Spatial, Temporal, and Dietary Variables Associated with Elevated Mercury Exposure in Peruvian Riverine Communities Upstream and Downstream of Artisanal and Small-Scale Gold Mining
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Background and Population
2.2. Study Design and Data Collection
2.3. Household Food Consumption
2.4. Hair Mercury Analysis
2.5. Nail Selenium Analysis
2.6. Statistical Analysis
2.6.1. Relationship between Environmental and Human Exposure
2.6.2. Temporal Variation in Hair Mercury
2.6.3. Risk Factors Related to Hair Mercury
3. Results
3.1. Population Characteristics for Hair Mercury
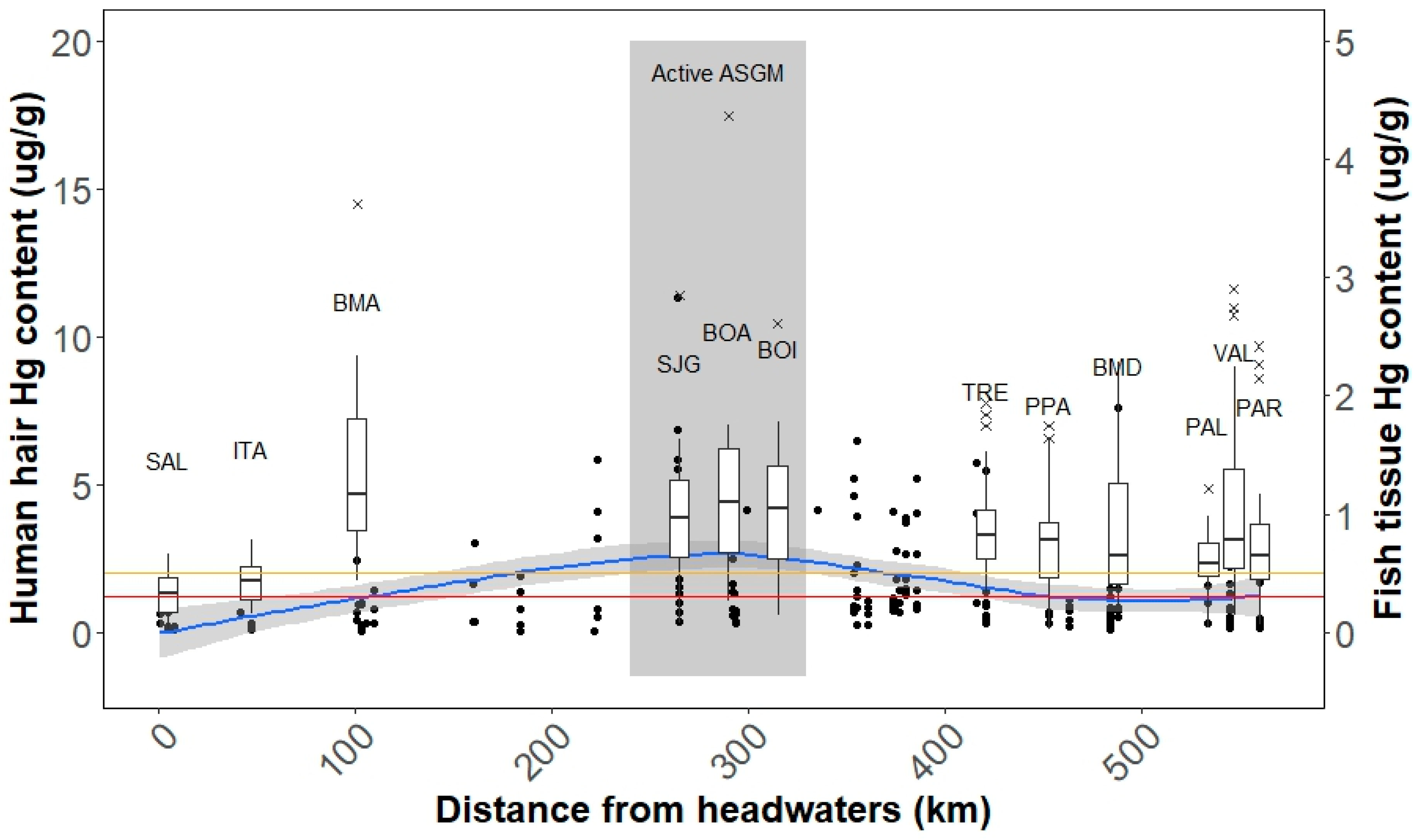
3.2. Relationship between Environmental and Human Exposure Levels
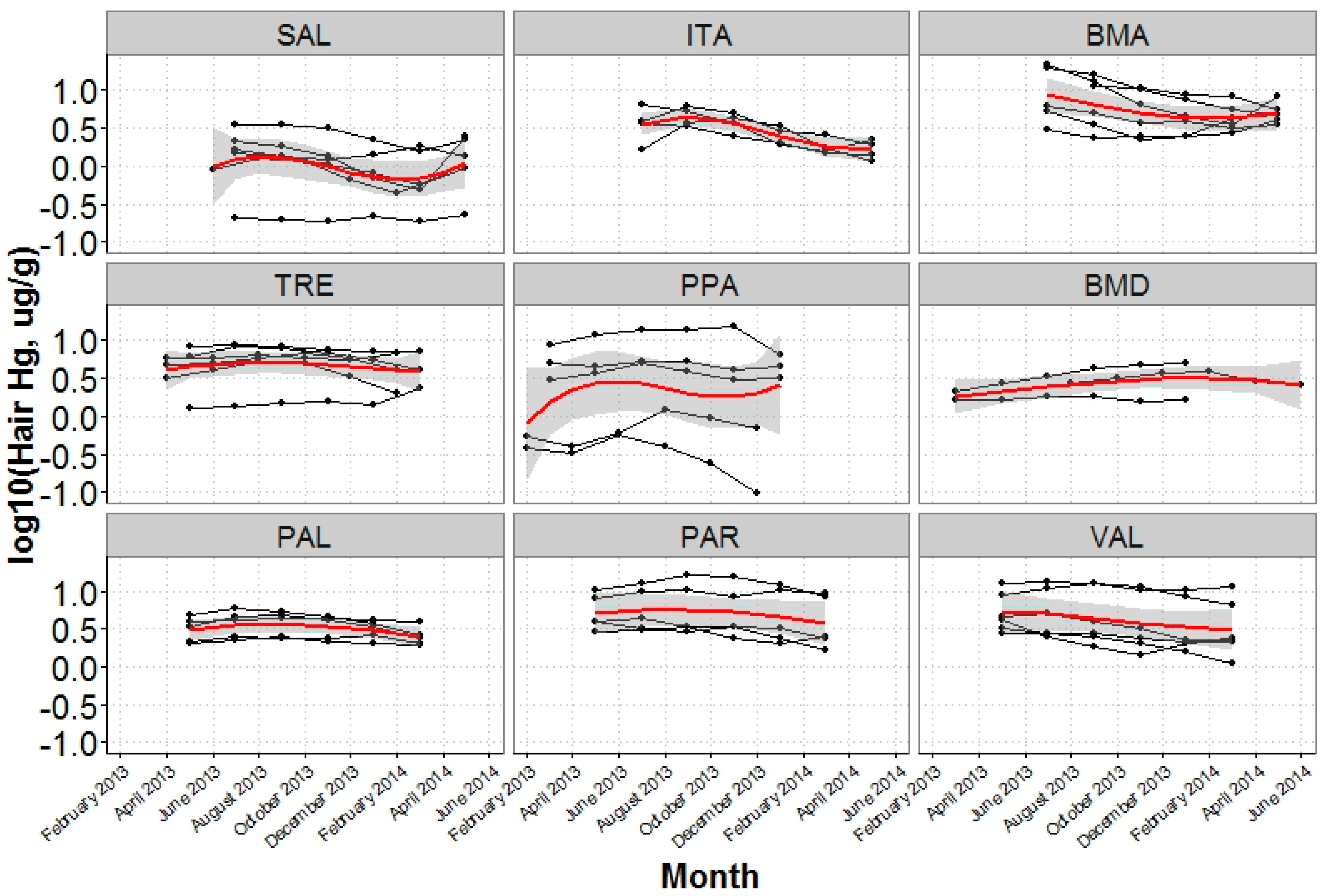
3.3. Temporal Variation in Hair Mercury
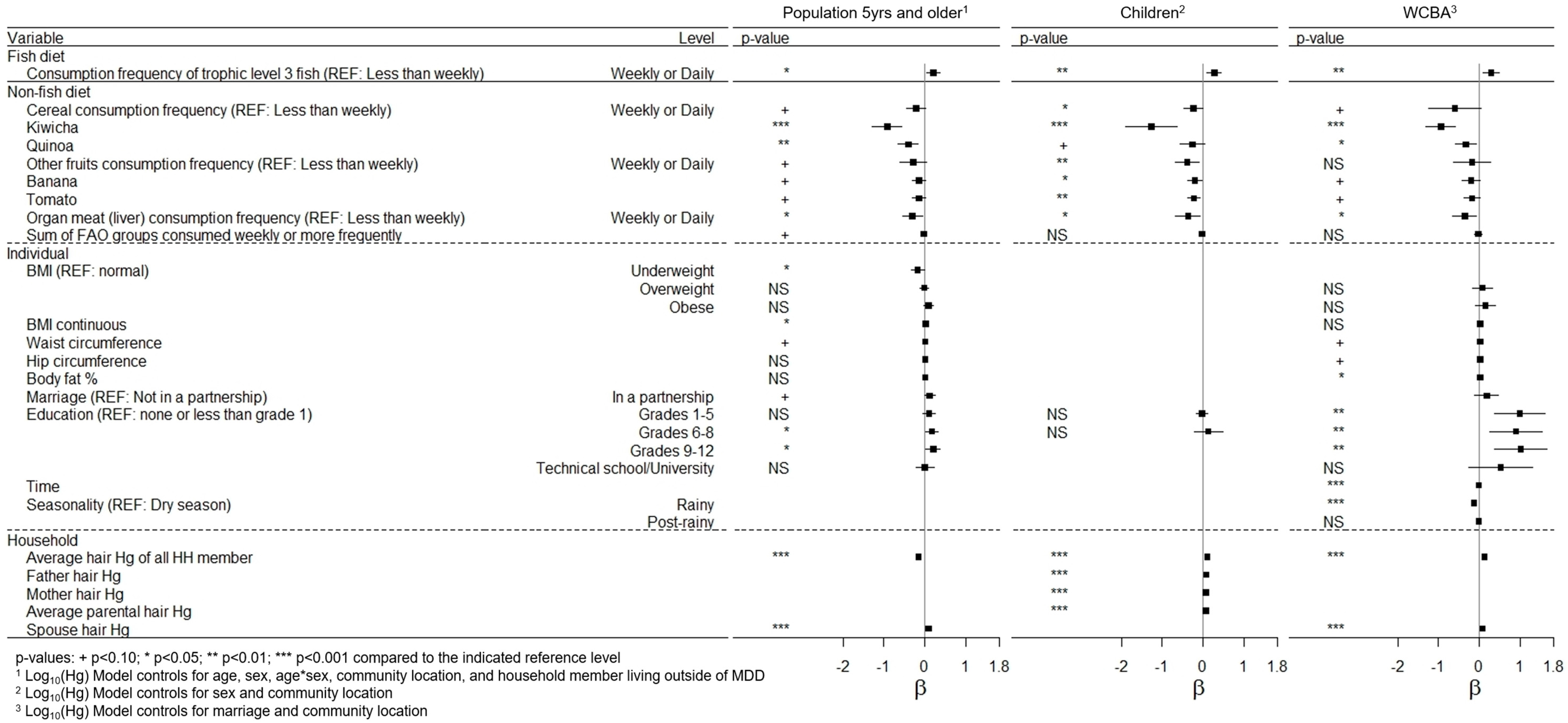
3.4. Risk Factors Related to Hair Mercury
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Swain, E.B.; Jakus, P.M.; Rice, G.; Lupi, F.; Maxson, P.A.; Pacyna, J.M.; Penn, A.; Spiegel, S.J.; Veiga, M.M. Socioeconomic consequences of mercury use and pollution. Ambio 2007, 36, 45–61. [Google Scholar] [CrossRef]
- United Nations Environment Programme. Global Mercury Assessment 2013: Sources, Emissions, Releases and Environmental Transport; UNEP Chemicals Branch: Geneva, Switzerland, 2013. [Google Scholar]
- Beliveau, A.; Lucotte, M.; Davidson, R.; Lopes, L.O.; Paquet, S. Early Hg mobility in cultivated tropical soils one year after slash-and-burn of the primary forest, in the Brazilian Amazon. Sci. Total Environ. 2009, 407, 4480–4489. [Google Scholar] [CrossRef] [PubMed]
- Kocman, D.; Wilson, S.J.; Amos, H.M.; Telmer, K.H.; Steenhuisen, F.; Sunderland, E.M.; Mason, R.P.; Outridge, P.; Horvat, M. Toward an Assessment of the Global Inventory of Present-Day Mercury Releases to Freshwater Environments. Int. J. Environ. Res. Public Health 2017, 14, 138. [Google Scholar] [CrossRef] [PubMed]
