Ambient Concentrations of Metabolic Disrupting Chemicals and Children’s Academic Achievement in El Paso, Texas
Abstract
:1. Introduction
Research Questions and Proposed Model
2. Materials and Methods
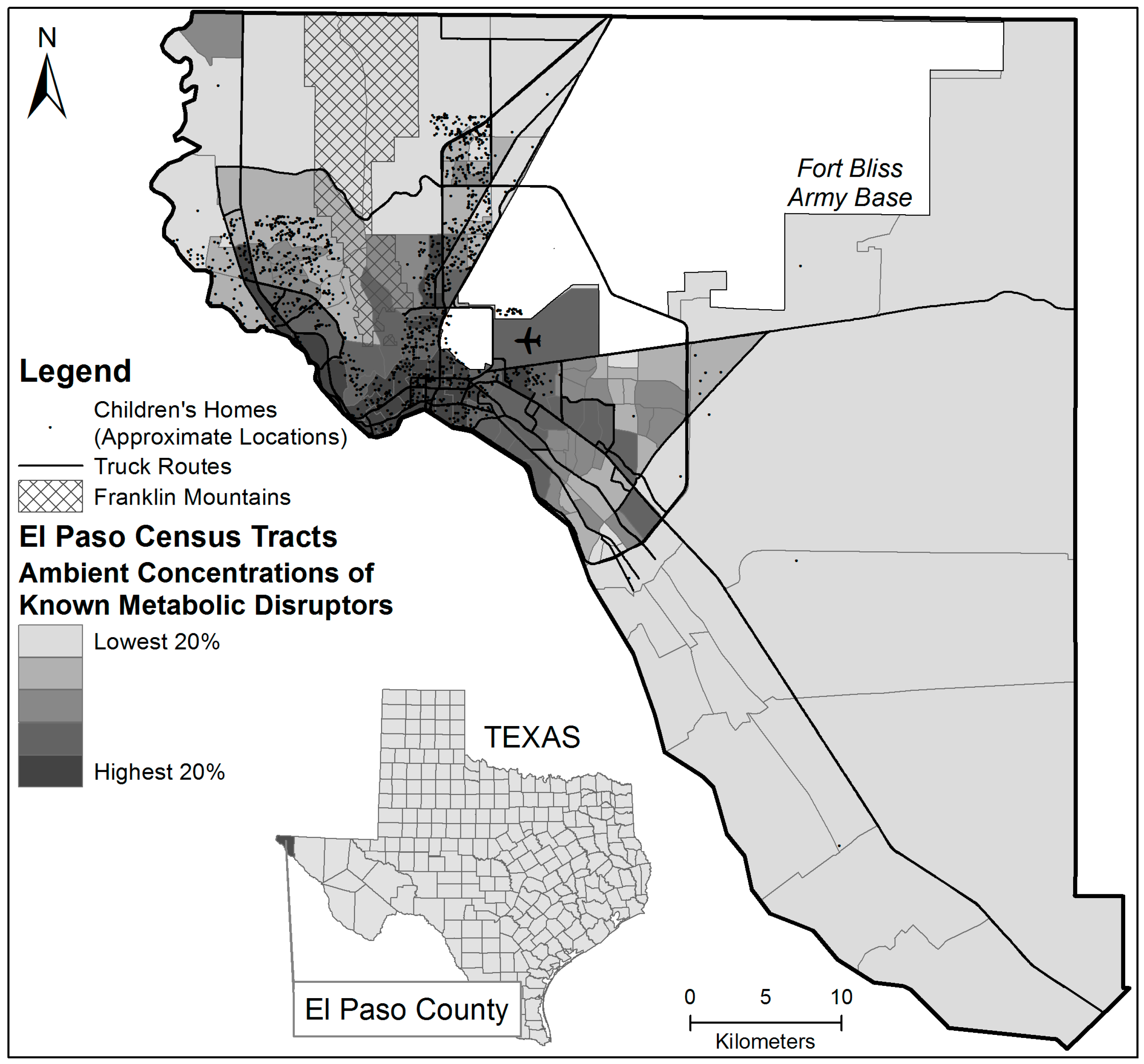
2.1. Study Context
2.2. Data Collection
2.3. Variables
2.3.1. Children’s Academic Performance
2.3.2. Concentration of Metabolic Disruptors
2.3.3. Body Mass Index
2.3.4. Control Variables
2.4. Analysis Methods
3. Results
3.1. Correlations
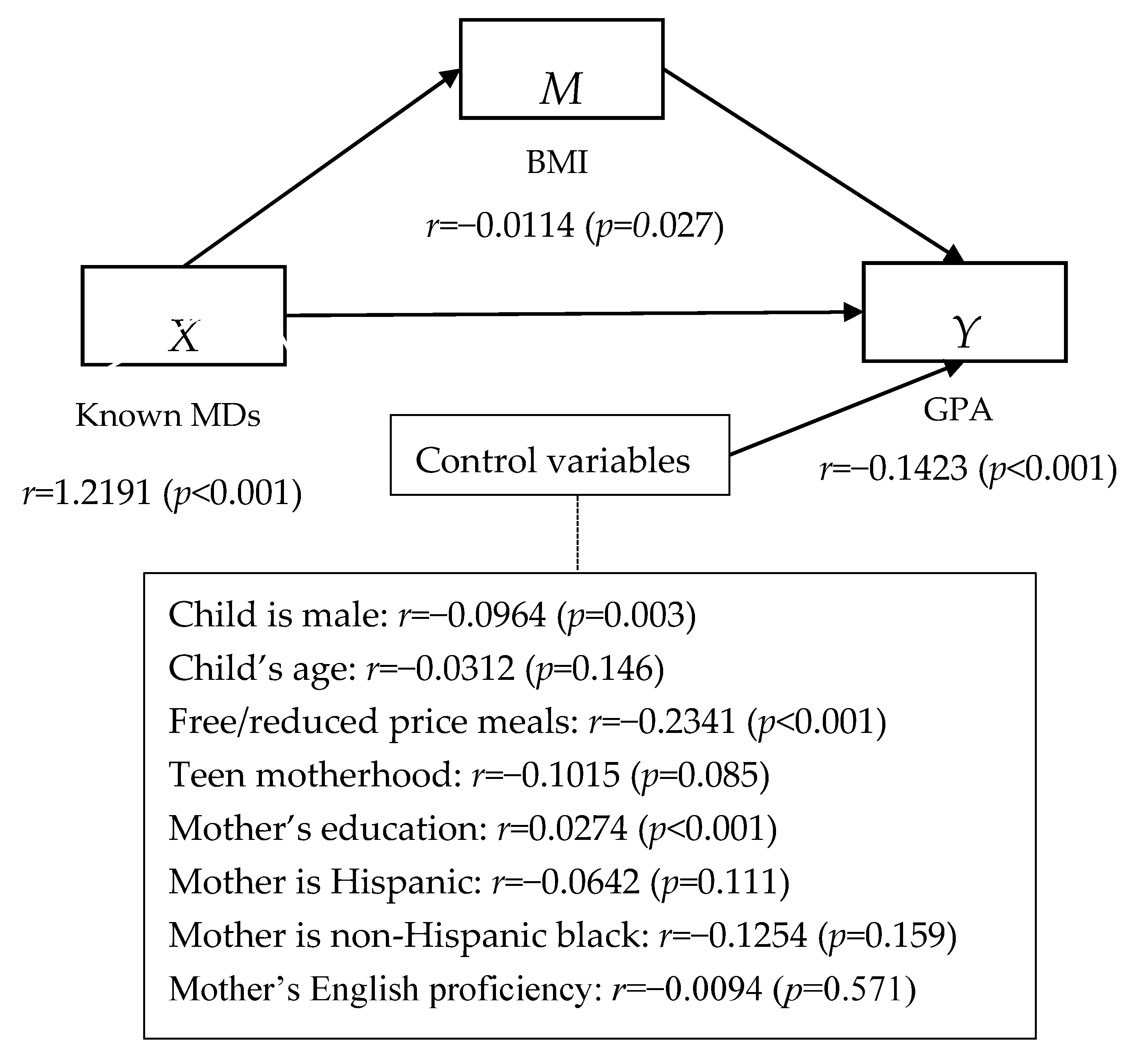
3.2. Mediation Model
4. Discussion
Limitations and Directions of Future Research
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gaffron, P.; Niemeier, D. School locations and traffic emissions-environmental (in)justice findings using a new screening method. Int. J. Environ. Res. Public Health 2015, 12, 2009–2025. [Google Scholar] [CrossRef] [PubMed]
- Legot, C.; London, B.; Shandra, J. Environmental ascription: High-volume polluters, schools, and human capital. Organ. Environ. 2010, 23, 271–290. [Google Scholar] [CrossRef]
- Lucier, C.; Rosofsky, A.; London, B.; Scharber, H.; Shandra, J. Toxic pollution and school performance scores: Environmental ascription in East Baton Rouge Parish, Louisiana. Organ. Environ. 2011, 24, 1–21. [Google Scholar] [CrossRef]
- Scharber, H.; Lucier, C.; London, B.; Rosofsky, A.; Shandra, J. The consequences of exposure to, neurological, and respiratory toxins for school performance: A closer look at environmental ascription in East Baton Rouge, Louisiana. Popul. Environ. 2013, 35, 205–224. [Google Scholar] [CrossRef]
