Association between Long-Term Exposure to Particulate Matter Air Pollution and Mortality in a South Korean National Cohort: Comparison across Different Exposure Assessment Approaches
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Individual- and Area-level Covariates
2.3. Mortality
2.4. Exposure Assessment for Long-Term PM10 Concentration
2.5. Statistical Analysis
3. Results
3.1. Study Population and Long-Term PM10 Concentration
3.2. Association between Long-Term PM10 and Mortality
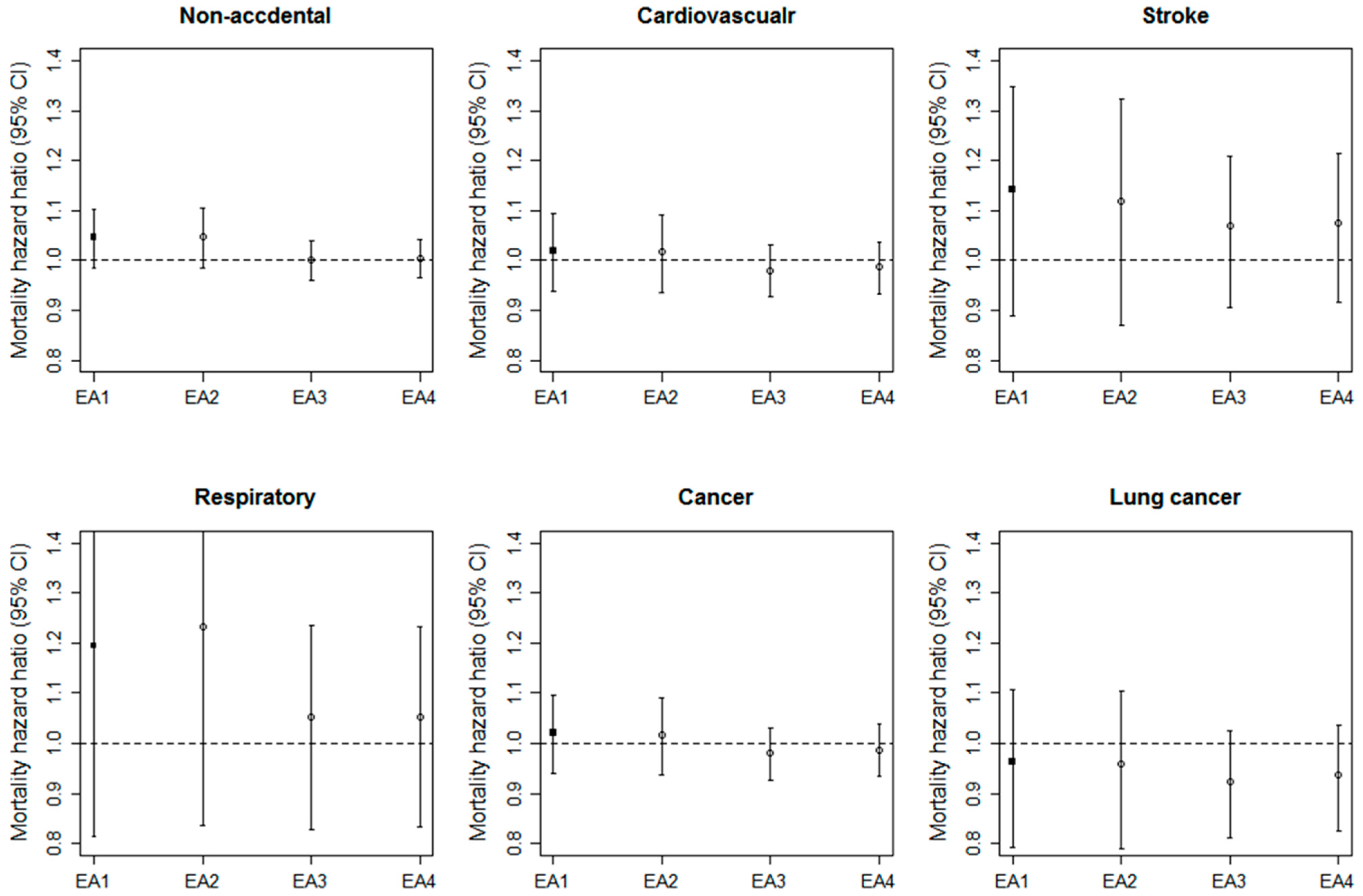
3.3. Comparisons of the Associations across Four EAs
3.4. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Beelen, R.; Raaschou-Nielsen, O.; Stafoggia, M.; Andersen, Z.J.; Weinmayr, G.; Hoffmann, B.; Wolf, K.; Samoli, E.; Fischer, P.; Nieuwenhuijsen, M.; et al. Effects of long-term exposure to air pollution on natural-cause mortality: An analysis of 22 European cohorts within the multicentre ESCAPE project. Lancet 2014, 383, 785–795. [Google Scholar] [CrossRef]
- Hoek, G.; Krishnan, R.M.; Beelen, R.; Peters, A.; Ostro, B.; Brunekreef, B.; Kaufman, J.D. Long-term air pollution exposure and cardio- respiratory mortality: A review. Environ. Health 2014, 12, 43. [Google Scholar] [CrossRef] [PubMed]
- Di, Q.; Wang, Y.; Zanobetti, A.; Wang, Y.; Koutrakis, P.; Choirat, C.; Dominici, F.; Schwartz, J.D. Air pollution and mortality in the Medicare population. N. Engl. J. Med. 2017, 376, 2513–2522. [Google Scholar] [CrossRef] [PubMed]
- Ueda, K.; Nagasawa, S.; Nitta, H.; Miura, K.; Ueshima, H. Exposure to particulate matter and long-term risk of cardiovascular mortality in Japan: NIPPON DATA80. J. Atheroscler. Thromb. 2012, 19, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Yang, C.; Li, J.; Chen, R.; Chen, B.; Gu, D.; Kan, H. Association between long-term exposure to outdoor air pollution and mortality in China: A cohort study. J. Hazard. Mater. 2011, 186, 1594–1600. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Dong, G.; Sun, B.; Zhang, L.; Chen, X.; Ma, N.; Yu, F.; Guo, H.; Huang, H.; Lee, Y.L.; et al. Long-term exposure to ambient air pollution and mortality due to cardiovascular disease and cerebrovascular disease in Shenyang, China. PLoS ONE 2011, 6, e20827. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Liu, Y.; Wang, L.; Kuang, X.; Xu, X.; Kan, H. Particulate air pollution and mortality in a cohort of Chinese men. Environ. Pollut. 2014, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.M.; Lai, H.K.; Tsang, H.; Thach, T.Q.; Thomas, G.N.; Lam, K.B.H.; Chan, K.P.; Yang, L.; Lau, A.K.H.; Ayres, J.G.; et al. Satellite-based estimates of long-term exposure to fine particles and association with mortality in elderly Hong Kong residents. Environ. Health Perspect. 