, G.; Malo, A.M.; Velly, A.M. A clinical and microbiological evaluation of denture cleansers for geriatric patients in long-term care institutions. J. Can. Dent. Assoc. 2002, 68, 39–45. [Google Scholar] [PubMed]
- Panzeri, H.; Lara, E.H.; Paranhos, H.; de, F.; Lovato da Silva, C.H.; de Souza, R.F.; de Souza Gugelmin, M.C.; Tirapelli, C.; Cruz, P.C.; de Andrade, I.M. In vitro and clinical evaluation of specific dentifrices for complete denture hygiene. Gerodontology 2009, 26, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Nalbant, A.D.; Kalkanci, A.; Filiz, B.; Kustimur, S. Effectiveness of denture cleaning agents against the colonization of Candida spp and the in vitro detection of the adherence of these yeast cells to denture acrylic surfaces. Yonsei. Med. J. 2008, 49, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Douglas, W.; Walker, D. Nystatin in denture liners—An alternative treatment of denture stomatitis. Br. Dent. J. 1973, 135, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Nutt, G. The in vitro fungicidal properties of Visco-gel, alone and combined with nystatin and amphotericin B. J. Oral Rehabil. 1978, 5, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Ren, B.; Zhou, X.; Xu, H.H.; Chen, Y.; Han, Q.; Li, B.; Weir, M.D.; Li, M.; Feng, M.; et al. Effect of Antimicrobial Denture Base Resin on Multi-Species Biofilm Formation. Int. J. Mol. Sci. 2016, 17, E1033. [Google Scholar] [CrossRef] [PubMed]
- Kiesow, A.; Sarembe, S.; Pizzey, R.L.; Axe, A.S.; Bradshaw, D.J. Material compatibility and antimicrobial activity of consumer products commonly used to clean dentures. J. Prosthet. Dent. 2016, 115, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Polychronakis, N.C.; Polyzois, G.L.; Lagouvardos, P.E.; Papadopoulos, T.D. Effects of cleansing methods on 3-D surface roughness, gloss and color of a polyamide denture base material. Acta Odontol. Scand. 2015, 73, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Peracini, A.; Davi, L.R.; de Queiroz Ribeiro, N.; de Souza, R.F.; Lovato da Silva, C.H.; de Freitas Oliveira Paranhos, H. Effect of denture cleansers on physical properties of heat-polymerized acrylic resin. J. Prosthodont. Res. 2010, 54, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Nikawa, H.; Iwanaga, H.; Hamada, T.; Yuhta, S. Effects of denture cleansers on direct soft denture lining materials. J. Prosthet. Dent. 1994, 72, 657–662. [Google Scholar] [CrossRef]
- Kuhn, D.; George, T.; Chandra, J.; Mukherjee, P.; Ghannoum, M. Antifungal susceptibility of Candida biofilms: Unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrob. Agents Chemother. 