Inorganic Macro- and Micronutrients in “Superberries” Black Chokeberries (Aronia melanocarpa) and Related Teas
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Glassware
2.2. Samples
2.3. Sample Preparation
2.4. ICP-AES Measurements
2.5. Inductively Coupled Plasma—Mass Spectrometry (ICP-MS) Measurements
2.6. Optimisation and Characterisation of the Analytical Method
3. Results and Discussion
3.1. Analytical Methods
3.2. Elemental Analysis of Berries
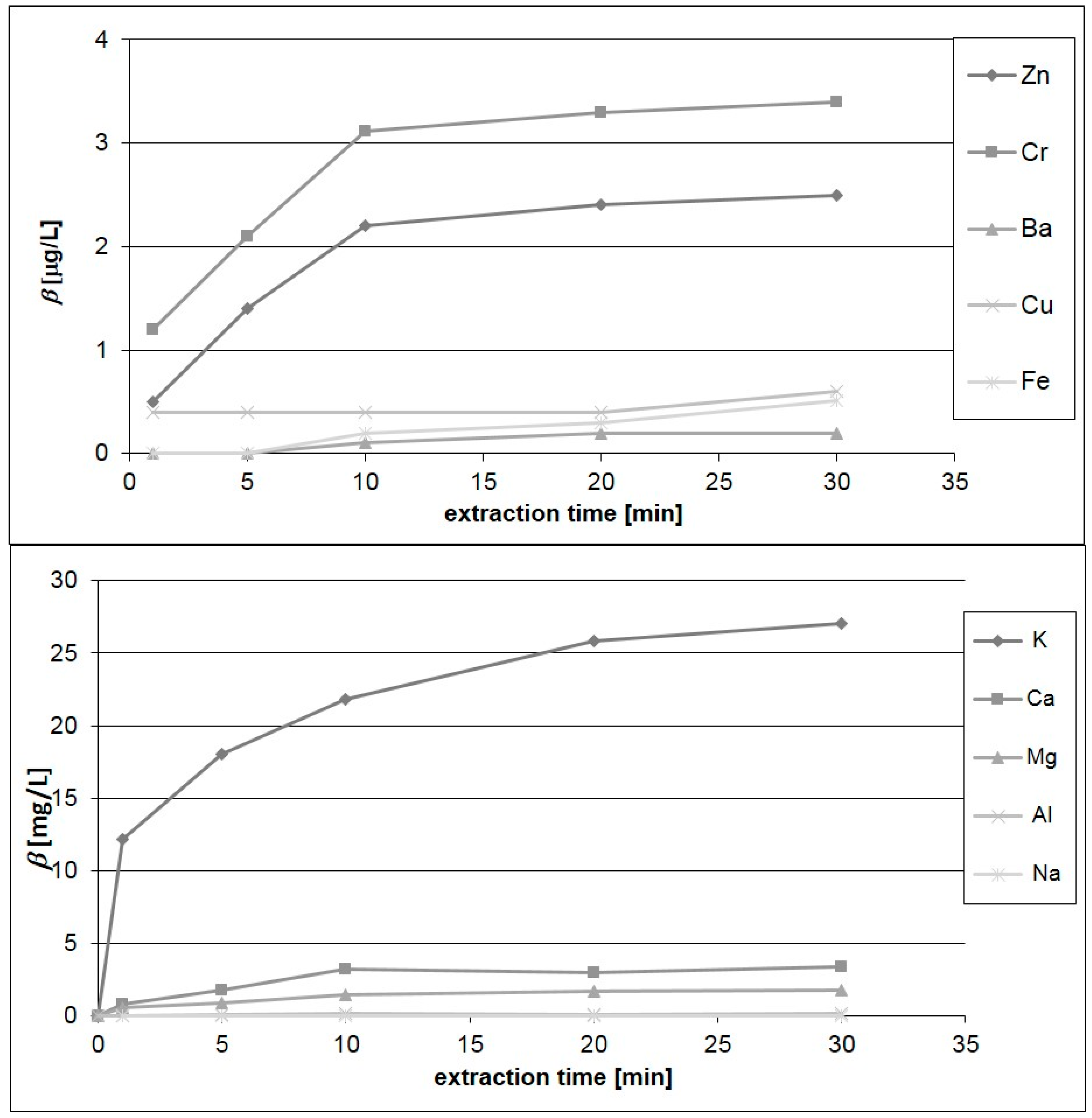
3.3. Elemental Analysis of Infusions
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Kokotkiewicz, A.; Jaremicz, Z.; Luczkiewicz, M. Aronia plants: A review of traditional use, biological activities, and perspectives for modern medicine. J. Med. Food 2010, 13, 255–269. [Google Scholar] [CrossRef] [PubMed]
- WertvollJournal for Members of “Mein Clubhaus” Ausgabe, 2015 p 12. Available online: http://www.mein-clubhaus.com/at/aktuell/wertvoll (accessed on 5 May 2016).
