Bioaugmentation: An Emerging Strategy of Industrial Wastewater Treatment for Reuse and Discharge
Abstract
:1. Introduction
2. Bioaugmentation to Remove Recalcitrant Pollutants in Industrial Wastewater
3. Applications of Bioaugmentation
3.1. Chlorinated and Fluorinated Compounds Removal
3.2. Lignin Removal
3.3. Quinoline and Pyridine
3.4. Synthetic Dyes
3.5. Cyanides
3.6. Nicotine
3.7. Diethylene Glycol Monobutyl Ether (DGBE)
3.8. Polycyclic Aromatic Hydrocarbons and Heterocyclic Compounds
4. Limitations of Bioaugmentation Technologies
4.1. Protozoan Grazing
4.2. Inoculum Size
4.3. Bacteriophage Infection
5. Potential Strategy to Improve the Efficiency of Bioaugmentation
5.1. Immobilized or Entrapped Cells in Bioaugmentation
5.2. Quorum Sensing (QS)
5.3. Genetically Modified Microorganisms and Gene Transfer
5.4. Plasmid Mediated Bioaugmentation
5.5. Nanotechnology in the Context of Bioaugmentation
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zheng, X.; Zhang, Z.; Yu, D.; Chen, X.; Cheng, R.; Min, S.; Wang, J.; Xiao, Q.; Wang, J. Overview of membrane technology applications for industrial wastewater treatment in China to increase water supply. Resour. Conserv. Recycl. 2015, 105, 1–10. [Google Scholar] [CrossRef]
- Norton-Brandao, D.; Scherrenberg, S.M.; van Lier, J.B. Reclamation of used urban waters for irrigation purposes—A review of treatment technologies. J. Environ. Manag. 2013, 122, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Bixio, D.; Wintgens, T. Water Reuse System Management Manual AQUAREC. Directorate-General for Research; European Commission: Brussels, Belgium, 2006. [Google Scholar]
- Fung, K.Y.; Lee, C.M.; Ng, K.M.; Wibowo, C.; Deng, Z.; Wei, C. Process development of treatment plants for dyeing wastewater. AIChe J. 2012, 58, 2726–2742. [Google Scholar] [CrossRef]
- Subba Rao, A.N.; Venkatarangaiah, V.T. Metal oxide-coated anodes in wastewater treatment. Environ. Sci. Pollut. Res. Int. 2014, 21, 3197–3217. [Google Scholar] [CrossRef] [PubMed]
- Bagal, M.V.; Gogate, P.R. Wastewater treatment using hybrid treatment schemes based on cavitation and Fenton chemistry: A review. Ultrason. Sonochem. 2014, 21, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, H.; Joss, A. Review on the fate of organic micropollutants in wastewater treatment and water reuse with membranes. Water Sci. Technol. 2012, 66, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Semrany, S.; Favier, L.; Djelal, H.; Taha, S.; Amrane, A. Bioaugmentation: Possible solution in the treatment of Bio-Refractory Organic Compounds (Bio-ROCs). Biochem. Eng. J. 2012, 69, 75–86. [Google Scholar] [CrossRef]
- Tyagi, M.; da Fonseca, M.M.; de Carvalho, C.C. Bioaugmentation and biostimulation strategies to improve the effectiveness of bioremediation processes. Biodegradation 2011, 22, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Mrozik, A.; Piotrowska-Seget, Z. Bioaugmentation as a strategy for cleaning up of soils contaminated with aromatic compounds. Microbiol. Res. 2010, 165, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Lebeau, T.; Braud, A.; Jezequel, K. Performance of bioaugmentation-assisted phytoextraction applied to metal contaminated soils: A review. Environ. Pollut. 2008, 153, 497–522. [Google Scholar] [CrossRef] [PubMed]
- Thompson, I.P.; van der Gast, C.J.; Ciric, L.; Singer, A.C. Bioaugmentation for bioremediation: The challenge of strain selection. Environ. Microbiol. 2005, 7, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.C.; van der Gast, C.J.; Thompson, I.P. Perspectives and vision for strain selection in bioaugmentation. Trends Biotechnol. 2005, 23, 74–77. [Google Scholar] [CrossRef] [PubMed]
- El Fantroussi, S.; Agathos, S.N. Is bioaugmentation a feasible strategy for pollutant removal and site remediation? Curr. Opin. Microbiol. 2005, 8, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Herrero, M.; Stuckey, D.C. Bioaugmentation and its application in wastewater treatment: A review. Chemosphere 2015, 140, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Providenti, M.A.; Lee, H.; Trevors, J.T. Selected factors limiting the microbial degradation of recalcitrant compounds. J. Ind. Microbiol. 1993, 12, 379–395. [Google Scholar] [CrossRef]