- Mainville, N.; Webb, J.; Lucotte, M.; Davidson, R.; Betancourt, O.; Cueva, E.; Mergler, D. Decrease of soil fertility and release of mercury following deforestation in the Andean Amazon, Napo River Valley, Ecuador. Sci. Total Environ. 2006, 368, 88–98. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Toxicological Effects of Methylmercury; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Grandjean, P.; Budtz-Jorgensen, E.; White, R.F.; Jorgensen, P.J.; Weihe, P.; Debes, F.; Keiding, N. Methylmercury exposure biomarkers as indicators of neurotoxicity in children aged 7 years. Am. J. Epidemiol. 1999, 150, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; White, R.F.; Nielsen, A.; Cleary, D.; Santos, E.C.D. Methylmercury neurotoxicity in Amazonian children downstream from gold mining. Environ. Health Perspect. 1999, 107, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Morel, F.M.M.; Kraepiel, A.M.L.; Amyot, M. The chemical cycle and bioaccumulation of mercury. Annu. Rev. Ecol. Syst. 1998, 29, 543–566. [Google Scholar] [CrossRef]
- Alvarez-Berros, N.L.; Aide, T.M. Global demand for gold is another threat for tropical forests. Environ. Res. Lett. 2015, 10, 029501. [Google Scholar] [CrossRef]
- Asner, G.P.; Llactayo, W.; Tupayachi, R.; Luna, E.R. Elevated rates of gold mining in the Amazon revealed through high-resolution monitoring. Proc. Natl. Acad. Sci. USA 2013, 110, 18454–18459. [Google Scholar] [CrossRef] [PubMed]
- Emel, J.; Plisinski, J.; Rogan, J. Monitoring geomorphic and hydrologic change at mine sites using satellite imagery: The Geita Gold Mine in Tanzania. Appl. Geogr. 2014, 54, 243–249. [Google Scholar] [CrossRef]
- Peterson, G.D.; Heemskerk, M. Deforestation and forest regeneration following small-scale gold mining in the Amazon: The case of Suriname. Environ. Conserv. 2001, 28, 117–126. [Google Scholar] [CrossRef]
- Schueler, V.; Kuemmerle, T.; Schroder, H. Impacts of Surface Gold Mining on Land Use Systems in Western Ghana. Ambio 2011, 40, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Snapir, B.; Simms, D.M.; Waine, T.W. Mapping the expansion of galamsey gold mines in the cocoa growing area of Ghana using optical remote sensing. Int. J. Appl. Earth Obs. Geoinf. 2017, 58, 225–233. [Google Scholar] [CrossRef]