- Kulling, S.E.; Rawel, H.M. Chokeberry (Aronia melanocarpa)—A review on the characteristic components and potential health effects. Planta Med. 2008, 74, 1625–1634. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Kim, G.-S.; Park, S.; Kim, Y.-H.; Kim, M.-B.; Lee, W.S.; Jeong, S.W.; Lee, S.J.; Jin, J.S.; Shin, S.C. Determination of chokeberry (Aronia melanocarpa) polyphenol components using liquid chromatography-tandem mass spectrometry: Overall contribution to antioxidant activity. Food Chem. 2014, 146, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, J.I.; Lee, I.; Lee, S.; Hwang, M.-W.; Bae, J.-Y.; Heo, J.; Kim, D.; Han, S.-Z.; Park, M.-S. Aronia melanocarpa and its components demonstrate antiviral activity against influenza viruses. Biochem. Biophys. Res. Commun. 2013, 440, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Šavikin, K.; Zdunić, G.; Janković, T.; Gođevac, D.; Stanojković, T.; Pljevlakušić, D. Berry fruit teas: Phenolic composition and cytotoxic activity. Food Res. Int. 2014, 62, 677–683. [Google Scholar] [CrossRef]
- Lala, G.; Malik, M.; Zhao, C.; He, J.; Kwon, Y.; Giusti, M.M.; Magnuson, B.A. Anthocyaninrich extracts inhibit multiple biomarkers of colon cancer in rats. Nutr. Cancer 2006, 54, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez-Soto, M.J.; Larrosa, M.; García-Cantalejo, J.; Espín, J.C.; Tomás-Barberan, F.A.; García-Conesa, M.T. Up-regulation of tumor suppressor carcinoembryonic antigen-related cell adhesion molecule 1 in human colon cancer Caco-2 cells following repetitive exposure to dietary levels of a polyphenol-rich chokeberry juice. J. Nutr. Biochem. 2007, 18, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin composition of different wild and cultivated berry species. Food Sci. Technol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Nakajima, J.; Tanaka, I.; Seo, S.; Yamazaki, M.; Saito, K. LC/PDA/ESI-MS profiling and radical scavenging activity of anthocyanins in various berries. J. Biomed. Biotechnol. 2004, 5, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Šnebergrová, J.; Čižková, H.; Neradová, E.; Kapci, B.; Rajchl, A.; Voldřich, M. Variability of characteristic components of Aronia. Czech J. Food Sci. 2014, 32, 25–30. [Google Scholar]
- Nile, S.H.; Park, S.W. Edible berries: Bioactive components and their effect on human health. Nutrition 2014, 30, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Chandler, F.B. Composition and uses of blueberries. Maine Agric. Exp. Station Bull. 1944, 428, 1–39. [Google Scholar]
- Konieczynski, P.; Arceusz, A.; Wesolowski, M. Relationships between flavonoids and selected elements in infusions of medicinal herbs. Open Chem. 2015, 13, 68–74. [Google Scholar] [CrossRef]
- Skupien, K.; Oszmianski, J. The effect of mineral fertilization on nutritive value and biological activity of chokeberry fruit. Agric. Food Sci. 2007, 16, 46–55. [Google Scholar] [CrossRef]
- Borycka, B.; Stachowiak, J. Relations between cadmium and magnesium and aronia fractional dietary fibre. Food Chem. 2008, 107, 44–48. [Google Scholar] [CrossRef]
- Nawirska, A. Binding of heavy metals to pomace fibers. Food Chem. 2005, 90, 395–400. [Google Scholar] [CrossRef]
- Soylak, M.; Tuzen, M.; Souza, A.S.; das Graças Andrade, K.M.; Ferreira, S.L. Optimization of microwave assisted digestion procedure for the determination of zinc, copper and nickel in tea samples employing flame atomic absorption spectrometry. J. Hazard. Mater. 2007, 149, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Boumans, P.W.J.M. Basic concepts and characteristics of ICP-AES. In Inductively Coupled Plasma Emission Spectroscopy Part I Methodology, Instrumentation, and Performance; Boumans, P.W.J.M., Ed.; Wiley: New York, NY, USA, 1987; pp. 100–257. [Google Scholar]