- Mattes, T.E.; Alexander, A.K.; Coleman, N.V. Aerobic biodegradation of the chloroethenes: Pathways, enzymes, ecology, and evolution. FEMS Microbiol. Rev. 2010, 34, 445–475. [Google Scholar] [CrossRef] [PubMed]
- Loffler, F.E.; Ritalahti, K.M.; Zinder, S.H. Dehalococcoides and reductive dechlorination of chlorinated solvents. In Bioaugmentation for Groundwater Remediation; Ward, C.H., Ed.; Springer Science and Business Media New York: New York, NY, USA, 2013; pp. 39–88. [Google Scholar]
- Boon, N.; Goris, J.; De Vos, P.; Verstraete, W.; Top, E.M. Bioaugmentation of activated sludge by an indigenous 3-chloroaniline-degrading Comamonas testosteroni strain, I2gfp. Appl. Environ. Microbiol. 2000, 66, 2906–2913. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xu, J.; Wang, J.; Wang, S.; Feng, H.; Shentu, J.; Shen, D. Differences between 4-fluoroaniline degradation and autoinducer release by Acinetobacter sp. TW: Implications for operating conditions in bacterial bioaugmentation. Environ. Sci. Pollut. Res. Int. 2013, 20, 6201–6209. [Google Scholar] [CrossRef] [PubMed]
- Quan, X.; Shi, H.; Liu, H.; Lv, P.; Qian, Y. Enhancement of 2,4-dichlorophenol degradation in conventional activated sludge systems bioaugmented with mixed special culture. Water Res. 2004, 38, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Puyol, D.; Monsalvo, V.M.; Sanchis, S.; Sanz, J.L.; Mohedano, A.F.; Rodriguez, J.J. Comparison of bioaugmented EGSB and GAC-FBB reactors and their combination with aerobic SBR for the abatement of chlorophenols. Chem. Eng. J. 2015, 259, 277–285. [Google Scholar] [CrossRef]
- Biermann, C.J. Essentials of Pulping and Papermaking; Academic Press, Inc.: San Diego, CA, USA, 1993. [Google Scholar]
- Wu, J.; Xiao, Y.Z.; Yu, H.Q. Degradation of lignin in pulp mill wastewaters by white-rot fungi on biofilm. Bioresour. Technol. 2005, 96, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Chai, L.Y.; Yang, Z.H.; Tang, C.J.; Chen, Y.H.; Shi, Y. Enhanced remediation of black liquor by activated sludge bioaugmented with a novel exogenous microorganism culture. Appl. Microbiol. Biotechnol. 2013, 97, 6525–6535. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.Y.; Chen, Y.; Huang, Y.; Yang, Z. Diversity of culturable microorganisms from erosive bamboo slips of Kingdom Wu. J. Cent. South Univ. 2010, 41, 1674–1679. (In Chinese) [Google Scholar]
- Tuo, B.H.; Yan, J.B.; Fan, B.A.; Yang, Z.H.; Liu, J.Z. Biodegradation characteristics and bioaugmentation potential of a novel quinoline-degrading strain of Bacillus sp. isolated from petroleum-contaminated soil. Bioresour. Technol. 2012, 107, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Jianlong, W.; Xiangchun, Q.; Libo, W.; Yi, Q.; Hegemann, W. Bioaugmentation as a tool to enhance the removal of refractory compound in coke plant wastewater. Process Biochem. 2002, 38, 777–781. [Google Scholar] [CrossRef]
- Bai, Y.; Sun, Q.; Zhao, C.; Wen, D.; Tang, X. Simultaneous biodegradation of pyridine and quinoline by two mixed bacterial strains. Appl. Microbiol. Biotechnol. 2009, 82, 963–973. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Sun, Q.; Xing, R.; Wen, D.; Tang, X. Removal of pyridine and quinoline by bio-zeolite composed of mixed degrading bacteria and modified zeolite. J. Hazard. Mater. 2010, 181, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Sun, Q.; Zhao, C.; Wen, D.; Tang, X. Bioaugmentation treatment for coking wastewater containing pyridine and quinoline in a sequencing batch reactor. Appl. Microbiol. Biotechnol. 2010, 87, 1943–1951. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Sun, Q.; Sun, R.; Wen, D.; Tang, X. Bioaugmentation and adsorption treatment of coking wastewater containing pyridine and quinoline using zeolite-biological aerated filters. Environ. Sci. Technol. 2011, 45, 1940–1948. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, Y.; Zhang, X.; Jiang, X.; Wu, S.; Shen, J.; Sun, X.; Li, J.; Lu, L.; Wang, L. Aerobic granulation strategy for bioaugmentation of a sequencing batch reactor (SBR) treating high strength pyridine wastewater. J. Hazard. Mater. 2015, 295, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Wen, D.; Zhang, J.; Xiong, R.; Liu, R.; Chen, L. Bioaugmentation with a pyridine-degrading bacterium in a membrane bioreactor treating pharmaceutical wastewater. J. Environ. Sci. 2013, 25, 2265–2271. [Google Scholar] [CrossRef]