2015, 123, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Tseng, E.; Ho, W.C.; Lin, M.H.; Cheng, T.J.; Chen, P.C.; Lin, H.H. Chronic exposure to particulate matter and risk of cardiovascular mortality: Cohort study from Taiwan. BMC Public Health 2015, 15, 936. [Google Scholar] [CrossRef] [PubMed]
- Katanoda, K.; Sobue, T.; Satoh, H.; Tajima, K.; Suzuki, T.; Nakatsuka, H.; Takezaki, T.; Nakayama, T.; Nitta, H.; Tanabe, K.; et al. An association between long-term exposure to ambient air pollution and mortality from lung cancer and respiratory diseases in Japan. J. Epidemiol. 2011, 21, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Brauer, M.; Amann, M.; Burnett, R.T.; Cohen, A.; Dentener, F.; Ezzati, M.; Henderson, S.B.; Krzyzanowski, M.; Martin, R.V.; van Dingenen, R.; et al. Exposure assessment for estimation of the global burden of disease attributable to outdoor air pollution. Environ. Sci. Technol. 2012, 46, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMarzroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Apte, J.S.; Marshall, J.D.; Cohen, A.J.; Brauer, M. Addressing global mortality from ambient PM2.5. Environ. Sci. Technol. 2015, 49, 8057–8066. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jiang, X.; Tong, D.; Davis, S.J.; Zhao, H.; Geng, G.; Feng, T.; Zheng, B.; Lu, Z.; Streets, D.G.; et al. Transboundary health impacts of transported global air pollution and international trade. Nature 2017, 543, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Krewski, D.; Jerrett, M.; Burnett, R.T.; Ma, R.; Hughes, E.; Shi, Y.; Turner, M.C.; Pope, C.A., III; Thurston, G.; Calle, E.E.; et al. Extended Follow-Up and Spatial Analysis of the American Cancer Society Study Linking Particulate Air Pollution and Mortality. HEI Research Report 140; Health Effects Institute: Boston, MA, USA, 2009. [Google Scholar]
- Pope, C.A., III; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurston, G.D. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA 2002, 287, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Laden, F.; Schwartz, J.; Speizer, F.E.; Dockery, D.W. Reduction in fine particulate air pollution and mortality extended follow-up of the Harvard six cities study. Am. J. Respir. Crit. Care Med. 2006, 173, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, J.S.; Park, S.H.; Shin, S.A.; Kim, K. Cohort profile: The National Health Insurance Service-National Sample Cohort (NHIS-NSC), South Korea. Int. J. Epidemiol. 