Quantitative Microbial Risk Assessment in Occupational Settings Applied to the Airborne Human Adenovirus Infection
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Settings
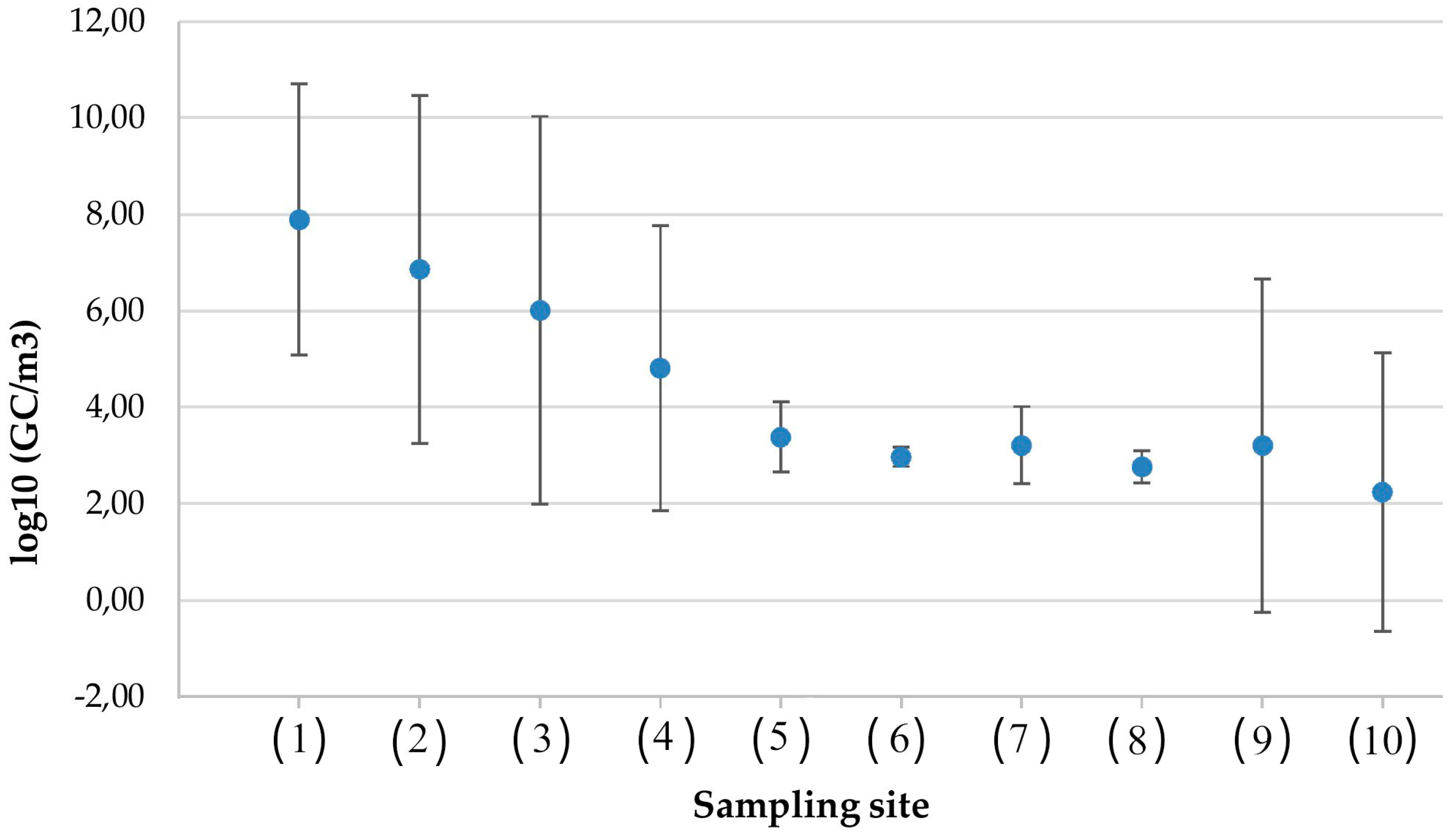
- Toilets in healthcare settings and offices. The airborne spreading of HAdV can occur through the aerosol and droplets produced by toilet flushing, contaminating the surrounding environment. The environmental monitoring of air and selected surfaces was carried out with five replicate sampling sessions, in three toilets in a hospital ward (i.e., one toilet for a four-bed patient room, one toilet for a two-bed patient room, and one toilet for healthcare personnel) and two toilets in an office building, for a total of 43 samples [7].