- Mohai, P.; Kweon, B.; Lee, S.; Ard, K. Air Pollution around Schools is Linked to Poorer Student Health and Academic Performance. Health Affair. 2011, 30, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Pastor, M.; Sadd, J.; Morello-Frosch, R. Reading, writing, and toxics: Children’s health, academic performance, and environmental justice in Los Angeles. Environ. Plann. C 2004, 22, 271–290. [Google Scholar]
- Pastor, M.; Sadd, J.; Morello-Frosch, R. Breathless: Schools, Air Toxics, and Environmental Justice in California. Policy Stud. J. 2006, 34, 337–362. [Google Scholar] [CrossRef]
- Rosofsky, A.; Lucier, C.A.; London, B.; Scharber, H.; Borges-Mendez, R.B.; Shandra, J. Environmental ascription in Worcester County, MA: Toxic pollution and education outcomes. Local Environ. Int. J. Justice Sustain. 2014, 19, 283–299. [Google Scholar] [CrossRef]
- Clark-Reyna, S.; Grineski, S.E.; Collins, T.W. Residential exposure to air toxics is linked to lower GPAs among school children. Popul. Environ. 2015. [Google Scholar] [CrossRef]
- Grineski, S.; Clark-Reyna, S.; Collins, T. School-based exposure to hazardous air pollutants and grade point average: A cross-sectional study. Environ. Res. 2016, 147, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J.; Vom Saal, F.S.; Blumberg, B.; Bovolin, P.; Calamandrei, G.; Ceresini, G.; Cohn, B.A.; Fabbri, E.; Gioiosa, L.; Kassotis, C.; et al. Parma Consensus statement on metabolic disruptors. Environ. Health 2015, 14, 1–7. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Environmental Health Sciences. Endocrine Disruptors. Available online: http://www.niehs.nih.gov/health/topics/agents/endocrine/ (accessed on 15 December 2015).
- Bergman, A.; Heindel, J.J.; Jobling, S.; Kidd, K.A.; Zoeller, T. State of the Science of Endocrine Disrupting Chemicals—2012. Available online: http://unep.org/pdf/9789241505031_eng.pdf (accessed on 29 August 2016).
- Baillie-Hamilton, P.F. Chemical toxins: A hypothesis to explain the global obesity epidemic. J. Altern. Complement. Med. 2002, 8, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Newbold, R.R.; Padilla-Banks, E.; Jefferson, W.N. Environmental estrogens and obesity. Mol. Cell. Endocrinol. 2009, 304, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Janesick, A.; Blumberg, B. Endocrine disrupting chemicals and the developmental programming of adipogenesis and obesity. Birth Defects Res. Part C Embryo Today Rev. 2011, 93, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Grün, F.; Blumberg, B. Environmental obesogens: Organotins and endocrine disruption via nuclear receptor signaling. Endocrinology 2006, 147, S50–S55. [Google Scholar] [CrossRef] [PubMed]
- Curtis, K.; Wilding, B.C. Is It in Us? Chemical Contamination in Our Bodies. Available online: http://www.isitinus.org/documents/Is%20It%20In%20Us%20Report.pdf (accessed on 19 January 2016).
