Association between Serum Uric Acid and Elevated Alanine Aminotransferase in the General Population
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.3. Blood Pressure Measurements
2.4. Anthropometric Measurements
2.5. Biochemical Measurements
2.6. Definitions
2.7. Statistical Analysis
3. Results
3.1. Subject Characteristics
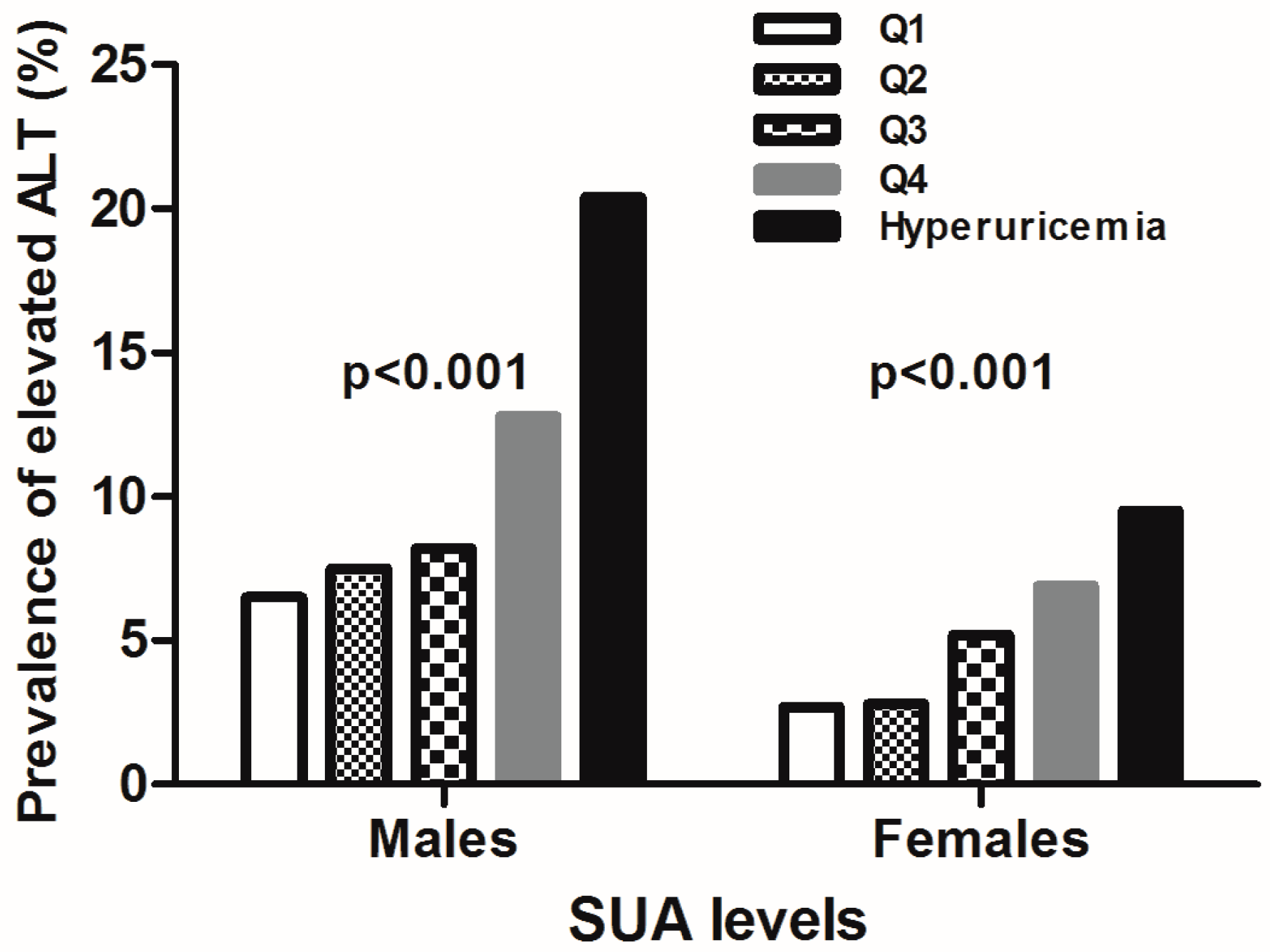
3.2. Prevalence of Elevated ALT Categorized by SUA Levels for both Sexes
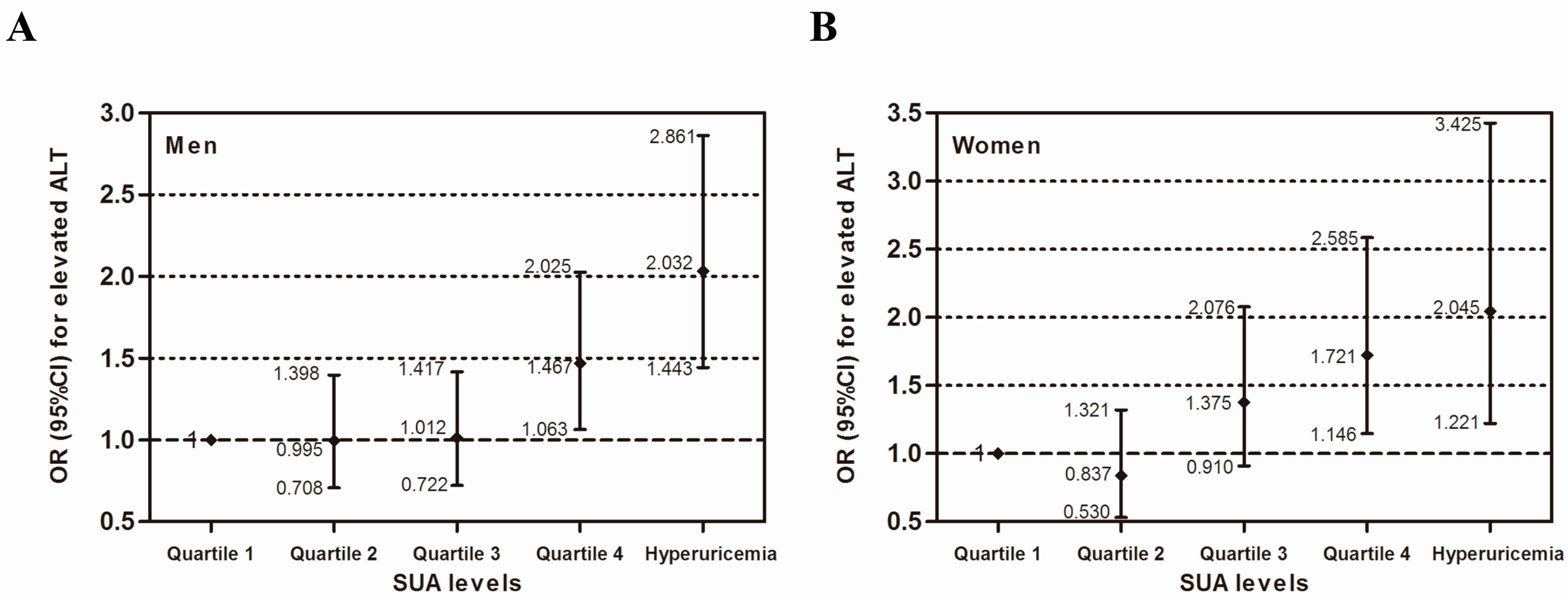
3.3. Multivariate Analysis of the Independent Association between SUA and Elevated Serum ALT
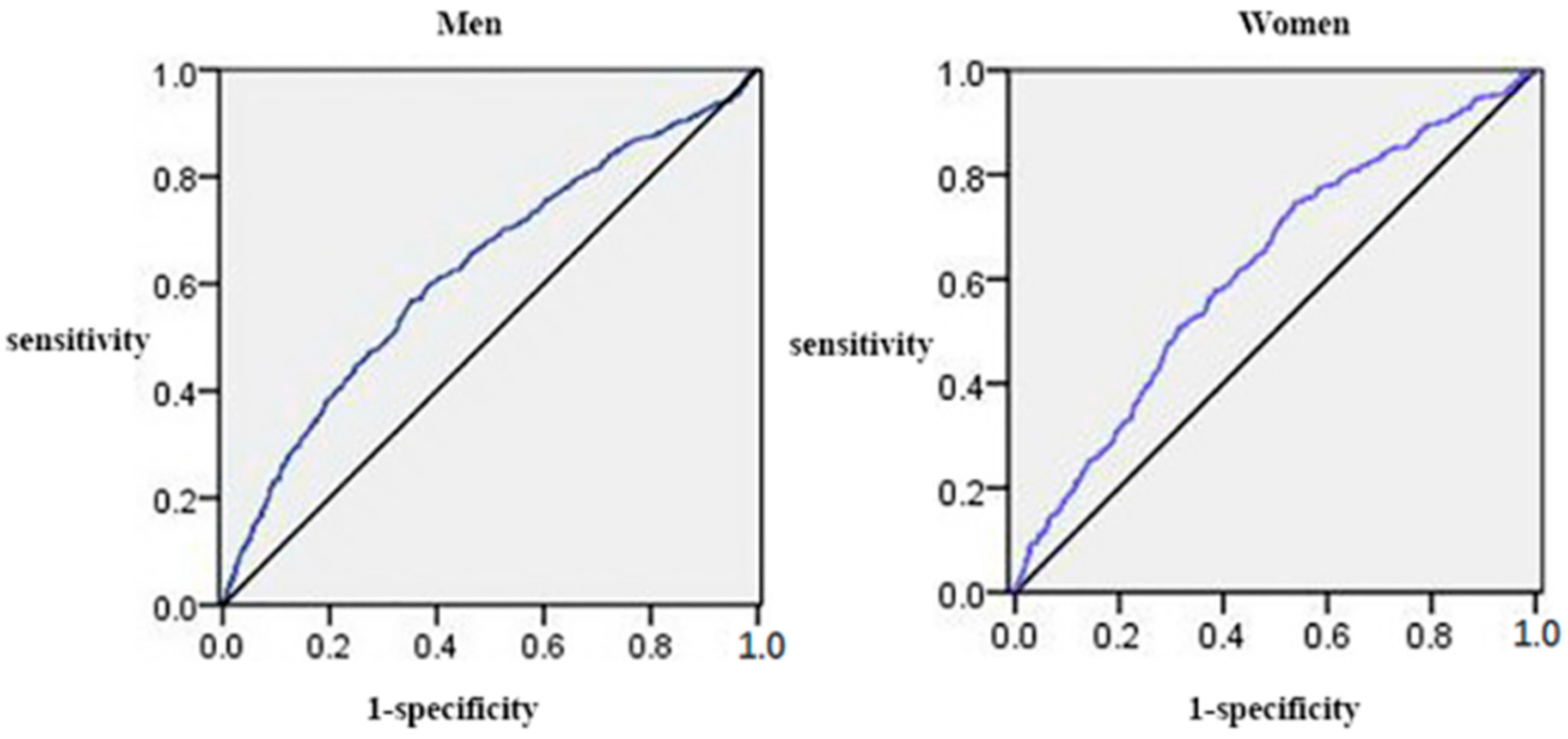
3.4. The Area under the Receiver Operating Characteristic Curves (AUCs) (and 95% CIs) of Anthropometric Measures for the Presence of Elevated ALT
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SBP | Systolic blood pressure |
| DBP | Diastolic blood pressure |
| WC | Waist circumference |
| BMI | Body mass index |
| TC | Total cholesterol |
| TG | Triacylglycerol |
| HDL-C | High-density lipoprotein cholesterol |
| LDL-C | Low-density lipoprotein cholesterol |
| FPG | Fasting plasma glucose |
| ALT | Alanine aminotransferase |
| CVD | Cardiovascular diseases |
| NAFLD | Nonalcoholic fatty liver disease |
| MetS | Metabolic syndrome |
| SUA | Serum uric acid |
| AUC | Area under the receiver operating characteristic curve |
References
- Kaplan, M.M. Alanine aminotransferase levels: What’s normal? Ann. Intern. Med. 2002, 137, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.M.; Tung, T.H.; Liu, J.H.; Chen, V.T.; Lin, C.H.; Hsu, C.T.; Chou, P. A community-based epidemiological study of elevated serum alanine aminotransferase levels in Kinmen, Taiwan. World J. Gastroenterol. 2005, 11, 1616–1622. [Google Scholar] [CrossRef] [PubMed]
- Hanley, A.J.; Williams, K.; Festa, A.; Wagenknecht, L.E.; D’Agostino, R.B., Jr.; Kempf, J.; Zinman, B.; Haffner, S.M. Elevations in markers of liver injury and risk of type 2 diabetes: The insulin resistance atherosclerosis study. Diabetes 2004, 53, 2623–2632. [Google Scholar] [CrossRef] [PubMed]