- Wastewater treatment plants (unpublished data). Aerosol produced by public sewage treatment plants may contain HAdVs that—because of their high stability under environmental conditions and likely transmission by the aerosol route—constitute a potential health hazard for plant workers and nearby residents. During a monitoring aimed to evidence the airborne contamination of 20 wastewater treatment plants, twenty-five samples were collected in areas that are at greatest risk of bioaerosol production: (i) entrance sewage as entry point of wastewater treatment plant; (ii) sludge treatment systems; (iii) biological oxidation tank; and (iv) side-entrance manhole.
- Solid waste landfill. The workers involved in the management of solid waste are at risk of exposure to bioaerosol which is a mixture of particles of biological origin or with biological effects such as bacteria, fungi, and viruses. To evaluate the presence of HAdV, sixteen samples were collected from eight sampling sites that were chosen based on their relevance to worker exposure [18]. Four of these sites were in the recycling paper area, and four were in an outside area.
2.2. Sampling Procedures and Virological Analysis
2.3. Statistical Analysis and Simulation Tool
2.4. QMRA Framework
2.4.1. Hazard Identification
2.4.2. Exposure Assessment
2.4.3. Dose-Response Relationship
2.4.4. Risk Characterization
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| QMRA | Quantitative Microbial Risk Assessment |
| HAdV | Human Adenovirus |
References
- Corrao, C.R.N.; Mazzotta, A.; La Torre, G.; De Giusti, M. Biological risk and occupational health. Ind. Health 2012, 50, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Reponen, T. Methodologies for assessing bioaerosol exposures. Encylopedia Environ. Health 2011, 3, 722–730. [Google Scholar]
- Douwes, J.; Thorne, P.; Pearce, N.; Heederik, D. Bioaerosol health effects and exposure assessment: Progress and prospects. Ann. Occup. Hyg. 2003, 47, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Eduard, W.; Heederik, D.; Duchaine, C.; Green, B.J. Bioaerosol exposure assessment in the workplace: The past, present and recent advances. J. Environ. Monit. 2012, 14, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Napoli, C.; Marcotrigiano, V.; Montagna, M.T. Air sampling procedures to evaluate microbial contamination: A comparison between active and passive methods in operating theatres. BMC Public Health 2012, 12, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gerba, C.P.; Goyal, S.M.; LaBelle, R.; Cech, I.; Bodgan, G.F. Failure of indicator bacteria to reflect the occurrence of enteroviruses in marine waters. Am. J. Public Health 1979, 69, 1116–1119. [Google Scholar] [CrossRef] [PubMed]
- Verani, M.; Bigazzi, R.; Carducci, A. Viral contamination of aerosol and surfaces through toilet use in health care and other settings. Am. J. Infect. Control 2014, 42, 758–762. [Google Scholar] [CrossRef] [PubMed]
- Górny, R.L.; Cyprowski, M.; Ławniczek-Wałczyk, A.; Gołofit-Szymczak, M.; Zapór, L. Biohazards in the indoor environment—A role for threshold limit values in exposure assessment. In Management of Indoor Air Quality; Dudzińska, M.R., Ed.; CRC Press/Balkema: Boca Raton, FL, USA, 2011; pp. 1–20. [Google Scholar]
- Bichai, F.; Smeets, P.W. Using QMRA-based regulation as a water quality management tool in the water security challenge: Experience from The Netherlands and Australia. Water Res. 2013, 47, 7315–7326. [Google Scholar] [CrossRef] [PubMed]
- Havelaar, A. Recommendations for Addressing Quantitative Microbiological Risk Assessment at the European Level. External Scientific Report 2005. Available online: www.efsa.europa.eu/en/af060303/docs/af060303-ax2.pdf (accessed on 7 August 2015).