2017, 46, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Song, I. National-scale exposure prediction for long-term concentrations of particulate matter and nitrogen dioxide in South Korea. Environ. Pollut. 2017, 226, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Song, I.; Kim, S.Y. Estimation of representative area-level concentrations of particulate matter (PM10) in Seoul, Korea (in Korean). J. Korean Assoc. Geogr. Inf. Stud. 2016, 19, 118–129. [Google Scholar]
- KOSIS. Available online: http://kosis.kr/statHtml/statHtml.do?orgId=101&tblId=DT_1INOO02&conn_path=I2 (accessed on 25 July 2017).
- KOSIS. Available online: http://kosis.kr/statHtml/statHtml.do?orgId=202&tblId=DT_F10101&conn_path=I2 (accessed on 25 July 2017).
- Pope, C.A., III; Dockery, D.W. Health effects of fine particulate air pollution: Lines that connect. J. Air & Waste Manage. Assoc. 2006, 56, 709–742. [Google Scholar]
- U.S. EPA. Integrated Science Assessment for Particulate Matter (Report No. EPA/600/R-08/139F); U.S. Environmental Protection Agency, Office of Research and Development: Research Triangle Park, Chapel Hill, NC, USA, 2009.
- NIER. Annual Report of Air Quality in Korea 2014 (Report No. NIER-GP2015-087); Air Quality Research Division Korean; National Institute of Environmental Research: Incheon, Korea, 2014.
- Fischer, P.H.; Marra, M.; Ameling, C.B.; Hoek, G.; Beelen, R.; de Hoogh, K.; Breugelmans, O.; Kruize, H.; Janssen, N.A.; Houthuijs, D. Air pollution and mortality in seven million adults: The Dutch Environmental Longitudinal Study (DUELS). Environ. Health Perspect. 2015, 123, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Thurston, G.D.; Ahn, J.; Cromar, K.R.; Shao, Y.; Reynolds, H.R.; Jerrett, M.; Lim, C.C.; Shanley, R.; Park, Y.; Hayes, R.B. Ambient particulate matter air pollution exposure and mortality in the NIH-AARP diet and health cohort. Environ. Health Perspect. 2016, 124, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Ostro, B.; Lipsett, M.; Reynolds, P.; Goldberg, D.; Hertz, A.; Garcia, C.; Henderson, K.D.; Bernstein, L. Long-term exposure to constituents of fine particulate air pollution and mortality: Results from the California Teachers Study. Environ. Health Perspect. 2010, 118, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.J.; Kim, H.; Kim, S.Y. Exploration and application of regulatory PM10 measurement data for developing long-term prediction models in South Korea (in Korean). J. Korean Soc. Atmos. Environ. 2016, 32, 114–126. [Google Scholar] [CrossRef]
- Crouse, D.L.; Peters, P.A.; Hystad, P.; Brook, J.R.; van Donkelaar, A.; Martin, R.V.; Villeneuve, P.J.; Jerrett, M.; Goldberg, M.S.; Pope, C.A., III; et al. Ambient PM2.5, O3 and NO2 exposures and associations with mortality over 16 years of follow-up in the Canadian Census Health and Environment Cohort (CanCHEC). Environ. Health Perspect. 2015, 123, 1180–1186. [Google Scholar] [CrossRef] [PubMed]