- Swenson, J.J.; Carter, C.E.; Domec, J.C.; Delgado, C.I. Gold Mining in the Peruvian Amazon: Global Prices, Deforestation, and Mercury Imports. PLoS ONE 2011, 6, e18875. [Google Scholar] [CrossRef] [PubMed]
- Boffetta, P.; Sallsten, G.; Garcia-Gomez, M.; Pompe-Kirn, V.; Zaridze, D.; Bulbulyan, M.; Caballero, J.D.; Ceccarelli, F.; Kobal, A.B.; Merler, E. Mortality from cardiovascular diseases and exposure to inorganic mercury. Occup. Environ. Med. 2001, 58, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Gump, B.B.; MacKenzie, J.A.; Dumas, A.K.; Palmer, C.D.; Parsons, P.J.; Segu, Z.M.; Mechref, Y.S.; Bendinskas, K.G. Fish consumption, low-level mercury, lipids, and inflammatory markers in children. Environ. Res. 2012, 112, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Ilback, N.G.; Wesslen, L.; Fohlman, J.; Friman, G. Effects of methyl mercury on cytokines, inflammation and virus clearance in a common infection (Coxsackie B3 myocarditis). Toxicol. Lett. 1996, 89, 19–28. [Google Scholar] [CrossRef]
- Rizzetti, D.A.; Torres, J.G.D.; Escobar, A.G.; Pecanha, F.M.; Santos, F.W.; Puntel, R.L.; Alonso, M.J.; Briones, A.M.; Salaices, M.; Vassallo, D.V.; et al. Apocynin Prevents Vascular Effects Caused by Chronic Exposure to Low Concentrations of Mercury. PLoS ONE 2013, 8, e55806. [Google Scholar] [CrossRef] [PubMed]
- Via, C.S.; Nguyen, P.; Niculescu, F.; Papadimitriou, J.; Hoover, D.; Silbergeld, E.K. Low-dose exposure to inorganic mercury accelerates disease and mortality in acquired murine lupus. Environ. Health Perspect. 2003, 111, 1273–1277. [Google Scholar] [CrossRef] [PubMed]
- Wiggers, G.A.; Pecanha, F.M.; Briones, A.M.; Perez-Giron, J.V.; Miguel, M.; Vassallo, D.V.; Cachofeiro, V.; Alonso, M.J.; Salaices, M. Low mercury concentrations cause oxidative stress and endothelial dysfunction in conductance and resistance arteries. Am. J. Physiol.-Heart Circ. Physiol. 2008, 295, H1033–H1043. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry. Toxicological Profile for Mercury; US Department of Health and Human Services: Washington, DC, USA, 1999.
