Long Non-Coding RNA as Potential Biomarker for Prostate Cancer: Is It Making a Difference?
Abstract
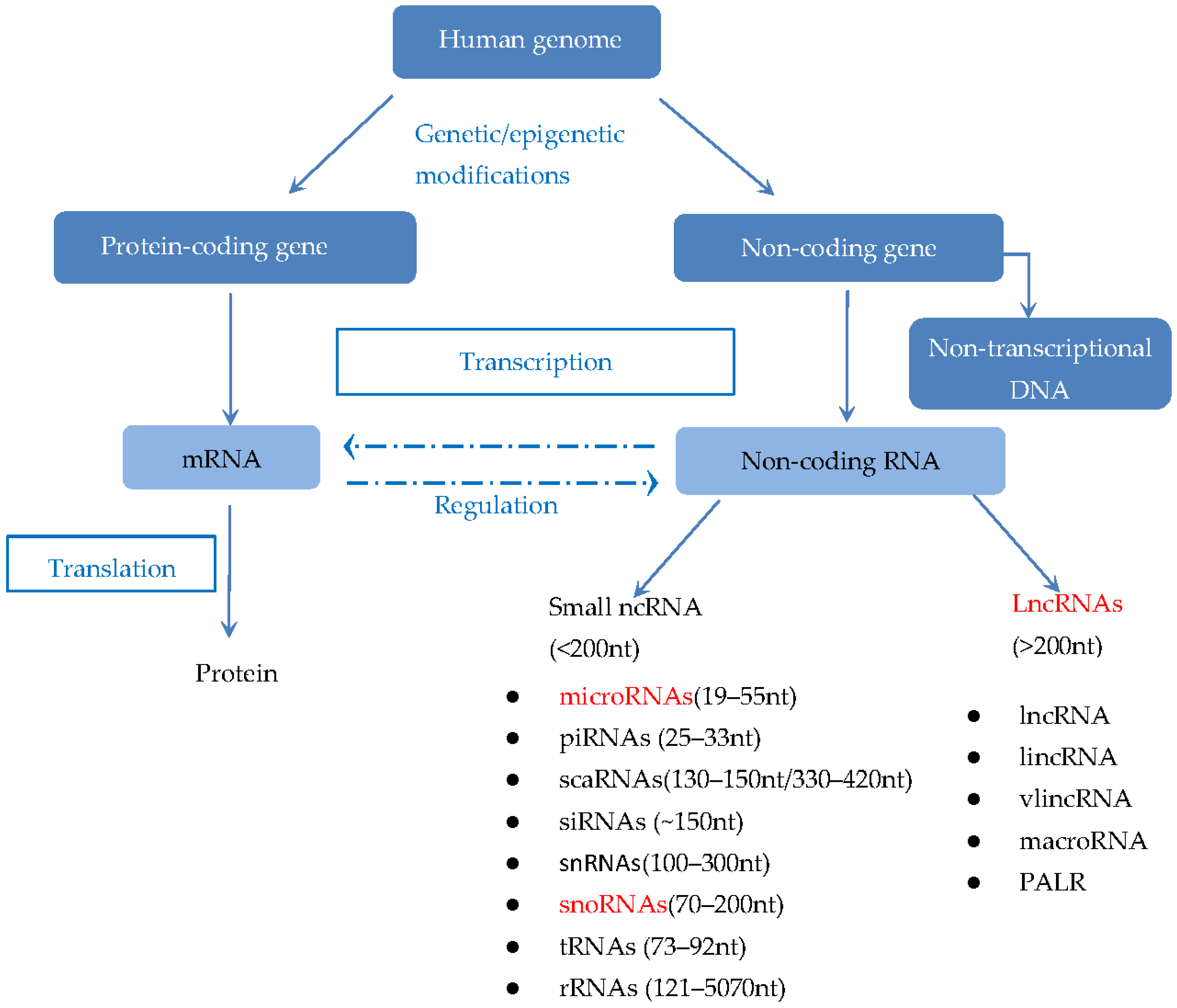
:1. Introduction
1.1. Prostate Cancer Diagnosis and Monitoring
1.2. LncRNAs in Tumor Biology
1.3. Biomarkers
2. Commercially-Available Tests for RNAs in Prostate Cancer
3. PCA3 and Its Combinations of Multiple Biomarkers
3.1. PCA3
3.2. Combinations of Multiple Biomarkers
4. Potential LncRNA Biomarkers under Investigation
4.1. LncRNAs and Prostate Cancer Risk Prediction
4.2. LncRNAs as Potential Diagnostic Biomarkers
4.3. LncRNA as Potential Prognostic Biomarkers
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- PDQ Screening and Prevention Editorial Board. Prostate Cancer Screening (PDQ®): Health Professional Version. PDQ Cancer Information Summaries; National Cancer Institute (US): Bethesda, MD, USA, 2002.
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Prostate-Specific Antigen (PSA) Test. Available online: https://www.cancer.gov/types/prostate/psa-fact-sheet (accessed on 20 October 2016).
