Modelling the Ozone-Based Treatments for Inactivation of Microorganisms
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material Matrix
2.2. Microorganisms
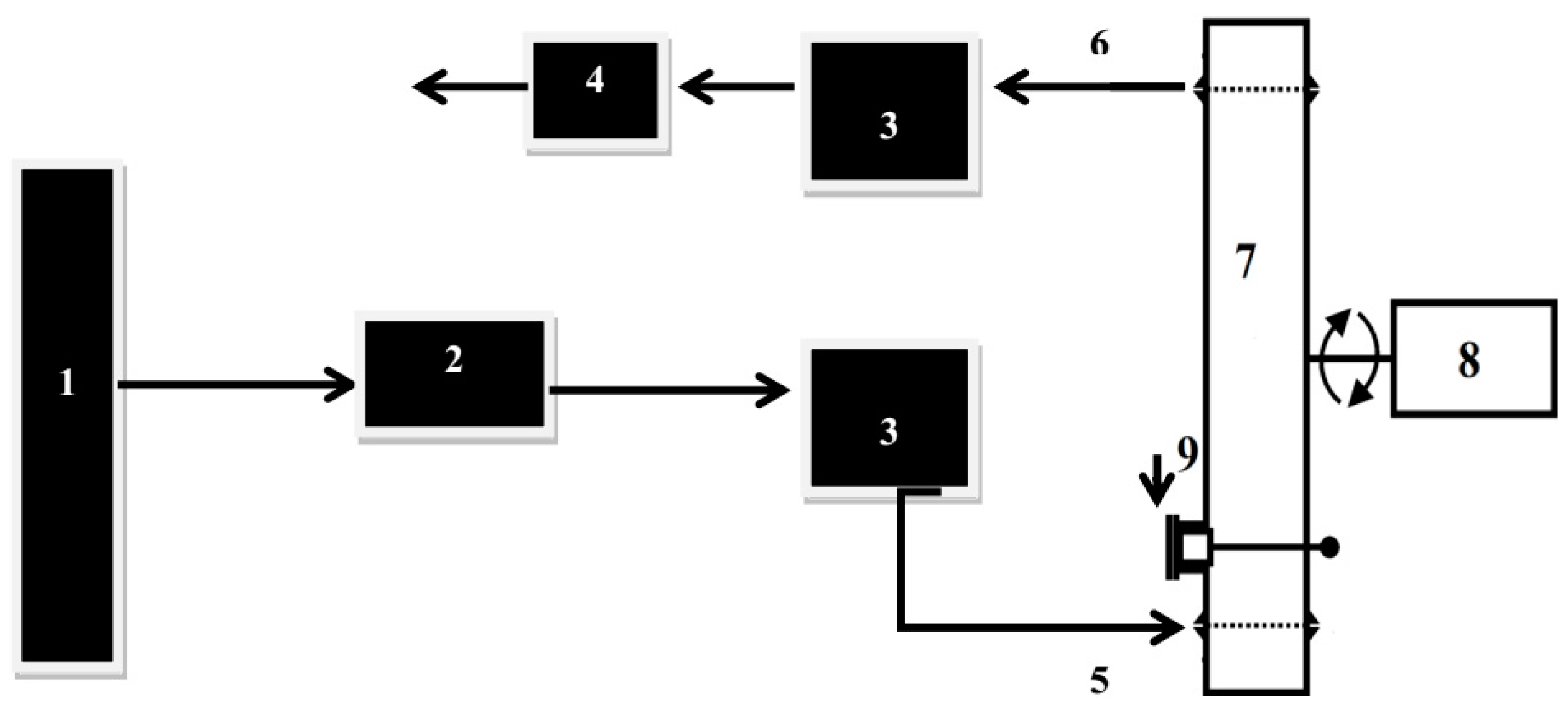
2.3. Ozone Treatment in Dynamic Bed
2.4. Inoculation of Plant Material
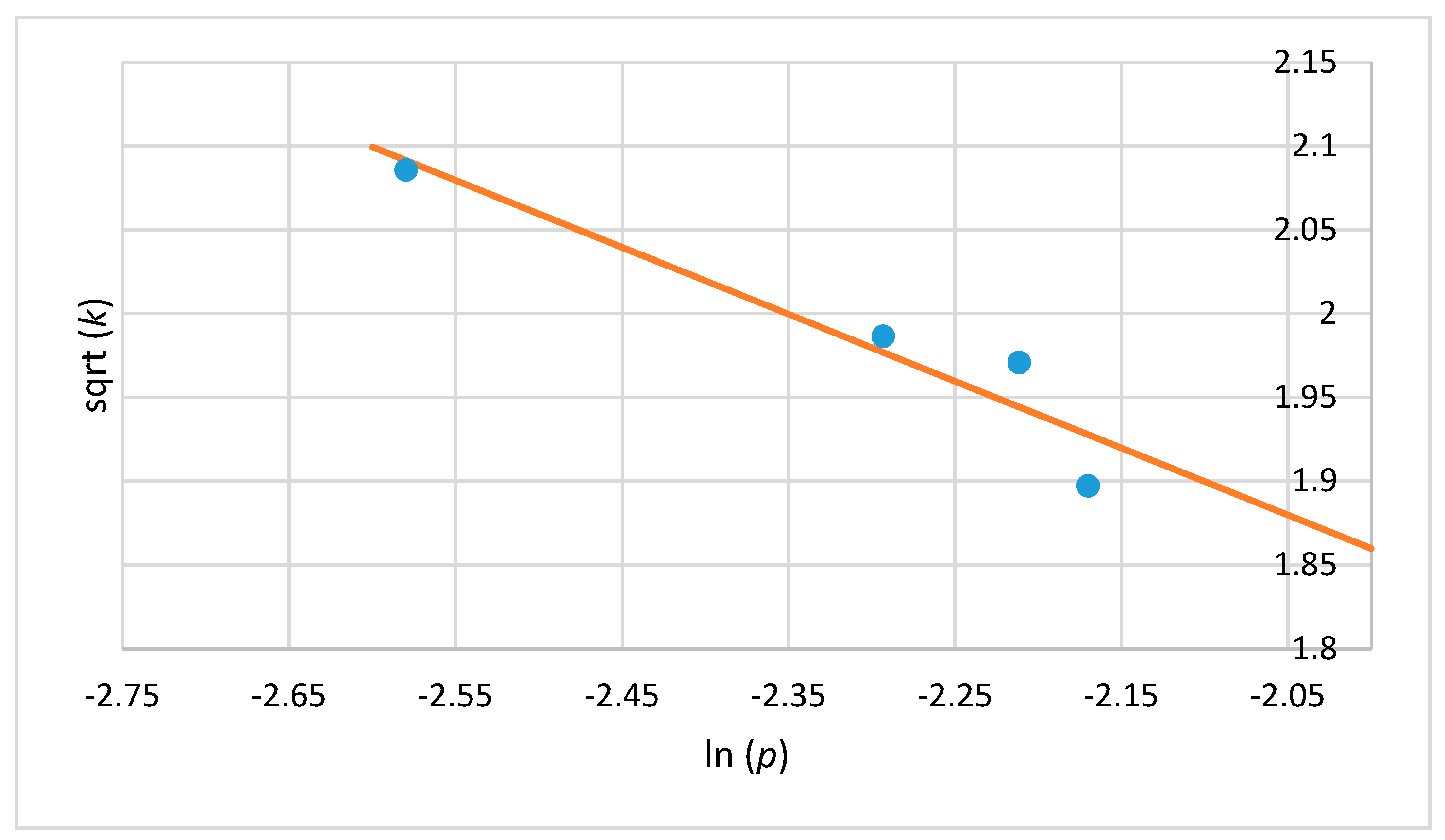
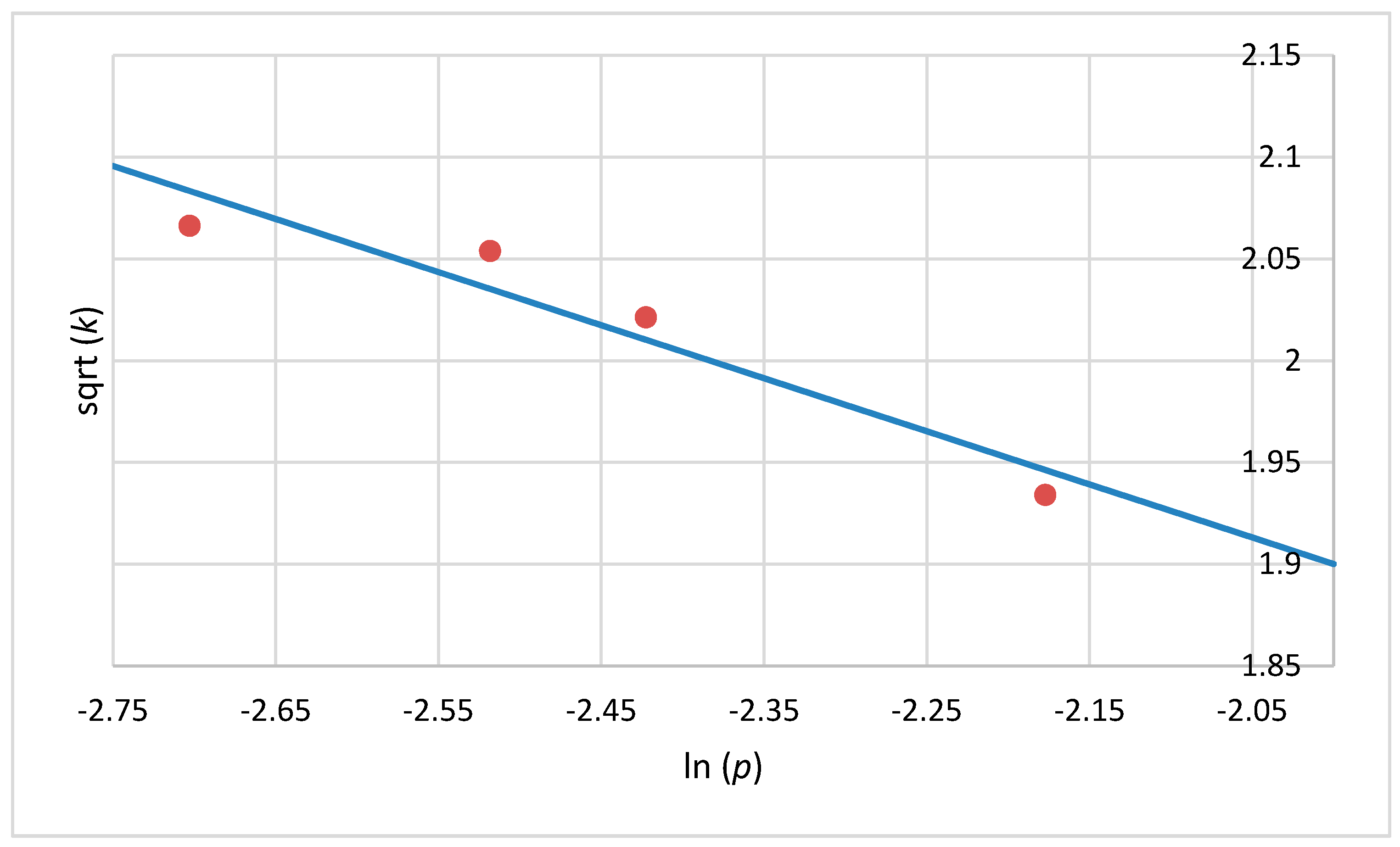
2.5. Mathematical Model
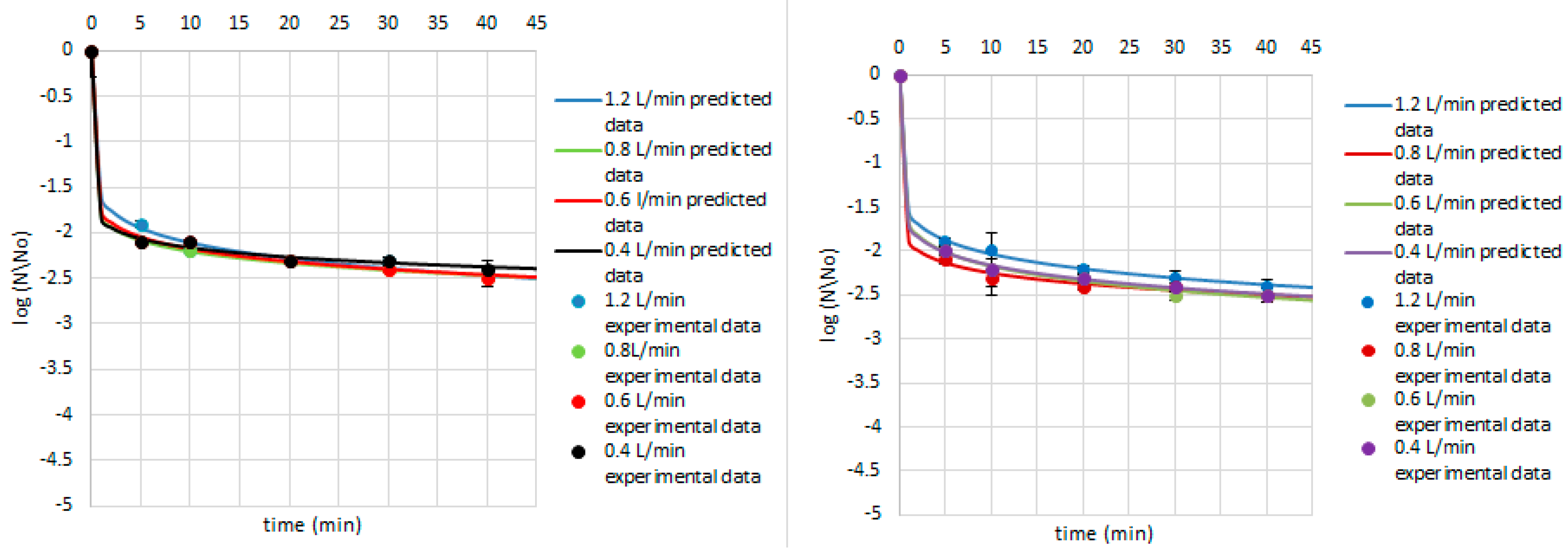
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Banerjee, M.; Sarkar, P.K. Microbiological quality of some retail spices in India. Food Res. Int. 2003, 36, 469–474. [Google Scholar] [CrossRef]
- Le Marc, Y.; Buchovec, I.; George, S.M.; Baranyi, J.; Luksiene, Z. Modelling the photosensitization-based inactivation of Bacillus cereus. J. Appl. Microbiol. 2009, 107, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Valdramidis, V.P.; Cullen, P.J.; Tiwari, B.K. Modelling approaches for ozone processing. In Ozone in Food Processing, 1st ed.; O’Donnell, C., Tiwari, B.K., Cullen, P.J., Rice, R.G., Eds.; Blackwell Publishing Ltd.: Chichester, UK, 2012; pp. 241–263. [Google Scholar]
- Jay, J.M. Indicators of food safety and quality, principles of quality control, and microbial criteria. In Modern Food Microbiology, 6th ed.; Jay, J.M., Ed.; Aspen Publishers, Inc.: Gaithersburg, MD, USA, 2000; pp. 384–402. [Google Scholar]
