Mercury Levels in Human Hair and Farmed Fish near Artisanal and Small-Scale Gold Mining Communities in the Madre de Dios River Basin, Peru
Abstract
:1. Introduction
1.1. Overview of Artisanal and Small-Scale Gold Mining
1.2. ASGM and Mercury
1.3. ASGM in Madre de Dios
2. Materials and Methods
2.1. Site Selection and Participant Recruitment
2.2. Human Subjects Survey
2.3. Human Hair Sample Collection and Analysis
2.4. Fish Sample Site Selection and Sample Collection
2.5. Fish Sample Laboratory Analysis
2.6. Statistical Analysis
3. Results
3.1. Human Survey Results
3.2. Human Hair Mercury Results
3.3. Fish Sample Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hentschel, T.; Hruschka, F.; Priester, M. Global Report on Artisanal & Small-Scale Mining. Avaliable online: http://pubs.iied.org/pdfs/G00723.pdf (accessed on 11 March 2017).
- United Nations Environment Programme. Reducing Mercury Use in arTisanal and Small-Scale: A Practical Guide; United Nations Environment Programme: Geneva, Switzerland, 2012. [Google Scholar]
- International Labour Organization. Report for Discussion at the Tripartite Meeting on Social and Labour Issues in Small-Scale Mines; International Labour Organization: Geneva, Switzerland, 1999. [Google Scholar]
- Damonte, G.H. Taming the “Wilderness”: Government Quest for Forrmalization and Conflict among Small-Scale Miners in the Peruvian Amazon. Antipode 2016, 48, 956–976. [Google Scholar] [CrossRef]
- United Nations Environment Programme (UNEP). Analysis of Formalization Approaches in the Artisanal and Small-Scale Gold Mining Sector Based on Experiences in Ecuador, Mongolia, Peru, Tanzania and Uganda: Peru Case Study; UNEP: Geneva, Switzerland, 2012. [Google Scholar]
- El Instituto Nacional de Estadística e Informática (INEI). PERÚ: Estimaciones y Proyecciones de Población Departamental, por Años Calendario y Edades Simples 1995–2025; Boletín Es, 1950–2050; INEI: Lima, Peru, 2010. [Google Scholar]
- Brooks, W.E.; Sandoval, E.; Yepez, M.A.; Howell, H. Peru Mercury Inventory 2006; U.S. Geological Survey: Reston, VA, USA, 2007.
- VERITE. Risk Analysis of Indicators of Forced Labor and Human Trafficking in Illegal Gold Mining in Peru; VERITE: Amherst, MA, USA, 2013. [Google Scholar]
- Swenson, J.J.; Carter, C.E.; Domec, J.C.; Delgado, C.I. Gold mining in the peruvian amazon: Global prices, deforestation, and mercury imports. PLoS ONE 2011, 6, e18875. [Google Scholar] [CrossRef] [PubMed]
- Asner, G.P.; Llactayo, W.; Tupayachi, R.; Luna, E.R. Elevated rates of gold mining in the Amazon revealed through high-resolution monitoring. Proc. Natl. Acad. Sci. USA 2013, 110, 18454–18459. [Google Scholar] [CrossRef] [PubMed]
- Veiga, M.M.; Maxson, P.A.; Hylander, L.D. Origin and consumption of mercury in small-scale gold mining. J. Clean. Prod. 2006, 14, 436–447. [Google Scholar] [CrossRef]
- Álvares, J.; Sotero, V.; Brack, E.A.; Ipenza, P.C.A. Minería Aurífera en Madre de Dios y Contaminación con Mercurio; Ministerio del Ambiente: Lima, Peru, 2011.