- Goessling, W.; Massaro, J.M.; Vasan, R.S.; D’Agostino, R.B., Sr.; Ellison, R.C.; Fox, C.S. Aminotransferase levels and 20-year risk of metabolic syndrome, diabetes, and cardiovascular disease. Gastroenterology 2008, 135, 1935–1944. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A.; Waters, O.R.; Knuiman, M.W.; Elliott, R.R.; Olynyk, J.K. NAFLD as a risk factor for the development of diabetes and the metabolic syndrome: An eleven-year follow-up study. Am. J. Gastroenterol. 2009, 104, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.K.; Nam, H.S.; Rhee, J.A.; Shin, J.H.; Kim, J.M.; Cho, K.H. Metabolic syndrome and ALT: A community study in adult Koreans. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Marchesini, G.; Brizi, M.; Bianchi, G.; Tomassetti, S.; Bugianesi, E.; Lenzi, M.; McCullough, A.J.; Natale, S.; Forlani, G.; Melchionda, N. Nonalcoholic fatty liver disease: A feature of the metabolic syndrome. Diabetes 2001, 50, 1844–1850. [Google Scholar] [CrossRef] [PubMed]
- Angelico, F.; del Ben, M.; Conti, R.; Francioso, S.; Feole, K.; Maccioni, D.; Antonini, T.M.; Alessandri, C. Non-alcoholic fatty liver syndrome: A hepatic consequence of common metabolic diseases. J. Gastroenterol. Hepatol. 2003, 18, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Cortez-Pinto, H.; Camilo, M.E.; Baptista, A.; de Oliveira, A.G.; de Moura, M.C. Non-alcoholic fatty liver: Another feature of the metabolic syndrome? Clin. Nutr. (Edinb. Scotl.) 1999, 18, 353–358. [Google Scholar] [CrossRef]
- Marchesini, G.; Forlani, G. NASH: From liver diseases to metabolic disorders and back to clinical hepatology. Hepatology (Baltimore MD) 2002, 35, 497–499. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Guo, X.; Zhang, X.; Yu, S.; Yang, H.; Jiang, M.; Sun, G.; Sun, Y. Association between elevated serum alanine aminotransferase and cardiometabolic risk factors in rural Chinese population: A cross-sectional study. BMC Cardiovasc. Disord. 2015, 15, 65. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Guo, X.; Yu, S.; Zhou, Y.; Li, Z.; Sun, Y. Metabolic Syndrome and Serum Liver Enzymes in the General Chinese Population. Int. J. Environ. Res. Public Health 2016, 13, 223. [Google Scholar] [CrossRef] [PubMed]
