Accumulation of De-Icing Salt and Leaching in Spanish Soils Surrounding Roadways
Abstract
:1. Introduction
2. Experimental
2.1. Research Area and Soil Sampling
- (1)
- Winter (application of de-icing salt): from January to February 2014.
- (2)
- Spring (melting time): from April to June 2014.
- (3)
- Late summer (control): September 2014.
2.2. Experimental Procedure
2.2.1. Technical Characterisation
2.2.2. Parameters Considered for Characterisation and Evaluation of the Effect of Salt on the Studied Soil
3. Results and Discussion
3.1. Salinity of the Studied Soils
3.2. Sodicity of Studied Soils
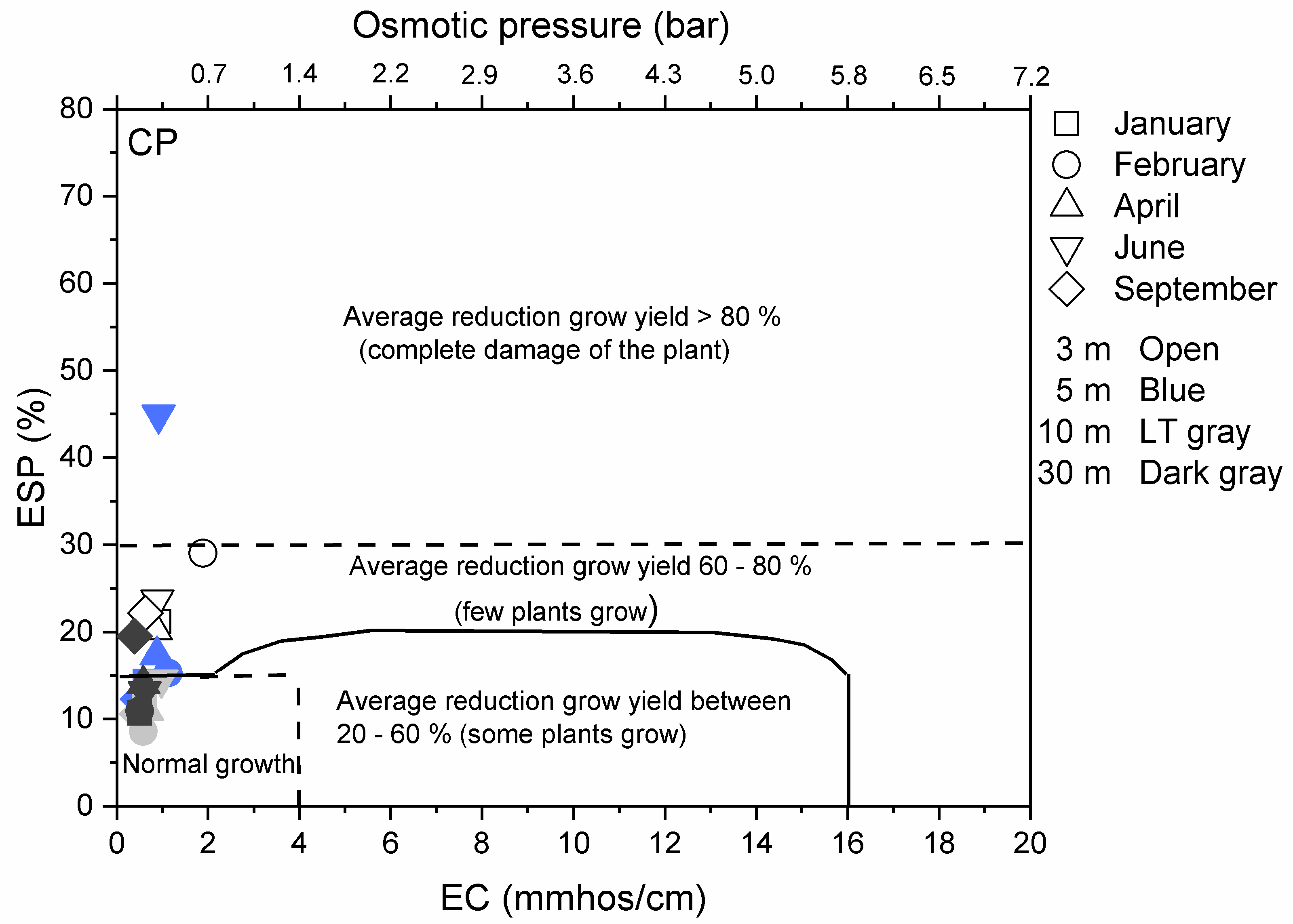
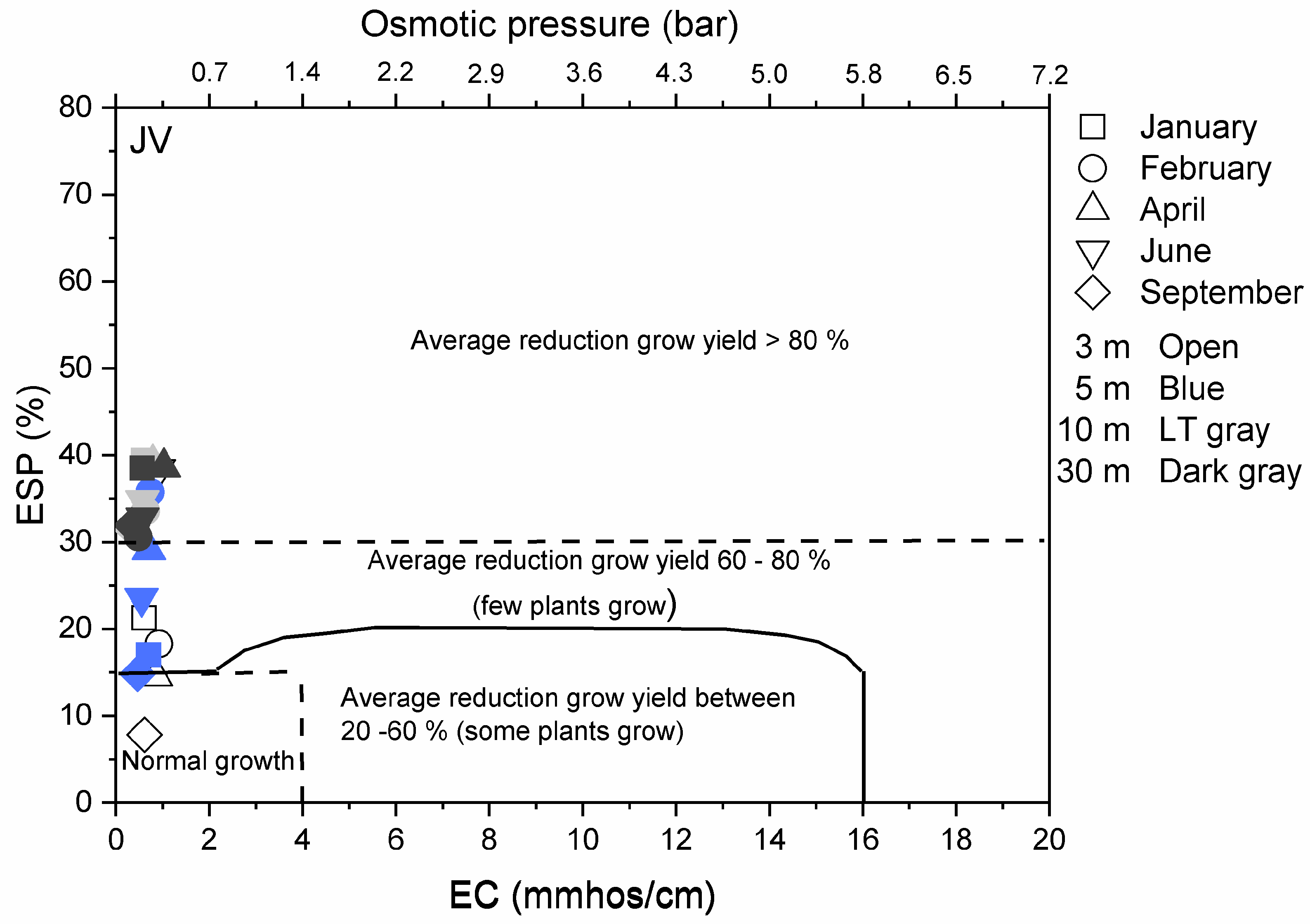
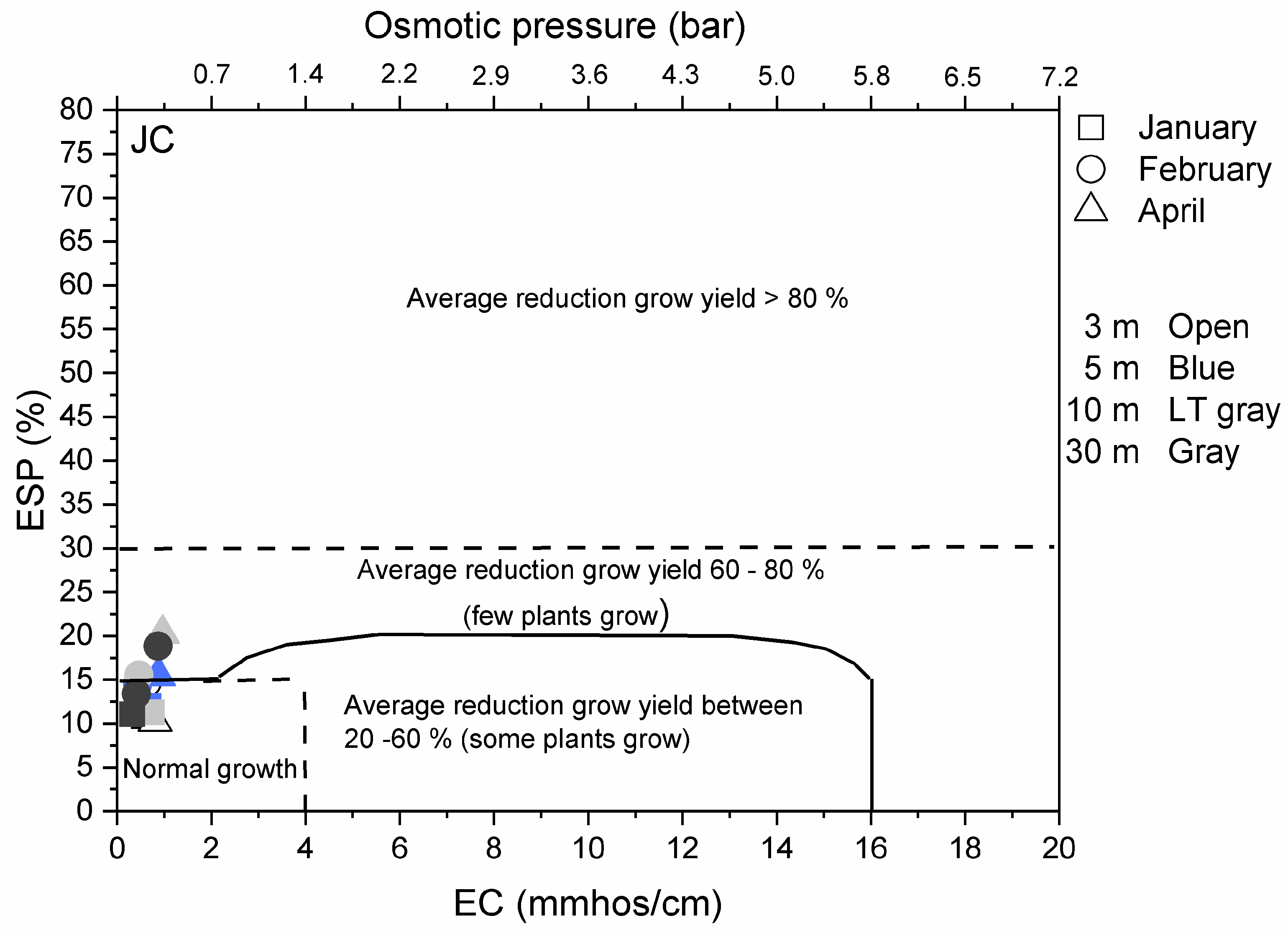
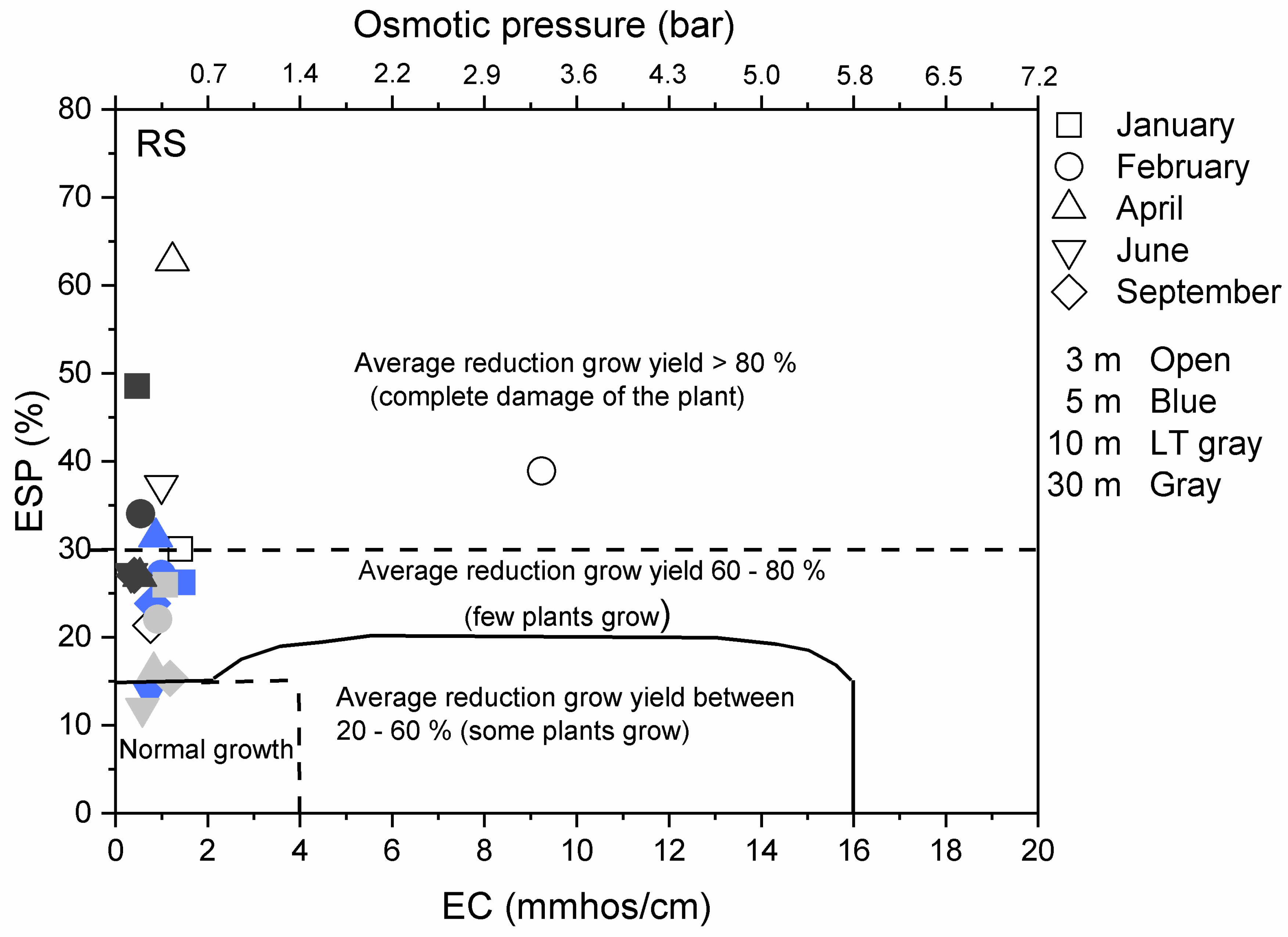
3.3. Environmental Implications
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CEC | Cation Exchangeable Capacity |
| CP | Canfranc (sampling point) |
| CSPS | Soil Property Databases of Spanish Soils |
| DGT | Direction General of Traffic of Spain |
| DPSIR | Force-pressure-state-impact-response |
| DS | De-icing salts |
| EC | Electrical Conductivity (measurement of salinity) |
| EEA | European Environmental Agency |
| ESP | Exchangeable Sodium Percentage (measurement of sodicity) |
| ESR | Exchangeable Sodium Ratio |
| JC | Jaca (Sampling Point) |
| NRCS | Natural Resources Conservation Service |
| LT gray | Light gray |
| RS | Rioseta (sampling point) |