- Olson, D.M.; Dinerstein, E. The Global 200: A Representation Approach to Conserv. Conserv. Biol. 1998, 12, 502–515. [Google Scholar] [CrossRef]
- Drake, P.L.; Rojas, M.; Reh, C.M.; Mueller, C.A.; Jenkins, F.M. Occupational exposure to airborne mercury during gold mining operations near El Callao, Venezuela. Int. Arch. Occup. Environ. Health 2001, 74, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Winfrey, R.; Rudd, W.M. Formation of methylmercury in low pH lakes. Environ. Toxicol. Chem. 1990, 9, 853–869. [Google Scholar] [CrossRef]
- Bloom, N.S. On the Chemical Form of Mercury in Edible Fish and Marine Invertebrate Tissue. Can. J. Fish. Aquat. Sci. 1992, 49, 1010–1017. [Google Scholar] [CrossRef]
- Pfeiffer, W.C.; Lacerda, L.D. De Mercury inputs into the Amazon Region, Brazil. Environ. Technol. Lett. 1988, 9, 325–330. [Google Scholar] [CrossRef]
- National Research Council Dose Estimation. Toxicological Effects of Methylmercury; National Academies Press: Washington, DC, USA, 2000; pp. 105–146. [Google Scholar]
- Pirrone, N.; Cinnirella, S.; Feng, X.; Finkelman, R.B.; Friedli, H.R.; Leaner, J.; Mason, R.; Mukherjee, A.B.; Stracher, G.B.; Streets, D.G.; et al. Global mercury emissions to the atmosphere from anthropogenic and natural sources. Atmos. Chem. Phys. 2010, 10, 5951–5964. [Google Scholar] [CrossRef]
- United Nations Environment Programme (UNEP). Global Mercury Assessment 2013: Sources, Emissions, Releases, and Environmental Transport; UNEP: Geneva, Switzerland, 2013. [Google Scholar]
- United Nations Environment Programme (UNEP). Minamata Convention on Mercury; UNEP: Geneva, Switzerland, 2013. [Google Scholar]
- Piñeiro, V.; Thomas, J.; Elverdin, P. The Agricultural Sector as an Alternative to Illegal Mining in Peru: A Case Study of Madre de Dios. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=2884337 (accessed on 11 March 2017).
- Finer, M.; Olexy, T.; Novoa, S. Gold Mining Deforestation within Tambopata National Reserve Exceeds 450 Hectares. Available online: http://maaproject.org/2016/tambopata450/ (accessed on 11 March 2017).
- Fraser, B. Peru’s gold rush raises health fears. Nature 2016, 534, 162. [Google Scholar] [CrossRef] [PubMed]
- Lebel, J.; Mergler, D.; Branches, F.; Lucotte, M.; Amorim, M.; Larribe, F.; Dolbec, J. Neurotoxic effects of low-level methylmercury contamination in the Amazonian Basin. Environ. Res. 1998, 79, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Zahir, F.; Rizwi, S.J.; Haq, S.K.; Khan, R.H. Low dose mercury toxicity and human health. Environ. Toxicol. Pharmacol. 2005, 20, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Salonen, J.T.; Seppanen, K.; Lakka, T.A.; Salonen, R.; Kaplan, G.A. Mercury accumulation and accelerated progression of carotid atherosclerosis: A population-based prospective 4-year follow-up study in men in eastern Finland. Atherosclerosis 2000, 148, 265–273. [Google Scholar] [CrossRef]
- Oken, E.; Wright, R.O.; Kleinman, K.P.; Bellinger, D.; Amarasiriwardena, C.J.; Hu, H.; Rich-Edwards, J.W.; Gillman, M.W. Maternal fish consumption, hair mercury, and infant cognition in a U.S. cohort. Environ. Health Perspect. 2005, 113, 1376–1380. [Google Scholar] [CrossRef] [PubMed]
- Jarosińska, D.; Horvat, M.; Sällsten, G.; Mazzolai, B.; Dabkowska, B.; Prokopowicz, A.; Biesiada, M.; Barregård, L. Urinary mercury and biomarkers of early renal dysfunction in environmentally and occupationally exposed adults: A three-country study. Environ. Res. 2008, 108, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Schwenk, M.; Klein, R.; Templeton, D.M. Immunological effects of mercury (IUPAC Technical Report). Pure Appl. Chem. 2009, 81, 153–167. [Google Scholar] [CrossRef]
- Basu, N.; Goodrich, J.M.; Head, J. Ecogenetics of mercury: From genetic polymorphisms and epigenetics to risk assessment and decision-making. Environ. Toxicol. Chem. 2014, 33, 1248–1258. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Sun, R.; Sun, Y.; Chen, Z.; Li, L. Psychological effects of low exposure to mercury vapor: Application of a computer-administered neurobehavioral evaluation system. Environ. Res. 1993, 60, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Fourth National Report on Human Exposure to Environmental Chemicals. Available online: https://www.cdc.gov/biomonitoring/pdf/fourthreport_updatedtables_feb2015.pdf (accessed on 11 March 2017).