- Hediger, M.A.; Johnson, R.J.; Miyazaki, H.; Endou, H. Molecular physiology of urate transport. Physiology (Bethesda MD) 2005, 20, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.H.; Min, H.G.; Kim, I.J.; Kim, Y.K.; Son, S.M. Comparison of alanine aminotransferase, white blood cell count, and uric acid in their association with metabolic syndrome: A study of Korean adults. Endocr. J. 2008, 55, 1093–1102. [Google Scholar] [CrossRef] [PubMed]
- Feig, D.I.; Kang, D.H.; Johnson, R.J. Uric acid and cardiovascular risk. N. Engl. J. Med. 2008, 359, 1811–1821. [Google Scholar] [CrossRef] [PubMed]
- Sirota, J.C.; McFann, K.; Targher, G.; Johnson, R.J.; Chonchol, M.; Jalal, D.I. Elevated serum uric acid levels are associated with non-alcoholic fatty liver disease independently of metabolic syndrome features in the United States: Liver ultrasound data from the National Health and Nutrition Examination Survey. Metabolism 2013, 62, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Lee, H.R.; Lee, J.H.; Shin, Y.H.; Shim, J.Y. Association between serum uric acid and non-alcoholic fatty liver disease in Korean adults. Clin. Chem. Lab. Med. 2010, 48, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, C.; Yu, C.; Xu, L.; Miao, M. Association of serum uric acid level with non-alcoholic fatty liver disease: A cross-sectional study. J. Hepatol. 2009, 50, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Ballestri, S.; Marchesini, G.; Angulo, P.; Loria, P. Nonalcoholic fatty liver disease: A precursor of the metabolic syndrome. Dig. Liver Dis. 2015, 47, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Ben-Assuli, O.; Rabinowich, L.; Goldstein, A.; Magid, A.; Shalev, V.; Shibolet, O.; Chodick, G. The association between the serum levels of uric acid and alanine aminotransferase in a population-based cohort. Liver Int. 2015, 35, 2408–2415. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, E.; Petrie, J.; Littler, W.; de Swiet, M.; Padfield, P.L.; Altman, D.G.; Bland, M.; Coats, A.; Atkins, N. An outline of the revised British Hypertension Society protocol for the evaluation of blood pressure measuring devices. J. Hypertens. 1993, 11, 677–679. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.W.; Chen, L.Y.; Dai, H.L.; Chen, J.H.; Fang, L.Z. Relationship between alanine aminotransferase levels and metabolic syndrome in nonalcoholic fatty liver disease. J. Zhejiang Univ. Sci. B 2008, 9, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Culleton, B.F.; Larson, M.G.; Kannel, W.B.; Levy, D. Serum uric acid and risk for cardiovascular disease and death: The Framingham Heart Study. Ann. Intern. Med. 1999, 131, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Alderman, M.H. Serum uric acid and cardiovascular mortality the NHANES I epidemiologic follow-up study, 1971–1992. National Health and Nutrition Examination Survey. JAMA 2000, 283, 2404–2410. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Yu, C.; Xu, L.; Miao, M.; Li, Y. High serum uric acid increases the risk for nonalcoholic Fatty liver disease: A prospective observational study. PLoS ONE 2010, 5, e11578. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.C.; Huang, Y.F.; Wang, J.D. Hyperferritinemia and hyperuricemia may be associated with liver function abnormality in obese adolescents. PLoS ONE 2012, 7, e48645. [Google Scholar] [CrossRef] [PubMed]
- Afzali, A.; Weiss, N.S.; Boyko, E.J.; Ioannou, G.N. Association between serum uric acid level and chronic liver disease in the United States. Hepatology (Baltimore MD) 2010, 52, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Niskanen, L.K.; Laaksonen, D.E.; Nyyssonen, K.; Alfthan, G.; Lakka, H.M.; Lakka, T.A.; Salonen, J.T. Uric acid level as a risk factor for cardiovascular and all-cause mortality in middle-aged men: A prospective cohort study. Arch. Intern. Med. 2004, 164, 1546–1551. [Google Scholar] [CrossRef] [PubMed]
- Lehto, S.; Niskanen, L.; Ronnemaa, T.; Laakso, M. Serum uric acid is a strong predictor of stroke in patients with non-insulin-dependent diabetes mellitus. Stroke J. Cereb. Circ. 1998, 29, 635–639. [Google Scholar] [CrossRef]
- Zhang, J.X.; Zhang, Y.P.; Wu, Q.N.; Chen, B. Uric acid induces oxidative stress via an activation of the renin-angiotensin system in 3T3-L1 adipocytes. Endocrine 2015, 48, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N.; Cathcart, R.; Schwiers, E.; Hochstein, P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: A hypothesis. Proc. Natl. Acad. Sci. USA 1981, 78, 6858–6862. [Google Scholar] [CrossRef] [PubMed]