- Catalona, W.J.; Smith, D.S.; Ratliff, T.L.; Dodds, K.M.; Coplen, D.E.; Yuan, J.J.; Petros, J.A.; Andriole, G.L. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. N. Engl. J. Med. 1991, 324, 1156–1161. [Google Scholar] [CrossRef] [PubMed]
- Oesterling, J.E. Prostate specific antigen: A critical assessment of the most useful tumor marker for adenocarcinoma of the prostate. J. Urol. 1991, 145, 907–923. [Google Scholar] [CrossRef] [PubMed]
- Barry, M.J. Clinical practice. Prostate-specific-antigen testing for early diagnosis of prostate cancer. N. Engl. J. Med. 2001, 344, 1373–1377. [Google Scholar] [CrossRef] [PubMed]
- Lilja, H.; Ulmert, D.; Vickers, A.J. Prostate-specific antigen and prostate cancer: Prediction, detection and monitoring. Nat. Rev. Cancer 2008, 8, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Schröder, F.H.; Carter, H.B.; Wolters, T.; van den Bergh, R.C.; Gosselaar, C.; Bangma, C.H.; Roobol, M.J. Early detection of prostate cancer in 2007. Part 1: PSA and PSA kinetics. Eur. Urol. 2008, 53, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Schröder, F.H.; Hugosson, J.; Roobol, M.J.; Tammela, T.L.; Ciatto, S.; Nelen, V.; Kwiatkowski, M.; Lujan, M.; Lilja, H.; Zappa, M.; et al. Screening and prostate-cancer mortality in a randomized European study. N. Engl. J. Med. 2009, 360, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Andriole, G.L.; Crawford, E.D.; Grubb, R.L.; Buys, S.S.; Chia, D.; Church, T.R.; Fouad, M.N.; Gelmann, E.P.; Kvale, P.A.; Reding, D.J.; et al. Mortality results from a randomized prostate-cancer screening trial. N. Engl. J. Med. 2009, 360, 1310–1319. [Google Scholar] [CrossRef] [PubMed]
- Hugosson, J.; Carlsson, S.; Aus, G.; Bergdahl, S.; Khatami, A.; Lodding, P.; Pihl, C.G.; Stranne, J.; Holmberg, E.; Lilja, H. Mortality results from the Göteborg randomised population-based prostate-cancer screening trial. Lancet Oncol. 2010, 11, 725–732. [Google Scholar] [CrossRef]
- Lin, H.M.; Castillo, L.; Mahon, K.L.; Chiam, K.; Lee, B.Y.; Nguyen, Q.; Boyer, M.J.; Stockler, M.R.; Pavlakis, N.; Marx, G.; et al. Circulating microRNAs are associated with docetaxel chemotherapy outcome in castration-resistant prostate cancer. Br. J. Cancer 2014, 110, 2462–2471. [Google Scholar] [CrossRef] [PubMed]
- Tannock, I.F.; de Wit, R.; Berry, W.R.; Horti, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.; Théodore, C.; James, N.D.; Turesson, I.; et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhang, H.; Wan, X.; Yang, X.; Zhu, C.; Wang, A.; He, L.; Miao, R.; Chen, S.; Zhao, H. Long noncoding RNA plays a key role in metastasis and prognosis of hepatocellular carcinoma. BioMed Res. Int. 2014, 2014, 780521. [Google Scholar] [CrossRef] [PubMed]
- Iyer, M.K.; Niknafs, Y.S.; Malik, R.; Singhal, U.; Sahu, A.; Hosono, Y.; Barrette, T.R.; Prensner, J.R.; Evans, J.R.; Zhao, S.; et al. The landscape of long noncoding RNAs in the human transcriptome. Nat. Genet. 2015, 47, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Cabili, M.N.; Trapnell, C.; Goff, L.; Koziol, M.; Tazon-Vega, B.; Regev, A.; Rinn, J.L. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev. 2011, 25, 1915–1927. [Google Scholar] [CrossRef] [PubMed]
- Rönnau, C.G.; Verhaegh, G.W.; Luna-Velez, M.V.; Schalken, J.A. Noncoding RNAs as novel biomarkers in prostate cancer. BioMed Res. Int. 2014, 2014, 591703. [Google Scholar] [CrossRef] [PubMed]
- St Laurent, G.; Wahlestedt, C.; Kapranov, P. The Landscape of long noncoding RNA classification. Trends Genet. 2015, 31, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Chinnaiyan, A.M. The emergence of lncRNAs in cancer biology. Cancer Discov. 2011, 1, 391–407. [Google Scholar] [CrossRef] [PubMed]
- Wilusz, J.E.; Sunwoo, H.; Spector, D.L. Long noncoding RNAs: Functional surprises from the RNA world. Genes Dev. 2009, 23, 1494–1504. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.D.; Sung, S. Long noncoding RNA: Unveiling hidden layer of gene regulatory networks. Trends Plant Sci. 2012, 17, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.H.; Abdelmohsen, K.; Gorospe, M. Posttranscriptional gene regulation by long noncoding RNA. J. Mol. Biol. 2013, 425, 3723–3730. [Google Scholar] [CrossRef] [PubMed]