- Romero-Barrios, P.; Hempen, M.; Messens, W.; Stella, P.; Hugas, M. Quantitative microbiological risk assessment (QMRA) of food-borne zoonoses at the European level. Food Control 2013, 29, 343–349. [Google Scholar] [CrossRef]
- Westrell, T.; Schonning, C.; Stenstrom, T.A.; Ashbolt, N.J. QMRA (quantitative microbial risk assessment) and HACCP (hazard analysis and critical control points) for management of pathogens in wastewater and sewage sludge treatment and reuse. Water Sci. Technol. 2004, 50, 23–30. [Google Scholar] [PubMed]
- Haas, C.N.; Rose, J.B.; Gerba, C.P. Quantitative Microbial Risk Assessment; John Wiley & Sons, Inc.: New York, NY, USA, 1999. [Google Scholar]
- National Academy Press (NRC). Risk Assessment in the Federal Government: Managing the Process; NRC: Washington, DC, USA, 1983. [Google Scholar]
- The NRC Risk Assessment Paradigm. Available online: http://www2.epa.gov/fera/nrc-risk-assessment-paradigm (accessed on 23 November 2015).
- Mackay, I.M.; Arden, K.E.; Nitsche, A. Real-time PCR in virology. Nucleic Acids Res. 2002, 30, 1292–1305. [Google Scholar] [CrossRef] [PubMed]
- Carducci, A.; Battistini, R.; Rovini, E.; Verani, M. Viral removal by wastewater treatment: Monitoring of indicators and pathogens. Food Environ. Virol. 2009, 1, 85–91. [Google Scholar] [CrossRef]
- Carducci, A.; Federigi, I.; Verani, M. Virus occupational exposure in solid waste processing facilities. Ann. Occup. Hyg. 2013, 57, 1115–1127. [Google Scholar] [CrossRef] [PubMed]
- Albinana-Gimenez, N.; Miagostovich, M.P.; Calgua, B.; Huguet, J.M.; Matia, L.; Girones, R. Analysis of adenoviruses and polyomaviruses quantified by qPCR as indicators of water quality in source and drinking-water treatment plants. Water Res. 2009, 43, 2011–2019. [Google Scholar] [CrossRef] [PubMed]
- Wyer, M.D.; Wyn-Jones, A.P.; Kay, D.; Au-Yeung, H.K.C.; Girones, R.; Lopez-Pila, J.; de Roda Husman, A.M.; Rutjes, S.; Schneider, O. Relationships between human adenoviruses and faecal indicator organisms in European recreational waters. Water Res. 2012, 46, 4130–4141. [Google Scholar] [CrossRef] [PubMed]
- Kundu, A.; McBride, G.; Wuertz, S. Adenovirus-associated health risks for recreational activities in a multi-use coastal watershed based on site-specific quantitative microbial risk assessment. Water Res. 2013, 47, 6309–6325. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, J.; Greening, G.E.; Leonard, M.; Lewis, G.D. Evaluation of human adenovirus and human polyomavirus as indicators of human sewage contamination in the aquatic environment. Water Res. 2013, 47, 6750–6761. [Google Scholar] [CrossRef] [PubMed]
- Couch, R.B.; Cate, T.R.; Douglas, R.G., Jr.; Gerone, P.J.; Knight, V. Effect of route of inoculation on experimental respiratory viral disease in volunteers and evidence for airborne transmission. Bacteriol. Rev. 1966, 30, 517–529. [Google Scholar] [PubMed]
- Carducci, A.; Arrighi, S.; Ruschi, A. Detection of coliphages and enteroviruses in sewage and aerosol from an activated sludge wastewater treatment plant. Lett. Appl. Microbiol. 1995, 21, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Bofill-Mas, S.; Calgua, B.; Clemente-Casares, P.; La Rosa, G.; Iaconelli, M.; Muscillo, M.; Rutjes, S.; Husman, A.M.D.; Grunert, A.; Graber, I.; et al. Quantification of human adenoviruses in European recreational waters. Food Environ. Virol. 2010, 2, 101–109. [Google Scholar] [CrossRef]
- Nordgren, J.; Matussek, A.; Mattsson, A.; Svensson, L.; Lindgren, P.E. Prevalence of norovirus and factors influencing virus concentrations during one year in a full-scale wastewater treatment plant. Water Res. 2009, 43, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Ventana Systems Inc. Vensim PLE User’s Guide, 6th ed.; Ventana Systems Inc.: Cambridge, MA, USA, 2012. [Google Scholar]
- Cioni, L. Introduction to System Dynamics; Technical Report TR-10-09; Computer Science Department, University of Pisa: Pisa, Italy, 2010; Available online: http://compass2.di.unipi.it/TR/Files/TR-10-09.ps.gz (accessed on 10 August 2015).