- Gujer, W.; von Gunten, U. A stochastic model of an ozonation reactor. Water Res. 2003, 37, 1667–1677. [Google Scholar] [CrossRef]
- Hunt, N.K.; Mariñas, B.J. Kinetics of Escherichia coli inactivation with ozone. Water Res. 1997, 31, 1355–1362. [Google Scholar] [CrossRef]
- Steenstrup, L.D.; Floros, J.D. Inactivation of E. coli 0157:H7 in apple cider by ozone at various temperatures and concentrations. J. Food Process Preserv. 2004, 28, 103–116. [Google Scholar] [CrossRef]
- Van Boekel, M.A.J.S. On the use of the Weilbull model to describe thermal inactivation of microbial vegetative cells. Int. J. Food Microbiol. 2002, 74, 139–159. [Google Scholar] [CrossRef]
- Li, H.B.; Finch, G.R.; Smith, D.W.; Belosevic, M. Sequential inactivation of Cryptosporidium parvum using ozone and chlorine. Water Res. 2001, 35, 4339–4348. [Google Scholar] [CrossRef]
- Selma, M.V.; Beltrán, D.; Allende, A.; Chacón-Vera, E.; Gil, M.I. Elimination by ozone of Shigella sonnei in shredded lettuce and water. Food Microbiol. 2007, 24, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.; Valdramidis, V.P.; Cullen, P.J.; Frias, J.; Bourke, P. Ozone treatment of apple juice at different pH levels; effects on the microbial responses of Escherichia coli. Food Microbiol. 2010, 27, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Adamski, W. Modelowanie Systemów Oczyszczania Wód; Wyd. Naukowe PWN: Warszawa, Poland, 2002; p. 253. [Google Scholar]