- Basu, N.; Clarke, E.; Green, A.; Calys-Tagoe, B.; Chan, L.; Dzodzomenyo, M.; Fobil, J.; Long, R.N.; Neitzel, R.L.; Obiri, S.; et al. Integrated Assessment of Artisanal and Small-Scale Gold Mining in Ghana-Part 1: Human Health Review. Int. J. Environ. Res. Public Health 2015, 12, 5143–5176. [Google Scholar] [CrossRef] [PubMed]
- Crompton, P.; Ventura, A.M.; de Souza, J.M.; Santos, E.; Strickland, G.T.; Silbergeld, E. Assessment of mercury exposure and malaria in a Brazilian Amazon riverine community. Environ. Res. 2002, 90, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Peplow, D.; Augustine, S. Community-directed risk assessment of mercury exposure from gold mining in Suriname. Revista Panamericana Salud Publica 2007, 22, 202–210. [Google Scholar] [CrossRef]
- Hacon, S.; Barrocas, P.R.; Vasconcellos, A.C.; Barcellos, C.; Wasserman, J.C.; Campos, R.C.; Ribeiro, C.; Azevedo-Carloni, F.B. An overview of mercury contamination research in the Amazon basin with an emphasis on Brazil. Cadernos Saude Publica 2008, 24, 1479–1492. [Google Scholar] [CrossRef]
- Doering, S.; Bose-O’Reilly, S.; Berger, U. Essential Indicators Identifying Chronic Inorganic Mercury Intoxication: Pooled Analysis across Multiple Cross-Sectional Studies. PLoS ONE 2016, 11, e0160323. [Google Scholar] [CrossRef] [PubMed]
- Dolbec, J.; Mergler, D.; Larribe, F.; Roulet, M.; Lebel, J.; Lucotte, M. Sequential analysis of hair mercury levels in relation to fish diet of an Amazonian population, Brazil. Sci. Total Environ. 2001, 271, 87–97. [Google Scholar] [CrossRef]
- Lebel, J.; Roulet, M.; Mergler, D.; Lucotte, M.; Larribe, F. Fish diet and mercury exposure in a riparian Amazonian population. Water Air Soil Pollut. 1997, 97, 31–44. [Google Scholar] [CrossRef]
- Passos, C.J.; Mergler, D.; Gaspar, E.; Morais, S.; Lucotte, M.; Larribe, F.; Davidson, R.; de Grosbois, S. Eating tropical fruit reduces mercury exposure from fish consumption in the Brazilian Amazon. Environ. Res. 2003, 93, 123–130. [Google Scholar] [CrossRef]
- Ouedraogo, O.; Amyot, M. Effects of various cooking methods and food components on bioaccessibility of mercury from fish. Environ. Res. 2011, 111, 1064–1069. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.-M.; Ferruzzi, M.G.; Kim, Y.-C.; Janle, E.M.; Santerre, C.R. Impact of Phytochemical-Rich Foods on Bioaccessibility of Mercury from Fish. Food Chem. 2009, 112, 46–50. [Google Scholar] [CrossRef]
- Ballatori, N.; Wang, W.; Lieberman, M.W. Accelerated methylmercury elimination in gamma-glutamyl transpeptidase-deficient mice. Am. J. Pathol. 1998, 152, 1049–1055. [Google Scholar] [PubMed]
- Deceaurriz, J.; Payan, J.P.; Morel, G.; Brondeau, M.T. Role of extracellular glutathione and gamma-glutamyl-transpepidase in the disposition and kidney toxicity of inorganic mercury in rats. J. Appl. Toxicol. 1994, 14, 201–206. [Google Scholar] [CrossRef]
- Zalups, R.K.; Barfuss, D.W. Accumulation and handeling of inorganic mercury in the kidney after coadministration with glutathione. J. Toxicol. Environ. Health 1995, 44, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Steuerwald, U.; Weihe, P.; Jorgensen, P.J.; Bjerve, K.; Brock, J.; Heinzow, B.; Budtz-Jorgensen, E.; Grandjean, P. Maternal seafood diet, methylmercury exposure, and neonatal neurologic function. J. Pediatr. 2000, 136, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, P.; Andersen, B.W.; Rankin, J.C. Retention of methyl mercury and inorganic mercury in rainbow trout Oncorhynchus mykiss (W): Effect of dietary selenium. Aquat. Toxicol. 1999, 45, 171–180. [Google Scholar] [CrossRef]
- Dang, F.; Wang, W.X. Antagonistic Interaction of Mercury and Selenium in a Marine Fish Is Dependent on Their Chemical Species. Environ. Sci. Technol. 2011, 45, 3116–3122. [Google Scholar] [CrossRef] [PubMed]
- Deng, D.F.; Teh, F.C.; Teh, S.J. Effect of dietary methylmercury and seleno-methionine on Sacramento Splittail Larvae. Sci. Total Environ. 2008, 407, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Frery, N.; Maury-Brachet, R.; Maillot, E.; Deheeger, M.; de Merona, B.; Boudou, A. Gold-mining activities and mercury contamination of native Amerindian communities in French Guiana: Key role of fish in dietary uptake. Environ. Health Perspect. 2001, 109, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Malm, O.; Branches, F.J.P.; Akagi, H.; Castro, M.B.; Pfeiffer, W.C.; Harada, M.; Bastos, W.R.; Kato, H. Mercury and methylmercury in fish and human hair from the Tapajos river basin, Brazil. Sci. Total Environ. 1995, 175, 141–150. [Google Scholar] [CrossRef]