- Gilbert, S.G.; Grant-Webster, K.S. Neurobehavioral effects of developmental methylmercury exposure. Environ. Health Perspect. 1995, 103, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Steuerwald, U.; Weihe, P.; Jørgensen, P.J.; Bjerve, K.; Brock, J.; Heinzow, B.; Budtz-Jørgensen, E.; Grandjean, P. Maternal seafood diet, methylmercury exposure, and neonatal neurologic function. J. Pediatr. 2000, 136, 599–605. [Google Scholar] [CrossRef] [PubMed]
- International Programme on Chemical Safety, WHO. Methylmercury (Environmental Health Criteria 101); WHO: Geneva, Switzerland, 1990. [Google Scholar]
- Caldwell, K.L.; Mortensen, M.E.; Jones, R.L.; Caudill, S.P.; Osterloh, J.D. Total blood mercury concentrations in the U.S. population: 1999–2006. Int. J. Hyg. Environ. Health 2009, 212, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Veiga, M. Mercury in artisanal gold mining in Latin America: Facts, fantasies and solutions. In Proceedings of the UNIDO—Expert Group Meeting—Introducing New Technologies for Abatement of Global Mercury Pollution Deriving from Artisanal Gold Mining, Vienna, Austria, 1–3 July 1997; p. 23.
- McDowell, M.A.; Dillon, C.F.; Osterloh, J.; Bolger, P.M.; Pellizzari, E.; Fernando, R.; Montes de Oca, R.; Schober, S.E.; Sinks, T.; Jones, R.L.; et al. Hair mercury levels in U.S. children and women of childbearing age: Reference range data from NHANES 1999–2000. Environ. Health Perspect. 2004, 112, 1165–1171. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Environmental Health Criteria, 101: Methylmercury; World Health Organization: Geneva, Switzerland, 1990. [Google Scholar]
- Joint FAO/WHO Expert Committee on Food Additives. Sixty-First Meeting. Summary and Conclusions; Joint FAO/WHO Expert Committee on Food Additives: Rome, Italy, 2003. [Google Scholar]
- Sheehan, M.C.; Burke, T.A.; Navas-Acien, A.; Breysse, P.N.; McGready, J.; Fox, M.A. Global methylmercury exposure from seafood consumption and risk of developmental neurotoxicity: A systematic review. Bull. World Health Organ. 2014, 92, 254–269. [Google Scholar] [CrossRef] [PubMed]
- Silva-Forsberg, M.C.; Forsberg, B.R.; Zeidemann, V.K. Mercury Contamination in Humans Linked to River Chemistry in the Amazon Basin P. Ambio 1999, 28, 519–521. [Google Scholar]
- Lechler, P.J.; Miller, J.R.; Lacerda, L.D.; Vinson, D.; Bonzongo, J.C.; Lyons, W.B.; Warwick, J.J. Elevated mercury concentrations in soils, sediments, water, and fish of the Madeira River basin, Brazilian Amazon: A function of natural enrichments? Sci. Total Environ. 2000, 260, 87–96. [Google Scholar] [CrossRef]
- Centro de Atención Médica Especializada y Preventiva. Mercury in Madre de Dios—Mercury Concentrations in Fish and Humans in Puerto Maldonado; CAMEP: Vista Alegre, Panama, 2013. [Google Scholar]
- Ashe, K. Elevated mercury concentrations in humans of madre de dios, Peru. PLoS ONE 2012, 7, e33305. [Google Scholar] [CrossRef] [PubMed]
- Yard, E.E.; Horton, J.; Schier, J.G.; Caldwell, K.; Sanchez, C.; Lewis, L.; Gastaňaga, C. Mercury Exposure Among Artisanal Gold Miners in Madre de Dios, Peru: A Cross-sectional Study. J. Med. Toxicol. 2012, 8, 441–448. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Water Quality Criterion for the Protection of Human Health: Methylmercury Final; EPA: Washington, DC, USA, 2001.
- Gutleb, A.C.; Schenck, C.; Staib, E. Giant otter (Pteronura brasiliensis) at risk? Total mercury and methylmercury levels in fish and otter scats, Peru. Ambio 1997, 26, 511–514. [Google Scholar]
- Luis Fernández, V.G. Niveles del Mercurio en Peces de Madre de Dios. Available online: http://www.minam.gob.pe/mineriailegal/wp-content/uploads/sites/43/2013/10/Carnegie-mercurio-Madre-de-Dios.pdf (accessed on 13 March 2017).