- Guxens, M.; Sunyer, J. A review of epidemiological studies on neuropsychological effects of air pollution. Swiss. Med. Wkly. 2012, w13322. [Google Scholar] [CrossRef]
- Roberts, C.K.; Freed, B.; McCarthy, W.J. Low aerobic fitness and obesity are associated with lower standardized test scores in children. J. Pediatr. 2010, 156, 711.e1–718.e1. [Google Scholar] [CrossRef] [PubMed]
- Datar, A.; Sturm, R. Childhood overweight and elementary school outcomes. Int. J. Obes. 2006, 30, 1449–1460. [Google Scholar] [CrossRef] [PubMed]
- Castelli, D.M.; Hillman, C.H.; Buck, S.M.; Erwin, H.E. Physical fitness and academic achievement in third- and fifth-grade students. J. Sport Exerc. Psychol. 2007, 29, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Caird, J.; Kavanagh, J.; Oliver, K.; Oliver, S.; O’Mara, A.; Stansfeild, C.; Thomas, J. Childhood Obesity and Educational Attainment. Available online: http://eprints.ioe.ac.uk/16316/1/Caird_et_al._2011._Childhood_obesity_and_educational_attainment._a_systematic_review.pdf (accessed on 19 January 2016).
- Centers for Disease Control and Prevention. Overweight and Obesity. 2014. Available online: http://www.cdc.gov/obesity/data/adult.html (accessed on 12 January 2016). [Google Scholar]
- National Institutes of Health, National Heart, Lung, and Blood Institute. Disease and Conditions Index: What Are Overweight and Obesity? Available online: http://www.nhlbi.nih.gov/health/health-topics/topics/obe (accessed on 30 June 2016).
- Khan, N.A.; Raine, L.B.; Donovan, S.M.; Hillman, C.H. The cognitive implications of obesity and nutrition in childhood. Monogr. Soc. Res. Child Dev. 2014, 79, 51–71. [Google Scholar] [CrossRef] [PubMed]
- Kamijo, K.; Pontifex, M.B.; Khan, N.A.; Raine, L.B.; Scudder, M.R.; Drollette, E.S.; Evans, E.M.; Castelli, D.M.; Hillman, C.H. The association of childhood obesity to neuroelectric indices of inhibition. Psychophysiology 2012, 49, 1361–1371. [Google Scholar] [CrossRef] [PubMed]
- Kamijo, K.; Pontifex, M.B.; Khan, N.A.; Raine, L.B.; Scudder, M.R.; Drollette, E.S.; Evans, E.M.; Castelli, D.M.; Hillman, C.H. The negative association of childhood obesity to cognitive control of action monitoring. Cereb. Cortex. 2014, 24, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Hillman, C.H.; Kamijo, K.; Pontifex, M.B. The relation of ERP indices of exercise to brain health and cognition. In Functional Neuroimaging in Exercise and Sport Sciences; Boecker, H., Hillman, C.H., Scheef, L., Struder, H.K., Eds.; Springer: New York, NY, USA, 2012; pp. 419–446. [Google Scholar]
- Li, Y.; Dai, Q.; Jackson, J.C.; Zhang, J. Overweight is associated with decreased cognitive functioning among school-age children and adolescents. Obesity 2008, 16, 1809–1815. [Google Scholar] [CrossRef] [PubMed]
- Bull, R.; Scerif, G. Executive functioning as a predictor of children’s mathematics ability: Inhibition, switching, and working memory. Dev. Neuropsychol. 2001, 19, 273–293. [Google Scholar] [CrossRef] [PubMed]
- Taras, H.; Potts-Datema, W. Obesity and Student Performance at School. J. School Health 2005, 75, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.K.; Kleinjan, M.; Engels, R.C.M.E.; Fisher, J.O.; Hermans, R.C.J. Higher Weight, Lower Education: A Longitudinal Association Between Adolescents’ Body Mass Index and Their Subsequent Educational Achievement Level? J. School Health 2014, 84, 769–776. [Google Scholar] [CrossRef] [PubMed]
- MacCann, C.; Roberts, R.D. Just as smart but not as successful: Obese students obtain lower school grades but equivalent test scores to nonobese students. Int. J. Obes. 2013, 37, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Kristjánsson, A.L.; Sigfúsdóttir, I.D.; Allegrante, J.P. Health Behavior and Academic Achievement among Adolescents: The Relative Contribution of Dietary Habits, Physical Activity, Body Mass Index, and Self-Esteem. Health Educ. Behav. 2008. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.P.; Sherry, B.; Park, S.; Blanck, H.M. The Association of Obesity and School Absenteeism Attributed to Illness or Injury among Adolescents in the United States, 2009. J. Adolesc. Health 2012, 52, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Morello-Frosch, R.; Zuk, M.; Jerrett, M.; Shamasunder, B.; Kyle, A.D. Understanding the Cumulative Impacts of Inequalities in Environmental Health: Implications for Policy. Health Aff. 2011, 30, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Mohai, P.; Lantz, P.M.; Morenoff, J.; House, J.S.; Mero, R.P. Racial and Socioeconomic Disparities in Residential Proximity to Polluting Industrial Facilities: Evidence from the Americans’ Changing Lives Study. Am. J. Public Health 2009, 99, S649–S656. [Google Scholar] [CrossRef] [PubMed]
- US Bureau of the Census. Quick Facts: United States. Available online: http://quickfacts.census.gov/qfd/states/48/48141.html (accessed on 23 January 2016).