- Maurice-Bourgoin, L.; Quiroga, I.; Chincheros, J.; Courau, P. Mercury distribution in waters and fishes of the upper Madeira rivers and mercury exposure in riparian Amazonian populations. Sci. Total Environ. 2000, 260, 73–86. [Google Scholar] [CrossRef]
- Ashe, K. Elevated Mercury Concentrations in Humans of Madre de Dios, Peru. PLoS ONE 2012, 7, e33305. [Google Scholar] [CrossRef] [PubMed]
- Langeland, A.L.; Hardin, R.D.; Neitzel, R.L. Mercury Levels in Human Hair and Farmed Fish near Artisanal and Small-Scale Gold Mining Communities in the Madre de Dios River Basin, Peru. Int. J. Environ. Res. Public Health 2017, 14, 302. [Google Scholar] [CrossRef] [PubMed]
- Diringer, S.E.; Feingold, B.J.; Ortiz, E.J.; Gallis, J.A.; Araujo-Flores, J.M.; Berky, A.; Pan, W.K.Y.; Hsu-Kim, H. River transport of mercury from artisanal and small-scale gold mining and risks for dietary mercury exposure in Madre de Dios, Peru. Environ. Sci. Process. Impacts 2015, 17, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Estadística e Informática. Estado de la Población Peruana 2015; INEI: Lima, Peru, 2015. [Google Scholar]
- MINAM. Cuantification y Analisis de la Deforestacion en la Amazonia Peruana en el Periodo 2010–2011–2013–2014; MINAM: Lima, Peru, 2015. [Google Scholar]
- Lu, F.; Gray, C.; Bilsborrow, R.E.; Mena, C.F.; Erlien, C.M.; Bremner, J.; Barbieri, A.; Walsh, S.J. Contrasting Colonist and Indigenous Impacts on Amazonian Forests. Conserv. Biol. 2010, 24, 881–885. [Google Scholar] [CrossRef] [PubMed]
- Tallman, P.S. The Index of Vulnerability: An anthropological method linking social-ecological systems to mental and physical health outcomes. Soc. Sci. Med. 2016, 162 (Suppl. C), 68–78. [Google Scholar] [CrossRef] [PubMed]
- Tello, R. Settlement Patterns and Ecosystem Pressures in the Peruvian Rainforest: Understanding the Impacts of Indigenous Peoples on Biodiversity; Amakella Publishing: Arlington, VA, USA, 2016; p. 88. [Google Scholar]
- Scanlon, J. Indigenous Communities in Peru and the Peruvian Nation State. Bachelor’s Thesis, Pace University, New York, NY, USA, 2012. [Google Scholar]
- Takasaki, Y.; Barham, B.; Coomes, O. Amazonian Peasants, Rain Forest Use, and Income Generation: The Role of Wealth and Geographical Factors. Soc. Nat. Resour. 2001, 14, 291–308. [Google Scholar] [CrossRef]
- Coomes, O.T.; Takasaki, Y.; Abizaid, C.; Barham, B.L. Floodplain fisheries as natural insurance for the rural poor in tropical forest environments: Evidence from Amazonia. Fish. Manag. Ecol. 2010, 17, 513–521. [Google Scholar] [CrossRef]
- LeBeau, M.A.; Montgomery, M.A.; Brewer, J.D. The role of variations in growth rate and sample collection on interpreting results of segmental analyses of hair. Forensic Sci. Int. 2011, 210, 110–116. [Google Scholar] [CrossRef] [PubMed]
- SENAMHI. Meteorológica Datos de Puerto Maldonado. In Hidrología. Available online: http://www.senamhi.gob.pe (accessed on 2 October 2017).
- Espinoza Villar, J.C.; Ronchail, J.; Guyot, J.L.; Cochonneau, G.; Naziano, F.; Lavado, W.; De Oliveira, E.; Pombosa, R.; Vauchel, P. Spatio-temporal rainfall variability in the Amazon basin countries (Brazil, Peru, Bolivia, Colombia, and Ecuador). Int. J. Climatol. 2009, 29, 1574–1594. [Google Scholar] [CrossRef]
- Espinoza, J.C.; Chavez, S.; Ronchail, J.; Junquas, C.; Takahashi, K.; Lavado, W. Rainfall hotspots over the southern tropical Andes: Spatial distribution, rainfall intensity, and relations with large-scale atmospheric circulation. Water Resour. Res. 2015, 51, 3459–3475. [Google Scholar] [CrossRef]
- Hamilton, J.B.; Terada, H.; Mestler, G.E. Studies of growth throughout the lifespan in Japanese: Growth and size of nails and their relationship to age, sex, heredity, and other factors. J. Gerontol. 1955, 10, 401–415. [Google Scholar] [CrossRef] [PubMed]
- Yaemsiri, S.; Hou, N.; Slining, M.M.; He, K. Growth rate of human fingernails and toenails in healthy American young adults. J. Eur. Acad. Dermatol. Venereol. 2010, 24, 420–423. [Google Scholar] [CrossRef] [PubMed]
- He, K. Trace Elements in Nails as Biomarkers in Clinical Research. Eur. J. Clin. Investig. 2011, 41, 98–102. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Integrated Risk Information System: Methylmercury (MeHg) (CASRN 22967-92-6); USEPA: Washington, DC, USA, 2001.
- United States Environmental Protection Agency. Title 21—Food and Drugs—Chapter I Subchapter B—Food for Human Consumption; USEPA: Washington, DC, USA, 2016.