- Chen, H.; Hoover, D.G. Pressure inactivation kinetics of Yersinia enterocolitica ATCC 35669. Int. J. Food Microbiol. 2003, 87, 161–171. [Google Scholar] [CrossRef]
- Fan, L.; Song, J.; McRae, K.B.; Walker, B.A.; Sharpe, D. Gaseous ozone treatment inactivates Listeria innocua in vitro. J. Appl. Microbiol. 2007, 103, 2657–2663. [Google Scholar] [CrossRef] [PubMed]
- Bialka, K.L.; Demirci, A.; Puri, V.M. Modeling the inactivation of Escherichia coli O157:H7 and Salmonella enterica on raspberries and strawberries resulting from exposure to ozone or pulsed UV-light. J. Food Eng. 2008, 85, 444–449. [Google Scholar] [CrossRef]
- Patil, S.; Valdramidis, V.P.; Cullen, P.J.; Frias, J.; Bourke, P. Ozone inactivation of Listeria monocytogenes and Listeria innocua in orange juice using a bubble column. Food Control. 2010, 21, 1723–1730. [Google Scholar] [CrossRef]
- Restaino, L.; Frampton, E.; Hemphill, J.B.; Palnikar, P. Efficacy of ozonated water against various food related microorganisms. Appl. Environ. Microbiol. 1995, 61, 3471–3475. [Google Scholar] [PubMed]
- Miller, F.A.; Silva, C.L.M.; Brandao, T.R.S. A review on ozone-based treatments for fruit and vegetables preservation. Food Eng. Rev. 2013, 5, 77–106. [Google Scholar] [CrossRef]
- Kawon. Available online: http://www.kawon.com.pl (accessed on 31 March 2017).