- Fan, L.; Zhu, S.; Liu, D.; Ni, J. Decolorization mechanism of 1-amino-4-bromoanthraquinone-2-sulfonic acid using Sphingomonas herbicidovorans FL. Dyes Pigments 2008, 78, 34–38. [Google Scholar] [CrossRef]
- Pandey, A.; Singh, P.; Iyengar, L. Bacterial decolorization and degradation of azo dyes. Int. Biodeterior. Biodegrad. 2007, 59, 73–84. [Google Scholar] [CrossRef]
- Wang, J.; Liu, G.-F.; Lu, H.; Jin, R.-F.; Zhou, J.-T.; Lei, T.-M. Biodegradation of Acid Orange 7 and its auto-oxidative decolorization product in membrane-aerated biofilm reactor. Int. Biodeterior. Biodegrad. 2012, 67, 73–77. [Google Scholar] [CrossRef]
- Fan, L.; Ni, J.; Wu, Y.; Zhang, Y. Treatment of bromoamine acid wastewater using combined process of micro-electrolysis and biological aerobic filter. J. Hazard. Mater. 2009, 162, 1204–1210. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Liu, D.Q.; Zhu, S.N.; Mai, J.X.; Ni, J.R. Degradation characteristics of bromoamine acid by Sphingomonas sp. FL. Huan Jing Ke Xue 2008, 29, 2618–2623. (In Chinese) [Google Scholar] [PubMed]
- Qu, Y.; Zhou, J.; Wang, J.; Song, Z.; Xing, L.; Fu, X. Bioaugmentation of bromoamine acid degradation with Sphingomonas xenophaga QYY and DNA fingerprint analysis of augmented systems. Biodegradation 2006, 17, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Zhou, J.; Wang, J.; Fu, X.; Xing, L. Microbial community dynamics in bioaugmented sequencing batch reactors for bromoamine acid removal. FEMS Microbiol. Lett. 2005, 246, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.Y.; Zhou, J.T.; Wang, J.; Xing, L.L.; Jiang, N.; Gou, M.; Salah Uddin, M. Population dynamics in bioaugmented membrane bioreactor for treatment of bromoamine acid wastewater. Bioresour. Technol. 2009, 100, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Lee, D.S.; Kim, Y.M.; Park, J.M. Bioaugmentation of cyanide-degrading microorganisms in a full-scale cokes wastewater treatment facility. Bioresour. Technol. 2008, 99, 2092–2096. [Google Scholar] [CrossRef] [PubMed]
- Talhout, R.; Schulz, T.; Florek, E.; van Benthem, J.; Wester, P.; Opperhuizen, A. Hazardous compounds in tobacco smoke. Int. J. Environ. Res. Public Health 2011, 8, 613–628. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.; Cardinale, A.; Margaritora, S.; Cesario, A. Nicotinic receptor and tobacco-related cancer. Life Sci. 2012, 91, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Zhu, C.; Shu, M.; Sun, K.; Zhao, L.; Wang, C.; Ye, Z.; Chen, J. Degradation of nicotine in tobacco waste extract by newly isolated Pseudomonas sp. ZUTSKD. Bioresour. Technol. 2010, 101, 6935–6941. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.; Mackay, J.; Ross, H. The Tobacco Atlas; American Cancer Society: Atlanta, GA, USA, 2012. [Google Scholar]
- Wang, M.Z.; Yang, G.Q.; Wang, X.; Yao, Y.L.; Min, H.; Lv, Z.M. Nicotine degradation by two novel bacterial isolates of Acinetobacter sp. TW and Sphingomonas sp. TY and their responses in the presence of neonicotinoid insecticides. World J. Microbiol. Biotechnol. 2011, 27, 1633–1640. [Google Scholar] [CrossRef]
- Wang, J.H.; He, H.Z.; Wang, M.Z.; Wang, S.; Zhang, J.; Wei, W.; Xu, H.X.; Lv, Z.M.; Shen, D.S. Bioaugmentation of activated sludge with Acinetobacter sp. TW enhances nicotine degradation in a synthetic tobacco wastewater treatment system. Bioresour. Technol. 2013, 142C, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yang, G.; Min, H.; Lv, Z.; Jia, X. Bioaugmentation with the nicotine-degrading bacterium Pseudomonas sp. HF-1 in a sequencing batch reactor treating tobacco wastewater: Degradation study and analysis of its mechanisms. Water Res. 2009, 43, 4187–4196. [Google Scholar] [CrossRef] [PubMed]