- Feingold, B.J.; Hsu-Kim, H.; Porter, K.; Ortiz, E.; Rogers, L.; Salmon-Mulanovich, G.; Diringer, S.; Pan, W. Mercury Exposure in Madre De Dios, Peru: Connecting Environmental Contamination To Human Exposure in A Region of Small Scale Gold Mining. Int. Soc. Environ. Epidemiol. 2015, 3484, 2015–3484. [Google Scholar]
- Conger, R.D.; Conger, K.J.; Martin, M.J. Socioeconomic Status, Family Processes, and Individual Development. J. Marriage Fam. 2010, 72, 685–704. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization. Guidelines for Measuring Household and Individual Dietary Diversity; FAO: Rome, Italy, 2010. [Google Scholar]
- Budtz-Jorgensen, E.; Grandjean, P.; Weihe, P. Separation of risks and benefits of seafood intake. Environ. Health Perspect. 2007, 115, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, J.L.; Muckle, G.; Ayotte, P.; Dewailly, E.; Jacobson, S.W. Relation of Prenatal Methylmercury Exposure from Environmental Sources to Childhood IQ. Environ. Health Perspect. 2015, 123, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Lebel, J.; Mergler, D.; Branches, F.; Lucotte, M.; Amorim, M.; Larribe, F.; Dolbec, J. Neurotoxic effects of low-level methylmercury contamination in the Amazonian Basin. Environ. Res. 1998, 79, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Marinho, J.S.; Lima, M.O.; de Oliveria Santos, E.C.; de Jesus, I.M.; da Conceicão, N.; Pinheiro, M.; Alves, C.N.; Muller, R.C. Mercury speciation in hair of children in three communities of the Amazon, Brazil. Biomed. Res. Int. 2014, 2014, 945963. [Google Scholar]
- Lebel, J.; Mergler, D.; Lucotte, M.; Amorim, M.; Dolbec, J.; Miranda, D.; Arantes, G.; Rheault, I.; Pichet, P. Evidence of early nervous system dysfunction in Amazonian populations exposed to low-levels of methylmercury. Neurotoxicology 1996, 17, 157–167. [Google Scholar] [PubMed]
- Santos, E.C.; Camara, V.M.; Jesus, I.M.; Brabo, E.S.; Loureiro, E.C.; Mascarenhas, A.F.; Fayal, K.F.; Sa Filho, G.C.; Sagica, F.E.; Lima, M.O.; et al. A contribution to the establishment of reference values for total mercury levels in hair and fish in Amazonia. Environ. Res. 2002, 90, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Dolbec, J.; Mergler, D.; Sousa Passos, C.J.; de Morais, S.S.; Lebel, J. Methylmercury exposure affects motor performance of a riverine population of the Tapajós river, Brazilian Amazon. Int. Arch. Occup. Environ. Health 2000, 73, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Oken, E.; Wright, R.O.; Kleinman, K.P.; Bellinger, D.; Amarasiriwardena, C.J.; Hu, H.; Rich-Edwards, J.W.; Gillman, M.W. Maternal fish consumption, hair mercury, and infant cognition in a US cohort. Environ. Health Perspect. 2005, 113, 1376–1380. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Weihe, P.; White, R.F.; Debes, F.; Araki, S.; Yokoyama, K.; Murata, K.; Sorensen, N.; Dahl, R.; Jorgensen, P.J. Cognitive deficit in 7-year-old children with prenatal exposure to methylmercury. Neurotoxicol. Teratol. 1997, 19, 417–428. [Google Scholar] [CrossRef]
- Trasande, L.; Schechter, C.; Haynes, K.A.; Landrigan, P.J. Applying cost analyses to drive policy that protects children—Mercury as a case study. Ann. NY Acad. Sci. 2006, 1076, 911–923. [Google Scholar] [CrossRef] [PubMed]
- Debes, F.; Weihe, P.; Grandjean, P. Cognitive deficits at age 22 years associated with prenatal exposure to methylmercury. Cortex 2016, 74, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Bellanger, M.; Pichery, C.; Aerts, D.; Berglund, M.; Castaño, A.; Čejchanová, M.; Crettaz, P.; Davidson, F.; Esteban, M.; Fischer, M.E.; et al. Economic benefits of methylmercury exposure control in Europe: Monetary value of neurotoxicity prevention. Environ. Health 2013, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Carrier, G.; Brunet, R.C.; Caza, M.; Bouchard, M. A toxicokinetic model for predicting the tissue distribution and elimination of organic and inorganic mercury following exposure to methyl mercury in animals and humans. I. Development and validation of the model using experimental data in rats. Toxicol. Appl. Pharmacol. 2001, 171, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Phelps, R.W.; Clarkson, T.W.; Kershaw, T.G.; Wheatley, B. Interrelationships of blood and hair mercury concentrations in a North American population exposed to methylmercury. Arch. Environ. Health 1980, 35, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Laffont, L.; Sonke, J.E.; Maurice, L.; Monrroy, S.L.; Chincheros, J.; Amouroux, D.; Behra, P. Hg speciation and stable isotope signatures in human hair as a tracer for dietary and occupational exposure to mercury. Environ. Sci. Technol. 2011, 45, 9910–9916. [Google Scholar] [CrossRef] [PubMed]