- Tanaka, T.; Tanaka, A. Chemical components and characteristics of black chokeberry. J. Jpn. Soc. Food Sci. Technol. 2001, 48, 606–610. [Google Scholar] [CrossRef]
- Ognik, K.; Rusinek, E.; Sembratowicz, I.; Truchlinski, J. Contents of heavy metal, nitrate (V), and nitrate (III) in fruits of elderberry and black chokeberry depending on harvest site and vegetation period. Rocz. Państw. Zakl. Hig. 2006, 57, 235–241. [Google Scholar] [PubMed]
- Juranović Cindrić, I.; Zeiner, M.; Mihajlov-Konanov, D.; Stingeder, G. Metal characterization of white hawthorn organs and infusions. J. Agric. Food Chem. 2015, 63, 1798–1802. [Google Scholar] [CrossRef] [PubMed]
- Pliszka, B.; Huszcza-Ciołkowska, G.; Wierzbicka, E. Effects of extraction conditions on the content of anthocyanins and bioelements in berry fruit extracts. Commun. Soil Sci. Plant Anal. 2008, 39, 753–762. [Google Scholar] [CrossRef]
- Zeiner, M.; Juranović Cindrić, I.; Majić, B.; Stingeder, G. Study of the accumulation of Toxic and Essential Ultra-Trace Elements in Fruits of Sorbus domestica L. Int. J. Environ. Res. Public Health 2017, 14, 341. [Google Scholar] [CrossRef] [PubMed]
- Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes, Standing Committee on the Scientific Evaluation of Dietary Reference Intakes; National Academies Press: Washington, DC, USA, 1997. [Google Scholar]
- World Health Organization (WHO). Evaluation of Certain Food Additives and Contaminants (41st Report of the Joint FAO/WHO Expert Committee on Food Additives); WHO Technical Report Series No 837; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- WHO. Chromium in Drinking-Water, Guidelines for Drinking Water Quality, 2nd ed.; World Health Organization: Geneva, Switzerland, 1996; Volume 2. [Google Scholar]
| Instrument | Prodigy High Dispersive ICP |
|---|---|
| Spectrometer | High resolution Echelle polychromator Large format programmable array detector (L-PAD) |
| RF-Generator | 40 MHz “free-running” |
| Output power | 1.1 kW |
| Argon flow | Coolant:18 L/min, Auxiliary: 0.8 L/min, Nebulizer: 1 L/min |
| Peristaltic pump | 1.0 mL min−1 |
| Nebulizer | Pneumatic (glass concentric) |
| Spray chamber | Glass cyclonic |
| Plasma viewing | Axial |
| Replicates for each analysis run | 3 |
| Sample uptake delay | 30 s |
| Content [mg/kg] | Al * | As | Ba | Ca | Cd | Co | Cr | Cu * | Fe | K | Li * | Mg | Mn | Mo | Na * | Ni | Pb | Se * | Sr | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| found | 1036 | 0.278 | 112 | 15,181 | 0.186 | 0.459 | 1.87 | 8.7 | 791 | 20,313 | 0.623 | 3829 | 162 | 0.300 | 218 | 2.30 | 1.78 | 0.347 | 63.8 | 21.5 |
| certified value | 1000 | 0.28 | 107 | 15,300 | 0.17 | 0.47 | 2.15 | 10 | 818 | 19,600 | 0.7 | 3770 | 171 | 0.32 | 210 | 2.6 | 1.8 | 0.04 | 64 | 24 |
| confidence interval | 0.07 | 10 | 700 | 0.04 | 0.11 | 0.34 | 48 | 1000 | 170 | 10 | 0.08 | 0.7 | 0.4 | 6 | 5 | |||||
| method | AES | MS | AES | AES | MS | MS | MS | MS | AES | AES | MS | AES | AES | MS | AES | MS | MS | MS | MS | AES |
| recovery in % | 104 | 99 | 105 | 99 | 109 | 98 | 87 | 87 | 97 | 104 | 89 | 102 | 95 | 94 | 104 | 85 | 99 | 87 | 100 | 90 |
| Concentration [mg/L] | Al | Ba | Ca | Cd | Co | Cr | Cu | Fe | K | Mg | Mn | Na | Ni | Pb | Sr | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| spiked | 1 | 1 | 5 | 1 | 1 | 1 | 1 | 3 | 5 | 3 | 1 | 5 | 1 | 1 | 1 | 1 |
| found | 1.02 | 1.03 | 4.87 | 1.06 | 1.04 | 1.02 | 0.976 | 2.84 | 5.12 | 3.08 | 0.957 | 4.73 | 0.981 | 1.02 | 0.924 | 0.976 |
| recovery in % | 102 | 103 | 97 | 106 | 104 | 102 | 98 | 95 | 102 | 103 | 96 | 95 | 98 | 102 | 92 | 98 |