- City of El Paso Department of Public Health. Community Health Assessment Final Report. Available online: https://www.elpasotexas.gov/~/media/files/coep/public%20health/community%20health%20assessment%20final%20report.ashx?la=en (accessed on 17 January 2016).
- Texas Department of State Health Services. Texas Overweight and Obesity Statistics. Available online: Dshs.texas.gov/obesity/pdf/DataFacts.pdf (accessed on 21 January 2016).
- Seagrave, J.C.; Mcdonald, J.D.; Reed, M.D.; Seilkop, S.K.; Mauderly, J.L. Responses to subchronic inhalation of low concentrations of diesel exhaust and hardwood smoke measured in rat bronchoalveolar lavage fluid. Inhal. Toxicol. 2005, 17, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Kurita, M. The masculinization of the fetus during pregnancy due to inhalation of diesel exhaust. Environ. Health Perspect. 2001, 109, 111–119. [Google Scholar] [CrossRef] [PubMed]
- US Customs and Border Protection. Locate a Port of Entry. Available online: http://www.cbp.gov/contact/ports (accessed on 21 January 2016).
- USA Today. The Smokestack Effect: Toxic Air and America’s Schools. Available online: http://content.usatoday.com/news/nation/environment/smokestack/index (accessed on 19 December 2015).
- Grineski, S.E.; Collins, T.W.; Chavez-Payan, P.; Jimenez, A.; Kim, Y.; Gaines, M.; Clark-Reyna, S. Social disparities in children’s respiratory health in El Paso, Texas. Int. J. Environ. Res. Public Health 2014, 11, 2941–2957. [Google Scholar] [CrossRef] [PubMed]
- Dillman, D.A.; Smyth, J.D.; Christian, L.M. Internet, Mail, and Mixed-Mode Surveys: The Tailored Design Method, 3rd ed.; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Curtin, R.; Presser, S.; Singer, E. The effects of response rate changes on the index of consumer sentiment. Public Opin. Quart. 2000, 64, 413–428. [Google Scholar] [CrossRef]
- Holbrook, A.; Krosnick, J.A.; Pfent, A. The causes and consequences of response rates in surveys by the news media and government contractor survey research. In Advances in Telephone Survey Methodology; Lepkowski, J.M., Tucker, N.C., Brick, J.M., DeLeeuw, E., Japec, L., Lavrakas, P.J., Link, M.W., Sangster, R.L., Eds.; Wiley: New York, NY, USA, 2008; pp. 499–528. [Google Scholar]
- Keeter, S.; Kennedy, C.; Dimock, M.; Best, J.; Craighill, P. Gauging the impact of growing non response on estimates from a national RDD telephone survey. Public Opin. Quart. 2006, 70, 759–779. [Google Scholar] [CrossRef]
- Visser, P.S.; Krosnick, J.A.; Marquette, J.; Curtin, M. Mail surveys for election forecasting? An evaluation of the Columbus Dispatch Poll. Public Opin. Quart. 1996, 60, 181–227. [Google Scholar] [CrossRef]
- US Department of Education. Digest of Education Statistics. Available online: Http://nces.ed.gov/programs/digest/d13/tables/dt13_225.80.asp (accessed on 17 January 2016).
- Environmental Protection Agency. National Air Toxics Assessments. Available online: http://www.epa.gov/nata/ (accessed on 16 December 2015).