- Bolton, E.M.; Tuzova, A.V.; Walsh, A.L.; Lynch, T.; Perry, A.S. Noncoding RNAs in prostate cancer: The long and the short of it. Clin. Cancer Res. 2014, 20, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Rubin, M.A.; Wei, J.T.; Chinnaiyan, A.M. Beyond PSA: The next generation of prostate cancer biomarkers. Sci. Transl. Med. 2012, 4, 127rv3. [Google Scholar] [CrossRef] [PubMed]
- Fraser, M.; Berlin, A.; Bristow, R.G.; van der Kwast, T. Genomic, pathological, and clinical heterogeneity as drivers of personalized medicine in prostate cancer. Urol. Oncol. 2015, 33, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Cuzick, J.; Swanson, G.P.; Fisher, G.; Brothman, A.R.; Berney, D.M.; Reid, J.E.; Mesher, D.; Speights, V.O.; Stankiewicz, E.; Foster, C.S.; et al. Prognostic value of an RNA expression signature derived from cell cycle proliferation genes in patients with prostate cancer: A retrospective study. Lancet Oncol. 2011, 12, 245–255. [Google Scholar] [CrossRef]
- Bishoff, J.T.; Freedland, S.J.; Gerber, L.; Tennstedt, P.; Reid, J.; Welbourn, W.; Graefen, M.; Sangale, Z.; Tikishvili, E.; Park, J.; et al. Prognostic utility of the cell cycle progression score generated from biopsy in men treated with prostatectomy. J. Urol. 2014, 192, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Cuzick, J.; Berney, D.M.; Fisher, G.; Mesher, D.; Moller, H.; Reid, J.E.; Perry, M.; Park, J.; Younus, A.; Gutin, A.; et al. Prognostic value of a cell cycle progression signature for prostate cancer death in a conservatively managed needle biopsy cohort. Br. J. Cancer 2012, 106, 1095–1099. [Google Scholar] [CrossRef] [PubMed]
- Erho, N.; Crisan, A.; Vergara, I.A.; Mitra, A.P.; Ghadessi, M.; Buerki, C.; Bergstralh, E.J.; Kollmeyer, T.; Fink, S.; Haddad, Z.; et al. Discovery and validation of a prostate cancer genomic classifier that predicts early metastasis following radical prostatectomy. PLoS ONE 2013, 8, e66855. [Google Scholar] [CrossRef] [PubMed]
- Den, R.B.; Feng, F.Y.; Showalter, T.N.; Mishra, M.V.; Trabulsi, E.J.; Lallas, C.D.; Gomella, L.G.; Kelly, W.K.; Birbe, R.C.; McCue, P.A.; et al. Genomic prostate cancer classifier predicts biochemical failure and metastases in patients after postoperative radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 1038–1046. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.A.; Yousefi, K.; Haddad, Z.; Choeurng, V.; Buerki, C.; Stephenson, A.J.; Li, J.; Kattan, M.W.; MagiGalluzzi, C.; Davicioni, E. A Genomic Classifier Improves Prediction of Metastatic Disease within 5 Years after Surgery in Node-negative High-risk Prostate Cancer Patients Managed by Radical Prostatectomy without Adjuvant Therapy. Eur. Urol. 2015, 67, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.A.; Cooperberg, M.R.; Magi-Galluzzi, C.; Simko, J.P.; Falzarano, S.M.; Maddala, T.; Chan, J.M.; Li, J.; Cowan, J.E.; Tsiatis, A.C.; et al. A 17-gene Assay to Predict Prostate Cancer Aggressiveness in the Context of Gleason Grade Heterogeneity, Tumor Multifocality, and Biopsy Undersampling. Eur. Urol. 2014, 66, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Knezevic, D.; Goddard, A.D.; Natraj, N.; Cherbavaz, D.B.; Clark-Langone, K.M.; Snable, J.; Watson, D.; Falzarano, S.M.; Magi-Galluzzi, C.; Klein, E.A.; et al. Analytical validation of the Oncotype DX prostate cancer assay—A clinical RT-PCR assay optimized for prostate needle biopsies. BMC Genom. 2013, 14, 690. [Google Scholar] [CrossRef] [PubMed]
- Cullen, J.; Rosner, I.L.; Brand, T.C.; Zhang, N.; Tsiatis, A.C.; Moncur, J.; Ali, A.; Chen, Y.; Knezevic, D.; Maddala, T.; et al. A Biopsy-based 17-gene Genomic Prostate Score Predicts Recurrence after Radical Prostatectomy and Adverse Surgical Pathology in a Racially Diverse Population of Men with Clinically Low- and Intermediate-risk Prostate Cancer. Eur. Urol. 2015, 68, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Groskopf, J.; Aubin, S.M.; Deras, I.L.; Blase, A.; Bodrug, S.; Clark, C.; Brentano, S.; Mathis, J.; Pham, J.; Meyer, T.; et al. APTIMA PCA3 molecular urine test: Development of a method to aid in the diagnosis of prostate cancer. Clin. Chem. 2006, 52, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Bussemakers, M.J.; van Bokhoven, A.; Verhaegh, G.W.; Smit, F.P.; Karthaus, H.F.; Schalken, J.A.; Debruyne, F.M.; Ru, N.; Isaacs, W.B. DD3: A new prostate-specific gene, highly overexpressed in prostate cancer. Cancer Res. 1999, 59, 5975–5979. [Google Scholar] [PubMed]