- Glantzounis, G.K.; Tsimoyiannis, E.C.; Kappas, A.M.; Galaris, D.A. Uric acid and oxidative stress. Curr. Pharm. Des. 2005, 11, 4145–4151. [Google Scholar] [CrossRef] [PubMed]
- Sautin, Y.Y.; Nakagawa, T.; Zharikov, S.; Johnson, R.J. Adverse effects of the classic antioxidant uric acid in adipocytes: NADPH oxidase-mediated oxidative/nitrosative stress. Am. J. Physiol. Cell Physiol. 2007, 293, C584–C596. [Google Scholar] [CrossRef] [PubMed]
- Marchesini, G.; Brizi, M.; Morselli-Labate, A.M.; Bianchi, G.; Bugianesi, E.; McCullough, A.J.; Forlani, G.; Melchionda, N. Association of nonalcoholic fatty liver disease with insulin resistance. Am. J. Med. 1999, 107, 450–455. [Google Scholar] [CrossRef]
- Angelico, F.; del Ben, M.; Conti, R.; Francioso, S.; Feole, K.; Fiorello, S.; Cavallo, M.G.; Zalunardo, B.; Lirussi, F.; Alessandri, C.; et al. Insulin resistance, the metabolic syndrome, and nonalcoholic fatty liver disease. J. Clin. Endocrinol. Metab. 2005, 90, 1578–1582. [Google Scholar] [CrossRef] [PubMed]
- Quinones Galvan, A.; Natali, A.; Baldi, S.; Frascerra, S.; Sanna, G.; Ciociaro, D.; Ferrannini, E. Effect of insulin on uric acid excretion in humans. Am. J. Physiol. 1995, 268, E1–E5. [Google Scholar] [PubMed]
- Choi, S.S.; Diehl, A.M. Hepatic triglyceride synthesis and nonalcoholic fatty liver disease. Curr. Opin. Lipidol. 2008, 19, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Lanaspa, M.A.; Sanchez-Lozada, L.G.; Choi, Y.J.; Cicerchi, C.; Kanbay, M.; Roncal-Jimenez, C.A.; Ishimoto, T.; Li, N.; Marek, G.; Duranay, M.; et al. Uric acid induces hepatic steatosis by generation of mitochondrial oxidative stress: Potential role in fructose-dependent and -independent fatty liver. J. Biol. Chem. 2012, 287, 40732–40744. [Google Scholar] [CrossRef] [PubMed]
- Salomone, F.; Li Volti, G.; Rosso, C.; Grosso, G.; Bugianesi, E. Unconjugated bilirubin, a potent endogenous antioxidant, is decreased in patients with non-alcoholic steatohepatitis and advanced fibrosis. J. Gastroenterol. Hepatol. 2013, 28, 1202–1208. [Google Scholar] [CrossRef] [PubMed]