- Brodowska, A.J.; Śmigielski, K.; Nowak, A.; Czyżowska, A.; Otlewska, A. The impact of ozone treatment in dynamic bed parameters on changes in biologically active substances of juniper berries. PLoS ONE 2015. [Google Scholar] [CrossRef] [PubMed]
- Śmigielski, K.; Piątkowski, M.; Nowak, A.; Brodowska, A. Komora do Dekontaminacji Surowców Roślinnych Ozonem w Złożu Dynamicznym. Patent Application No. 409933, 27 September 2014. [Google Scholar]
- Brodowska, A.J.; Nowak, A.; Śmigielski, K. Ozone in the food industry: Principles of ozone treatment, mechanisms of action, and applications: An overview. Crit. Rev. Food Sci. 2017, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Akbas, M.Y.; Ozdemir, M. Effect of gaseous ozone on microbial inactivation and sensory of flaked red peppers. Int. J. Food Sci. Technol. 2008, 43, 1657–1662. [Google Scholar] [CrossRef]
- Peleg, M.; Cole, M.B. Reinterpretation of microbial survival curves. Crit. Rev. Food Sci. 1998, 38, 353–380. [Google Scholar] [CrossRef] [PubMed]
- Dätwyler, C.; Stucki, T. Parametric Survival Models. Available online: http://data.princeton.edu/pop509/ParametricSurvival.pdf (accessed on 18 August 2017).
- Yuanfang, C.; Yu, H.; van Gelder, P.; Zhigui, S. Study of parameter estimation methods for Pearson-III distribution in flood frequency analysis. Extrem. Extrem. Extraordinary Floods 2002, 271, 263–269. [Google Scholar]
- Fritze, D.; Pukall, R. Culture media for Bacillus spp. and related genera relevant to foods. In Handbook of Culture Media for Food and Water Microbiology, 3rd ed.; Corry, J.E.L., Curtis, G.D.W., Baird, R.M., Eds.; RSC Publishing: Cambridge, UK, 2012; pp. 90–114. [Google Scholar]