2002, 46, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Yodmongkol, S.; Chantarachindawong, R.; Thaweboon, S.; Thaweboon, B.; Amornsakchai, T.; Srikhirin, T. The effects of silane-SiO2 nanocomposite films on Candida albicans adhesion and the surface and physical properties of acrylic resin denture base material. J. Prosthet. Dent. 2014, 112, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- Izumida, F.E.; Moffa, E.B.; Vergani, C.E.; Machado, A.L.; Jorge, J.H.; Giampaolo, E.T. In vitro evaluation of adherence of Candida albicans, Candida glabrata, and Streptococcus mutans to an acrylic resin modified by experimental coatings. Biofouling 2014, 30, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A.; Alharbi, F.A.; Suresh, C.S. Effectiveness of coating acrylic resin dentures on the Candida adhesion. J. Prosthodont. 2013, 22, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E.; Raj, P.A.; Loza, J.C. Effect of surface-charged poly(methyl methacrylate) on the adhesion of Candida albicans. J. Prosthodont. 2003, 12, 249–254. [Google Scholar] [CrossRef]
- Nawasrah, A.; AlNimr, A.; Ali, A.A. Antifungal Effect of Henna against Candida albicans Adhered to Acrylic Resin as a Possible Method for Prevention of Denture Stomatitis. Int. J. Environ. Res. Public Health 2016, 13, E520. [Google Scholar] [CrossRef] [PubMed]
- Singla, S.; Gupta, R.; Puri, A.; Singh, V.; Roy, S. Comparison of anticandidal activity of Punica granatum (Pomegranate) and Lawsonia inermis (Henna leaves): An in vitro study. Int. J. Dent. Res. 2013, 1, 8–13. [Google Scholar] [CrossRef]
- Entok, E.; Ustuner, M.C.; Ozbayer, C.; Tekin, N.; Akyuz, F.; Yangi, B.; Kurt, H.; Degirmenci, I.; Gunes, H.V. Anti-inflammatuar and anti-oxidative effects of Nigella sativa L.: FDG-PET imaging of inflammation. Mol. Biol. Rep. 2014, 41, 2827–2834. [Google Scholar] [CrossRef] [PubMed]
- Bakathir, H.A.; Abbas, N.A. Detection of the antibacterial effect of Nigella sativa ground seeds with water. Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 159–164. [Google Scholar] [PubMed]
- Hajhashemi, V.; Ghannadi, A.; Jafarabadi, H. Black cumin seed essential oil, as a potent analgesic and antiinflammatory drug. Phytother. Res. 2004, 18, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.S.; Rao, M.V.; Kaneez, F.S.; Qadri, S.; Al-Marzouqi, A.H.; Chandranath, I.S.; Adem, A. Nigella sativa extract as a potent antioxidant for petrochemical-induced oxidative stress. J. Chromatogr. Sci. 2011, 49, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Aikemu, A.; Xiaerfuding, X.; Shiwenhui, C.; Abudureyimu, M.; Maimaitiyiming, D. Immunomodulatory and anti-tumor effects of Nigella glandulifera freyn and sint seeds on ehrlich ascites carcinoma in mouse model. Pharmacogn. Mag. 2013, 9, 187–191. [Google Scholar] [PubMed]
- El-Sayed, W.M. Upregulation of chemoprotective enzymes and glutathione by Nigella sativa (black seed) and thymoquinone in CCl4-intoxicated rats. Int. J. Toxicol. 2011, 30, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Kundu, J.; Kim, D.H.; Kundu, J.K.; Chun, K.S. Thymoquinone induces heme oxygenase-1 expression in HaCaT cells via Nrf2/ARE activation: Akt and AMPKalpha as upstream targets. Food Chem. Toxicol. 2014, 65, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Alhebshi, A.H.; Gotoh, M.; Suzuki, I. Thymoquinone protects cultured rat primary neurons against amyloid beta-induced neurotoxicity. Biochem. Biophys. Res. Commun. 2013, 433, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, N.A. Effect of Nigella sativa L. extracts against streptococcus mutans and Streptococcus mitis in vitro. J. Baghdad Coll. Dent. 2012, 24, 154–157. [Google Scholar]