| SAR | Sodium Adsorption Ratio |
| SP | Saturation Percentage |
| UTM | Universal Transverse Mercator coordinate system |
| VJ | Villajuanita (sampling point) |
Appendix A
Appendix A.1. Estimation of Saturation Percentage (SP) of Studied Soils
| Point (zone) | Clay Average (%) | Silt Average (%) | Sand Average (%) | SP ± 6.89 |
|---|---|---|---|---|
| RS and CP | 25 | 38 | 37 | 48 |
| VJ and JC | 16 | 11 | 70 | 57 |
Appendix A.2. Environmental Implications
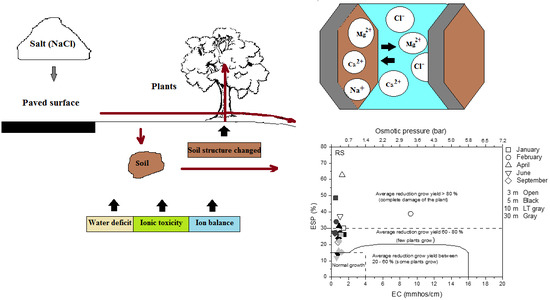
Appendix A.2.1. Main Factors Affecting Plant Growth
Appendix A.2.2. Force-PRESSURE-State-Impact-Response (DPSIR) Approach
Appendix A.2.3. Results on Overall Environmental Impact of De-Icing Salt on the Vegetation Surrounding the Road
References
- Hall, R.; Hofstra, G.; Lumis, G.P. Effects of deicing salt on eastern white pine: Foliar injury, growth suppression and seasonal changes in foliar concentrations of sodium and chloride. Can. J. Forest Res. 1972, 2, 244–249. [Google Scholar] [CrossRef]
- D’Itri, F.M. Chemical Deicers and the Environment; Taylor & Francis-Lewis Publishers: Abingdon, UK, 1992. [Google Scholar]
- Munck, I.A.; Bennett, C.M.; Camilli, K.S.; Nowak, R.S. Long-term impact of de-icing salts on tree health in the Lake Tahoe Basin: Environmental influences and interactions with insects and diseases. For. Ecol. Manag. 2010, 260, 1218–1229. [Google Scholar] [CrossRef]