| Sample/Method | Determined Value | Al | As | Ba | Ca | Cd | Co | Cr | Cu | Fe | K | Li | Mg | Mn | Mo | Na | Ni | Pb | Se | Sr | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aronia berries | LOD [mg/kg] | 0.91 | 0.020 | 0.86 | 0.12 | 0.0005 | 0.0037 | 0.0068 | 0.0072 | 0.05 | 0.27 | 0.0085 | 0.21 | 0.32 | 0.011 | 0.059 | 0.0063 | 0.013 | 0.0047 | 0.06 | 0.35 |
| analytical method | - | AES | MS | AES | AES | MS | MS | MS | MS | AES | AES | MS | AES | AES | MS | AES | MS | MS | MS | MS | AES |
| Aronia berries | content [mg/kg] | 158 | a | a | 1212 | 0.055 | 0.019 | 0.029 | 1.58 | 1.32 | 6790 | 0.012 | 669 | 0.829 | 0.039 | 4.27 | 0.38 | 0.041 | a | 1.66 | 0.55 |
| SD [mg/kg] | 72 | 310 | 0.027 | 0.001 | 0.018 | 0.07 | 0.05 | 308 | 0.001 | 89 | 0.123 | 0.016 | 1.22 | 0.02 | 0.021 | 0.40 | 0.13 | ||||
| Aronia berries [11] | content in fresh fruits [mg/kg] | 272 | 2903 | 155 | |||||||||||||||||
| Aronia berries [20] reported in [3] | content in fresh fruits [mg/kg] | 322 | 9.3 | 2180 | 162 | 26 | 1.47 | ||||||||||||||
| Aronia berries [21] | content [mg/kg] unpolluted area (2003/04) | 0.047/0.045 | 0.012/0.014 | ||||||||||||||||||
| (fresh fruits) | content [mg/kg] polluted area (2003/04) | 0.047/0.046 | 0.043/0.048 | ||||||||||||||||||
| Hawthorn berries [22] | content [mg/kg] | 917 | 0.012 | 4.17 | 3722 | 0.045 | 0.058 | 1.31 | 0.55 | 2.4 | 4223 | 0.51 | 987 | 1.88 | 0.097 | 6.34 | 0.067 | 0.027 | 0.41 | 1.12 |
| Sample/Method | Determined Value | Al | Ba | Ca | Cd | Co | Cr | Cu | Fe | K | Mg | Mn | Na | Ni | Pb | Sr | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| infusions from Aronia berries | LOD [µg/L] | 0.58 | 0.09 | 2.1 | 0.41 | 0.57 | 0.77 | 0.40 | 0.21 | 0.05 | 2.8 | 0.08 | 0.09 | 1.0 | 5.1 | 0.07 | 0.81 |
| analytical method | a | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES | AES |
| infusions from Aronia berries | Concentration [µg/L] | 160 | 0.10 | 3235 | b | b | 3.2 | 3.3 | 0.31 | 21,821 | 1485 | 0.40 | 8.7 | 2.3 | b | b | 2.2 |
| SD [µg/L] | 14 | 0.04 | 89 | 0.4 | 0.03 | 0.02 | 264 | 72 | 0.02 | 0.3 | 0.1 | 0.1 | |||||
| extraction yield in % | 4.3 | n.c. | 7.6 | 25 | 27 | 9.5 | 11 | 7.2 | 4.0 | 44 | 20 | 23 | |||||
| extracts from Aronia berries 50 g berries in 200 mL solution [23] | concentration [µg/L] | 29,530 | 620 | 37,490 | 2750 |
| Element | RDA [mg/day] | Element | RDA [mg/day] |
|---|---|---|---|
| Ca | 1000 | Mg | 400 |
| Cr | 0.120 | Mn | 2 |
| Cu | 2 | ||
| Fe | 8 | Mo | 0.120 |
| Zn | 11 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juranović Cindrić, I.; Zeiner, M.; Mihajlov-Konanov, D.; Stingeder, G. Inorganic Macro- and Micronutrients in “Superberries” Black Chokeberries (Aronia melanocarpa) and Related Teas. Int. J. Environ. Res. Public Health 2017, 14, 539. https://doi.org/10.3390/ijerph14050539
Juranović Cindrić I, Zeiner M, Mihajlov-Konanov D, Stingeder G. Inorganic Macro- and Micronutrients in “Superberries” Black Chokeberries (Aronia melanocarpa) and Related Teas. International Journal of Environmental Research and Public Health. 2017; 14(5):539. https://doi.org/10.3390/ijerph14050539
Chicago/Turabian StyleJuranović Cindrić, Iva, Michaela Zeiner, Darija Mihajlov-Konanov, and Gerhard Stingeder. 2017. "Inorganic Macro- and Micronutrients in “Superberries” Black Chokeberries (Aronia melanocarpa) and Related Teas" International Journal of Environmental Research and Public Health 14, no. 5: 539. https://doi.org/10.3390/ijerph14050539
APA StyleJuranović Cindrić, I., Zeiner, M., Mihajlov-Konanov, D., & Stingeder, G. (2017). Inorganic Macro- and Micronutrients in “Superberries” Black Chokeberries (Aronia melanocarpa) and Related Teas. International Journal of Environmental Research and Public Health, 14(5), 539. https://doi.org/10.3390/ijerph14050539