- Sitarek, K.; Gromadzińska, J.; Lutz, P.; Stetkiewicz, J.; Świercz, R.; Wąsowicz, W. Fertility and developmental toxicity studies of diethylene glycol monobutyl ether (DGBE) in rats. Int. J. Occup. Med. Environ. Health 2012, 25, 404–417. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Fan, R.; Zou, W.; Zhou, H.; Tan, Z.; Li, X. Bioaugmentation for treatment of full-scale diethylene glycol monobutyl ether (DGBE) wastewater by Serratia sp. BDG-2. J. Hazard. Mater. 2016, 309, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Ma, W.; Han, H.; Jia, S.; Hou, B. Isolation of a naphthalene-degrading strain from activated sludge and bioaugmentation with it in a MBR treating coal gasification wastewater. Bull. Environ. Contam. Toxicol. 2015, 94, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Liu, R.; Liu, C.; Chen, L. Bioaugmentation with isolated strains for the removal of toxic and refractory organics from coking wastewater in a membrane bioreactor. Biodegradation 2015, 26, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Qu, Y.; Ma, Q.; Zhang, X.; Zhou, J.; Ma, F. Performance and microbial community dynamics in bioaugmented aerated filter reactor treating with coking wastewater. Bioresour. Technol. 2015, 190, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Han, H.; Zhao, Q.; Xu, C.; Zhang, L. Bioaugmentation of biological contact oxidation reactor (BCOR) with phenol-degrading bacteria for coal gasification wastewater (CGW) treatment. Bioresour. Technol. 2013, 150, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Lyon, D.Y.; Vogel, T. Bioaugmentation for groundwater remediation: An overview. In Bioaugmentation for Groundwater Remediation; Ward, C.H., Ed.; Springer Science and Business Media: New York, NY, USA, 2013; pp. 1–37. [Google Scholar]
- Kalogerakis, N. Bioaugmentation—Is It Really Needed for the Bioremediation of Contaminated Sites? Available online: http://www.srcosmos.gr/srcosmos/showpub.aspx?aa=8113 (accessed on 23 June 2016).
- Stroo, H.F.; Leeson, A.; Ward, C.H. Bioaugmentation for Groundwater Remediation; Springer Science and Business Media: New York, NY, USA, 2013; p. 389. [Google Scholar]
- Bouchez, T.; Patureau, D.; Dabert, P.; Juretschko, S.; Dore, J.; Delgenes, P.; Moletta, R.; Wagner, M. Ecological study of a bioaugmentation failure. Environ. Microbiol. 2000, 2, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, R.M.; Mallory, L.M.; Alexander, M. Reasons for possible failure of inoculation to enhance biodegradation. Appl. Environ. Microbiol. 1985, 50, 977–983. [Google Scholar] [PubMed]
- Eberl, L.; Schulze, R.; Ammendola, A.; Geisenberger, O.; Erhart, R.; Sternberg, C.; Molin, S.; Amann, R. Use of green fluorescent protein as a marker for ecological studies of activated sludge communities. FEMS Microbiol. Lett. 1997, 149, 77–83. [Google Scholar] [CrossRef]
- Ramadan, M.A.; el-Tayeb, O.M.; Alexander, M. Inoculum size as a factor limiting success of inoculation for biodegradation. Appl. Environ. Microbiol. 1990, 56, 1392–1396. [Google Scholar] [PubMed]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, O.H.; Kushmaro, A. Bacteriophage ecology in environmental biotechnology processes. Curr. Opin. Biotechnol. 2011, 22, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Fan, H.; Liu, S.; Liu, Y.; Liu, Z. A bioaugmentation failure caused by phage infection and weak biofilm formation ability. J. Environ. Sci. 2009, 21, 1153–1161. [Google Scholar] [CrossRef]
- Barr, J.J.; Slater, F.R.; Fukushima, T.; Bond, P.L. Evidence for bacteriophage activity causing community and performance changes in a phosphorus-removal activated sludge. FEMS Microbiol. Ecol. 2010, 74, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Otawa, K.; Onuki, M.; Satoh, H.; Mino, T. Population dynamics of phage-host system of Microlunatus phosphovorus indigenous in activated sludge. J. Microbiol. Biotechnol. 2007, 17, 1704–1707. [Google Scholar] [PubMed]
- Shi, S.; Qu, Y.; Ma, F.; Zhou, J. Bioremediation of coking wastewater containing carbazole, dibenzofuran and dibenzothiphene by immobilized naphthalene-cultivated Arthrobacter sp. W1 in magnetic gellan gum. Bioresour. Technol. 2014, 166, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Danquah, M.K.; Zheng, C.; Potumarthi, R.; Chen, X.D.; Lu, Y. NaCS–PDMDAAC immobilized autotrophic cultivation of Chlorella sp. for wastewater nitrogen and phosphate removal. Chem. Eng. J. 2012, 187, 185–192. [Google Scholar] [CrossRef]