- Mason, C.F.; Norton, S.A.; Fernandez, I.J.; Katz, L.E. Deconstruction of the chemical effects of road salt on stream water chemistry. J. Environ. Qual. 1999, 28, 82. [Google Scholar] [CrossRef]
- Novotny, E.V.; Murphy, D.; Stefan, H.G. Increase of urban lake salinity by road deicing salt. Sci. Total Environ. 2008, 406, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Rosenberry, D.O.; Bukaveckas, P.A.; Buso, D.C.; Likens, G.E.; Shapiro, A.M.; Winter, T.C. Movement of road salt to a small New Hampshire Lake. Water Air Soil Pollut. 1999, 109, 179–206. [Google Scholar] [CrossRef]
- Sadashivaiah, C.; Ramakrishnaiah, C.; Ranganna, G. Hydrochemical analysis and evaluation of groundwater quality in Tumkur Taluk, Karnataka State, India. Int. J. Environ. Res. Public Health 2008, 5, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Trombulak, S.C.; Frissell, C.A. Review of ecological effects of roads on Terrestrial and Aquatic (Communities Revisión de los Efectos de Carreteras en Comunidades Terrestres y Acuáticas). Conserv. Biol. 2000, 14, 18–30. [Google Scholar] [CrossRef]
- Schweiger, A.H.; Audorff, V.; Beierkuhnlein, C. Salt in the wound: The interfering effect of road salt on acidified forest catchments (Supplement C). Sci. Total Environ. 2015, 532, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Bryson, G.M.; Barker, A.V. Sodium accumulation in soils and plants along Massachusetts roadsides. Commun. Soil Sci. Plant Anal. 2002, 33, 67–78. [Google Scholar] [CrossRef]