- Hart, J.E.; Liao, X.; Hong, B.; Puett, R.C.; Yanosky, J.D.; Suh, H.; Kioumourtzoglou, M.A.; Spiegelman, D.; Laden, F. The association of long-term exposure to PM2.5 on all-cause mortality in the Nurses’ Health Study and the impact of measurement-error correction. Environ. Health 2015, 14, 38. [Google Scholar] [CrossRef] [PubMed]
- Pope, C.A., III. Mortality effects of longer term exposures to fine particulate air pollution: Review of recent epidemiological evidence. Inhal. Toxicol. 2007, 19, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Pelucchi, C.; Negri, E.; Gallus, S.; Boffetta, P.; Tramacere, I.; la Vechhia, C. Long-term particulate matter exposure and mortality: A review of European epidemiological studies. BMC Public Health 2009, 9, 453. [Google Scholar] [CrossRef] [PubMed]
- Dockery, D.W.; Pope, C.A., III; Xu, X.; Spengler, J.D.; Ware, J.H.; Fay, M.E.; Ferris, B.G.; Speizer, F.E. An association between air pollution and mortality in six U.S. cities. N. Engl. J. Med. 1993, 329, 1753–1759. [Google Scholar] [CrossRef] [PubMed]
- Migration Statistics. KOSIS. Available online: http://kostat.go.kr/portal/korea/kor_nw/2/2/4/index.board?bmode=read&aSeq=311470 (accessed on 26 July 2017).
- 2010 Census. KOSIS. Available online: http://kostat.go.kr/portal/korea/kor_nw/2/2/1/index.board?bmode=read&aSeq=250515&pageNo=21&rowNum=10&amSeq=&sTarget=&sTxt= (accessed on 25 July 2017).
- Carey, I.M.; Atkinson, R.W.; Kent, A.J.; van Staa, T.; Cook, D.G.; Andersen, H.R. Mortality associations with long-term exposure to outdoor air pollution in a national English cohort. Am. J. Respir. Crit. Care Med. 2013, 187, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, J.; Thiering, E.; Rzehak, P.; Krämer, U.; Hochadel, M.; Rauchfuss, K.M.; Gehring, U.; Wichmann, H.E. Long-term exposure to NO2 and PM10 and all-cause and cause-specific mortality in a prospective cohort of women. Occup. Environ. Med. 2013, 70, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Szpiro, A.A.; Sheppard, L.; Lumley, T. Efficient measurement error correction with spatially misaligned data. Biostatistics 2011, 12, 610–623. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Number of Subjects | PM10 Concentration (µg/m3) | ||||||
|---|---|---|---|---|---|---|---|---|
| Total (39.2–72.1) | Quintile 1 (39.2–49.3) | Quintile 2 (49.3–53.9) | Quintile 3 (53.9–59.5) | Quintile 4 (59.5–61.9) | Quintile 5 (61.9–72.1) | |||