- Roach, K.A.; Jacobsen, N.F.; Fiorello, C.V.; Stronza, A.; Winemiller, K.O. Gold mining and mercury bioaccumulation in a floodplain lake and main channel of the Tambopata River, Perú. J. Environ. Prot. 2013, 4, 51–60. [Google Scholar] [CrossRef]
- Kohler, C.C.; Kohler, S.T.; Alcantara, F.; Isern, E.R. Development of sustainable pond aquaculture practices for Piaractus brachypomus in the Peruvian amazon. In Pond Dynamics/Aquaculture Collaborative Research Support Program Fifthteenth Annual Technical Report; Pond Dynamics/Aquaculture CRSP Management Office, Office of International Research and Development, Oregon State University: Corvallis, OR, USA, 1997; Volume 15, pp. 105–107. [Google Scholar]
- Diana, J.S. Aquaculture Production and Biodiversity Conservation. Bioscience 2009, 59, 27–38. [Google Scholar] [CrossRef]
- African Cashew Alliance. Annual Report 2012. Available online: http://www.africancashewalliance.com/sites/default/files/documents/aca-annual-report-2012.pdf (accessed on 11 March 2017).
- Flores, H.G.; Bocanegra, F.A.; Riveiro, H.S.; Quiroz, S.A. Hibridacion de paco, Piaractus brachypomus (Cuvier, 1818) por gamitana, Colossoma macropomum (Cuvier, 1818) En Iquitos -Peru. Folia Amaz. 1992, 4, 107–114. [Google Scholar]
- Morel, F.M.M.; Kraepiel, A.M.L.; Amyot, M. The Chemical Cycle and Bioaccumulation of Mercury. Annu. Rev. Ecol. Syst. 1998, 29, 543–566. [Google Scholar] [CrossRef]
- Ziesler, R.; Ardizzone, G.D. The Inland Waters of Latin America; Food and Agriculture Organization of the United Nations: Rome, Italy, 1979. [Google Scholar]
- AABP. Atrium Biodiversity Information System for the Andes to Amazon Biodiversity Program at the Botanical Research Institute of Texas. Available online: http://www.atrium-biodiversity.org/ (accessed on 13 March 2017).