- Viskari, E.-L.; Kärenlampi, L. Roadside scots pine as an indicator of deicing salt use—A comparative study from two consecutive winters. Water Air Soil Pollut. 2000, 122, 405–419. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, G.; Yin, X.; Liu, Z. Field-scale spatial variation of saline-sodic soil and its relation with environmental factors in Western Songnen Plain of China. Int. J. Environ. Res. Public Health 2011, 8, 374–387. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Zhang, Y.; Fan, Z.; Oh, K. Accumulation of de-icing salts and its short-term effect on metal mobility in urban roadside soils. Bull. Environ. Contam. Toxicol. 2015, 94, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Pająk, M.; Krzaklewski, W.; Duda, K.; Gruba, P. Spatial and temporal variation in soil salinity as a result of chemical de-icing of road in Krakow, Poland. Fresenius Environ. Bull. 2015, 24, 3363–3370. [Google Scholar]
- Fay, L.; Shi, X. Environmental impacts of chemicals for snow and ice control: State of the knowledge. Water Air Soil Pollut. 2012, 223, 2751–2770. [Google Scholar] [CrossRef]
- Environment-and-Climate-Change-Canada, Road Salts. Available online: http://www.ec.gc.ca/sels-salts/ (accessed on 11 November 2017).
- Diaz, D.R.; Presley, D. Management of Saline and Sodic Soils. Available online: www.bookstore.ksre.ksu.edu (accessed on 11 November 2017).