- De Kok, J.B.; Verhaegh, G.W.; Roelofs, R.W.; Hessels, D.; Kiemeney, L.A.; Aalders, T.W.; Swinkels, D.W.; Schalken, J.A. DD3(PCA3), a very sensitive and specific marker to detect prostate tumors. Cancer Res. 2002, 62, 2695–2698. [Google Scholar] [PubMed]
- Deras, I.L.; Aubin, S.M.; Blase, A.; Day, J.R.; Koo, S.; Partin, A.W.; Ellis, W.J.; Marks, L.S.; Fradet, Y.; Rittenhouse, H.; et al. PCA3: A molecular urine assay for predicting prostate biopsy outcome. J. Urol. 2008, 179, 1587–1592. [Google Scholar] [CrossRef] [PubMed]
- Vlaeminck-Guillem, V.; Bandel, M.; Cottancin, M.; Rodriguez-Lafrasse, C.; Bohbot, J.M.; Sednaoui, P. Chronic prostatitis does not influence urinary PCA3 score. Prostate 2012, 72, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hessels, D.; Klein Gunnewiek, J.M.; van Oort, I.; Karthaus, H.F.; van Leenders, G.J.; van Balken, B.; Kiemeney, L.A.; Witjes, J.A.; Schalken, J.A. DD3(PCA3)-based molecular urine analysis for the diagnosis of prostate cancer. Eur. Urol. 2003, 44, 8–15. [Google Scholar] [CrossRef]
- Neveu, B.; Jain, P.; Têtu, B.; Wu, L.; Fradet, Y.; Pouliot, F. A PCA3 gene-based transcriptional amplification system targeting primary prostate cancer. Oncotarget 2016, 7, 1300–1310. [Google Scholar] [PubMed]
- Nicholson, A.; Mahon, J.; Boland, A.; Beale, S.; Dwan, K.; Fleeman, N.; Hockenhull, J.; Dundar, Y. The clinical effectiveness and cost-effectiveness of the PROGENSA® prostate cancer antigen 3 assay and the Prostate Health Index in the diagnosis of prostate cancer: A systematic review and economic evaluation. Health Technol. Assess. 2015, 19, 1–191. [Google Scholar] [CrossRef] [PubMed]
- Aubin, S.M.; Reid, J.; Sarno, M.J.; Blase, A.; Aussie, J.; Rittenhouse, H.; Rittmaster, R.; Andriole, G.L.; Groskopf, J. PCA3 molecular urine test for predicting repeat prostate biopsy outcome in populations at risk: Validation in the placebo arm of the dutasteride REDUCE trial. J. Urol. 2010, 184, 1947–1952. [Google Scholar] [CrossRef] [PubMed]
- Crawford, E.D.; Rove, K.O.; Trabulsi, E.J.; Qian, J.; Drewnowska, K.P.; Kaminetsky, J.C.; Huisman, T.K.; Bilowus, M.L.; Freedman, S.J.; Glover, W.L.; et al. Diagnostic performance of PCA3 to detect prostate cancer in men with increased prostate specific antigen: A prospective study of 1962 cases. J. Urol. 2012, 188, 1726–1731. [Google Scholar] [CrossRef] [PubMed]
- Gittelman, M.C.; Hertzman, B.; Bailen, J.; Williams, T.; Koziol, I.; Henderson, R.J.; Efros, M.; Bidair, M.; Ward, J.F. PCA3 molecular urine test as a predictor of repeat prostate biopsy outcome in men with previous negative biopsies: A prospective multicenter clinical study. J. Urol. 2013, 190, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Ramos, C.G.; Valdevenito, R.; Vergara, I.; Anabalon, P.; Sanchez, C.; Fulla, J. PCA3 sensitivity and specificity for prostate cancer detection in patients with abnormal PSA and/or suspicious digital rectal examination. First Latin American experience. Urol. Oncol. 2013, 31, 1522–1526. [Google Scholar] [CrossRef] [PubMed]
- De la Taille, A.; Irani, J.; Graefen, M.; Chun, F.; de Reijke, T.; Kil, P.; Gontero, P.; Mottaz, A.; Haese, A. Clinical evaluation of the PCA3 assay in guiding initial biopsy decisions. J. Urol. 2011, 185, 2119–2125. [Google Scholar] [CrossRef] [PubMed]
- Durand, X.; Moutereau, S.; Xylinas, E.; de la Taille, A. Progensa™ PCA3 test for prostate cancer. Expert Rev. Mol. Diagn. 2011, 11, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.; Auprich, M.; Ahyai, S.A.; de la Taille, A.; van Poppel, H.; Marberger, M.; Stenzl, A.; Mulders, P.F.; Huland, H.; Fisch, M.; et al. Initial prostate biopsy: Development and internal validation of a biopsy-specific nomogram based on the prostate cancer antigen 3 assay. Eur. Urol. 2013, 63, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Tombal, B.; Andriole, G.L.; de la Taille, A.; Gontero, P.; Haese, A.; Remzi, M.; Speakman, M.; Smets, L.; Stoevelaar, H. Clinical judgment versus biomarker prostate cancer gene 3: Which is best when determining the need for repeat prostate biopsy? Urology 2013, 81, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.T.; Feng, Z.; Partin, A.W.; Brown, E.; Thompson, I.; Sokoll, L.; Chan, D.W.; Lotan, Y.; Kibel, A.S.; Busby, J.E.; et al. Can Urinary PCA3 Supplement PSA in the Early Detection of Prostate Cancer? J. Clin. Oncol. 2014, 32, 4066–4072. [Google Scholar] [CrossRef] [PubMed]