- US Environmental Protection Agency. Method 7473, mercury in solids and solutions by thermal decomposition, amalgamation, and atomic absorption spectometry. In Test Methods for Evaluating Solid Waste, Physical/Chemical Methods; U.S. Government Printing Office: Washington, DC, USA, 2007; pp. 1–17. [Google Scholar]
- World Health Organization. Guidance for Identifying Populations at Risk from Mercury Exposure; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Sun, G.W.; Shook, T.L.; Kay, G.L. Inappropriate use of bivariable analysis to screen risk factors for use in multivariable analysis. J. Clin. Epidemiol. 1996, 49, 907–916. [Google Scholar] [CrossRef]
- Maldonado, G.; Greenland, S. Simulation study of confounder-selection strategies. Am. J. Epidemiol. 1993, 138, 923–936. [Google Scholar] [CrossRef] [PubMed]
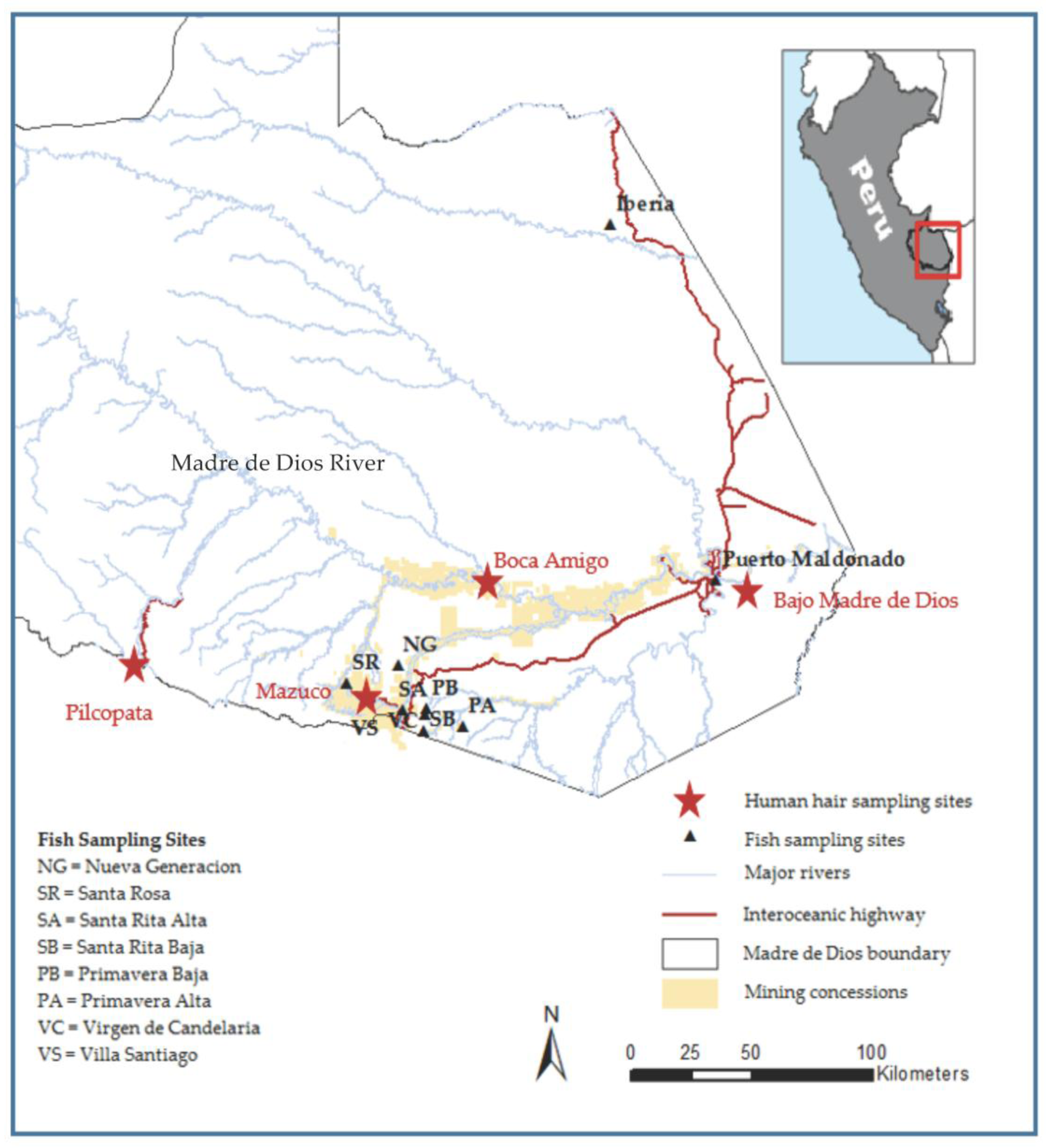
| Study Site | GPS Coordinates | Distance 1 to Headwaters (km) | District | Region | Illegal Mining Activity |
|---|---|---|---|---|---|
| Bajo Madre de Dios | 12°35′639″ S 69°09′197″ W | 228.1 | Tambopata | Madre de Dios | Yes |
| Boca Amigo | 12°36′161″ S 70°05′288″ W | 142.8 | Madre de Dios | Madre de Dios | Yes |
| Mazuco | 13°06′558″ S 70°22′770″ W | 69.4 | Inambari | Madre de Dios | Yes |
| Pilcopata | 12°54′367″ S 71°24′178″ W | 53.4 | Kosñipata | Cusco | No |
| Site | No. of Farms | No. of Samples | GPS |
|---|---|---|---|
| Iberia | 2 | 10 | 11°24′629″ S 69°23′026″ W |
| Puerto Maldonado | 3 | 10 | 29°39′033″ S 69°19′290″ W |
| Mazuco | 1 | 5 | 13°03′968″ S 70°21′145″ W |
| Nueva Generación | 1 | 5 | 12°50′225″ S 70°11′766″ W |
| Primavera Alta | 2 | 10 | 12°54′765″ S 70°09′002″ W |
| Primavera Baja | 4 | 17 | 12°54′805″ S 70°10′285″ W |
| Santa Rosa | 2 | 10 | 12°53′227″ S 70°18′669″ W |
| Santa Rita Alta | 3 | 14 | 12°54′556″ S 70°14′529″ W |
| Santa Rita Baja | 2 | 9 | 12°54′814″ S 70°12′360″ W |
| Virgen de la Candalaria | 2 | 11 | 12°52′735″ S 70°01′987″ W |
| Villa Santiago | 2 | 10 | 13°01′621″ S 70°20′954″ W |
| Total | 24 | 111 |
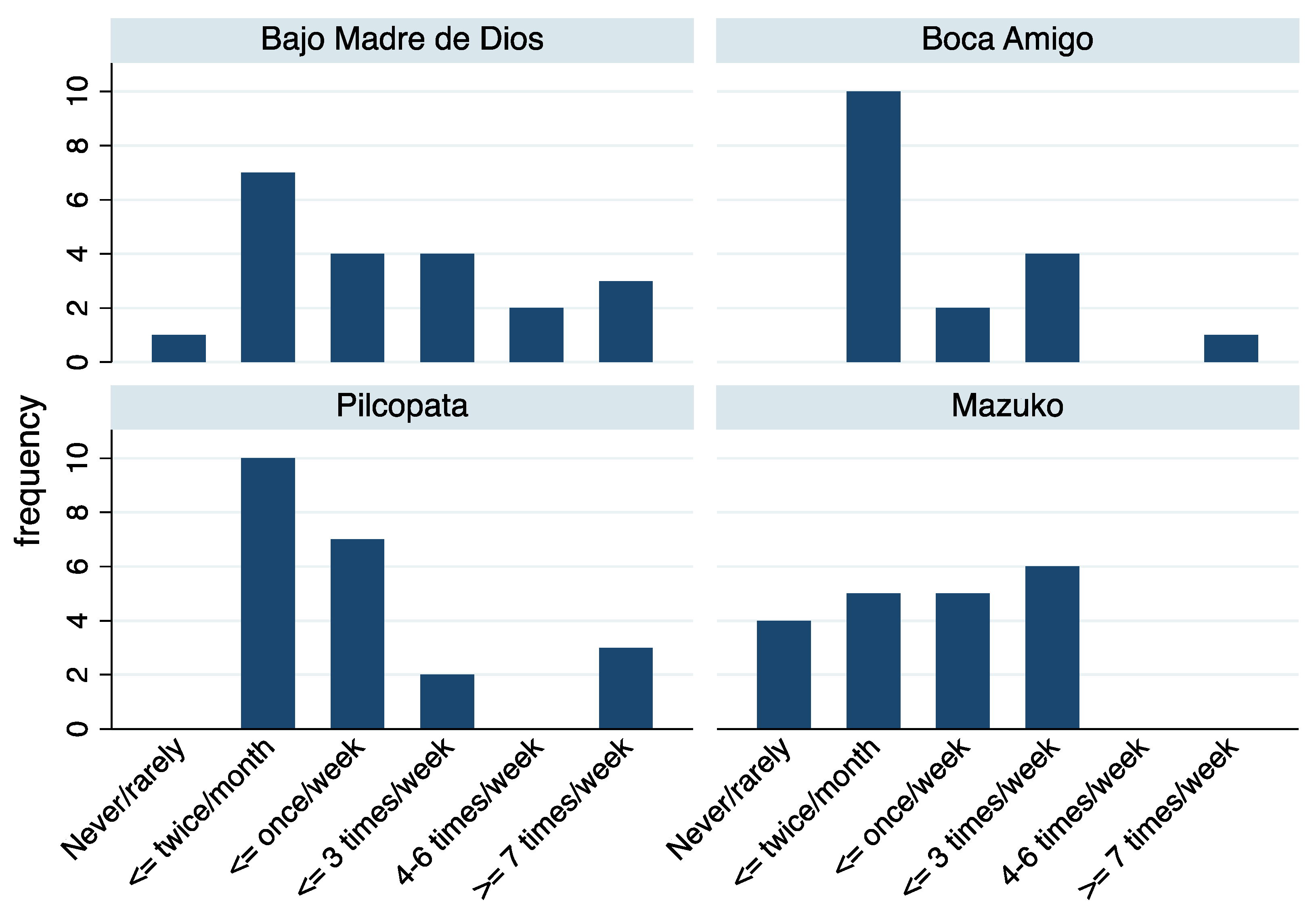