| Variables | Quartiles of Normal SUA | Hyperuricemia | p or p for Trend | |||
|---|---|---|---|---|---|---|
| Q1 (n = 1115) | Q2 (n = 1146) | Q3 (n = 1128) | Q4 (n = 1167) | (n = 800) | ||
| SUA, mg/dL | 3.9 ± 0.5 | 4.9 ± 0.2 | 5.5 ± 0.2 | 6.4 ± 0.3 | 8.0 ± 1.0 | <0.001 * |
| ALT, U/L | 22.7 ± 18.8 | 23.5 ± 17.9 | 24.4 ± 18.2 | 27.7 ± 31.9 | 31.6 ± 22.3 | <0.001 * |
| Age, year | 56.1 ± 10.4 | 55.0 ± 10.7 | 53.9 ± 10.8 | 53.4 ± 10.8 | 53.0 ± 11.0 | <0.001 * |
| Race(Han), % | 1051 (94.3) | 1079 (94.2) | 1070 (94.9) | 1104 (94.6) | 766 (95.8) | 0.573 |
| Current smokers, % | 662 (59.4) | 677 (59.1) | 649 (57.5) | 656 (56.2) | 411 (51.4) | 0.004 # |
| Current drinkers, % | 434 (38.9) | 499 (43.5) | 501 (44.4) | 595 (51.0) | 404 (50.5) | <0.001 * |
| High school or above, % | 97 (8.7) | 128 (11.2) | 136 (12.1) | 143 (12.3) | 109 (13.6) | 0.006 # |
| Physical activity, % | 0.015 # | |||||
| High | 59 (5.3) | 72 (6.3) | 67 (5.9) | 67 (5.7) | 32 (4.0) | |
| Moderate | 796 (71.4) | 844 (73.6) | 810 (71.8) | 851 (72.9) | 550 (68.8) | |
| Low | 260 (23.3) | 230 (21.1) | 251 (22.3) | 249 (21.3) | 218 (27.3) | |
| Family income > 20,000 CNY/y, % | 314 (28.2) | 354 (30.9) | 388 (34.4) | 434 (37.2) | 277 (34.6) | <0.001 * |
| History of CVD, % | 124 (11.4) | 94 (8.4) | 113 (10.3) | 113 (9.8) | 114 (14.5) | 0.001 # |
| SBP, mm Hg | 140.2 ± 22.4 | 140.7 ± 22.9 | 141.6 ± 22.2 | 141.8 ± 22.2 | 143.7 ± 22.5 | 0.004 # |
| DBP, mm Hg | 81.2 ± 11.1 | 81.9 ± 11.9 | 82.4 ± 11.9 | 83.9 ± 12.2 | 86.4 ± 12.8 | <0.001 * |
| WC, cm | 80.5 ± 9.3 | 82.1 ± 9.1 | 83.5 ± 9.4 | 85.5 ± 9.5 | 88.9 ± 9.8 | <0.001 * |
| BMI, kg/m2 | 23.8 ± 3.3 | 24.2 ± 3.3 | 24.6 ± 3.5 | 25.2 ± 3.4 | 26.3 ± 3.8 | <0.001 * |
| TC, mmol/L | 5.0 ± 0.9 | 5.1 ± 1.0 | 5.1 ± 1.0 | 5.2 ± 1.0 | 5.4 ± 1.1 | <0.001 * |
| TG, mmol/L | 1.2 ± 0.9 | 1.4 ± 1.1 | 1.6 ± 1.5 | 1.8 ± 1.8 | 2.4 ± 2.4 | <0.001 * |
| HDL-C, mmol/L | 1.5 ± 0.5 | 1.4 ± 0.4 | 1.4 ± 0.4 | 1.3 ± 0.4 | 1.1 ± 0.2 | <0.001 * |
| LDL-C, mmol/L | 2.8 ± 0.8 | 2.8 ± 0.8 | 2.9 ± 0.8 | 3.0 ± 0.8 | 3.2 ± 0.8 | <0.001 * |
| FPG, mmol/L | 5.9 ± 1.4 | 5.9 ± 1.5 | 5.9 ± 1.5 | 6.0 ± 1.4 | 6.1 ± 2.3 | 0.001 # |
