Vitamin D Concentration in Maternal and Umbilical Cord Blood by Season
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Laboratory Analysis and Data Collection
2.3. Statistical Analysis
3. Results
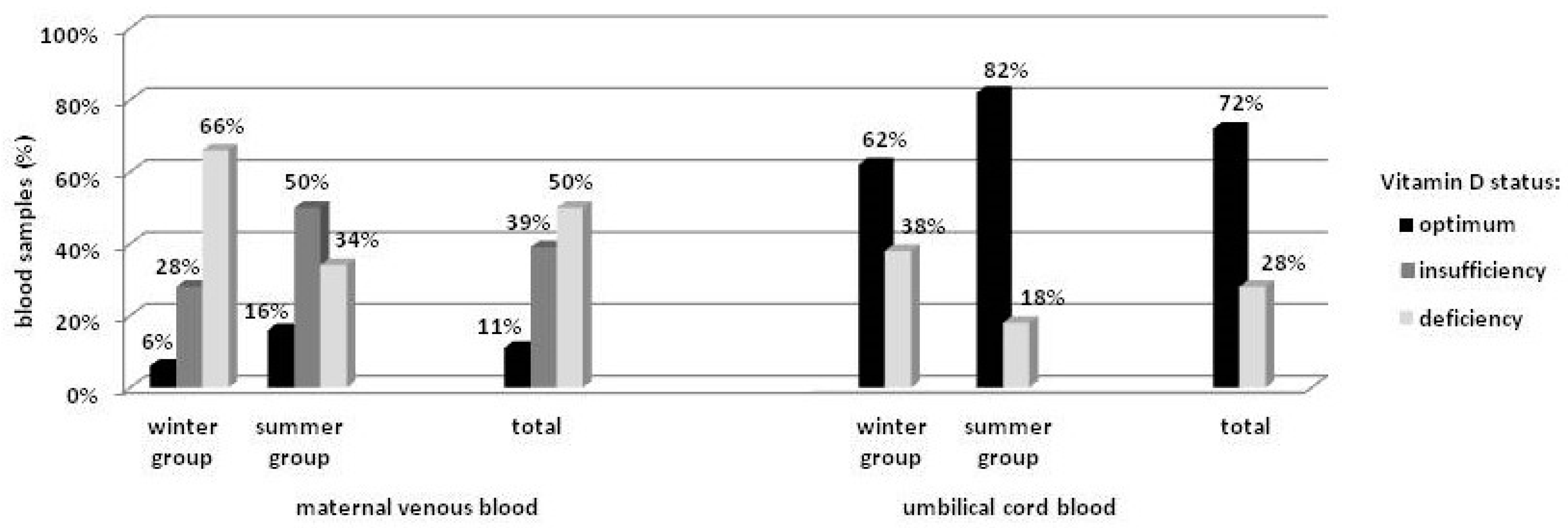
3.1. Maternal Venous Blood by Season
3.2. Umbilical Cord Blood by Season
3.3. Maternal vs. Umbilical Cord Blood
3.4. Other Factors Related to Serum Vitamin D Concentration
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Saraf, R.; Morton, S.M.; Camargo, C.A.; Grant, C.C. Global summary of maternal and newborn vitamin D status—Systematic review. Matern. Child. Nutr. 2016, 12, 647–668. [Google Scholar] [CrossRef] [PubMed]
- Karras, S.N.; Anagnostis, P.; Annweiler, C.; Naughton, D.P.; Petroczi, A.; Bili, E.; Harizopoulou, V.; Tarlatzis, B.C.; Persinkai, A.; Papadopoulou, F.; et al. Maternal vitamin D status during pregnancy: The Mediterranean reality. Eur. J. Clin. Nutr. 2014, 68, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Skowrońska-Jóźwiak, E.; Lebiedzińska, K.; Smyczyńska, J.; Lewandowski, K.C.; Głowacka, E.; Lewiński, A. Effects of maternal vitamin D status on pregnancy outcomes, health of pregnant women and their offspring. Neuroendocrinol. Lett. 2014, 35, 367–372. [Google Scholar] [PubMed]
- Wei, S.Q.; Qi, H.P.; Luo, Z.C.; Fraser, W.D. Maternal vitamin D status and pregnancy outcomes: A systematic review and meta-analysis. J. Matern. Fetal Neonatal Med. 2013, 26, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Aghajafari, F.; Nagulesapilai, T.; Ronksely, P.; Tough, S.; O´Beirne, M.; Rabi, D. Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: Systematic review and meta-analysis of observational studies. BMJ 2013, 346, f1169. [Google Scholar] [CrossRef] [PubMed]
- Bartoszewicz, Z.; Kondracka, A.; Krasnodębska-Kiljańska, M.; Niedźwiecka, B.; Popow, M.; Ambroziak, U.; Bednarczuk, T. Vitamin D insufficiency in healthy pregnant women living in Warsaw. Ginekol. Pol. 2013, 84, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Dovnik, A.; Mujezinović, F.; Treiber, M.; Pečovnik Balon, B.; Gorenjak, M.; Maver, U.; Takač, I. Seasonal variations of vitamin D concentrations in pregnant women and neonates in Slovenia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 181, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Płudowski, P.; Karczmarewicz, E.; Bayer, M.; Carter, G.; Chlebna-Sokół, D.; Czech-Kowalska, J.; Dębski, R.; Decsi, T.; Dobrzańska, A.; Franek, E.; et al. Practical guidelines for the supplementation of vitamin D and the treatment of deficits in Central Europe—Recommended vitamin D intakes in the general population and groups at risk of vitamin D deficiency. Endokrynol. Pol. 2013, 64, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Xu, Y.; Lv, L.; Zhang, M. Association between vitamin D status and the risk of gestational diabetes mellitus: A meta-analysis. Arch. Gynecol. Obstet. 2016, 293, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L. New insights into the vitamin D requirements during pregnancy. Bone Res. 2017, 5, 17030. [Google Scholar] [CrossRef] [PubMed]