| Site | N | n Male (%) | Mean (SD) Age (Years) | Mean (SD) Residence (Years) | Mean (SD) n Fish Servings in Past 3 Days (SD) 1 | Mean n (SD) in Household | |
| Total | Children 2 | ||||||
| Overall | 81 | 39 (48.2) | 43.3 (17.7) | 16.5 (16.9) | 1.7 (2.1) | 3.9 (2.5) | 1.4 (1.5) |
| Bajo Madre de Dios | 21 | 13 (61.9) | 58.8 (16.3) | 23.5 (18.9) | 1.3 (2.2) | 3.1 (2.2) | 0.9 (1.3) |
| Boca Amigo | 17 | 7 (41.2) | 34.2 (10.7) | 3.2 (2.5) | 3.0 (2.7) | 3.5 (2.0) | 1.0 (0.9) |
| Mazuco | 23 | 10 (43.5) | 41.5 (18.0) | 24.6 (19.0) | 1.9 (2.0) | 4.3 (2.6) | 1.5 (1.5) |
| Pilcopata | 20 | 6 (30.0) | 36.6 (13.5) | 11.1 (8.8) | 0.9 (1.0) | 4.6 (3.1) | 2.1 (1.8) |
| Site | N (%) Eating Fish Species | ||||||
| Paco | Bagre | Boca chico 3 | Sabalo 3 | Doncella 3 | Zungaro 3 | ||
| 37 (45.7) | 28 (34.6) | 28 (34.6) | 18 (22.2) | 21 (25.9) | 20 (24.7) | ||
| Overall | 13 (61.9) | 10 (47.6) | 6 (28.6) | 7 (33.3) | 10 (47.6) | 8 (38.1) | |
| Bajo Madre de Dios | 7 (41.2) | 8 (47.1) | 5 (29.4) | 0 (0.0) | 10 58.8) | 9 (52.9) | |
| Boca Amigo | 7 (30.4) | 3 (13.0) | 17 (73.9) | 10 (43.5) | 0 (0.0) | 1 (4.3) | |
| Mazuco | 10 (50.0) | 7 (35.0) | 0 (0.0) | 1 (5.0) | 1 (5.0) | 2 (10) | |
| Pilcopata | |||||||
| Site/Sex | n | Range | Arithmetic Mean ± SD | Geometric Mean ± GSD | No. (%) ≥ Reference Level of 2.2 µg Hg/g Hair |
|---|---|---|---|---|---|
| Overall 1 | 80 | 0.3, 11.0 | 3.4 ± 3.0 | 2.0 ± 2.5 | 36 (45.0) 3 |
| Male | 39 | 0.3, 11.0 | 3.1 ± 2.8 | 2.0 ± 2.8 | 19 (48.7) |
| Female | 41 | 0.5, 10.1 | 3.6 ± 3.4 | 2.1 ± 2.3 | 17 (41.5) |
| Bajo Madre de Dios | 21 | 0.6, 11.0 | 4.3 ± 2.8 | 3.3 ± 2.2 2 | 14 (66.7) |
| Male | 13 | 0.6, 11.0 | 4.4 ± 0.6 | 3.2 ± 2.5 | 9 (69.2) |
| Female | 8 | 1.3, 6.5 | 4.0 ± 0.5 | 3.5 ± 1.8 | 5 (62.5) |
| Boca Amigo 1 | 17 | 2.1, 10.1 | 5.0 ± 0.4 | 4.5 ± 1.6 2 | 15 (93.8) |
| Male | 7 | 2.3, 7.9 | 4.9 ± 0.5 | 4.6 ± 1.5 | 7 (100.0) |
| Female | 10 | 2.1, 10.1 | 5.1 ± 0.6 | 4.5 ± 1.7 | 8 (88.9) |
| Pilcopata | 23 | 0.3, 2.3 | 0.9 ± 0.5 | 0.8 ± 1.7 2 | 1 (4.4) |