- Bäckström, M.; Karlsson, S.; Bäckman, L.; Folkeson, L.; Lind, B. Mobilisation of heavy metals by deicing salts in a roadside environment. Water Res. 2004, 38, 720–732. [Google Scholar] [CrossRef] [PubMed]
- Lundmark, A.; Olofsson, B. Chloride deposition and distribution in soils along a deiced highway—Assessment using different methods of measurement. Water Air Soil Pollut. 2007, 182, 173–185. [Google Scholar] [CrossRef]
- Amrhein, C.; Strong, J.E.; Mosher, P.A. Effect of deicing salts on metal and organic matter mobilization in roadside soils. Environ. Sci. Technol. 1992, 26, 703–709. [Google Scholar] [CrossRef]
- Nikolaeva, O.; Rozanova, M.; Karpukhin, M. Distribution of traffic-related contaminants in urban. topsoils across a highway in moscow. J. Soils Sediments 2017, 17, 1045–1053. [Google Scholar] [CrossRef]
- Deutsches Institut für Normung. UNE-77308:2001. Soil Quality. Determination of the Specific Electrical Conductivity; Deutsches Institut für Normung: Berlin, Germany, 2001. [Google Scholar]
- Sonon, L.S.; Saha, U.; Kissel, D.E. Soil Salinity Testing, Data Interpretation and Recommendations; University of Gerogia: Athens, GA, USA, 2012; pp. 1–6. [Google Scholar]
- Bourrie, G. Swelling clays and salt-affected soils: Demixing of Na/Ca clays as the rationale for discouraging the use of sodium adsorption ratio (SAR). Eurasian J. Soil Sci. 2014, 3, 245–253. [Google Scholar] [CrossRef]
- Sibbett, G.S.; Ferguson, L.; Lindstrand, M. Olive Production Manual; Agriculture and Natural Resources, University of California: Oakland, CA, USA, 2005. [Google Scholar]
- United-States-Department-of-Agriculture. Diagnosis and Improvement of Saline and Alkali Soils; Handbook 60; United-States-Department-of-Agriculture: Washington, DC, USA, 1954.