- Roberts, A.L.; Lyall, K.; Hart, J.E.; Laden, F.; Just, A.C.; Bobb, J.F.; Koenen, K.C.; Ascherio, A.; Weisskopf, M.G. Perinatal Air pollutant exposures and autism spectrum disorder in the children of Nurses’ Health Study II participants. Environ. Health Persp. 2013, 121. [Google Scholar] [CrossRef] [PubMed]
- Linder, S.H.; Marko, D.; Sexton, K. Cumulative cancer risk from air pollution in Houston: Disparities in risk burden and social disadvantage. Environ. Sci. Technol. 2008, 42, 4312–4322. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.C.; O’Brien, T.E.; Charrier, J.G.; Hafner, H.R. Characterization of the chronic risk and hazard of hazardous air pollutants in the United States using ambient monitoring data. Environ. Health Persp. 2009, 117, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.D.; Swor, K.R.; Nguyen, N.P. Prioritizing environmental justice and equality: Diesel emissions in Southern California. Environ. Sci. Technol. 2014, 48, 4063–4068. [Google Scholar] [CrossRef] [PubMed]
- Su, J.G.; Morello-Frosch, R.; Jesdale, B.M.; Kyle, A.D.; Shamasunder, B.; Jerrett, M. An index for assessing demographic inequalities in cumulative environmental hazards with application to Los Angeles, California. Environ. Sci. Technol. 2009, 43, 7626–7634. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency. 2005 NATA: Assessment Methods. Available online: https://www.epa.gov/national-air-toxics-assessment/2005-nata-assessment-methods (accessed on 15 January 2016).
- The Endocrine Disruption Exchange. TEDX List of Potential Endocrine Disruptors. Available online: http://endocrinedisruption.org/endocrine-disruption/tedx-list-of-potential-endocrine-disruptors/overview (accessed on 13 December 2015).
- Institute for Environment and Health. Chemicals Purported to Be Endocrine Disruptors: A Compilation of Published Lists. Available online: http://www.le.ac.uk/ieh/ (accessed on 15 December 2015).
- Environmental Protection Agency List of Air Toxics in the 2005 NATA Assessment. Available online: http://www.epa.gov/ttn/atw/nata2005/05pdf/2005polls.pdf (accessed on 18 December 2015).
- Reardon, S.F.; Galindo, C. The Hispanic-White achievement gap in math and reading in the elementary grades. Am. Educ. Res. J. 2009, 46, 853–891. [Google Scholar] [CrossRef]
- US Department of Agriculture Food and Nutrition Service. Income Eligibility Guidelines. Available online: http://www.fns.usda.gov/school-meals/income-eligibility-guidelines (accessed on 26 January 2016).
- Magnuson, K. Maternal education and children’s academic achievement during middle childhood. Dev. Psychol. 2007, 43, 1497–1512. [Google Scholar] [CrossRef] [PubMed]
- Duncan, G.J.; Magnuson, K.A. Can family socioeconomic resources account for racial and ethnic test score gaps? Future Child. 2005, 1, 35–54. [Google Scholar] [CrossRef]
- Kao, G.; Thompson, J.S. Racial and ethnic stratification in educational achievement and attainment. Ann. Rev. Sociol. 2003, 29, 417–442. [Google Scholar] [CrossRef]
- Bali, V.A.; Alvarez, M.R. Schools and educational outcomes: What causes the “race gap” in student test scores? Soc. Sci. Quart. 2003, 84, 485–507. [Google Scholar] [CrossRef]
- Fisher, C.B.; Wallace, S.A.; Fenton, R.E. Discrimination distress during adolescence. J. Youth Adolesc. 2000, 29, 679–695. [Google Scholar] [CrossRef]
- Penn, D.A. Estimating missing values from the general social survey: An application of multiple imputation. Soc. Sci. Quart. 2007, 88, 573–584. [Google Scholar] [CrossRef]