- Streym, S.; Moller, U.K.; Rejnmark, L.; Heickendorff, L.; Mosekilde, L.; Vestergaard, P. Maternal and infant vitamin D status during the first 9 months of infant life—A cohort study. Eur. J. Clin. Nutr. 2013, 67, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- Nicolaidou, P.; Hatzistamatiou, Z.; Papadopoulou, A.; Kaleyias, J.; Floropoulou, E.; Lagona, E.; Tsagris, V.; Costalos, C.; Antsaklis, A. Low vitamin D status in mother-newborn pairs in Greece. Calcif. Tissue Int. 2006, 78, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Flynn, A.; Hirvonen, T.; Mensink, G.B.; Ocké, M.C.; Serra-Majem, L.; Stoś, K.; Szponar, L.; Tetens, I.; Turrini, A.; Fletcher, R.; et al. Intake of selected nutrients from foods, from fortification and from supplements in various European countries. Food Nutr. Res. 2009, 1, 1–51. [Google Scholar] [CrossRef] [PubMed]
- Wierzejska, R. Dose of the vitamin D in prenatal supplements and the current prevention its deficiency at mothers and newborns. Gin. Pol. Med. Project 2015, 37, 49–53. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Kiely, M.; O’Donovan, S.M.; Kenny, L.C.; O´B Hourihane, J.; Irvine, A.D.; Murray, D.M. Vitamin D metabolite concentrations in umbilical cord blood serum and associations with clinical characteristics in large prospective mother-infant cohort in Ireland. J. Steroid Biochem. Mol. Biol. 2017, 167, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Zasimovich, A.; Fijałkowska, A.; Chełchowska, M.; Maciejewski, T. Maternal serum vitamin D and parathormone concentrations during gestation and in umbilical cord blood—Pilot study. J. Matern. Fetal Neonatal Med. 2017, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Domaracki, P.; Sadlecki, P.; Odrowąż-Sypniewska, G.; Dzikowska, E.; Walentowicz, P.; Siodmiak, J.; Grabiec, M.; Walentowicz-Sadlecka, M. Serum 25(OH) vitamin D levels in Polish women during pregnancies complicated by hypertensive disorders and gestational diabetes. Int. J. Mol. Sci. 2016, 17, 1574. [Google Scholar] [CrossRef] [PubMed]
- Baczyńska-Strzecha, M.; Kalinka, J. Assessment of correlation between vitamin D level and prevalence of preterm births in the population of pregnant women in Poland. Int. J. Occup. Med. Environ. Health 2017, 30. [Google Scholar] [CrossRef] [PubMed]
- Gellert, S.; Ströhle, A.; Bitterlich, N.; Hahn, A. Higher prevalence of vitamin D deficiency in German pregnant women compared to non-pregnant women. Arch. Gynecol. Obstet. 2017, 296, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Godang, K.; Frøslie, K.F.; Henriksen, T.; Qvigstad, E.; Bollerslev, J. Seasonal variation in maternal and umbilical cord 25(OH) vitamin D and their associations with neonatal adiposity. Eur. J. Endocrinol. 2014, 170, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Carlson, C.R.; Uriu-Adams, J.Y.; Chambers, C.D.; Yevtushok, L.; Zymak-Zakutnya, N.; Chan, P.H.; Schafer, J.J.; Wertelecki, W.; Keen, C.L. Vitamin D deficiency in pregnant Ukrainian women: Effects of alcohol consumption on vitamin D status. J. Am. Coll. Nutr. 2017, 36, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Halicioglu, O.; Aksit, S.; Koc, F.; Sezin, A.; Akman, S.A.; Albudak, E.; Yaprak, I.; Coker, I.; Colak, A.; Ozturk, C.; et al. Vitamin D deficiency in pregnant woman and their neonates in spring time in western Turkey. Paediatr. Perinat. Epidemiol. 