| Total | 275,337 | 100.0 | 19.8 | 20.2 | 19.6 | 20.5 | 19.9 | |
| Sex | Male | 149,735 | 54.4 | 53.3 | 54.2 | 54.9 | 55.0 | 54.5 |
| Female | 125,602 | 45.6 | 46.7 | 45.8 | 45.1 | 45.0 | 45.5 | |
| Age at baseline (years) | 20–24 | 24,943 | 9.1 | 7.8 | 9.5 | 10.1 | 9.3 | 8.6 |
| 25–29 | 31,843 | 11.6 | 8.5 | 10.4 | 12.9 | 13.3 | 12.6 | |
| 30–34 | 32,092 | 11.7 | 9.9 | 11.2 | 11.8 | 12.9 | 12.6 | |
| 35–39 | 35,837 | 13.0 | 12.4 | 13.2 | 12.7 | 13.6 | 13.2 | |
| 40–44 | 43,769 | 15.9 | 16.8 | 15.6 | 15.6 | 16.0 | 15.4 | |
| 45–49 | 35,816 | 13.0 | 13.7 | 12.8 | 13.2 | 12.6 | 12.6 | |
| 50–54 | 27,692 | 10.1 | 11.4 | 10.0 | 10.0 | 9.3 | 9.6 | |
| 55–59 | 22,072 | 8.0 | 9.4 | 8.1 | 7.4 | 7.1 | 8.1 | |
| 60–64 | 21,273 | 7.7 | 10.1 | 9.1 | 6.3 | 5.9 | 7.2 | |
| Income 1 (%) | <20 | 39,246 | 14.3 | 14.6 | 15.5 | 13.3 | 14.0 | 13.9 |
| 20–50 | 72,764 | 26.4 | 26.6 | 27.2 | 24.9 | 26.4 | 27.0 | |
| 50–80 | 93,126 | 33.8 | 33.2 | 34.1 | 33.0 | 34.5 | 34.4 | |
| >80 | 70,201 | 25.5 | 25.6 | 23.2 | 28.8 | 25.2 | 24.8 | |
| Type of health insurance | Self-insured | 94,475 | 34.3 | 37.0 | 35.1 | 32.3 | 32.1 | 35.2 |
| Employee-insured | 180,041 | 65.4 | 62.6 | 64.6 | 67.5 | 67.6 | 64.6 | |
| Medical aid | 821 | 0.3 | 0.4 | 0.4 | 0.3 | 0.3 | 0.2 | |
| Smoking | Non-smoker | 179,428 | 65.2 | 67.5 | 66.3 | 64.9 | 63.3 | 64.0 |
| Ex-smoker | 13,092 | 4.8 | 4.1 | 4.4 | 5.3 | 5.1 | 4.8 | |
| Current smoker | 82,817 | 30.1 | 28.4 | 29.3 | 29.8 | 31.6 | 31.2 | |
| Alcohol use (>3 times/week) | 25,935 | 9.4 | 10.5 | 9.3 | 8.7 | 9.1 | 9.5 | |
| Exercise (<3 times/week) | 227,960 | 82.8 | 82.3 | 83.8 | 82.2 | 83.0 | 82.7 | |
| Obese (BMI > 25.0) | 83,894 | 30.5 | 31.3 | 30.5 | 29.2 | 30.0 | 31.4 | |
| Co-morbidity | Cardiovascular | 2377 | 0.9 | 1.1 | 0.9 | 0.8 | 0.7 | 0.8 |
| Hypertension | 13,681 | 5.0 | 5.6 | 5.2 | 4.5 | 4.5 | 5.1 | |
| Diabetes | 6919 | 2.5 | 2.8 | 2.7 | 2.3 | 2.3 | 2.6 | |
| Percent of the high school completed or more 2 | <46.6 | 63,688 | 23.1 | 50.7 | 40.3 | 11.6 | 3.0 | 10.4 |
| 46.6–51.9 | 68,075 | 24.7 | 16.4 | 40.6 | 23.7 | 19.9 | 22.9 | |
| 51.9–55.2 | 72,227 | 26.2 | 32.7 | 11.0 | 20.8 | 39.9 | 26.5 | |
| >55.2 | 71,347 | 25.9 | 0.2 | 8.1 | 43.9 | 37.2 | 40.3 | |
| Percent of the elderly 2 (>65 years) | <4.8 | 71,231 | 25.9 | 14.1 | 20.3 | 27.0 | 47.0 | 20.4 |
| 4.8–6.0 | 70,978 | 25.8 | 10.6 | 16.3 | 38.9 | 28.3 | 35.0 | |
| 6.0–8.3 | 69,440 | 25.2 | 26.8 | 20.6 | 21.9 | 20.8 | 36.1 | |
| >8.3 | 63,688 | 23.1 | 48.5 | 42.8 | 12.2 | 4.0 | 8.5 | |
| GRDP 2,3 (per 1000 US dollar) | <2,665,207 | 69,362 | 25.2 | 42.8 | 33.1 | 13.9 | 14.4 | 15.9 |
| 2,665,207–5,145,783 | 68,853 | 25.0 | 24.8 | 35.2 | 38.1 | 14.4 | 13.8 | |