- Enders, C.K. Applied Missing Data Analysis; Guilford Press: New York, NY, USA, 2010. [Google Scholar]
- Hayes, A.F. Introduction to Mediation, Moderation, and Conditional Process Analysis: A Regression-Based Approach; Guilford Press: New York, NY, USA, 2008. [Google Scholar]
- Calderón-Garcidueñas, L.; Solt, A.C.; Henriquez-Roldan, C.; Torres-Jardon, R.; Nuse, B.; Herritt, L.; Villarreal-Calderón, R.; Osnaya, N.; Stone, I.; García, R.; et al. Long-term air pollution exposure is associated with neuroinflammation, an altered innate immune response, disruption of the blood-brain barrier, ultrafine particulate deposition, and accumulation of amyloid beta-42 and alpha-synuclein in children and young adults. Toxicol. Pathol. 2008, 36, 289–310. [Google Scholar] [PubMed]
- Case, A.; Fertig, A.; Paxson, C. The lasting impact of childhood health and circumstance. J. Health Econ. 2005, 24, 365–389. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, J.; Zandbergen, P.A. Children at risk: Measuring racial/ethnic disparities in potential exposure to air pollution at school and home. J. Epidemiol. Commun. Health 2007, 61, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Colburn, T.; Dumanoski, D.; Myers, J.P. Our Stolen Future: Are We Threatening Our Fertility, Intelligence, and Survival? A Scientific Detective Story; Plume Books: New York, NY, USA, 1997. [Google Scholar]
- Dodson, R.E.; Nishioka, M.; Standley, L.J.; Perovich, L.J.; Brody, J.G.; Rudel, R.A. Endocrine Disruptors and Asthma-Associated Chemicals in Consumer Products. Environ. Health Perspect. 2012, 120, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, K.M.; Rudel, R.A. Phthalates in Food Packaging, Consumer Products, and Indoor Environments. In Toxicants in Food Packaging and Household Plastics: Exposure and Health Risks to Consumers; Snedeker, S.M., Ed.; Humana Press: New York, NY, USA, 2014; pp. 31–59. [Google Scholar]
- Rudel, R.A.; Camann, D.E.; Spengler, J.D.; Korn, L.R.; Brody, J.G. Phthalates, Alkylphenols, Pesticides, Polybrominated Diphenyl Ethers, and Other Endocrine-Disrupting Compounds in Indoor Air and Dust. Environ. Sci. Technol. 2003, 37, 4543–4553. [Google Scholar] [CrossRef] [PubMed]
- Rudel, R.A.; Gray, M.E.; Engel, C.L.; Rawsthorne, T.W.; Dodson, R.E.; Ackerman, J.M.; Rizzo, J.; Nudelman, J.L.; Brody, J.G. Food Packaging and Bisphenol A and Bis(2-Ethyhexyl) Phthalate Exposure: Findings from a Dietary Intervention Environ. Health Perspect. 2011, 119, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Muncke, J. Exposure to endocrine disrupting compounds via the food chain: Is packaging a relevant source? Sci. Total Environ. 2009, 407, 4549–4559. [Google Scholar] [CrossRef] [PubMed]
- Weden, M.M.; Brownell, P.B.; Rendall, M.S.; Lau, C.; Fernandes, M.; Nazaroz, Z. Parent-reported height and weight as sources of bias in survey estimates of childhood obesity. Am. J. Epidemiol. 2013, 178, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Cockrell Skinner, A.; Miles, D.; Perrin, E.M.; Coyne-Beasley, T.; Ford, C. Source of Parental Reports of Child Height and Weight During Phone Interviews and Influence on Obesity Prevalence Estimates among Children Aged 3–17 Years. Public Health Rep. 2013, 128, 46–53. [Google Scholar]
- Ritz, B.; Wilhelm, M.; UCLA Institute of the Environment and Sustainability. Air Pollution Impacts on Infants and Children. Available online: http://www.environment.ucla.edu/reportcard/article1700.html (accessed on 7 August 2016).