- Castoldi, A.F.; Johansson, C.; Onishchenko, N.; Coccini, T.; Roda, E.; Vahter, M.; Ceccatelli, S.; Manzo, L. Human developmental neurotoxicity of methylmercury: Impact of variables and risk modifiers. Regul. Toxicol. Pharmacol. 2008, 51, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Golding, J.; Steer, C.D.; Hibbeln, J.R.; Emmett, P.M.; Lowery, T.; Jones, R. Dietary Predictors of Maternal Prenatal Blood Mercury Levels in the ALSPAC Birth Cohort Study. Environ. Health Perspect. 2013, 121, 1214–1218. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Weihe, P.; Jorgensen, P.J.; Clarkson, T.; Cernichiari, E.; Videro, T. Impact of maternal seafood diet on fetal exposure to mercury, selenium, and lead. Arch. Environ. Health 1992, 47, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Morrissette, J.; Takser, L.; St-Amour, G.; Smargiassi, A.; Lafond, J.; Mergler, D. Temporal variation of blood and hair mercury levels in pregnancy in relation to fish consumption history in a population living along the St. Lawrence River. Environ. Res. 2004, 95, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Abderrahim, F.; Huanatico, E.; Segura, R.; Arribas, S.; Gonzalez, M.C.; Condezo-Hoyos, L. Physical features, phenolic compounds, betalains and total antioxidant capacity of coloured quinoa seeds (Chenopodium quinoa Willd.) from Peruvian Altiplano. Food Chem. 2015, 183, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Repo-Carrasco-Valencia, R.; Hellstrom, J.K.; Pihlava, J.M.; Mattila, P.H. Flavonoids and other phenolic compounds in Andean indigenous grains: Quinoa (Chenopodium quinoa), kaniwa (Chenopodium pallidicaule) and kiwicha (Amaranthus caudatus). Food Chem. 2010, 120, 128–133. [Google Scholar] [CrossRef]
- Repo-Carrasco-Valencia, R.; Pena, J.; Kallio, H.; Salminen, S. Dietary fiber and other functional components in two varieties of crude and extruded kiwicha (Amaranthus caudatus). J. Cereal Sci. 2009, 49, 219–224. [Google Scholar] [CrossRef]
- Vega-Galvez, A.; Miranda, M.; Vergara, J.; Uribe, E.; Puente, L.; Martinez, E.A. Nutrition facts and functional potential of quinoa (Chenopodium quinoa willd.), an ancient Andean grain: A review. J. Sci. Food Agric. 2010, 90, 2541–2547. [Google Scholar] [CrossRef] [PubMed]
- Gagne, D.; Lauziere, J.; Blanchet, R.; Vezina, C.; Vaissiere, E.; Ayotte, P.; O’Brien, H.T. Consumption of tomato products is associated with lower blood mercury levels in Inuit preschool children. Food Chem. Toxicol. 2013, 51, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Nwokocha, C.R.; Nwokocha, M.I.; Aneto, I.; Obi, J.; Udekweleze, D.C.; Olatunde, B.; Owu, D.U.; Iwuala, M.O. Comparative analysis on the effect of Lycopersicon esculentum (tomato) in reducing cadmium, mercury and lead accumulation in liver. Food Chem. Toxicol. 2012, 50, 2070–2073. [Google Scholar] [CrossRef] [PubMed]
- Zefferino, R.; Leone, A.; Piccaluga, S.; Cincione, R.; Ambrosi, L. Mercury modulates interplay between IL-1 beta, TNF-alpha, and gap junctional intercellular communication in keratinocytes: Mitigation by lycopene. J. Immunotoxicol. 2008, 5, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Leopold, I.; Gunther, D.; Schmidt, J.; Neumann, D. Phytochelatins and heavy metal tolerance. Phytochemistry 1999, 50, 1323–1328. [Google Scholar] [CrossRef]
- Siddiqui, M.W.; Chakraborty, I.; Mishra, P.; Hazra, P. Bioactive attributes of tomatoes possessing dg, og(c), and rin genes. Food Funct. 2014, 5, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Tito, A.; Carola, A.; Bimonte, M.; Barbulova, A.; Arciello, S.; de Laurentiis, F.; Monoli, I.; Hill, J.; Gibertoni, S.; Colucci, G.; et al. A tomato stem cell extract, containing antioxidant compounds and metal chelating factors, protects skin cells from heavy metal-induced damages. Int. J. Cosmet. Sci. 2011, 33, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Songsermsakul, P. Comparison of Antioxidant Activity of Grape Seed Extract and Fruits Containing High β-Carotene, Vitamin C, and E. Int. J. Food Prop. 2013, 16, 643–648. [Google Scholar] [CrossRef]
- Wang, H.; Cao, G.; Prior, R.L. Total Antioxidant Capacity of Fruits. J. Agric. Food Chem. 1996, 44, 701–705. [Google Scholar] [CrossRef]
- Despres, C.; Beuter, A.; Richer, F.; Poitras, K.; Veilleux, A.; Ayotte, P.; Dewailly, T.; Saint-Amour, D.; Muckle, G. Neuromotor functions in Inuit preschool children exposed to Pb, PCBs, and Hg. Neurotoxicol. Teratol. 2005, 27, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Ganther, H.E.; Goudie, C.; Wagner, P.; Sunde, M.L.; Kopecky, M.J.; Oh, S.H.; Hoekstra, W.G. Selenium—Relation to decreased toxicity of methylmercury added to diets containing tuna. Science 1972, 175, 1122–1124. [Google Scholar] [CrossRef] [PubMed]
- Ralston, N.V.C. Selenium Health Benefit Values as Seafood Safety Criteria. Ecohealth 2008, 5, 442–455. [Google Scholar] [CrossRef] [PubMed]
- Ralston, N.V.C.; Ralston, C.R.; Blackwell, J.L.; Raymond, L.J. Dietary and tissue selenium in relation to methylmercury toxicity. Neurotoxicology 2008, 29, 802–811. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, L.; Diringer, S.; Rodgers, L.; Hsu-Kim, H.; Pan, W.; Meyer, J. Antagonistic effects of mercury and selenium on growth in Caenorhabditis elegans are species-dependent, occur at high aqueous concentrations of Se, and are not dependent on the internal Hg/Se ratio. Environ. Sci. Technol. 2016, 50, 3256–3264. [Google Scholar] [CrossRef] [PubMed]