- From, C.; Hormazabal, V.; Granum, P.E. Food poisoning associated with pumilacidin-producing Bacillus pumilus in rice. Int. J. Food Microbiol. 2007, 115, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Brodowska, A.J.; Śmigielski, K.; Nowak, A.; Brodowska, K.; Catthoor, R.; Czyżowska, A. The impact of ozone treatment on changes in biologically active substances of cardamom seeds. J. Food Sci. 2014, 79, C1649–C1655. [Google Scholar] [CrossRef] [PubMed]
- Sagoo, S.K.; Little, C.L.; Greenwood, M.; Mithani, V.; Grant, K.A.; McLauchlin, J.; de Pinna, E.; Threlfall, E.J. Assessment of the microbiological safety of dried spices and herbs from production and retail premises in the United Kingdom. Food Microbiol. 2009, 26, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Couvert, O.; Gaillard, S.; Savy, N.; Mafart, P.; Leguerinel, I. Survival curves of heated bacterial spores: Effect of environmental factors on Weibull parameters. Int. J. Food Microbiol. 2005, 101, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Akbas, M.Y.; Ozdemir, M. Application of gaseous ozone to control populations of Escherichia coli, Bacillus cereus and Bacillus cereus spores in dried figs. Food Microbiol. 2008, 25, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Akbas, M.Y.; Ozdemir, M. Effectiveness of ozone for inactivation of Escherichia coli and Bacillus cereus in pistachios. Int. J. Food Sci. Tech. 2006, 41, 513–519. [Google Scholar] [CrossRef]
- Naitoh, S.; Okada, Y.; Sakai, T. Studies on utilization of ozone in food preservation: III. Microbicidal properties of ozone on cereal grains, cereal grain powders, peas, beans and whole spices. J. Jpn. Soc. Food Sci. Technol. 1987, 34, 788–793. [Google Scholar] [CrossRef]
- Zagon, J.; Dehne, L.I.; Wirz, J.; Linke, B.; Boegl, K.W. Ozone treatment for removal of microorganisms from spices as an alternative to ethylene oxide fumigation or irradiation: Results of a practical study. Bundesgesundheitsblatt 1992, 35, 20–23. [Google Scholar]