2012, 26, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, L.M.; Catov, J.M.; Zmuda, J.M.; Cooper, M.E.; Parrott, M.S.; Roberts, J.M.; Marazita, M.L.; Simhan, H.N. Maternal serum 25-hydroxyvitamin D concentrations are associated with small-for-gestational age births in white women. J. Nutr. 2010, 140, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Vandevijvere, S.; Amsalkhir, S.; Van Oyen, H.; Moreno-Reyes, R. High prevalence of vitamin D deficiency in pregnant women: A national cross-sectional survey. PLoS ONE 2012, 7, e43868. [Google Scholar] [CrossRef] [PubMed]
- Jacquemyn, Y.; Ajaji, M.; Karepouan, N. Vitamin D levels in maternal serum and umbilical cord blood in a multi-ethnic population in Antwerp, Belgium. Facts Views Vis. ObGyn. 2013, 5, 3–5. [Google Scholar]
- Shakiba, M.; Iranmanesh, M.R. Vitamin D requirement in pregnancy to prevent deficiency in neonates: A randomized trial. Singap. Med. 2013, 54, 285–288. [Google Scholar] [CrossRef]
- Skowrońska-Jóźwiak, E.; Adamczewski, Z.; Tyszkiewicz, A.; Krawczyk-Rusiecka, K.; Lewandowski, K.; Lewiński, A. Assessment of adequacy of vitamin D supplementation during pregnancy. Ann. Agric. Environ. Med. 2014, 21, 198–200. [Google Scholar] [PubMed]
- EUR-Lex. Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002 on the Approximation of the Laws of the MEMBER States Relating to Food Supplements. OJ L183. 12 July 2002; 51–57. [Google Scholar]
- Li, W.; Green, T.J.; Innis, S.M.; Barr, S.I.; Whiting, S.J.; Shand, A.; von Dadelszen, P. Suboptimal vitamin D levels in pregnant women despite supplement use. Can. J. Public Health 2011, 102, 308–312. [Google Scholar] [PubMed]
- Dębski, R.; Karowicz-Bilińska, A.; Oszukowski, P.; Paszkowski, T.; Spaczyński, M. Recommendations of the Polish Gynecological Society concerning docosahexaenoic acid supplementation in the prevention of preterm birth. Ginekol. Pol. 2014, 85, 318–320. [Google Scholar]
- Cadario, F.; Savastio, S.; Pozzi, E.; Capelli, A.; Dondi, E.; Gatto, M.; Zaffaroni, M.; Bona, G. Vitamin D status in cord blood and newborns: Ethnic differences. Ital. J. Pediatr. 2013, 39, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Josefson, J.L.; Feinglass, J.; Rademaker, A.W.; Metzger, B.E.; Zeiss, D.M.; Price, H.E.; Langman, C.B. Maternal obesity and vitamin D sufficiency are associated with cord blood vitamin D insufficiency. J. Clin. Endocrinol. Metabol. 2013, 98, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Institute of Food and Nutrition. Dietary References Intake; Jarosz, M., Bułhak-Jachymczyk, B., Eds.; Wydawnictwo Lekarskie PZWL: Warsaw, Poland, 2008. [Google Scholar]
- Bojar, I.; Owoc, A.; Humeniuk, E.; Wierzba, W.; Fronczak, A. Inappropriate consumption of vitamins and minerals by pregnant women in Poland. Ann. Agric. Environ. Med. 2012, 19, 263–266. [Google Scholar] [PubMed]
- Lebiedzińska, A.; Rypina, M.; Czaja, J. Analysis of vitamin D content in daily food rations of Polish adults. Bromat. Chem. Toksykol. 2010, 43, 255–259. (In Polish) [Google Scholar]