| Male | 13 | 0.3, 2.3 | 0.9 ± 0.1 | 0.8 ± 0.6 | 1 (7.7) |
| Female | 10 | 0.5, 1.6 | 1.0 ± 0.8 | 0.9 ± 1.5 | 0 (0.0) |
| Mazuco | 20 | 0.6, 9.2 | 2.6 ± 2.3 | 1.9 ± 2.1 2 | 6 (30.0) |
| Male | 6 | 0.9, 9.2 | 3.2 ± 0.9 | 2.2 ± 2.3 | 2 (33.3) |
| Female | 14 | 0.6, 7.3 | 2.3 ± 0.4 | 1.8 ± 2.1 | 4 (28.6) |
| Model | Adjusted R2 | Variables | Coefficient (SE) | p Value |
| Linear regression, dependent variable log-transformed total hair mercury | ||||
| 1 | 0.50 | Site (Reference: Pilcopata) | ||
| Bajo Madre de Dios | 1.41 (0.22) | <0.0001 | ||
| Boca Amigo | 1.73 (0.17) | <0.0001 | ||
| Mazuco | 0.8 (0.20) | <0.0001 | ||
| 2 | 0.51 | Site (Reference: Pilcopata) | ||
| Bajo Madre de Dios | 1.45 (0.23) | <0.0001 | ||
| Boca Amigo | 1.71 (0.18) | <0.0001 | ||
| Mazuco | 0.84 (0.20) | <0.0001 | ||
| Age | −0.002 (0.005) | 0.74 | ||
| Sex (reference: Male) | −0.004 (0.15) | 0.98 | ||
| Model | Pseudo R2 | Variables | Odds Ratio (SE) | p Value |
| Logistic regression, dependent variable total hair mercury level above reference level | ||||
| 3 | 0.39 | Site (Reference: Pilcopata) | ||
| Bajo Madre de Dios | 3.78 (1.58–5.98) | 0.001 | ||
| Boca Amigo | 5.80 (2.95–8.64) | <0.0001 | ||
| Mazuco | 2.24 (0.02–4.46) | 0.04 | ||
| 4 | 0.40 | Site (Reference: Pilcopata) | ||
| Bajo Madre de Dios | 3.93 (1.58–6.28) | 0.001 | ||
| Boca Amigo | 5.91 (3.12–8.81) | <0.0001 | ||
| Mazuco | 2.40 (0.13–4.67) | 0.03 | ||
| Age | −0.007 (−0.05–0.04) | 0.73 | ||
| Sex (reference: Male) | −0.64 (−5.33–0.23) | 0.28 | ||
| Site | n | Range Ww [Hg] (mg/kg) | Arithmetic Mean ± SD Ww [Hg] (mg/kg) | Geometric Mean ± GSD Ww [Hg] (mg/kg) 2 | No. (%) ≥ EPA Tissue Residue Criterion of 0.3 mg/kg |
|---|---|---|---|---|---|
| Overall | 111 | 0.01, 0.23 | 0.05 ± 0.04 | 0.04 ± 1.3 | 0 (0) |
| Iberia | 10 | 0.01, 0.09 | 0.04 ± 0.03 | 0.03 ± 1.5 | 0 (0) |
| Mazuco | 5 | 0.02, 0.03 | 0.03 ± 0.01 | 0.03 ± 1.1 | 0 (0) |