- Kim, J.G.; Yousef, A.E.; Dave, S. Application of ozone for enhancing the microbiological safety and quality of foods: A review. J. Food Protect. 1999, 62, 1071–1087. [Google Scholar] [CrossRef]
- Victorin, K. Review of the genotoxicity of ozone. Mutat. Res. 1992, 277, 221–238. [Google Scholar] [CrossRef]
- Półjanowska, M.; Kędzia, A.; Kochańska, B. Wrażliwość bakterii mikroaerofilnych izolowanych z jamy ustnej na działanie ozonu. Badania in vitro. Ann. Acad. Med. Stetin. 2007, 3, 114–118. [Google Scholar]
- Palou, L.; Smilanick, J.L.; Crisosto, C.H.; Mansour, M. Effect of gaseous ozone exposure on the development of green and blue molds on cold stored citrus fruit. Plant Dis. 2001, 85, 632–638. [Google Scholar] [CrossRef]
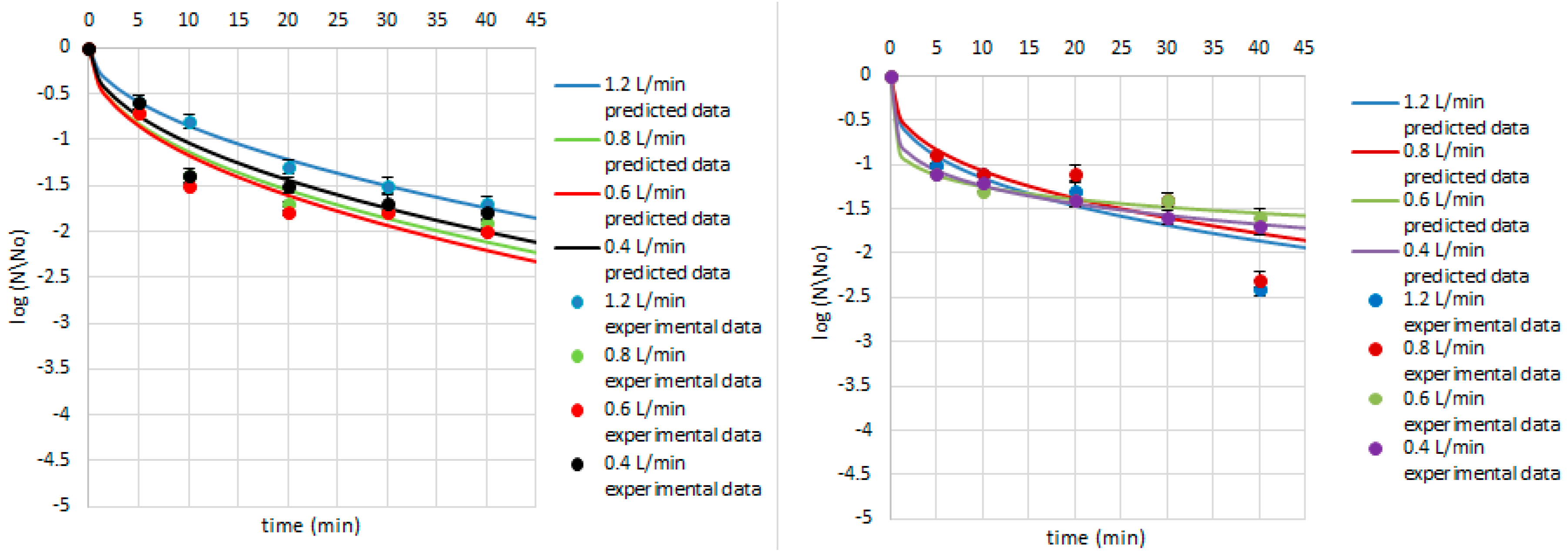
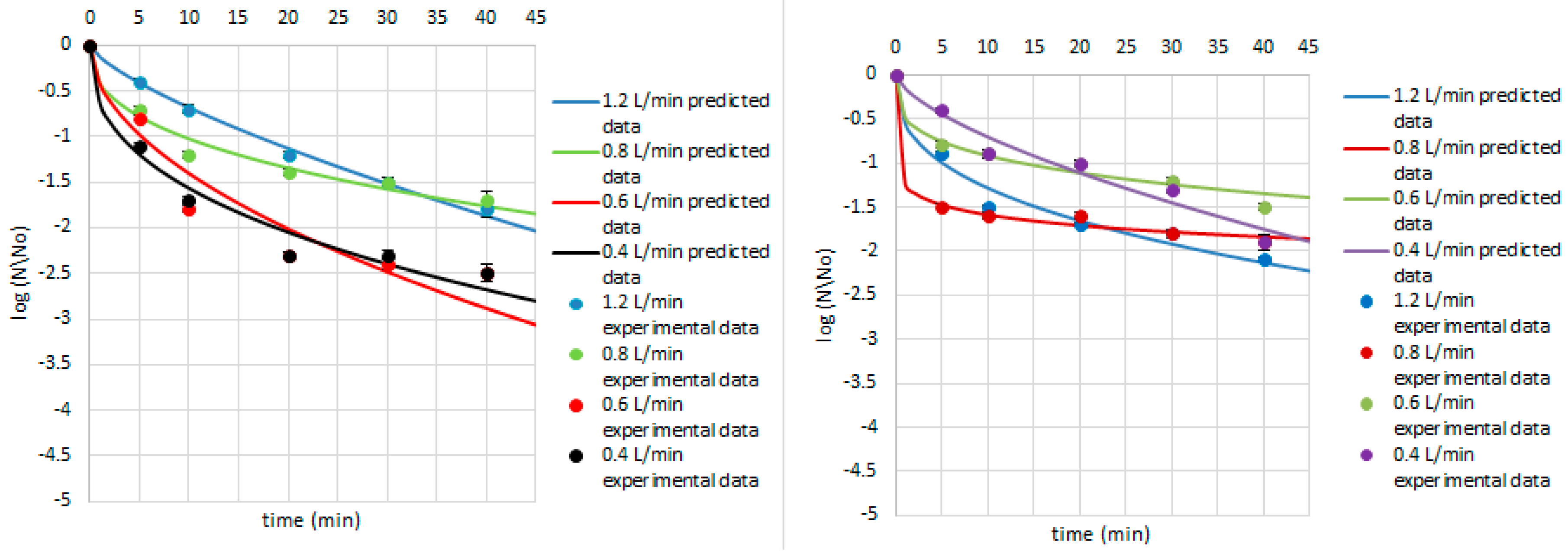
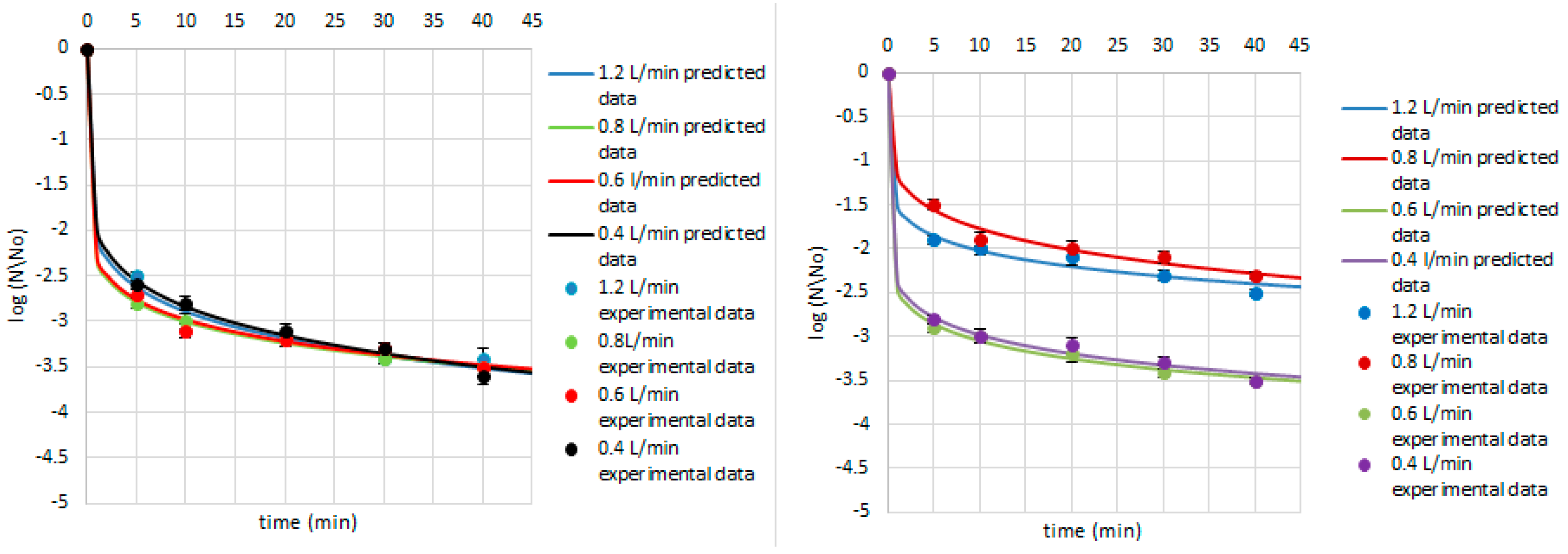
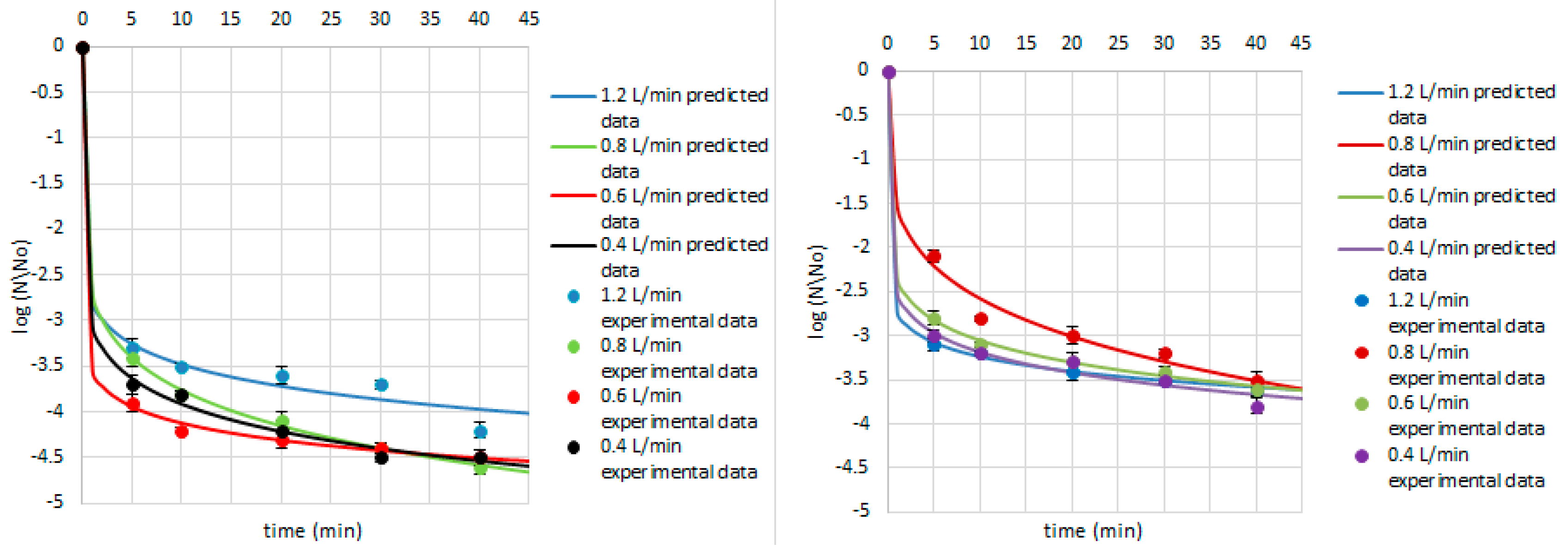
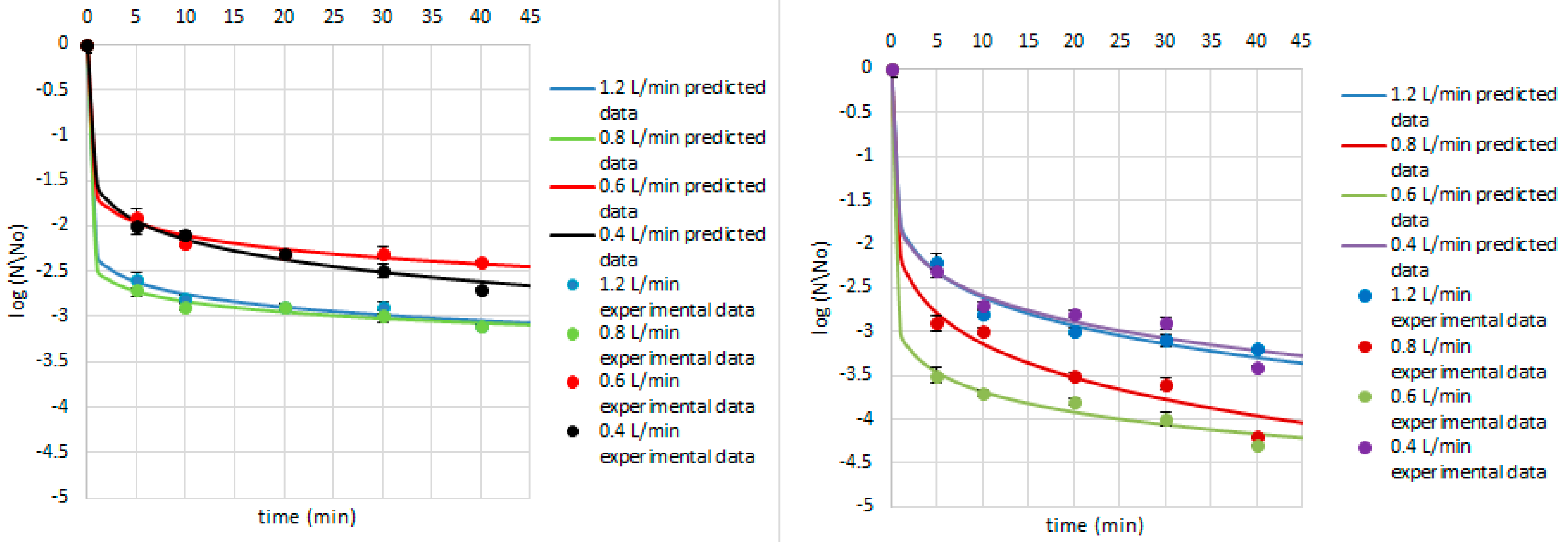
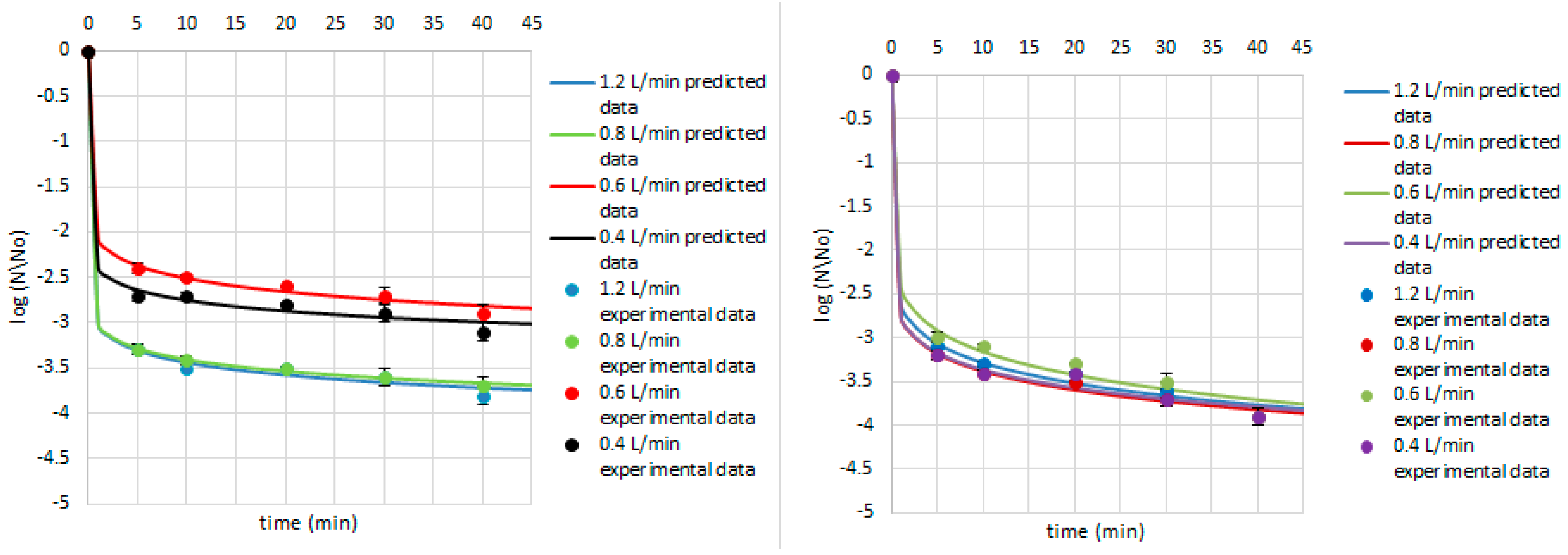
| Research Matrix | Microorganism | Ozone Flow Rate (L/min) | Weibull Model Parameters | Fitted Parameters | |||
|---|---|---|---|---|---|---|---|
| k | p | tR (min) | RMSE | R2 | |||
| Juniper berries | B. cereus | 1.2 | 3.60 | 0.11 | 0.02 | 0.01 | 0.99 |
| 0.8 | 4.35 | 0.08 | 0.00 | 0.02 | 0.93 | ||
| 0.6 | 3.88 | 0.11 | 0.01 | 0.02 | 0.97 | ||
| 0.4 | 3.95 | 0.10 | 0.01 | 0.01 | 0.98 | ||
| B. subtilis | 1.2 | 1.20 | 0.35 | 6.62 | 0.20 | 0.73 | |
| 0.8 | 1.07 | 0.36 | 8.25 | 0.22 | 0.73 | ||
| 0.6 | 2.01 | 0.16 | 2.37 | 0.05 | 0.92 | ||
| 0.4 | 1.74 | 0.22 | 3.69 | 0.03 | 0.98 | ||
| B. pumilus | 1.2 | 1.28 | 0.37 | 5.03 | 0.11 | 0.91 | |
| 0.8 | 2.87 | 0.11 | 0.12 | 0.04 | 0.84 | ||
| 0.6 | 1.12 | 0.28 | 13.62 | 0.10 | 0.89 | ||