- Moradi Sardareh, H.; Goodarzi, M.T.; Yadegar-Azari, R.; Poorolajal, J.; Mousavi-Bahar, S.H.; Saidijam, M. Prostate cancer antigen 3 gene expression in peripheral blood and urine sediments from prostate cancer and benign prostatic hyperplasia patients versus healthy individuals. Urol. J. 2014, 11, 1952–1958. [Google Scholar] [PubMed]
- Tomlins, S.A.; Day, J.R.; Lonigro, R.J.; Hovelson, D.H.; Siddiqui, J.; Kunju, L.P.; Dunn, R.L.; Meyer, S.; Hodge, P.; Groskopf, J.; et al. Urine TMPRSS2:ERG Plus PCA3 for Individualized Prostate Cancer Risk Assessment. Eur. Urol. 2016, 70, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Rigau, M.; Morote, J.; Mir, M.C.; Ballesteros, C.; Ortega, I.; Sanchez, A.; Colás, E.; Garcia, M.; Ruiz, A.; Abal, M.; et al. PSGR and PCA3 as biomarkers for the detection of prostate cancer in urine. Prostate 2010, 70, 1760–1767. [Google Scholar] [CrossRef] [PubMed]
- Cornu, J.N.; Cancel-Tassin, G.; Egrot, C.; Gaffory, C.; Haab, F.; Cussenot, O. Urine TMPRSS2:ERG fusion transcript integrated with PCA3 score, genotyping, and biological features are correlated to the results of prostatic biopsies in men at risk of prostate cancer. Prostate 2013, 73, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Leyten, G.H.; Hessels, D.; Jannink, S.A.; Smit, F.P.; de Jong, H.; Cornel, E.B.; de Reijke, T.M.; Vergunst, H.; Kil, P.; Knipscheer, B.C.; et al. Prospective multicentre evaluation of PCA3 and TMPRSS2-ERG gene fusions as diagnostic and prognostic urinary biomarkers for prostate cancer. Eur. Urol. 2014, 65, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.B.; Wang, Z.; Yeboah, E.D.; Tettey, Y.; Biritwum, R.B.; Adjei, A.A.; Tay, E.; Truelove, A.; Niwa, S.; Chung, C.C.; et al. A genome-wide association study of prostate cancer in West African men. Hum. Genet. 2014, 133, 509–521. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsson, J.; Sulem, P.; Manolescu, A.; Amundadottir, L.T.; Gudbjartsson, D.; Helgason, A.; Rafnar, T.; Bergthorsson, J.T.; Agnarsson, B.A.; Baker, A.; et al. Genome-wide association study identifies a second prostate cancer susceptibility variant at 8q24. Nat. Genet. 2007, 39, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Al Olama, A.A.; Kote-Jarai, Z.; Giles, G.G.; Guy, M.; Morrison, J.; Severi, G.; Leongamornlert, D.A.; Tymrakiewicz, M.; Jhavar, S.; Saunders, E.; et al. Multiple loci on 8q24 associated with prostate cancer susceptibility. Nat. Genet. 2009, 41, 1058–1060. [Google Scholar] [CrossRef] [PubMed]
- Ahmadiyeh, N.; Pomerantz, M.M.; Grisanzio, C.; Herman, P.; Jia, L.; Almendro, V.; He, H.H.; Brown, M.; Liu, X.S.; Davis, M.; et al. 8q24 prostate, breast, and colon cancer risk loci show tissue-specific long-range interaction with MYC. Proc. Natl. Acad. Sci. USA 2010, 107, 9742–9746. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Iyer, M.K.; Balbin, O.A.; Dhanasekaran, S.M.; Cao, Q.; Brenner, J.C.; Laxman, B.; Asangani, I.A.; Grasso, C.S.; Kominsky, H.D.; et al. Transcriptome sequencing across a prostate cancer cohort identifies PCAT-1, an unannotated lincRNA implicated in disease progression. Nat. Biotechnol. 2011, 29, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Chen, W.; Han, S.; Iyer, M.K.; Cao, Q.; Kothari, V.; Evans, J.R.; Knudsen, K.E.; Paulsen, M.T.; Ljungman, M.; et al. The long non-coding RNA PCAT-1 promotes prostate cancer cell proliferation through cMyc. Neoplasia 2014, 16, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Ahmed, M.; Zhang, F.; Yao, C.Q.; Li, S.; Liang, Y.; Hua, J.; Soares, F.; Sun, Y.; Langstein, J.; et al. Modulation of long noncoding RNAs by risk SNPs underlying genetic predispositions to prostate cancer. Nat. Genet. 2016, 48, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Srikantan, V.; Zou, Z.; Petrovics, G.; Xu, L.; Augustus, M.; Davis, L.; Livezey, J.R.; Connell, T.; Sesterhenn, I.A.; Yoshino, K.; et al. PCGEM1, a prostate-specific gene, is overexpressed in prostate cancer. Proc. Natl. Acad. Sci. USA 2000, 97, 12216–12221. [Google Scholar] [CrossRef] [PubMed]