- Strain, J.J.; Davidson, P.W.; Bonham, M.P.; Duffy, E.M.; Stokes-Riner, A.; Thurston, S.W.; Wallace, J.M.; Robson, P.J.; Shamlaye, C.F.; Georger, L.A.; et al. Associations of maternal long-chain polyunsaturated fatty acids, methyl mercury, and infant development in the Seychelles Child Development Nutrition Study. Neurotoxicology 2008, 29, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, C.M.; Smith, D.M.; Golightly, M.G.; Meliker, J.R. Total blood mercury and rubella antibody concentrations in US children aged 6–11 years, NHANES 2003–2004. Sci. Total Environ. 2013, 442, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, C.M.; Smith, D.M.; Meliker, J.R. Total blood mercury and serum measles antibodies in US children, NHANES 2003–2004. Sci. Total Environ. 2011, 410, 65–71. [Google Scholar] [CrossRef] [PubMed]

| Population Grouping | N | Geometric Mean Hair Hg (95% CI) | % Individuals above 1.2 µg/g a | % Individuals above 2.0 µg/g b | |
|---|---|---|---|---|---|
| Total | 231 | 2.6 (0.4, 10.5) | 85.7 | 68.4 | |
| Sex and age in years | |||||
| Female | <8 | 32 | 2.5 (0.4, 9.4) | 81.3 | 65.6 |
| 8–17 | 26 | 2.4 (0.9, 6.3) | 92.3 | 65.4 | |
| 18+ | 61 | 2.7 (0.2, 11.5) | 85.2 | 68.9 | |
| Male | <8 | 25 | 1.9 (0.4, 4.8) | 72.0 | 60.0 |
| 8–17 | 31 | 1.9 (0.4, 5.2) | 77.4 | 48.4 | |
| 18+ | 56 | 3.5 (1.2, 10.5) | 96.4 | 85.7 | |
| Community | |||||
| Upstream | SAL | 19 | 1.0 (0.2, 2.6) | 52.6 | 26.3 |
| ITA | 22 | 1.6 (0.7, 3.1) | 68.2 | 31.8 | |
| BMA | 23 | 4.8 (1.8, 11.7) | 100.0 | 91.3 | |
| Midriver (near mining) | SJG | 13 | 3.2 (0.8, 9.9) | 92.3 | 84.6 |
| BOA | 14 | 4.1 (1.4, 14.1) | 92.9 | 92.9 | |
| BOI | 10 | 3.5 (0.9, 9.7) | 90.0 | 80.0 | |
| Downstream | TRE | 23 | 3.3 (1.5, 7.5) | 100.0 | 91.3 |
| PPA | 19 | 2.2 (0.2, 6.8) | 84.2 | 63.2 | |
| BMD | 21 | 2.8 (1.0, 8.5) | 90.5 | 61.9 | |
| PAL | 25 | 2.1 (0.6, 4.3) | 88.0 | 64.0 | |
| PAR | 20 | 2.6 (0.5, 9.4) | 90.0 | 70.0 | |
| VAL | 22 | 3.1 (0.7, 11.3) | 81.8 | 77.3 | |
| Consumption Frequency | HH (N) | People (N) | Geometric Mean Hg All HH Individuals (95% CI) | Geometric Mean Hg WCBA (95% CI) | Geometric Mean Hg Children <10 Years (95% CI) | |
|---|---|---|---|---|---|---|
| Fish | Never/Seasonally/Sometimes | 30 | 113 | 2.9 (0.4, 9.6) | 2.7 (0.4, 7.7) | 2.2 (0.3, 6.7) |
| Weekly | 11 | 45 | 1.7 (0.3, 4.6) | 1.5 (0.3, 4.5) | 1.6 (0.8, 3.9) | |
| Daily | 18 | 73 | 2.9 (0.6, 11.1) | 3.4 (1.3, 10.6) | 2.3 (0.4, 9.7) | |
| Antioxidant-rich | Never/Seasonally/Sometimes | 31 | 120 | 3.0 (0.6, 11.0) | 2.9 (1.1, 10.8) | 2.6 (0.3, 8.9) |
| Weekly | 6 | 23 | 2.1 (0.7, 8.4) | 2.3 (0.9, 7.5) | 1.6 (0.6, 4.2) | |
| Daily | 22 | 88 | 2.2 (0.2, 8.8) | 2.4 (0.2, 7.8) | 1.6 (0.3, 6.3) | |
| Selenium-rich | Never/Seasonally/Sometimes | 55 | 215 | 2.6 (0.3, 10.6) | 2.5 (0.2, 9.0) | 2.1 (0.2, 8.7) |
| Weekly | 1 | 3 | 2.1 (0.9, 5.1) | 2.1 (2.1, 2.1) | 0.8 (0.8, 0.8) | |
| Daily | 3 | 13 | 2.4 (0.8, 7.3) | 3.3 (1.5, 8.3) | 2.6 (2.2, 3.0) | |
| Variable | Family and Sex Variables | Odds Ratio (OR) | 95% CI |
|---|---|---|---|
| Family | Paternal hair Hg exceeds level | 2.14 | (1.17, 3.93) † |
| Maternal hair Hg exceeds level | 2.39 | (1.86, 3.07) ††† | |
| Parent hair Hg exceeds level | 2.19 | (1.24, 3.90) †† | |
| Mean HH hair Hg exceeds level | 2.32 | (1.73, 3.12) ††† | |
| Mean HH adult hair Hg exceeds level | 2.29 | (1.54, 3.38) ††† | |
| Sex | Female | 1.12 | (0.92, 1.36) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wyatt, L.; Ortiz, E.J.; Feingold, B.; Berky, A.; Diringer, S.; Morales, A.M.; Jurado, E.R.; Hsu-Kim, H.; Pan, W. Spatial, Temporal, and Dietary Variables Associated with Elevated Mercury Exposure in Peruvian Riverine Communities Upstream and Downstream of Artisanal and Small-Scale Gold Mining. Int. J. Environ. Res. Public Health 2017, 14, 1582. https://doi.org/10.3390/ijerph14121582
Wyatt L, Ortiz EJ, Feingold B, Berky A, Diringer S, Morales AM, Jurado ER, Hsu-Kim H, Pan W. Spatial, Temporal, and Dietary Variables Associated with Elevated Mercury Exposure in Peruvian Riverine Communities Upstream and Downstream of Artisanal and Small-Scale Gold Mining. International Journal of Environmental Research and Public Health. 2017; 14(12):1582. https://doi.org/10.3390/ijerph14121582
Chicago/Turabian StyleWyatt, Lauren, Ernesto J. Ortiz, Beth Feingold, Axel Berky, Sarah Diringer, Ana Maria Morales, Elvis Rojas Jurado, Heileen Hsu-Kim, and William Pan. 2017. "Spatial, Temporal, and Dietary Variables Associated with Elevated Mercury Exposure in Peruvian Riverine Communities Upstream and Downstream of Artisanal and Small-Scale Gold Mining" International Journal of Environmental Research and Public Health 14, no. 12: 1582. https://doi.org/10.3390/ijerph14121582
APA StyleWyatt, L., Ortiz, E. J., Feingold, B., Berky, A., Diringer, S., Morales, A. M., Jurado, E. R., Hsu-Kim, H., & Pan, W. (2017). Spatial, Temporal, and Dietary Variables Associated with Elevated Mercury Exposure in Peruvian Riverine Communities Upstream and Downstream of Artisanal and Small-Scale Gold Mining. International Journal of Environmental Research and Public Health, 14(12), 1582. https://doi.org/10.3390/ijerph14121582