- Bohn, H.L.; Myer, R.A.; O’Connor, G.A. Soil Chemistry; Wiley: Hoboken, NJ, USA, 2002. [Google Scholar]
- Evangelou, V.P.; Coale, F.J. Dependence of the gapon coefficient on exchangeable sodium for mineralogically different soils1. Soil Sci. Soc. Am. J. 1987. [Google Scholar] [CrossRef]
- Harron, W.R.A.; Webster, G.R.; Cairns, R.R. Relationship between exchangeable sodium and sodium adsorption ratio in a solonetzic soil association. Can. J. Soil Sci. 1983, 63, 461–467. [Google Scholar] [CrossRef]
- Shainberg, I.; Shalhevet, J. Soil Salinity Under Irrigation: Processes and Management; Springer: Berlin, Germany, 1984. [Google Scholar]
- Poonia, S.R.; Talibudeen, O. Sodium-calcium exchange equilibria in salt-affected and normal soils. J. Soil Sci. 1977, 28, 276–288. [Google Scholar] [CrossRef]
- Oster, J.D.; Sposito, G. The gapon coefficient and the exchangeable sodium percentage-sodium adsorption ratio relation. Soil Sci. Soc. Am. J. 1980, 44, 258–260. [Google Scholar] [CrossRef]
- Frenkel, H.; Alperovitch, N. The effect of mineral weathering and soil solution concentration on ESR—SAR relationships of arid and semi-arid zone soils from Israel*. J. Soil Sci. 1984, 35, 367–372. [Google Scholar] [CrossRef]
- Hanes, R.E.; Zelazny, L.W.; Blaser, R.E. Effects of Deicing Salts on Water Quality and Biota : Litterature Review and Recommended Research; Highway Research Board, National Academy of Sciences: Washington, DC, USA, 1970. [Google Scholar]
- Fan, Y.; Weisberg, P.J.; Nowak, R.S. Spatio-temporal analysis of remotely-sensed forest mortality associated with road de-icing salts. Sci. Total Environ. 2014, 472, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Stiven, G.A.; Khan, M.A. Saturation percentage as a measure of soil texture in the lower indus basin. J. Soil Sci. 1966, 17, 255–273. [Google Scholar] [CrossRef]