- Petrovics, G.; Zhang, W.; Makarem, M.; Street, J.P.; Connelly, R.; Sun, L.; Sesterhenn, I.A.; Srikantan, V.; Moul, J.W.; Srivastava, S. Elevated expression of PCGEM1, a prostate-specific gene with cell growth-promoting function, is associated with high-risk prostate cancer patients. Oncogene 2004, 23, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Wang, M.; Kang, M.; Wang, Q.; Wu, B.; Chu, H.; Zhong, D.; Qin, C.; Yin, C.; Zhang, Z.; Wu, D. Association between lncRNA PCGEM1 polymorphisms and prostate cancer risk. Prostate Cancer Prostatic Dis. 2013, 16, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Liu, Y.; Xu, W.; Sun, Y.; Lu, J.; Wang, F.; Wei, M.; Shen, J.; Hou, J.; Gao, X.; et al. Long noncoding RNA MALAT-1 is a new potential therapeutic target for castration resistant prostate cancer. J. Urol. 2013, 190, 2278–2287. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Ren, S.; Chen, R.; Lu, J.; Shi, X.; Zhu, Y.; Zhang, W.; Jing, T.; Zhang, C.; Shen, J.; et al. Development and prospective multicenter evaluation of the long noncoding RNA MALAT-1 as a diagnostic urinary biomarker for prostate cancer. Oncotarget 2014, 5, 11091–11102. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Wang, F.; Shen, J.; Sun, Y.; Xu, W.; Lu, J.; Wei, M.; Xu, C.; Wu, C.; Zhang, Z.; et al. Long non-coding RNA metastasis associated in lung adenocarcinoma transcript 1 derived miniRNA as a novel plasma-based biomarker for diagnosing prostate cancer. Eur. J. Cancer 2013, 49, 2949–2959. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.; Zhou, C.X.; Shi, Y.B.; Lu, H.; He, X.Z. MD-miniRNA could be a more accurate biomarker for prostate cancer screening compared with serum prostate-specific antigen level. Tumour Biol. 2015, 36, 3541–3547. [Google Scholar] [CrossRef] [PubMed]
- Crea, F.; Watahiki, A.; Quagliata, L.; Xue, H.; Pikor, L.; Parolia, A.; Wang, Y.; Lin, D.; Lam, W.L.; Farrar, W.L.; et al. Identification of a long non-coding RNA as a novel biomarker and potential therapeutic target for metastatic prostate cancer. Oncotarget 2014, 5, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Peng, Z.; Mao, J.H.; Yu, Y.; Yin, C.; Gao, X.; Cui, Z.; Zhang, J.; Yi, K.; Xu, W.; et al. RNA-seq analysis of prostate cancer in the Chinese population identifies recurrent gene fusions, cancer-associated long noncoding RNAs and aberrant alternative splicings. Cell Res. 2012, 22, 806–821. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ren, S.C.; Shi, X.L.; Liu, Y.W.; Zhu, Y.S.; Jing, T.L.; Wang, F.B.; Chen, R.; Xu, C.L.; Wang, H.Q.; et al. A novel urinary long non-coding RNA transcript improves diagnostic accuracy in patients undergoing prostate biopsy. Prostate 2015, 75, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Mehra, R.; Shi, Y.; Udager, A.M.; Prensner, J.R.; Sahu, A.; Iyer, M.K.; Siddiqui, J.; Cao, X.; Wei, J.; Jiang, H.; et al. A novel RNA in situ hybridization assay for the long noncoding RNA SChLAP1 predicts poor clinical outcome after radical prostatectomy in clinically localized prostate cancer. Neoplasia 2014, 16, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Zhao, S.; Erho, N.; Schipper, M.; Iyer, M.K.; Dhanasekaran, S.M.; Magi-Galluzzi, C.; Mehra, R.; Sahu, A.; Siddiqui, J.; et al. RNA biomarkers associated with metastatic progression in prostate cancer: A multi-institutional high-throughput analysis of SChLAP1. Lancet Oncol. 2014, 15, 1469–1480. [Google Scholar] [CrossRef]
- Mehra, R.; Udager, A.M.; Ahearn, T.U.; Cao, X.; Feng, F.Y.; Loda, M.; Petimar, J.S.; Kantoff, P.; Mucci, L.A.; Chinnaiyan, A.M. Overexpression of the Long Non-coding RNA SChLAP1 Independently Predicts Lethal Prostate Cancer. Eur. Urol. 2016, 70, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cheng, G.; Li, X.; Pan, Y.; Qin, C.; Yang, H.; Hua, L.; Wang, Z. Overexpression of long non-coding RNA LOC400891 promotes tumor progression and poor prognosis in prostate cancer. Tumour Biol. 2016, 37, 9603–9613. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.Y.; Gao, Y.; Wang, X.J.; Ruan, Y.; Bei, X.Y.; Wang, X.H.; Jing, Y.F.; Zhao, W.; Jiang, Q.; Li, J.; et al. Long non-coding RNA lnc-MX1-1 is associated with poor clinical features and promotes cellular proliferation and invasiveness in prostate cancer. Biochem. Biophys. Res. Commun. 2016, 470, 721–727. [Google Scholar] [CrossRef] [PubMed]