| 0.4 | 0.36 | 0.66 | 17.04 | 0.18 | 0.92 | ||
| E. coli | 1.2 | 3.50 | 0.12 | 0.03 | 0.04 | 0.90 | |
| 0.8 | 2.68 | 0.18 | 0.44 | 0.05 | 0.93 | ||
| 0.6 | 5.67 | 0.09 | 0.00 | 0.02 | 0.97 | ||
| 0.4 | 5.46 | 0.10 | 0.00 | 0.02 | 0.95 | ||
| P. fluorescens | 1.2 | 6.30 | 0.07 | 0.00 | 0.01 | 0.99 | |
| 0.8 | 3.56 | 0.22 | 0.14 | 0.06 | 0.93 | ||
| 0.6 | 5.42 | 0.11 | 0.00 | 0.01 | 0.98 | ||
| 0.4 | 5.81 | 0.10 | 0.00 | 0.03 | 0.91 | ||
| E. cinnamopurpureum | 1.2 | 4.08 | 0.17 | 0.03 | 0.06 | 0.90 | |
| 0.8 | 4.91 | 0.17 | 0.01 | 0.06 | 0.90 | ||
| 0.6 | 6.93 | 0.09 | 0.00 | 0.03 | 0.91 | ||
| 0.4 | 4.16 | 0.16 | 0.02 | 0.06 | 0.88 | ||
| A. niger | 1.2 | 6.03 | 0.10 | 0.00 | 0.03 | 0.91 | |
| 0.8 | 6.36 | 0.09 | 0.00 | 0.02 | 0.95 | ||
| 0.6 | 5.58 | 0.12 | 0.00 | 0.04 | 0.87 | ||
| 0.4 | 6.36 | 0.09 | 0.00 | 0.03 | 0.86 | ||
| Research Matrix | Microorganism | Ozone Flow Rate (L/min) | Weibull Model Parameters | Fitted Parameters | |||
|---|---|---|---|---|---|---|---|
| k | p | tR (min) | RMSE | R2 | |||
| Cardamom seeds | B. cereus | 1.2 | 3.74 | 0.11 | 0.01 | 0.04 | 0.90 |
| 0.8 | 4.22 | 0.08 | 0.00 | 0.01 | 0.98 | ||
| 0.6 | 4.09 | 0.09 | 0.00 | 0.03 | 0.91 | ||
| 0.4 | 4.27 | 0.07 | 0.00 | 0.02 | 0.89 | ||
| B. subtilis | 1.2 | 0.59 | 0.52 | 13.75 | 0.05 | 0.99 | |
| 0.8 | 0.92 | 0.45 | 7.59 | 0.18 | 0.86 | ||
| 0.6 | 0.94 | 0.46 | 7.12 | 0.21 | 0.82 | ||
| 0.4 | 0.79 | 0.48 | 9.34 | 0.22 | 0.81 | ||
| B. pumilus | 1.2 | 0.30 | 0.73 | 16.89 | 0.05 | 0.99 | |
| 0.8 | 0.96 | 0.39 | 9.37 | 0.12 | 0.91 | ||
| 0.6 | 0.98 | 0.52 | 5.18 | 0.22 | 0.84 | ||
| 0.4 | 1.48 | 0.39 | 3.12 | 0.11 | 0.93 | ||
| E. coli | 1.2 | 4.82 | 0.14 | 0.00 | 0.05 | 0.87 | |
| 0.8 | 5.40 | 0.11 | 0.00 | 0.01 | 0.99 | ||
| 0.6 | 5.32 | 0.11 | 0.00 | 0.03 | 0.93 | ||
| 0.4 | 4.62 | 0.15 | 0.00 | 0.02 | 0.97 | ||