- Trueba, C.; Millán, R.; Schmid, T.; Lago, C.; Roquero, C.; Magister, M. Base de Datos de Propiedades Edafológicas de los Suelos Españoles; Departamento de Impacto Ambiental de la Energía: Aragon, Spain, 1999; Volume 15. [Google Scholar]
- Poljakoff-Mayber, A.; Gale, J. Plants in Saline Environments, Ecological Studies; Springer: Berlin, Germany, 1975. [Google Scholar]
- Swedish National Road Administration. Miljö och Säkerhet på Väg–en Investering för Framtiden; Artikelnummer VV 88227; Swedish National Road Administration: Borlänge, Sweden, 1999. (In Swedish)
- Blomqvist, G. Long-term effects of deicing salt on the roadside environment, Part I: Forestry. In Proceedings of the Ninth Maintenance Management Conference, Juneau, AK, USA, 16–20 July 2001; pp. 179–185. [Google Scholar]
- National Research Concil. Transportation research. In Proceedings of the Ninth Maintenance Management Conference, Juneau, AK, USA, 16–20 July 2001. [Google Scholar]

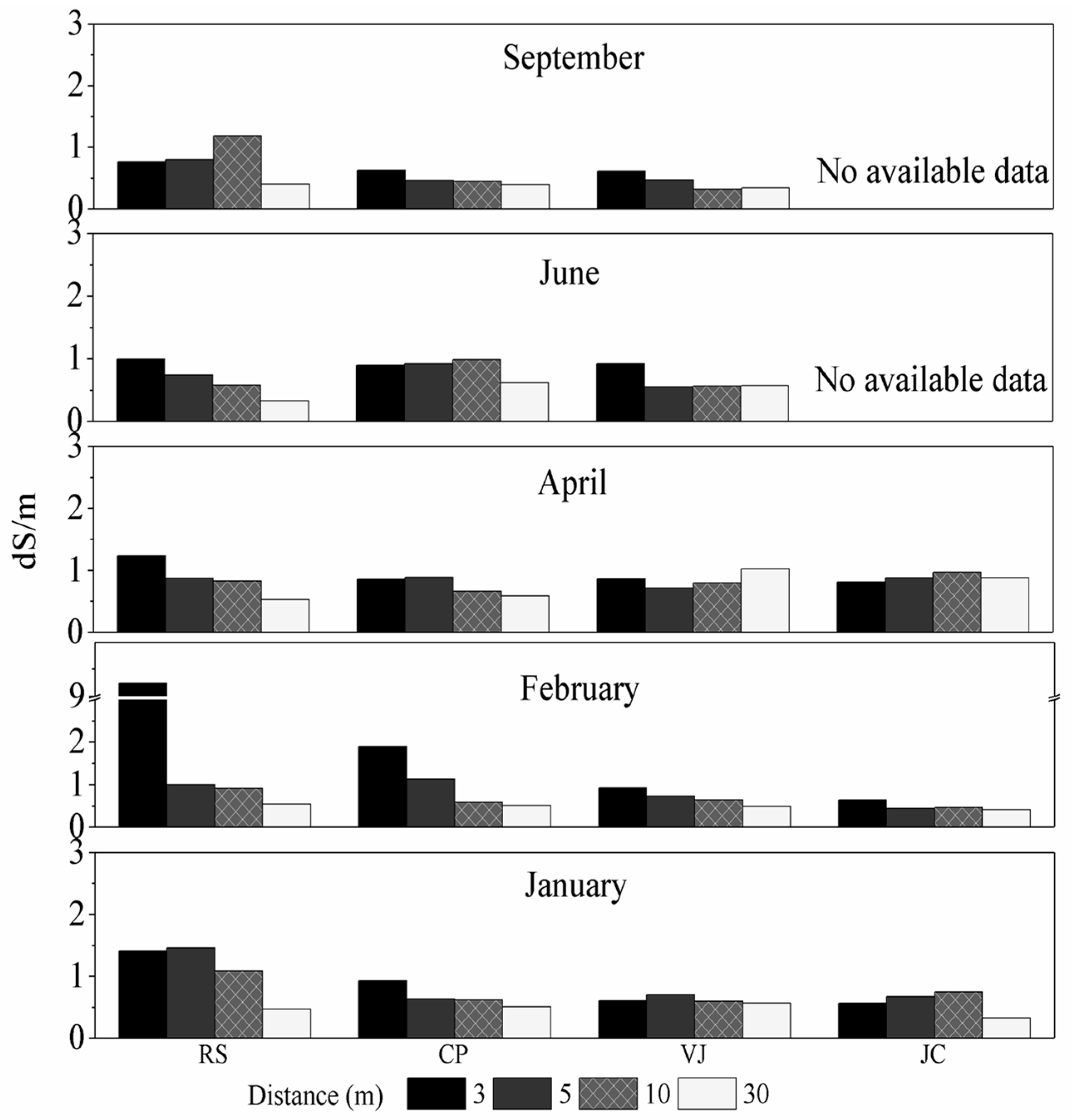
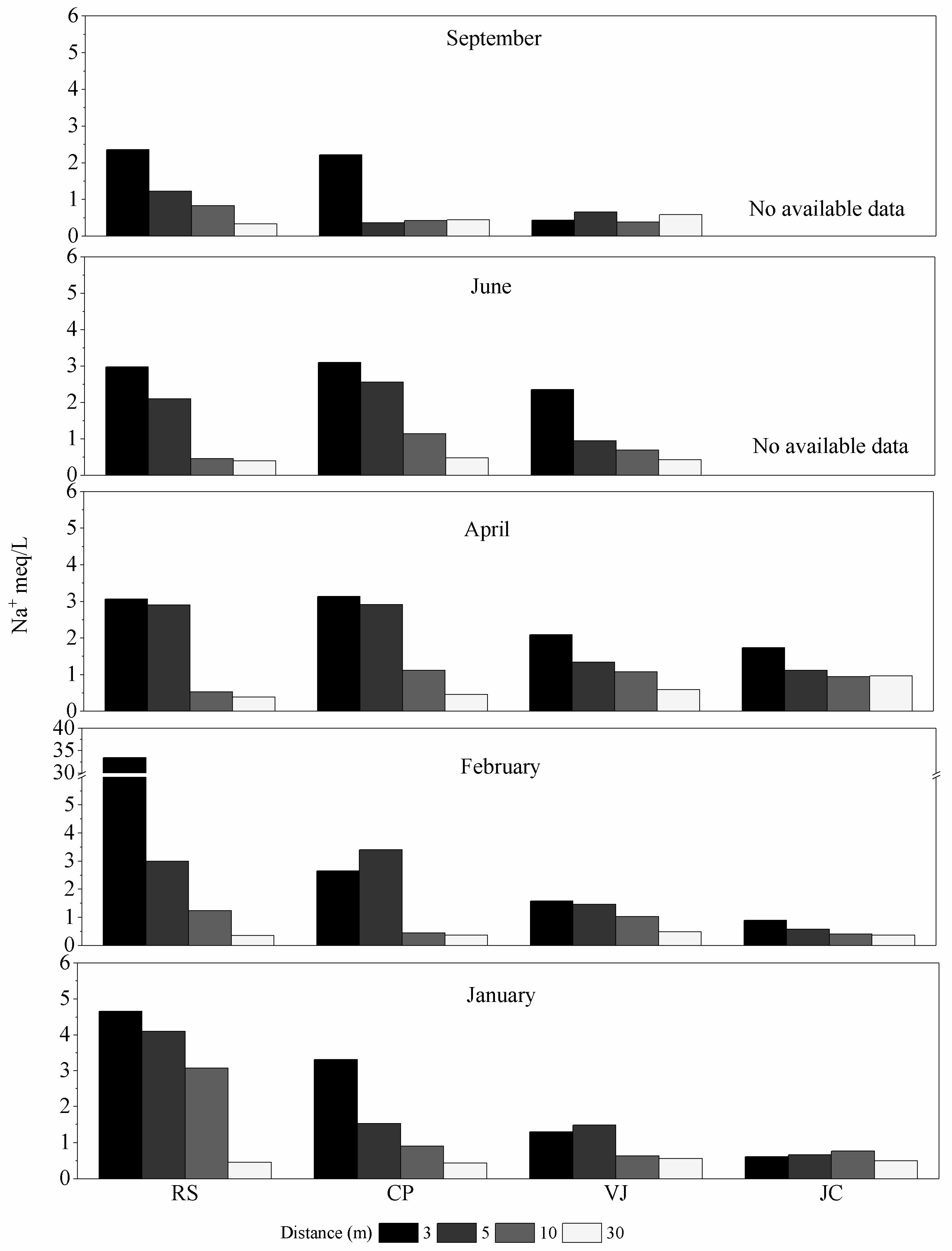
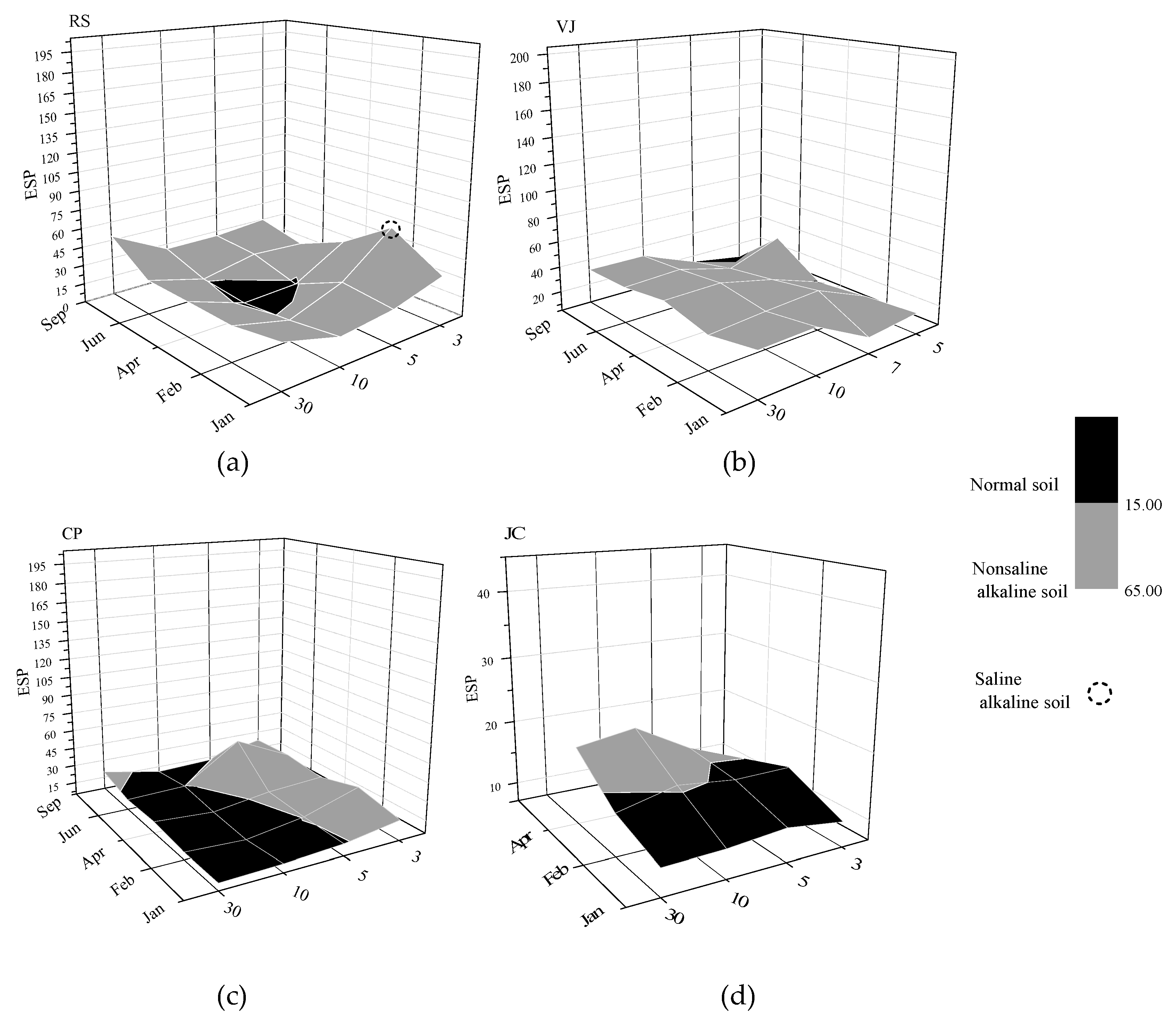
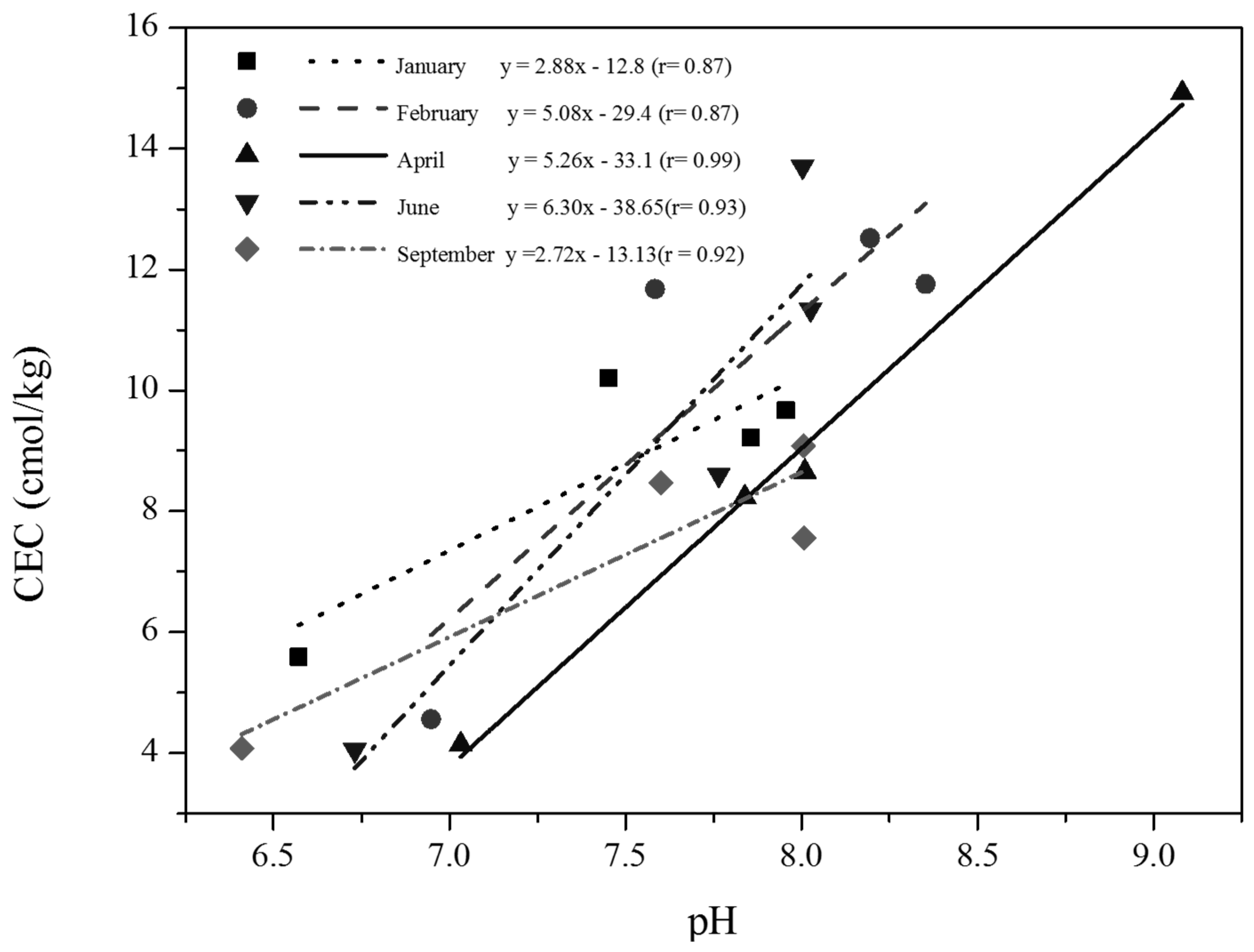
| Zone | Length (km) | Sampling Points | UTM Coordinates |
|---|---|---|---|
| 1 | 14.9 | Rioseta (RS) | Huso 30 X: 7013.53 m; Y: 4,738,572.32 m; Z: 1399 m |
| Canfranc (CP) | Huso 30 X: 702,476.7 m; Y: 4,730,979.37 m; Z: 1004 m | ||
| 2 | 13.8 | Villajuanita (VJ) | Huso 30 X: 700,127.46 m; Y: 4,725,426.96 m; Z: 922 m |
| 3 | 14.9 | Jaca (JC) | Huso 30 X: 696,368.79 m; Y: 4,714,147.81 m; Z: 724 m |