- White, N.M.; Zhao, S.G.; Zhang, J.; Rozycki, E.B.; Dang, H.X.; McFadden, S.D.; Eteleeb, A.M.; Alshalalfa, M.; Vergara, I.A.; Erho, N.; et al. Multi-institutional Analysis Shows that Low PCAT-14 Expression Associates with Poor Outcomes in Prostate Cancer. Eur. Urol. 2017, 71, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Zhang, X.; Niknafs, Y.S.; Xiao, L.; Mehra, R.; Cieślik, M.; Ross, A.; Schaeffer, E.; Malik, B.; Guo, S.; et al. Identification and Validation of PCAT14 as Prognostic Biomarker in Prostate Cancer. Neoplasia 2016, 18, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Işın, M.; Uysaler, E.; Özgür, E.; Köseoğlu, H.; Şanlı, Ö.; Yücel, Ö.B.; Gezer, U.; Dalay, N. Exosomal lncRNA-p21 levels may help to distinguish prostate cancer from benign disease. Front. Genet. 2015, 6, 168. [Google Scholar] [PubMed]
- Zheng, J.; Zhao, S.; He, X.; Zheng, Z.; Bai, W.; Duan, Y.; Cheng, S.; Wang, J.; Liu, X.; Zhang, G. The up-regulation of long non-coding RNA CCAT2 indicates a poor prognosis for prostate cancer and promotes metastasis by affecting epithelial-mesenchymal transition. Biochem. Biophys. Res. Commun. 2016, 480, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, P.; Wan, X.; Su, X.; Kong, Z.; Zhai, Q.; Xiang, X.; Li, L.; Li, Y. Downregulation of long non-coding RNA HCG11 predicts a poor prognosis in prostate cancer. Biomed. Pharmacother. 2016, 83, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Yi, X.M.; Tang, C.P.; Ge, J.P.; Zhang, Z.Y.; Zhou, W.Q. Long non-coding RNA ATB promotes growth and epithelial-mesenchymal transition and predicts poor prognosis in human prostate carcinoma. Oncol. Rep. 2016, 36, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; German, R.; King, J.; Joseph, D.; Thompson, T.; Wu, X.C.; Ajani, U.; Tai, E. Recent trends in prostate cancer testing and incidence among men under age of 50. Cancer Epidemiol. 2012, 36, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.L.; Gu, C.Y.; Zhu, Y.; Dai, B.; Zhang, H.L.; Shi, G.H.; Shen, Y.J.; Zhu, Y.P.; Ma, C.G.; Xiao, W.J.; et al. Polymorphisms at long non-coding RNAs and prostate cancer risk in an eastern Chinese population. Prostate Cancer Prostatic Dis. 2014, 17, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Ji, P.; Diederichs, S.; Wang, W.; Böing, S.; Metzger, R.; Schneider, P.M.; Tidow, N.; Brandt, B.; Buerger, H.; Bulk, E.; et al. MALAT-1, a novel noncoding RNA, and thymosin beta4 predict metastasis and survival in early-stage non-small cell lung cancer. Oncogene 2003, 22, 8031–8041. [Google Scholar] [CrossRef] [PubMed]
- Rak, J. Extracellular vesicles—Biomarkers and effectors of the cellular interactome in cancer. Front. Pharmacol. 2013, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Thakur, B.K.; Zhang, H.; Becker, A.; Matei, I.; Huang, Y.; Costa-Silva, B.; Zheng, Y.; Hoshino, A.; Brazier, H.; Xiang, J.; et al. Double stranded DNA in exosomes: A novel biomarker in cancer detection. Cell Res. 2014, 24, 766–769. [Google Scholar] [CrossRef] [PubMed]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Gezer, U.; Özgür, E.; Cetinkaya, M.; Isin, M.; Dalay, N. Long non-coding RNAs with low expression levels in cells are enriched in secreted exosomes. Cell Biol. Int. 2014, 38, 1076–1079. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Kikyo, N. Strategies to identify long noncoding RNAs involved in gene regulation. Cell Biosci. 2012, 2, 37. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Zhang, Z.; Bailey, T.L.; Perkins, A.C.; Tallack, M.R.; Xu, Z.; Liu, H. Prediction of novel long non-coding RNAs based on RNA-Seq data of mouse Klf1 knockout study. BMC Bioinf. 2012, 13, 331. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.M.; Ma, Z.W.; Wang, Q.; Wang, J.N.; Yang, J.W.; Li, X.D.; Li, H.; Men, T.Y. A new RNA-seq method to detect the transcription and non-coding RNA in prostate cancer. Pathol. Oncol. Res. 2014, 20, 43–50. [Google Scholar] [CrossRef] [PubMed]
| References | Name (Vendor) | Number of Genes | Discovery Approach |
|---|---|---|---|
| [26] | Prolaris® (Myriad Genetics) | 31 CCP test genes | Candidate gene approach, 126 cell cycle and cellular proliferation genes tested in prostatectomy tissues |
| [29] | Decipher® Prostate Cancer Classifier (GenomeDx) | 22 GC test genes | Whole transcriptome analysis Affymetrix GeneChip® Human Exon 1.0 ST Array used to assess prostatectomy tissues |