- Forrester, J.W. Principles of Systems; MIT Press: Cambridge, MA, USA, 1968. [Google Scholar]
- Richmond, B. An Introduction to Systems Thinking. High Performance Systems, Inc., 2005. Available online: http://www.fi.muni.cz/~xpelanek/IV109/jaro07/IST.pdf (accessed on 10 August 2015).
- Sterman, J.D. System Dynamics Modeling Tools for Learning in a Complex World. 2001. Available online: http://www.systemdynamics.org/conferences/2002/proceed/papers/CAREVIEW/C2STERMA.PDF (accessed on 10 August 2015).
- Wolstenholme, E.F. System Enquiry: A System Dynamics Approach; John Wiley & Sons, Inc.: New York, NY, USA, 1990. [Google Scholar]
- Haas, C.; Eisenberg, J.N.S. Risk assessment. In Water Quality Guidelines, Standards and Health: Assessment of Risk and Risk Management for Water-Related Infectious Disease; Fewtrell, L., Bartram, J., Eds.; IWA Publishing: London, UK, 2001; pp. 161–184. [Google Scholar]
- U.S. Environmental Protection Agency (EPA). Exposure Factors Handbook, 2011 ed.EPA/600/R-09/052F; National Center for Environmental Assessment: Washington, DC, USA, 2011.
- Bae, H.G.; Nitsche, A.; Teichmann, A.; Biel, S.S.; Niedrig, M. Detection of yellow fever virus: A comparison of quantitative real-time PCR and plaque assay. J. Virol. Methods 2003, 110, 185–191. [Google Scholar] [CrossRef]
- Donia, D.; Bonanni, E.; Diaco, L.; Divizia, M. Statistical correlation between enterovirus genome copy numbers and infectious viral particles in wastewater samples. Lett. Appl. Microbiol. 2010, 50, 237–240. [Google Scholar] [CrossRef] [PubMed]
- McBride, G.B.; Stott, R.; Miller, W.; Bambic, D.; Wuertz, S. Discharge-based QMRA for estimation of public health risks from exposure to stormwater-borne pathogens in recreational waters in the United States. Water Res. 2013, 47, 5282–5297. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.O.; Haas, C.N.; Gurian, P.L.; Gerba, C.P.; Panzl, B.M.; Rose, J.B. Application of quantitative microbial risk assessment for selection of microbial reduction targets for hard surface disinfectants. Am. J. Infect. Control 2014, 42, 1165–1172. [Google Scholar] [CrossRef] [PubMed]
- Azuma, K.; Uchiyama, I.; Okumura, J. Assessing the risk of Legionnaires’ disease: The inhalation exposure model and the estimated risk in residential bathrooms. Regul. Toxicol. Pharmacol. 2013, 65, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Buse, H.Y.; Schoen, M.E.; Ashbolt, N.J. Legionellae in engineered systems and use of quantitative microbial risk assessment to predict exposure. Water Res. 2012, 46, 921–933. [Google Scholar] [CrossRef] [PubMed]
- De Man, H.; Bouwknegt, M.; van Heijnsbergen, E.; Leenen, E.J.; van Knapen, F.; de Roda Husman, A.M. Health risk assessment for splash parks that use rainwater as source water. Water Res. 2014, 54, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Stellacci, P.; Liberti, L.; Notarnicola, M.; Haas, C.N. Hygienic sustainability of site location of wastewater treatment plants: A case study. II. Estimating airborne biological hazard. Desalination 2010, 253, 106–111. [Google Scholar] [CrossRef]
- Jahne, M.A.; Rogers, S.W.; Holsen, T.M.; Grimberg, S.J. Quantitative microbial risk assessment of bioaerosols from a manure application site. Aerobiologia 2015, 31, 73–87. [Google Scholar] [CrossRef]