| Puerto Maldonado 1 | 10 | 0.02, 0.10 | 0.04 ± 0.04 | 0.03 ± 1.4 | 0 (0) |
| Nueva Generación | 5 | 0.05, 0.11 | 0.07 ± 0.02 | 0.07 ± 1.1 | 0 (0) |
| Primavera Alta | 10 | 0.04, 0.12 | 0.07 ± 0.03 | 0.06 ± 1.2 | 0 (0) |
| Primavera Baja | 18 | 0.03, 0.08 | 0.05 ± 0.02 | 0.04 ± 1.4 | 0 (0) |
| Santa Rosa | 10 | 0.02, 0.05 | 0.03 ± 0.03 | 0.03 ± 1.2 | 0 (0) |
| Santa Rita Alta | 14 | 0.02, 0.07 | 0.04 ± 0.01 | 0.04 ± 1.2 | 0 (0) |
| Santa Rita Baja | 9 | 0.03, 0.09 | 0.04 ± 0.02 | 0.04 ± 1.2 | 0 (0) |
| Virgen de la Candalaria | 11 | 0.05, 0.23 | 0.12 ± 0.06 | 0.10 ± 1.3 | 0 (0) |
| Villa Santiago | 10 | 0.05, 0.06 | 0.05 ± 0.01 | 0.05 ± 1.0 | 0 (0) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langeland, A.L.; Hardin, R.D.; Neitzel, R.L. Mercury Levels in Human Hair and Farmed Fish near Artisanal and Small-Scale Gold Mining Communities in the Madre de Dios River Basin, Peru. Int. J. Environ. Res. Public Health 2017, 14, 302. https://doi.org/10.3390/ijerph14030302
Langeland AL, Hardin RD, Neitzel RL. Mercury Levels in Human Hair and Farmed Fish near Artisanal and Small-Scale Gold Mining Communities in the Madre de Dios River Basin, Peru. International Journal of Environmental Research and Public Health. 2017; 14(3):302. https://doi.org/10.3390/ijerph14030302
Chicago/Turabian StyleLangeland, Aubrey L., Rebecca D. Hardin, and Richard L. Neitzel. 2017. "Mercury Levels in Human Hair and Farmed Fish near Artisanal and Small-Scale Gold Mining Communities in the Madre de Dios River Basin, Peru" International Journal of Environmental Research and Public Health 14, no. 3: 302. https://doi.org/10.3390/ijerph14030302
APA StyleLangeland, A. L., Hardin, R. D., & Neitzel, R. L. (2017). Mercury Levels in Human Hair and Farmed Fish near Artisanal and Small-Scale Gold Mining Communities in the Madre de Dios River Basin, Peru. International Journal of Environmental Research and Public Health, 14(3), 302. https://doi.org/10.3390/ijerph14030302