| [32] | OncotypeDX® Prostate Cancer Assay (Genomic Health) | 12 GPS test genes | Candidate gene approach, 727 genes tested in prostatectomy tissues Refined panel of 81 genes tested in biopsies |
| Model | Prediction | n | AUC | p Value vs. PSA | p Value vs. T2:ERG or PCA3 |
|---|---|---|---|---|---|
| PSA | Cancer | 1225 | 0.585 | NA | NA |
| PSA plus T2:ERG | 0.693 | <0.001 | NA | ||
| PSA plus PCA3 | 0.726 | <0.001 | <0.05 | ||
| PSA plus T2:ERG plus PCA3 (MiPS) | 0.751 | <0.001 | <0.001, <0.001 | ||
| PSA | HG cancer | 1225 | 0.651 | NA | NA |
| PSA plus T2:ERG | 0.729 | <0.001 | NA | ||
| PSA plus PCA3 | 0.747 | <0.001 | NS | ||
| PSA plus T2:ERG plus PCA3 (MiPShg) | 0.772 | <0.001 | <0.01, <0.001 |
| Model | Prostate Biopsies Avoided (n = 443) n (%) | Prostate Cancers Missed (n = 196) n (%) | Prostate Cancers Gleason ≥ 7 Missed (n = 115) n (%) |
|---|---|---|---|
| PCA3 score ≥ 25 | 166 (37) | 37 (19) | 20 (17) |
| PCA3 score ≥ 35 | 211 (48) | 62 (32) | 36 (31) |
| TMPRSS2-ERG ≥ 10 | 382 (86) | 150 (77) | 75 (65) |
| PCA3-25 plus TMPRSS2-ERG | 153 (35) | 26 (13) | 11 (10) |
| PCA3-35 plus TMPRSS2-ERG | 195 (44) | 48 (24) | 24 (21) |
| References | lncRNA | Alteration in Prostate Cancer | Location | Clinical Association | Description |
|---|---|---|---|---|---|
| [57] | lncRNA RP11-543F8.2 | Unknown | Unknown | Risk prediction | A set of promising susceptibility loci |
| [58,59,60,61,62,63] | PCAT1 | Upregulation | Tissues, plasma | Risk prediction | Promoting prostate cancer cell proliferation and tumor growth |
| [64,65,66] | PCGEM1 | Upregulation | Tissues | Risk prediction | Polymorphisms were associated with an increased risk of prostate cancer |
| [67,68] | MALAT1 | Upregulation | Tissues, urinary | Diagnosis | Preventing unnecessary biopsies |
| [69,70] | MALAT-1 derived miniRNA (MD-miniRNA) | Upregulation | Plasma | Diagnosis | Higher sensitivity, specificity, and accuracy |
| [71] | PCAT-18 | Upregulation | Tissues, plasma | Diagnosis | A potential therapeutic target and biomarker for metastatic prostate cancer |
| [72,73] | lncRNA FR0348383 | Upregulation | Tissues | Diagnosis | More outstanding performance than PSA |
| [74,75,76] | SChLAP1 | Upregulation | Tissues | Prognosis | Independently predicting the poor clinical outcomes |
| [77] | lncRNA LOC400891 | Upregulation | Tissues | Prognosis | As an independent predictor for biochemical recurrence-free survival of PCa |
| [78] | lnc-MX1-1 | Upregulation | Tissues | Prognosis | Relevant to patients‘ clinical features |
| [79,80] | PCAT14 | Upregulation | Tissues | Prognosis | Highly expressed in low grade disease and loss of PCAT14 predicts for disease aggressiveness and recurrence. |
| [81] | lincRNA-p21 | Upregulation | Exosomes | Prognosis | A promising prognostic biomarker for the detection and stratification of PCa |
| [82] | CCAT2 | Upregulation | Tissues | Prognosis | High CCAT2 expression level had poorer overall survival and progression-free survival |
| [83] | HCG11 | Downregulation | Tissues | Prognosis | Downregulation of HCG11 expression in tissues was associated with poor survival of PCa patients. |
| [84] | ATB | Upregulation | Tissues | Prognosis | High lncRNA-ATB expression may be an independent prognostic factor for biochemical recurrence (BCR)-free survival in prostate cancer patients. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, J.; Tang, J.; Wang, G.; Zhu, Y.-S. Long Non-Coding RNA as Potential Biomarker for Prostate Cancer: Is It Making a Difference? Int. J. Environ. Res. Public Health 2017, 14, 270. https://doi.org/10.3390/ijerph14030270
Deng J, Tang J, Wang G, Zhu Y-S. Long Non-Coding RNA as Potential Biomarker for Prostate Cancer: Is It Making a Difference? International Journal of Environmental Research and Public Health. 2017; 14(3):270. https://doi.org/10.3390/ijerph14030270
Chicago/Turabian StyleDeng, Junli, Jie Tang, Guo Wang, and Yuan-Shan Zhu. 2017. "Long Non-Coding RNA as Potential Biomarker for Prostate Cancer: Is It Making a Difference?" International Journal of Environmental Research and Public Health 14, no. 3: 270. https://doi.org/10.3390/ijerph14030270
APA StyleDeng, J., Tang, J., Wang, G., & Zhu, Y.-S. (2017). Long Non-Coding RNA as Potential Biomarker for Prostate Cancer: Is It Making a Difference? International Journal of Environmental Research and Public Health, 14(3), 270. https://doi.org/10.3390/ijerph14030270