| Variables | Quartiles of Normal SUA | Hyperuricemia | p or p for Trend | |||
|---|---|---|---|---|---|---|
| Q1 (n = 1412) | Q2 (n = 1439) | Q3 (n = 1472) | Q4 (n = 1442) | (n = 451) | ||
| SUA, mg/dL | 3.0 ± 0.4 | 3.8 ± 0.2 | 4.4 ± 0.2 | 5.3 ± 0.3 | 6.8 ± 0.9 | <0.001 * |
| ALT, U/L | 17.7 ± 10.0 | 18.2 ± 11.7 | 20.2 ± 13.5 | 21.7 ± 15.4 | 23.0 ± 14.3 | <0.001 * |
| Age, year | 51.1 ± 9.8 | 52.2 ± 10.0 | 53.7 ± 10.3 | 55.1 ± 10.4 | 57.7 ± 10.5 | <0.001 * |
| Race(Han), % | 1312 (92.9) | 1363 (94.7) | 1396 (94.8) | 1386 (96.1) | 440 (97.6) | <0.001 * |
| Current smokers, % | 224 (15.9) | 225 (15.6) | 247 (16.8) | 255 (17.7) | 79 (17.5) | 0.549 |
| Current drinkers, % | 37 (2.6) | 42 (2.9) | 45 (3.1) | 42 (2.9) | 17 (3.8) | 0.797 |
| High school or above, % | 101 (7.2) | 126 (8.8) | 105 (7.1) | 113 (7.8) | 35 (7.8) | <0.001 * |
| Physical activity, % | <0.001 * | |||||
| High | 56 (4.0) | 85 (5.9) | 85 (5.8) | 98 (6.8) | 32 (7.1) | |
| Moderate | 890 (63.0) | 874 (60.7) | 842 (57.2) | 793 (55.0) | 229 (50.8) | |
| Low | 466 (33.0) | 480 (33.4) | 545 (37.0) | 551 (38.2) | 190 (42.1) | |
| Family income > 20,000 CNY/y, % | 466 (33.0) | 488 (33.9) | 497 (33.8) | 472 (32.7) | 136 (30.2) | 0.210 |
| History of CVD, % | 211 (15.2) | 234 (16.5) | 274 (19.0) | 300 (21.3) | 135 (30.7) | <0.001 * |
| SBP, mm Hg | 135.3 ± 23.7 | 134.9 ± 23.2 | 136.9 ± 23.3 | 140.5 ± 24.6 | 143.7 ± 24.4 | 0.202 |
| DBP, mm Hg | 78.0 ± 11.4 | 78.6 ± 11.5 | 79.6 ± 11.5 | 81.5 ± 12.0 | 82.9 ± 12.5 | <0.001 * |
| WC, cm | 78.1 ± 9.1 | 79.4 ± 9.1 | 81.4 ± 9.6 | 84.3 ± 9.3 | 87.1 ± 9.9 | <0.001 * |
| BMI, kg/m2 | 23.9 ± 3.7 | 24.3 ± 3.6 | 24.8 ± 3.7 | 25.9 ± 3.7 | 26.6 ± 3.9 | <0.001 * |
| TC, mmol/L | 5.0 ± 1.1 | 5.2 ± 1.1 | 5.3 ± 1.1 | 5.5 ± 1.1 | 5.8 ± 1.4 | <0.001 * |
| TG, mmol/L | 1.2 ± 0.7 | 1.4 ± 1.0 | 1.6 ± 1.3 | 1.9 ± 1.5 | 2.5 ± 2.2 | <0.001 * |
| HDL-C, mmol/L | 1.5 ± 0.4 | 1.4 ± 0.3 | 1.4 ± 0.3 | 1.3 ± 0.3 | 1.3 ± 0.3 | <0.001 * |
| LDL-C, mmol/L | 2.8 ± 0.8 | 2.9 ± 0.8 | 3.0 ± 0.8 | 3.1 ± 0.8 | 3.3 ± 0.9 | <0.001 * |
| FPG, mmol/L | 5.8 ± 1.8 | 5.8 ± 1.7 | 5.8 ± 1.4 | 5.9 ± 1.5 | 6.2 ± 1.6 | <0.001 * |
| SUA | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|