| 5,145,783–10,085,624 | 99,362 | 36.1 | 20.2 | 17.5 | 39.6 | 50.6 | 57.5 | |
| >10,085,624 | 37,760 | 13.7 | 12.2 | 14.2 | 8.5 | 20.6 | 12.8 | |
| Cause of death | Non-accidental | 3796 | 1.4 | 1.6 | 1.6 | 1.2 | 1.2 | 1.3 |
| Cardiovascular | 720 | 0.3 | 0.3 | 0.3 | 0.2 | 0.2 | 0.2 | |
| Cerebrovascular | 295 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | |
| Respiratory | 152 | 0.1 | 0.1 | 0.1 | 0.1 | 0.0 | 0.1 | |
| Cancer | 2179 | 0.8 | 0.9 | 0.9 | 0.7 | 0.7 | 0.7 | |
| Lung cancer | 461 | 0.2 | 0.2 | 0.2 | 0.1 | 0.2 | 0.1 | |
| Model 1 | HRs (95% CIs) | |||||
|---|---|---|---|---|---|---|
| Non-Accidental | Cardiovascular | Cerebrovascular | Respiratory | Cancer | Lung Cancer | |
| Model 1 | 0.95 (0.90, 0.99 ) | 0.88 (0.79, 0.98) | 0.89 (0.75, 1.05) | 0.96 (0.76, 1.22) | 0.97 (0.91, 1.03) | 0.86 (0.75, 0.98) |
| Model 2 | 0.98 (0.93, 1.03 ) | 0.91 (0.82, 1.02) | 0.93 (0.79, 1.11) | 1.05 (0.83, 1.33) | 0.98 (0.92, 1.05) | 0.88 (0.77, 1.01) |
| Model 3 | 0.97 (0.93, 1.02) | 0.91 (0.81, 1.01) | 0.93 (0.78, 1.10) | 1.05 (0.83, 1.32) | 0.98 (0.92, 1.05) | 0.89 (0.77, 1.01) |
| Model 4 | 1.05 (0.99, 1.11) | 1.02 (0.90, 1.16) | 1.14 (0.93, 1.39) | 1.19 (0.91, 1.57) | 1.02 (0.95, 1.10) | 0.96 (0.82, 1.13) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, O.-J.; Kim, S.-Y.; Kim, H. Association between Long-Term Exposure to Particulate Matter Air Pollution and Mortality in a South Korean National Cohort: Comparison across Different Exposure Assessment Approaches. Int. J. Environ. Res. Public Health 2017, 14, 1103. https://doi.org/10.3390/ijerph14101103
Kim O-J, Kim S-Y, Kim H. Association between Long-Term Exposure to Particulate Matter Air Pollution and Mortality in a South Korean National Cohort: Comparison across Different Exposure Assessment Approaches. International Journal of Environmental Research and Public Health. 2017; 14(10):1103. https://doi.org/10.3390/ijerph14101103
Chicago/Turabian StyleKim, Ok-Jin, Sun-Young Kim, and Ho Kim. 2017. "Association between Long-Term Exposure to Particulate Matter Air Pollution and Mortality in a South Korean National Cohort: Comparison across Different Exposure Assessment Approaches" International Journal of Environmental Research and Public Health 14, no. 10: 1103. https://doi.org/10.3390/ijerph14101103
APA StyleKim, O.-J., Kim, S.-Y., & Kim, H. (2017). Association between Long-Term Exposure to Particulate Matter Air Pollution and Mortality in a South Korean National Cohort: Comparison across Different Exposure Assessment Approaches. International Journal of Environmental Research and Public Health, 14(10), 1103. https://doi.org/10.3390/ijerph14101103