- Omar, O.M.; Khattab, N.M.; Khater, D.S. Nigella sativa oil as a pulp medicament for pulpotomized teeth: A histopathological evaluation. J. Clin. Pediatr. Dent. 2012, 36, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Al-Bayaty, F.; Kamaruddin, A.; Ismail, M.; Abdulla, M. Formulation and Evaluation of a New Biodegradable Periodontal Chip Containing Thymoquinone in a Chitosan Base for the Management of Chronic Periodontitis. J. Nanomater. 2013, 2013, 397308. [Google Scholar] [CrossRef]
- Al-Douri, A.; Al-Kazaz, S. The effect of Nigella sativa oil (black seed) on the healing of chemically induced oral ulcer in rabbit (experimental study). Al-Rafidain Dent. J. 2010, 10, 151–157. [Google Scholar]
- Sritrairat, N.; Nukul, N.; Inthasame, P.; Sansuk, A.; Prasirt, J.; Leewatthanakorn, T.; Piamsawad, U.; Dejrudee, A.; Panichayupakaranant, P.; Pangsomboon, K.; et al. Antifungal activity of lawsone methyl ether in comparison with chlorhexidine. J. Oral Pathol. Med. 2011, 40, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Harzallah, H.; Kouidhi, B.; Flamini, G.; Bakhrouf, A.; Mahjoub, T. Chemical composition, antimicrobial potential against cariogenic bacteria and cytotoxic activity of Tunisian Nigella sativa essential oil and thymoquinone. Food Chem. 2011, 129, 1469–1474. [Google Scholar] [CrossRef]
- Badary, O.A.; Al-Shabanah, O.A.; Nagi, M.N.; Al-Bekairi, A.M.; Elmazar, M.M.A. Acute and subchronic toxicity of thymoquinone in mice. Drug Dev. Res. 1998, 44, 56–61. [Google Scholar] [CrossRef]
- Baillie, G.S.; Douglas, L.J. Effect of growth rate on resistance of Candida albicans biofilms to antifungal agents. Antimicrob. Agents Chemother. 1998, 42, 1900–1905. [Google Scholar] [PubMed]
- Selmecki, A.; Forche, A.; Berman, J. Aneuploidy and isochromosome formation in drug-resistant Candida albicans. Science 2006, 313, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Abu-Elteen, K.; Abu-Alteen, R. The prevalence of Candida albicans populations in the mouths of complete denture wearers. New Microbiol. 1998, 21, 41–48. [Google Scholar] [PubMed]
- Khan, M.A.; Ashfaq, M.K.; Zuberi, H.S.; Mahmood, M.S.; Gilani, A.H. The in vivo antifungal activity of the aqueous extract from Nigella sativa seeds. Phytother. Res. 2003, 17, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Fareid, M.A. In vitro: Evaluation of inhibitory activity of some plant extracts against oral candidiasis. N. Y. Sci. J. 2014, 7, 66–76. [Google Scholar]