| Males | OR | 95% CI | p-Value | OR | 95% CI | p-Value |
| Quartile 1 (<4.5 mg/dL) | 1 | – | – | 1 | – | – |
| Quartile 2 (4.5–5.2 mg/dL) | 1.101 | 0.795–1.525 | 0.563 | 0.995 | 0.708–1.398 | 0.976 |
| Quartile 3 (5.2–5.9 mg/dL) | 1.145 | 0.830–1.580 | 0.410 | 1.012 | 0.722–1.417 | 0.947 |
| Quartile 4 (5.9–7.0 mg/dL) | 1.865 | 1.387–2.508 | <0.001 * | 1.467 | 1.063–2.025 | 0.020 # |
| Hyperuricemia (≥7.0 mg/dL) | 3.236 | 2.405–4.355 | <0.001 * | 2.032 | 1.443–2.861 | <0.001 * |
| Females | ||||||
| Quartile 1 (<3.5 mg/dL) | 1 | – | – | 1 | – | – |
| Quartile 2 (3.5–4.1 mg/dL) | 0.963 | 0.616–1.507 | 0.871 | 0.837 | 0.530–1.321 | 0.445 |
| Quartile 3 (4.1–4.7 mg/dL) | 1.906 | 1.289–2.818 | 0.001 | 1.375 | 0.910–2.076 | 0.130 |
| Quartile 4 (4.7–6.0) mg/dL) | 2.608 | 1.788–3.805 | <0.001 * | 1.721 | 1.146–2.585 | 0.009 # |
| Hyperuricemia (≥6.0 mg/dL) | 3.809 | 2.426–5.980 | <0.001 * | 2.045 | 1.221–3.425 | 0.007 # |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Guo, X.; Yu, S.; Sun, G.; Yang, H.; Li, Z.; Sun, Y. Association between Serum Uric Acid and Elevated Alanine Aminotransferase in the General Population. Int. J. Environ. Res. Public Health 2016, 13, 841. https://doi.org/10.3390/ijerph13090841
Chen S, Guo X, Yu S, Sun G, Yang H, Li Z, Sun Y. Association between Serum Uric Acid and Elevated Alanine Aminotransferase in the General Population. International Journal of Environmental Research and Public Health. 2016; 13(9):841. https://doi.org/10.3390/ijerph13090841
Chicago/Turabian StyleChen, Shuang, Xiaofan Guo, Shasha Yu, Guozhe Sun, Hongmei Yang, Zhao Li, and Yingxian Sun. 2016. "Association between Serum Uric Acid and Elevated Alanine Aminotransferase in the General Population" International Journal of Environmental Research and Public Health 13, no. 9: 841. https://doi.org/10.3390/ijerph13090841
APA StyleChen, S., Guo, X., Yu, S., Sun, G., Yang, H., Li, Z., & Sun, Y. (2016). Association between Serum Uric Acid and Elevated Alanine Aminotransferase in the General Population. International Journal of Environmental Research and Public Health, 13(9), 841. https://doi.org/10.3390/ijerph13090841