- Ogugbue, C.J.; Sawidis, T.; Oranusi, N.A. Evaluation of colour removal in synthetic saline wastewater containing azo dyes using an immobilized halotolerant cell system. Ecol. Eng. 2011, 37, 2056–2060. [Google Scholar] [CrossRef]
- Surkatti, R.; El-Naas, M.H. Biological treatment of wastewater contaminated with p-cresol using Pseudomonas putida immobilized in polyvinyl alcohol (PVA) gel. J. Water Process Eng. 2014, 1, 84–90. [Google Scholar] [CrossRef]
- Pakshirajan, K.; Kheria, S. Continuous treatment of coloured industry wastewater using immobilized Phanerochaete chrysosporium in a rotating biological contactor reactor. J. Environ. Manag. 2012, 101, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Khondee, N.; Tathong, S.; Pinyakong, O.; Powtongsook, S.; Chatchupong, T.; Ruangchainikom, C.; Luepromchai, E. Airlift bioreactor containing chitosan-immobilized Sphingobium sp. P2 for treatment of lubricants in wastewater. J. Hazard. Mater. 2012, 213–214, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Del Castillo, I.; Hernández, P.; Lafuente, A.; Rodríguez-Llorente, I.D.; Caviedes, M.A.; Pajuelo, E. Self-bioremediation of cork-processing wastewaters by (chloro)phenol-degrading bacteria immobilised onto residual cork particles. Water Res. 2012, 46, 1723–1734. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C. Quorum sensing inhibitors: An overview. Biotechnol. Adv. 2013, 31, 224–245. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Li, M. Quorum sensing inhibitors: A patent review. Expert Opin. Ther. Pat. 2013, 23, 867–894. [Google Scholar] [CrossRef] [PubMed]
- Valle, A.; Bailey, M.J.; Whiteley, A.S.; Manefield, M. N-acyl-l-homoserine lactones (AHLs) affect microbial community composition and function in activated sludge. Environ. Microbiol. 2004, 6, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Chong, G.; Kimyon, O.; Rice, S.A.; Kjelleberg, S.; Manefield, M. The presence and role of bacterial quorum sensing in activated sludge. Microb. Biotechnol. 2012, 5, 621–633. [Google Scholar] [CrossRef] [PubMed]
- Yeon, K.M.; Cheong, W.S.; Oh, H.S.; Lee, W.N.; Hwang, B.K.; Lee, C.H.; Beyenal, H.; Lewandowski, Z. Quorum sensing: A new biofouling control paradigm in a membrane bioreactor for advanced wastewater treatment. Environ. Sci. Technol. 2009, 43, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, A.M. Genetically-manipulated microorganisms and their products in the oil service industries. Trends Biotechnol. 1985, 3, 32–39. [Google Scholar] [CrossRef]
- McClure, N.C.; Fry, J.C.; Weightman, A.J. Survival and catabolic activity of natural and genetically engineered bacteria in a laboratory-scale activated-sludge unit. Appl. Environ. Microbiol. 1991, 57, 366–373. [Google Scholar] [PubMed]
- Nusslein, K.; Maris, D.; Timmis, K.; Dwyer, D.F. Expression and transfer of engineered catabolic pathways harbored by Pseudomonas spp. introduced into activated sludge microcosms. Appl. Environ. Microbiol. 1992, 58, 3380–3386. [Google Scholar] [PubMed]
- Wei, M.J.; Wang, H.; Liu, C.; Ning, D.L. Bioaugmentation with immobilized genetically engineered microorganism (GEM)/CAS process for treatment of atrazine wastewater. Huan Jing Ke Xue 2008, 29, 1555–1560. [Google Scholar] [PubMed]
- Jin, R.; Yang, H.; Zhang, A.; Wang, J.; Liu, G. Bioaugmentation on decolorization of C.I. Direct blue 71 by using genetically engineered strain Escherichia coli JM109 (pGEX-AZR). J. Hazard. Mater. 2009, 163, 1123–1128. [Google Scholar] [CrossRef] [PubMed]
- Okino, S.; Iwasaki, K.; Yagi, O.; Tanaka, H. Removal of mercuric chloride by a genetically engineered mercury-volatilizing bacterium Pseudomonas putida PpY101/pSR134. Bull. Environ. Contam. Toxicol. 2002, 68, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Wilson, D.B. Bioaccumulation of mercury from wastewater by genetically engineered Escherichia coli. Appl. Microbiol. Biotechnol. 2001, 56, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Almaguer-Cantu, V.; Morales-Ramos, L.H.; Balderas-Renteria, I. Biosorption of lead (II) and cadmium (II) using Escherichia coli genetically engineered with mice metallothionein I. Water Sci. Technol. 2011, 63, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Li, Q.B.; Lu, Y.H.; Sun, D.H.; Huang, Y.L. Uptake of nickel from industrial wastewater by genetically engineered Escherichia coli JM109. Sheng Wu Gong Cheng Xue Bao 2003, 19, 343–348. (In Chinese) [Google Scholar] [PubMed]