- Barker, D.J.P. The developmental origins of adult disease. J. Am. Coll. Nutr. 2004, 23 (Suppl. 6), 588S–595S. [Google Scholar] [CrossRef] [PubMed]
| Continuous Variables | N | % Missing | Min. | Max. | Mean | Standard Deviation |
| Grade Point Average (GPA) | 1240 | 6 | 0.20 | 4 | 3.40 | 0.70 |
| Ambient concentration of known metabolic disrupters (MDs) (ln) | 1319 | 0 | −0.07 | 2.48 | 0.84 | 0.43 |
| Known and suspected (ln) ambient concentration of MDs 1 | 1319 | 0 | −0.02 | 2.61 | 0.94 | 0.44 |
| Body Mass Index (BMI) | 1273 | 3.50 | 6.67 | 46.48 | 19.59 | 5.16 |
| Child’s age (years) | 1312 | 0.50 | 9 | 13 | 10.40 | 0.80 |
| Mother’s education (years) | 1208 | 8.40 | 1 | 21 | 13.70 | 3.60 |
| Mother speaks English | 1180 | 12.70 | 0 | 3 | 2.20 | 1 |
| Dichotomous Indicators | N | % Missing | Frequency % | Yes | No | |
| Child is male | 1299 | 1.50 | 49.60 | 644 | 655 | |
| Free/reduced priced meals | 1209 | 8.30 | 40.90 | 494 | 715 | |
| Teenage motherhood | 1178 | 8.40 | 8.10 | 95 | 1083 | |
| Mother is Hispanic | 1185 | 10.20 | 76.70 | 909 | 276 | |
| Mother is non-Hispanic black | 1198 | 9.20 | 3.00 | 36 | 1162 |
| Variables | GPA | Known MDs | Suspected/Known MD | Child’s BMI | Child Is Male | Age of the Child | Free Meals | Teenage Motherhood | Mother’s Education | Mother Is Hispanic | Mother Is Non-Hispanic Black |
|---|---|---|---|---|---|---|---|---|---|---|---|
| GPA | |||||||||||
| Known MDs (ln) | −0.226 ** | ||||||||||
| Suspected/known MDs (ln) | −0.228 ** | 0.999 ** | |||||||||
| Child’s BMI | −0.141 ** | 0.104 ** | 0.103 ** | ||||||||
| Child is male | −0.096 ** | 0.030 | 0.031 | 0.077 ** | |||||||
| Age of the child | −0.074 ** | 0.054 * | 0.054 * | 0.099 ** | 0.033 | ||||||
| Free meals | −0.316 ** | 0.329 ** | 0.328 ** | 0.149 ** | −0.008 | 0.089 ** | |||||
| Teenage motherhood | −0.100 ** | 0.083 ** | 0.088 ** | −0.008 | 0.015 | 0.049 | 0.116 ** | ||||
| Mother‘s education | 0.298 ** | −0.298 ** | −0.299 ** | −0.106 ** | 0.009 | −0.075 ** | −0.487 ** | −0.079 ** | |||
| Mother is Hispanic | −0.201 ** | 0.256 ** | 0.257 ** | 0.123 ** | −0.005 | 0.010 | 0.256 ** | 0.074 * | −0.238 ** | ||
| Mother is non-Hispanic black | 0.016 | −0.068 * | −0.067 * | −0.026 | 0.040 | 0.025 | −0.067 * | 0.020 | 0.060 * | −0.321 ** | |
| Mother’s English proficiency | −0.066 * | 0.136 ** | 0.137 ** | 0.042 | 0 | 0.048 | 0.112 ** | 0.012 | −0.030 | 0.327 ** | −0.078 ** |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark-Reyna, S.E.; Grineski, S.E.; Collins, T.W. Ambient Concentrations of Metabolic Disrupting Chemicals and Children’s Academic Achievement in El Paso, Texas. Int. J. Environ. Res. Public Health 2016, 13, 874. https://doi.org/10.3390/ijerph13090874
Clark-Reyna SE, Grineski SE, Collins TW. Ambient Concentrations of Metabolic Disrupting Chemicals and Children’s Academic Achievement in El Paso, Texas. International Journal of Environmental Research and Public Health. 2016; 13(9):874. https://doi.org/10.3390/ijerph13090874
Chicago/Turabian StyleClark-Reyna, Stephanie E., Sara E. Grineski, and Timothy W. Collins. 2016. "Ambient Concentrations of Metabolic Disrupting Chemicals and Children’s Academic Achievement in El Paso, Texas" International Journal of Environmental Research and Public Health 13, no. 9: 874. https://doi.org/10.3390/ijerph13090874
APA StyleClark-Reyna, S. E., Grineski, S. E., & Collins, T. W. (2016). Ambient Concentrations of Metabolic Disrupting Chemicals and Children’s Academic Achievement in El Paso, Texas. International Journal of Environmental Research and Public Health, 13(9), 874. https://doi.org/10.3390/ijerph13090874