| P. fluorescens | 1.2 | 6.44 | 0.10 | 0.00 | 0.05 | 0.80 | |
| 0.8 | 6.25 | 0.14 | 0.00 | 0.01 | 0.99 | ||
| 0.6 | 8.20 | 0.06 | 0.00 | 0.01 | 0.96 | ||
| 0.4 | 7.06 | 0.11 | 0.00 | 0.02 | 0.95 | ||
| E. cinnamopurpureum | 1.2 | 5.37 | 0.07 | 0.00 | 0.02 | 0.91 | |
| 0.8 | 5.71 | 0.06 | 0.00 | 0.02 | 0.90 | ||
| 0.6 | 3.84 | 0.10 | 0.01 | 0.04 | 0.88 | ||
| 0.4 | 3.57 | 0.14 | 0.05 | 0.03 | 0.95 | ||
| A. niger | 1.2 | 6.96 | 0.06 | 0.00 | 0.02 | 0.86 | |
| 0.8 | 6.95 | 0.05 | 0.00 | 0.01 | 0.97 | ||
| 0.6 | 4.78 | 0.08 | 0.00 | 0.02 | 0.91 | ||
| 0.4 | 5.51 | 0.06 | 0.00 | 0.03 | 0.78 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brodowska, A.J.; Nowak, A.; Kondratiuk-Janyska, A.; Piątkowski, M.; Śmigielski, K. Modelling the Ozone-Based Treatments for Inactivation of Microorganisms. Int. J. Environ. Res. Public Health 2017, 14, 1196. https://doi.org/10.3390/ijerph14101196
Brodowska AJ, Nowak A, Kondratiuk-Janyska A, Piątkowski M, Śmigielski K. Modelling the Ozone-Based Treatments for Inactivation of Microorganisms. International Journal of Environmental Research and Public Health. 2017; 14(10):1196. https://doi.org/10.3390/ijerph14101196
Chicago/Turabian StyleBrodowska, Agnieszka Joanna, Agnieszka Nowak, Alina Kondratiuk-Janyska, Marcin Piątkowski, and Krzysztof Śmigielski. 2017. "Modelling the Ozone-Based Treatments for Inactivation of Microorganisms" International Journal of Environmental Research and Public Health 14, no. 10: 1196. https://doi.org/10.3390/ijerph14101196
APA StyleBrodowska, A. J., Nowak, A., Kondratiuk-Janyska, A., Piątkowski, M., & Śmigielski, K. (2017). Modelling the Ozone-Based Treatments for Inactivation of Microorganisms. International Journal of Environmental Research and Public Health, 14(10), 1196. https://doi.org/10.3390/ijerph14101196