- Singh, J.S.; Abhilash, P.C.; Singh, H.B.; Singh, R.P.; Singh, D.P. Genetically engineered bacteria: An emerging tool for environmental remediation and future research perspectives. Gene 2011, 480, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Madhavi, V.; Lele, S. Laccase: Properties and applications. BioResources 2009, 4, 1694–1717. [Google Scholar]
- Ba, S.; Arsenault, A.; Hassani, T.; Jones, J.P.; Cabana, H. Laccase immobilization and insolubilization: From fundamentals to applications for the elimination of emerging contaminants in wastewater treatment. Crit. Rev. Biotechnol. 2013, 33, 404–418. [Google Scholar] [CrossRef] [PubMed]
- Blakely, G.W. Chapter 15—Mechanisms of horizontal gene transfer and DNA recombination. In Molecular Medical Microbiology, 2nd ed.; Schwartzman, Y.-W., Sussman, M., Liu, D., Poxton, L., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 291–302. [Google Scholar]
- Thomas, C.M.; Nielsen, K.M. Mechanisms of, and barriers to, horizontal gene transfer between bacteria. Nat. Rev. Microbiol. 2005, 3, 711–721. [Google Scholar] [CrossRef] [PubMed]
- McClure, N.C.; Weightman, A.J.; Fry, J.C. Survival of Pseudomonas putida UWC1 containing cloned catabolic genes in a model activated-sludge unit. Appl. Environ. Microbiol. 1989, 55, 2627–2634. [Google Scholar] [PubMed]
- Hausner, M.; Starek, M.; Bathe, S. 3.13—Bioremediation: Plasmid-mediated bioaugmentation of microbialcommunities—Experience from laboratory-scale bioreactors. In Treatise on Water Science; Wilderer, P., Ed.; Elsevier: Oxford, UK, 2011; pp. 369–376. [Google Scholar]
- Top, E.M.; Springael, D.; Boon, N. Catabolic mobile genetic elements and their potential use in bioaugmentation of polluted soils and waters. FEMS Microbiol. Ecol. 2002, 42, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Liao, R.; Zhang, X.-X.; Liu, B.; Li, Y.; Wu, B.; Li, A. Metagenomic insights into salinity effect on diversity and abundance of denitrifying bacteria and genes in an expanded granular sludge bed reactor treating high-nitrate wastewater. Chem. Eng. J. 2015, 277, 116–123. [Google Scholar] [CrossRef]
- Miao, Y.; Liao, R.; Zhang, X.-X.; Wang, Y.; Wang, Z.; Shi, P.; Liu, B.; Li, A. Metagenomic insights into Cr(VI) effect on microbial communities and functional genes of an expanded granular sludge bed reactor treating high-nitrate wastewater. Water Res. 2015, 76, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Shu, D.; He, Y.; Yue, H.; Wang, Q. Metagenomic and quantitative insights into microbial communities and functional genes of nitrogen and iron cycling in twelve wastewater treatment systems. Chem. Eng. J. 2016, 290, 21–30. [Google Scholar] [CrossRef]
- Tomazetto, G.; Wibberg, D.; Schlüter, A.; Oliveira, V.M. New FeFe-hydrogenase genes identified in a metagenomic fosmid library from a municipal wastewater treatment plant as revealed by high-throughput sequencing. Res. Microbiol. 2015, 166, 9–19. [Google Scholar] [CrossRef] [PubMed]
- de Morais, M.G.; Martins, V.G.; Steffens, D.; Pranke, P.; da Costa, J.A. Biological applications of nanobiotechnology. J. Nanosci. Nanotechnol. 2014, 14, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, G.; Momenpour, M.; Omidi, F.; Hosseini, M.R.; Ahani, M.; Barzegari, A. Applications of nanomaterials in water treatment and environmental remediation. Front. Environ. Sci. Eng. 2014, 8, 471–482. [Google Scholar] [CrossRef]
- Garner, K.L.; Keller, A.A. Emerging patterns for engineered nanomaterials in the environment: A review of fate and toxicity studies. J. Nanopart. Res. 2014, 16, 1–28. [Google Scholar] [CrossRef]