- Płudowski, P.; Ducki, C.; Konstantynowicz, J.; Jaworski, M. Vitamin D status in Poland. Pol. Arch. Med. Wewn. 2016, 126, 530–539. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Winter Group (n = 50) | Summer Group (n = 50) | p-Value |
|---|---|---|---|
| age (in years) mean ± SD | 29.7 ± 4.4 | 30.3 ± 4.4 | NS |
| education | |||
| higher, (%) | 64 | 68 | NS |
| other, (%) | 36 | 32 | |
| gravidity | |||
| primipara, (%) | 40 | 44 | NS |
| multipara, (%) | 60 | 56 | |
| pre-pregnancy BMI (mean) ± SD | 22.4 ± 3.3 | 23.1 ± 3.9 | NS |
| weight gain | |||
| inadequate, (%) | 24 | 28 | NS |
| normal, (%) | 34 | 32 | NS |
| excessive, (%) | 42 | 40 | NS |
| gestational diabetes, (%) | 14 | 8 | NS |
| pregnancy induced hypertension, (%) | 4 | 14 | 0.0500 |
| smoking during pregnancy, (%) | 14 | 16 | NS |
| professionally active during pregnancy, (%) | 56 | 60 | NS |
| supplementation with vitamin/mineral preparations, (%) | 88 | 90 | NS |
| supplementation with single-component vitamin D preparations, (%) | 16 | 14 | NS |
| fish consumption (at least once a week), (%) | 46 | 38 | NS |
| daily consumption of vitamin D—diet (µg), median (min–max) | 2.11 (0.24–6.58) | 2.07 (0.65–11.48) | NS |
| number of women with adequate daily consumption of vitamin D from the diet (≥ 5 µg), (%) | 6 | 6 | NS |
| daily consumption of vitamin D—diet and supplements (µg), median (min–max) | 14.35 (0.82–53.85) | 14.07 (0.91–98.93) | NS |
| time outside between 10 a.m. and 3 p.m. (mean) ± SD | 2 h 18 min ± 1 h 34 min | ||
| avoiding sun exposure between 10 a.m. and 3 p.m., (%) | 46 | ||
| sunscreen, (%) | 26 | ||
| maternal serum vitamin D concentration, (ng/mL) mean ± SD | 16.5 ± 8.2 | 22.2 ± 6.5 | <0.001 |
| umbilical cord blood vitamin D concentration, (ng/mL) mean ± SD | 22.7 ± 11.2 | 31.3 ± 9.4 | <0.0001 |
| sex of the newborn | |||
| male, (%) | 54 | 48 | |
| female, (%) | 46 | 52 | NS |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wierzejska, R.; Jarosz, M.; Sawicki, W.; Bachanek, M.; Siuba-Strzelińska, M. Vitamin D Concentration in Maternal and Umbilical Cord Blood by Season. Int. J. Environ. Res. Public Health 2017, 14, 1121. https://doi.org/10.3390/ijerph14101121
Wierzejska R, Jarosz M, Sawicki W, Bachanek M, Siuba-Strzelińska M. Vitamin D Concentration in Maternal and Umbilical Cord Blood by Season. International Journal of Environmental Research and Public Health. 2017; 14(10):1121. https://doi.org/10.3390/ijerph14101121
Chicago/Turabian StyleWierzejska, Regina, Mirosław Jarosz, Włodzimierz Sawicki, Michał Bachanek, and Magdalena Siuba-Strzelińska. 2017. "Vitamin D Concentration in Maternal and Umbilical Cord Blood by Season" International Journal of Environmental Research and Public Health 14, no. 10: 1121. https://doi.org/10.3390/ijerph14101121
APA StyleWierzejska, R., Jarosz, M., Sawicki, W., Bachanek, M., & Siuba-Strzelińska, M. (2017). Vitamin D Concentration in Maternal and Umbilical Cord Blood by Season. International Journal of Environmental Research and Public Health, 14(10), 1121. https://doi.org/10.3390/ijerph14101121