| Class of Soil | EC (dS/m) | SAR | ESP |
|---|---|---|---|
| Normal | Below 4.0 | Below 13 | Below 15 |
| Saline | Above 4.0 | Below 13 | Below 15 |
| Sodic | Below 4.0 | Above 13 | Above 15 |
| Saline-sodic | Above 4.0 | Above 13 | Above 15 |
| Point (Zone) | Equation | R2 |
|---|---|---|
| RS | ESR = 0.0354 + 0.0225 (SAR) | 0.18 |
| CP | ESR = 0.0104 + 0.1952 (SAR) | 0.04 |
| VJ | ESR = 0.6641 − 0.2179 (SAR) | 0.42 |
| JC | ESR = 0.1027 + 0.043 (SAR) | 0.43 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asensio, E.; Ferreira, V.J.; Gil, G.; García-Armingol, T.; López-Sabirón, A.M.; Ferreira, G. Accumulation of De-Icing Salt and Leaching in Spanish Soils Surrounding Roadways. Int. J. Environ. Res. Public Health 2017, 14, 1498. https://doi.org/10.3390/ijerph14121498
Asensio E, Ferreira VJ, Gil G, García-Armingol T, López-Sabirón AM, Ferreira G. Accumulation of De-Icing Salt and Leaching in Spanish Soils Surrounding Roadways. International Journal of Environmental Research and Public Health. 2017; 14(12):1498. https://doi.org/10.3390/ijerph14121498
Chicago/Turabian StyleAsensio, Esther, Víctor J. Ferreira, Gonzalo Gil, Tatiana García-Armingol, Ana M. López-Sabirón, and Germán Ferreira. 2017. "Accumulation of De-Icing Salt and Leaching in Spanish Soils Surrounding Roadways" International Journal of Environmental Research and Public Health 14, no. 12: 1498. https://doi.org/10.3390/ijerph14121498
APA StyleAsensio, E., Ferreira, V. J., Gil, G., García-Armingol, T., López-Sabirón, A. M., & Ferreira, G. (2017). Accumulation of De-Icing Salt and Leaching in Spanish Soils Surrounding Roadways. International Journal of Environmental Research and Public Health, 14(12), 1498. https://doi.org/10.3390/ijerph14121498