- Crane, R.A.; Scott, T.B. Nanoscale zero-valent iron: Future prospects for an emerging water treatment technology. J. Hazard. Mater. 2012, 211–212, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.; Dionysiou, D.D.; Liu, H. The use of zero-valent iron for groundwater remediation and wastewater treatment: A review. J. Hazard. Mater. 2014, 267, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Beyth, N.; Houri-Haddad, Y.; Domb, A.; Khan, W.; Hazan, R. Alternative antimicrobial approach: nano-antimicrobial materials. Evid.-Based Complement. Altern. Med. 2015, 2015, 246012. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, M.; Xu, X.; Liu, N. Effects of carbon nanotubes on atrazine biodegradation by Arthrobacter sp. J. Hazard. Mater. 2015, 287, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.A.; Pereira, M.F.R.; Alves, M.M.; Pereira, L. Carbon based materials as novel redox mediators for dye wastewater biodegradation. Appl. Catalysis B 2014, 144, 713–720. [Google Scholar] [CrossRef]
- Yang, S.; Chen, D.; Li, N.; Xu, Q.; Li, H.; He, J.; Lu, J. Surface-nanoengineered bacteria for efficient local enrichment and biodegradation of aqueous organic wastes: Using phenol as a model compound. Adv. Mater. 2016, 28, 2916–2922. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Liu, F.; Wu, N.; Ju, J.; Yu, B. Efficient biodegradation of chlorophenols in aqueous phase by magnetically immobilized aniline-degrading Rhodococcus rhodochrous strain. J. Nanobiotechnol. 2016, 14, 5. [Google Scholar] [CrossRef] [PubMed]
| Pollutant | Set Up | Medium for Bioaugmentation | Bioaugmented Bacteria | Ref. | |
|---|---|---|---|---|---|
| 3-Chloroaniline |  | Semi-continuous activated sludge (SCAS) (1 L) | Synthetic influent consisting of skim milk powder | Comamonas testosteroni | [19] |
| 4-Fluoroaniline |  | Batch reactor (BR) (250 mL) | Inorganic salt medium | Acinetobacter sp. | [20] |
| 2,4-Dichlorophenol (2,4-DCP) |  | Laboratory-scale continuous flow complete-mixed reactors (CFSTRs) (16 L) | Synthetic wastewater (SW) | Consortium of bacteria | [21] |
| 2,4,6-Trichloro-phenol | 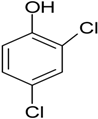 | fluidized bed biofilm reactor (FBBR) and expanded granular sludge bed (EGSB) | Industrial wastewater (IW) | Desulfitobacterium sp. | [22] |
| Quinoline | 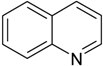 | Sequential Batch reactor (SBR) (250 mL) | Petroleum refinery wastewater | Bacillus sp. | [27] |
| Quinoline | SBR (2–7 L) | Coke plant wastewater | Burkholderia pickettii | [28] | |
| Pyridine and quinoline | 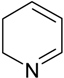 | BR (100 mL) | Inorganic medium and wastewater | Paracoccus sp. and Pseudomonas sp. | [29] |
| Quinoline and Pyridine | BR (250 mL) with modified zeolite | Coke wastewater | Paracoccus sp. and Pseudomonas sp. | [30] | |
| Quinoline and Pyridine | SBR | Coke wastewater | Consortium of Paracoccus sp. BW001, Shinella Zoogloeoids BC026, Pseudomonas sp. BC001 and BW003, | [31] | |
| Pyridine | SBR | Industrial wastewater | Rhizobium sp. | [33] | |
| Pyridine | 2 Membrane Bioreactors (MBR, 25 L each) | Pharmaceutical Wastewater | Paracoccus denitrificans | ||
| Acid Orange 7 dye | 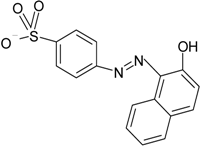 | Membrane-aerated biofilm reactor (MABR) (2 L) | SW | Shewanella sp. XB | [37] |
| Bromoamine | 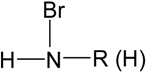 | MBR 10 L | SW | Sphingomonas xenophaga | [40] |
| Bromoamine | Combined process of microelectrolysis and biological aerated filter 1–3 L | Wastewater | Sphingomonas sp. | [39] | |
| Bromoamine | BR (250 mL) | Inorganic medium | Sphingomonas xenophaga | [40] | |
| Cyanide | 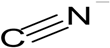 | Full scale Cokes wastewater treatment facility (fluidized bed type process) > 3 × 105 L | Cokes wastewater | Cryptococcus humicolus, and Unidentified cyanide-degrading microorganisms | [43] |
| Nicotine |  | 3 reactors of cylindrical shape Height: Bore size of 10:1 (2 L) | Synthetic tobacco wastewater | Acinetobacter sp. | [49] |
| Nicotine | SBR (2 L) | Tobacco wastewater diluted in tap water (7%) (g/mL) | Pseudomonas sp. | [50] | |
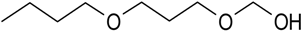
| Diethylene glycol butyl ether | SBR (2 L) Full scale Plug flow aerated tank (60,000 L) | Wastewater from silicon plate manufacture plant | Serratia sp. | [52] | |
 | |||||
| Lignin (highly complex polymer of phenol) | 2 L BR | Industrial wastewater | Comamonas and Pandoraea (bacteria), and Aspergillus (fungus) | [25] | |
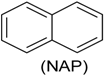
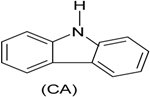
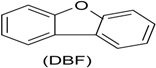