- Lim, K.Y.; Hamilton, A.J.; Jiang, S.C. Assessment of public health risk associated with viral contamination in harvested urban stormwater for domestic applications. Sci. Total Environ. 2015, 523, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Park, D.U.; Ryu, S.H.; Kim, S.B.; Yoon, C.S. An assessment of dust, endotoxin, and microorganism exposure during waste collection and sorting. J. Air Waste Manag. Assoc. 2011, 61, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.C. Human adenoviruses in water: Occurrence and health implications: A critical review. Environ. Sci. Technol. 2006, 40, 7132–7140. [Google Scholar] [CrossRef] [PubMed]
- Mena, K.D.; Gerba, C.P. Waterborne adenovirus. Rev. Environ. Contam. Toxicol. 2009, 198, 133–167. [Google Scholar] [PubMed]
- Carducci, A.; Verani, M.; Battistini, R.; Pizzi, F.; Rovini, E.; Andreoli, E.; Casini, B. Epidemiological surveillance of human enteric viruses by monitoring of different environmental matrices. Water Sci. Technol. 2006, 54, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Carducci, A.; Morici, P.; Pizzi, F.; Battistini, R.; Rovini, E.; Verani, M. Study of the viral removal efficiency in a urban wastewater treatment plant. Water Sci. Technol. 2008, 58, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Carducci, A.; Verani, M. Effects of bacterial, chemical, physical and meteorological variables on virus removal by a wastewater treatment plant. Food Environ. Virol. 2013, 5, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Korzeniewska, E. Emission of bacteria and fungi in the air from wastewater treatment plants—A review. Front. Biosci. 2011, 1, 393–407. [Google Scholar] [CrossRef]
- Sigari, G.; Panatto, D.; Lai, P.; Stefani, L.; Giuntini, A.; Carducci, A.; Gasparini, R. Virological investigation on aerosol from waste depuration plants. J. Prev. Med. Hyg. 2006, 47, 4–7. [Google Scholar] [PubMed]

| Parameter | Value | Reference |
|---|---|---|
| Recovery efficiency: reff | 40% | [18] |
| Single parameter-model for HAdV: r | 0.4172 | [33] 1 |
| Conversion factor: fconv | 10−3 PFU/GC | [37] 2 |
| Average inhalation rate: rin | 1.2 m3/h | [34] 3 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carducci, A.; Donzelli, G.; Cioni, L.; Verani, M. Quantitative Microbial Risk Assessment in Occupational Settings Applied to the Airborne Human Adenovirus Infection. Int. J. Environ. Res. Public Health 2016, 13, 733. https://doi.org/10.3390/ijerph13070733
Carducci A, Donzelli G, Cioni L, Verani M. Quantitative Microbial Risk Assessment in Occupational Settings Applied to the Airborne Human Adenovirus Infection. International Journal of Environmental Research and Public Health. 2016; 13(7):733. https://doi.org/10.3390/ijerph13070733
Chicago/Turabian StyleCarducci, Annalaura, Gabriele Donzelli, Lorenzo Cioni, and Marco Verani. 2016. "Quantitative Microbial Risk Assessment in Occupational Settings Applied to the Airborne Human Adenovirus Infection" International Journal of Environmental Research and Public Health 13, no. 7: 733. https://doi.org/10.3390/ijerph13070733
APA StyleCarducci, A., Donzelli, G., Cioni, L., & Verani, M. (2016). Quantitative Microbial Risk Assessment in Occupational Settings Applied to the Airborne Human Adenovirus Infection. International Journal of Environmental Research and Public Health, 13(7), 733. https://doi.org/10.3390/ijerph13070733