- Randhawa, M.A.; Gondal, M.; Al-Zahrani, A.; Rashid, S.G.; Ali, A. Synthesis, morphology and antifungal activity of nanoparticulated amphotericin-B, ketoconazole and thymoquinone against Candida albicans yeasts and Candida biofilm. J. Environ. Sci. Health A Toxic Hazard Subst. Environ. Eng. 2015, 50, 119–124. [Google Scholar] [CrossRef] [PubMed]
| Groups | Description |
|---|---|
| 0% (Control) | heat polymerized specimens |
| 0.5% | heat polymerized specimens incorporated with 0.5% TQ |
| 1% | heat polymerized specimens incorporated with 1% TQ |
| 1.5% | heat polymerized specimens incorporated with 1.5% TQ |
| 2% | heat polymerized specimens incorporated with 2% TQ |
| 2.5% | heat polymerized specimens incorporated with 2.5% TQ |
| 3% | heat polymerized specimens incorporated with 3% TQ |
| 5% | heat polymerized specimens incorporated with 5% TQ |
| Artificial Saliva | Composition |
|---|---|
| A.S. Orthana, Biofac A/S, Kastrup, Denmark | Mucin, methyl-4-hydroxybenzoate, benzalconium chloride, ethylenediaminetetraacetic acid (EDTA), H2O2, xylitol, peppermint oil, spearmint oil and mineral salts |
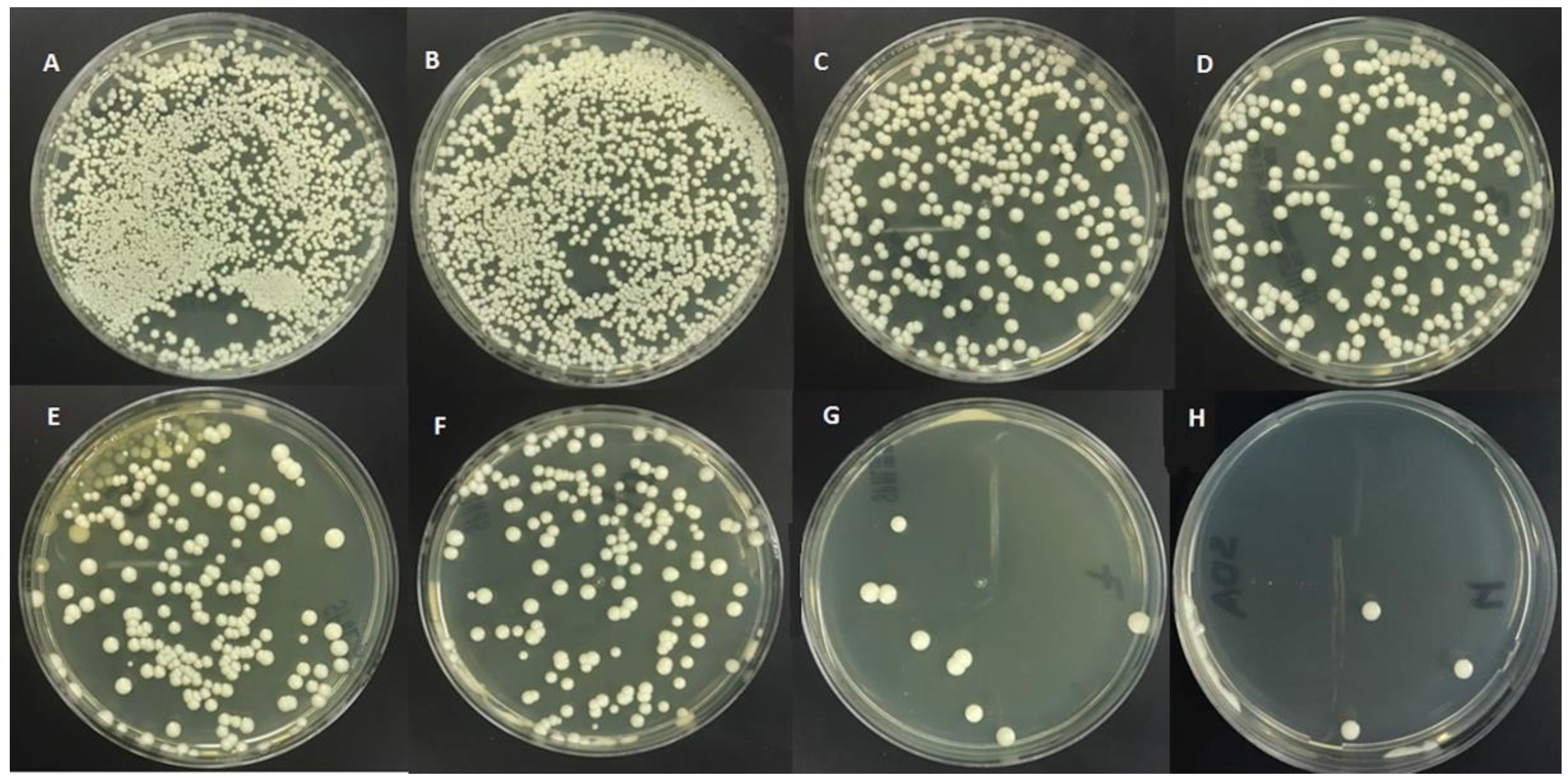
| TQ Concentration | Slide Count | Serial Dilution Test |
|---|---|---|
| Mean ± SD | Mean ± SD | |
| 0% Control | 5436.9 ± 266 | 4691.4 ± 176.8 |
| 0.5% | 3776.10 ± 98.8 | 3334.7 ± 121.2 |
| 1% | 3037.4 *** ± 39.2 | 2619.4 *** ± 50.1 |
| 1.5% | 980.2 ** ± 10.8 | 894.6 ** ± 32.3 |
| 2% | 466 ** ± 6.5 | 310.3 ** ± 8.2 |
| 2.5% | 166.5 * ± 6 | 91.9 * ± 4.5 |
| 3% | 0 * ± 0 | 53.9 * ± 2.0 |
| 5% | 0 * ± 0 | 32.4 * ± 1.7 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Thobity, A.M.; Al-Khalifa, K.S.; Gad, M.M.; Al-Hariri, M.; Ali, A.A.; Alnassar, T. In Vitro Evaluation of the Inhibitory Activity of Thymoquinone in Combatting Candida albicans in Denture Stomatitis Prevention. Int. J. Environ. Res. Public Health 2017, 14, 743. https://doi.org/10.3390/ijerph14070743
Al-Thobity AM, Al-Khalifa KS, Gad MM, Al-Hariri M, Ali AA, Alnassar T. In Vitro Evaluation of the Inhibitory Activity of Thymoquinone in Combatting Candida albicans in Denture Stomatitis Prevention. International Journal of Environmental Research and Public Health. 2017; 14(7):743. https://doi.org/10.3390/ijerph14070743
Chicago/Turabian StyleAl-Thobity, Ahmad M., Khalifa S. Al-Khalifa, Mohammed M. Gad, Mohammed Al-Hariri, Aiman A. Ali, and Talal Alnassar. 2017. "In Vitro Evaluation of the Inhibitory Activity of Thymoquinone in Combatting Candida albicans in Denture Stomatitis Prevention" International Journal of Environmental Research and Public Health 14, no. 7: 743. https://doi.org/10.3390/ijerph14070743
APA StyleAl-Thobity, A. M., Al-Khalifa, K. S., Gad, M. M., Al-Hariri, M., Ali, A. A., & Alnassar, T. (2017). In Vitro Evaluation of the Inhibitory Activity of Thymoquinone in Combatting Candida albicans in Denture Stomatitis Prevention. International Journal of Environmental Research and Public Health, 14(7), 743. https://doi.org/10.3390/ijerph14070743