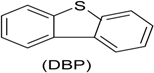
| Phenol (PH) and naphthalene (NAP) along with carbazole (CA), dibenzofuran (DBF), and dibenzothiophene | 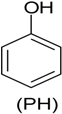 | BR (column of 10 × 50 cm) | Coking wastewater from a treatment plant | Immobilized phenol-utilizing Arthrobacter sp. | [55] |
 | |||||
 | |||||
 | |||||
 | |||||
| Naphthalene | MBR (8 L) | Coal gasification wastewater. | Streptomyces sp. | [53] | |
| Mixture of phenol, pyridine, quinoline, naphthalene and carbazole | A sequential system of anaerobic reactor(4.9 L), anoxic reactor, A2 (4.5 L), and an oxic MBR (9 L). MBR (9.0 L) | Coking wastewater | Consortium of 6 bacteria containing Paracoccus Denitrificans and 5 strains of Pseudomonas sp. | [54] | |
| Phenol | Biological contact oxidation reactor (BCOR) | Coal gasification wastewater | Mixture of phenol-degrading bacteria | [56] | |
| Main Objectives | Limitations | Potential Solutions | Remarks | References |
|---|---|---|---|---|
| Overcoming low growth or washout of bioaugmented bacteria | Low inocula can lead to limited survival of bioaugmented bacteria | Use of high inocula, at least 106–107 cells per mL. Monitoring of growth of bioaugmented bacteria (followed by the addition of new bacteria) | Has been tested with encouraging results in groundwater | [57] |
| Lysis of bacteria by viral (bacteriophage) infections | Monitoring of growth of bioaugmented bacteria (followed by the addition of new bacteria) | Several approaches exist to monitor bacterial levels in wastewater | [15,57] | |
| Limited bacterial growth as the result of low quorum sensing (QS) | Use of QS inducers to increase bacterial growth. Monitoring of growth of bioaugmented bacteria | Has been evaluated in laboratory scale, but cost may be a limitation in full scale treatment | [78,79,80] | |
| Increase of efficiency of bioaugmentation | Low biochemical ability of bioaugmented bacteria to biodegrade pollutants | Use of genetically modified organisms encoding catabolic efficient enzymes | Has been tested with encouraging results | [84,85,86,87,88,89] |
| Use of plasmids encoding catabolic efficient enzymes | Potentially attractive, but so far, not clear evidence of success due to the uncertainty of incorporation of plasmids into receiving organisms | [96,97] | ||
| Low ability of bioaugmented bacteria to biodegrade pollutants | Use of immobilized bioaugmented bacteria | Has been evaluated with encouraging results, but cost may be a limitation in full scale treatment | [72,73,74,75] | |
| Exploitation of nanotechnology with the use of nanomaterial (NM) along with bioaugmented bacteria to increase biodegradation | NM (at low concentration) increases bacterial growth and the rates of biochemical reactions. Approach is promising but more studies are still needed to ascertain this evidence. | [105,106,108,109] | ||
| Use of functionalized bioaugmented bacteria by fixing NM on their surface to increase bio-availability of pollutants | Promising approach, based one study, thus more studies are needed to support this technology | [110] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nzila, A.; Razzak, S.A.; Zhu, J. Bioaugmentation: An Emerging Strategy of Industrial Wastewater Treatment for Reuse and Discharge. Int. J. Environ. Res. Public Health 2016, 13, 846. https://doi.org/10.3390/ijerph13090846
Nzila A, Razzak SA, Zhu J. Bioaugmentation: An Emerging Strategy of Industrial Wastewater Treatment for Reuse and Discharge. International Journal of Environmental Research and Public Health. 2016; 13(9):846. https://doi.org/10.3390/ijerph13090846
Chicago/Turabian StyleNzila, Alexis, Shaikh Abdur Razzak, and Jesse Zhu. 2016. "Bioaugmentation: An Emerging Strategy of Industrial Wastewater Treatment for Reuse and Discharge" International Journal of Environmental Research and Public Health 13, no. 9: 846. https://doi.org/10.3390/ijerph13090846
APA StyleNzila, A., Razzak, S. A., & Zhu, J. (2016). Bioaugmentation: An Emerging Strategy of Industrial Wastewater Treatment for Reuse and Discharge. International Journal of Environmental Research and Public Health, 13(9), 846. https://doi.org/10.3390/ijerph13090846







