The Relation of Moderate Alcohol Consumption to Hyperuricemia in a Rural General Population
Abstract
:1. Introduction
2. Methods
2.1. Study Population
2.2. Data Collection and Measurements
2.3. Alcohol Consumption Assessment
2.4. Definitions
2.5. Statistical Analysis
3. Result
3.1. Baseline Characteristics of the Study Population by Gender
3.2. Levels of Alcohol Consumption and Serum Uric Acid
3.3. Association Between Alcohol Consumption and Hyperuricemia by Gender
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Robinson, P.C.; Taylor, W.J.; Merriman, T.R. Systematic review of the prevalence of gout and hyperuricaemia in Australia. Intern. Med. J. 2012, 42, 997–1007. [Google Scholar] [PubMed]
- Qu, Y.L.; Yu, L.; Li, Z.M.; Kong, L.H.; Xiao, T.C.; Li, M. Epidemiological status and risk factors of hyperuricemia in rural area of the Three Gorges. Chin. J. Cardiol. 2013, 41, 427–431. (In Chinese) [Google Scholar]
- Zhang, X.; Liu, L.; Liang, R.; Jin, S. Hyperuricemia is a biomarker of early mortality in patients with chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2015, 10, 2519–2523. [Google Scholar] [PubMed]
- Bobak, M.; Malyutina, S.; Horvat, P.; Pajak, A.; Tamosiunas, A.; Kubinova, R.; Marmot, M.G. Alcohol, drinking pattern and all-cause, cardiovascular and alcohol-related mortality in Eastern Europe. Eur. J. Epidemiol. 2016, 31, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Braga, F.; Pasqualetti, S.; Ferraro, S.; Panteghini, M. Hyperuricemia as risk factor for coronary heart disease incidence and mortality in the general population: A systematic review and meta-analysis. Clin. Chem. Lab. Med. 2016, 54, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.M.; Hsu, P.F.; Cheng, H.M.; Lu, D.Y.; Cheng, Y.L.; Guo, C.Y.; Chen, C.H. Determinants and prognostic impact of hyperuricemia in hospitalization for acute heart failure. Circ. J. 2016, 80, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.K.; Atkinson, K.; Karlson, E.W.; Willett, W.; Curhan, G. Alcohol intake and risk of incident gout in men: A prospective study. Lancet. 2004, 363, 1277–1281. [Google Scholar] [CrossRef]
- Bae, J.; Park, P.S.; Chun, B.Y.; Choi, B.Y.; Kim, M.K.; Shin, M.H.; Kim, S.K. The effect of coffee, tea, and caffeine consumption on serum uric acid and the risk of hyperuricemia in Korean Multi-Rural Communities Cohort. Rheumatol. Int. 2015, 35, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Markus, M.R.P.; Lieb, W.; Stritzke, J.; Siewert, U.; Troitzsch, P.; Koch, M.; Baumeister, S.E. Light to moderate alcohol consumption is associated with lower risk of aortic valve sclerosis: The study of health in pomerania (SHIP). Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.Y.; Sun, C.C.; Wei, J.C.C.; Tai, H.C.; Sun, C.A.; Chung, C.F.; Yang, T. Association between hyperuricemia and metabolic syndrome: An epidemiological study of a labor force population in Taiwan. BioMed Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Han, C.; Wu, D.; Xia, X.; Gu, J.; Guan, H.; Teng, W. Prevalence of hyperuricemia and gout in mainland China from 2000 to 2014: A systematic review and meta-analysis. BioMed Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Alatalo, P.I.; Koivisto, H.M.; Hietala, J.P.; Bloigu, R.S.; Niemelä, O.J. Gender-dependent impacts of body mass index and moderate alcohol consumption on serum uric acid—An index of oxidant stress status? Free Radical Biol. Med. 2009, 46, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.K.; Curhan, G. Beer, liquor, and wine consumption and serum uric acid level: The Third National Health and Nutrition Examination Survey. Arthritis Rheum. 2004, 51, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Guo, X.; Yang, H.; Zheng, L.; Sun, Y. An update on the prevalence of metabolic syndrome and its associated factors in rural northeast China. BMC Public Health 2014. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Li, Z.; Guo, L.; Zheng, L.; Yu, S.; Yang, H.; Zhang, Y. An update on overweight and obesity in rural Northeast China: From lifestyle risk factors to cardiometabolic comorbidities. BMC Public Health 2014. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.; Ma, M.; Ye, N.; Wang, J.; Chen, Y.; Dai, D.; Sun, Y. Diabetes mellitus is an independent risk factor for atrial fibrillation in a general Chinese population. J. Diabetes Investig. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.Z.; Li, Z.; Guo, L.; Zhou, Y.; Yang, H.M.; Sun, Y.X. High prevalence of dyslipidemia and associated risk factors among rural Chinese adults. Lipids Health Dis. 2014. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Shi, R.; Hou, B.; Wu, L.; Zheng, L.; Ding, L.; Yao, Y. Impact of alcohol consumption on substrate remodeling and ablation outcome of paroxysmal atrial fibrillation. J. Am. Heart Assoc. 2015. [Google Scholar] [CrossRef] [PubMed]
- National Institute on Alcohol Abuse and Alcoholism. The Physicians’ Guide to Helping Patients with Alcohol Problem; NIH: Bethesda, MD, USA, 1995.
- Shmerling, R.H. Management of gout: A 57-year-old man with a history of podagra, hyperuricemia, and mild renal insufficiency. JAMA 2012, 308, 2133–2141. [Google Scholar] [CrossRef] [PubMed]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L.; Roccella, E.J. Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef] [PubMed]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497. [Google Scholar]
- World Health Organization. Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycaemia; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Kumral, E.; Karaman, B.; Orman, M.; Kabaroglu, C. Association of uric acid and carotid artery disease in patients with ischemic stroke. Acta Neurol. Scand. 2014, 130, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Coresh, J. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Yang, H.; Guo, X.; Zhang, X.; Zhou, Y.; Ou, Q.; Sun, Y. Prevalence of hyperuricemia and its correlates in rural Northeast Chinese population: From lifestyle risk factors to metabolic comorbidities. Clin. Rheumatol. 2016, 35, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Elmadhun, N.Y.; Sadek, A.A.; Sabe, A.A.; Lassaletta, A.D.; Sellke, F.W. Alcohol and the heart: A proteomics analysis of pericardium and myocardium in a swine model of myocardial ischemia. Ann. Thorac. Surg. 2015, 100, 1627–1635. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Lan, J.L.; Cheng, C.F.; Liang, W.M.; Lin, H.Y.; Tsay, G.J.; Pan, W.H. Effect of urate-lowering therapy on all-cause and cardiovascular mortality in hyperuricemic patients without Gout: A case-matched cohort study. PLoS ONE 2015. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Hernandez, A.; Gea, A.; Ruiz-Canela, M.; Toledo, E.; Beunza, J.J.; Bes-Rastrollo, M.; Martinez-Gonzalez, M.A. Mediterranean alcohol-drinking pattern and the incidence of cardiovascular Disease and cardiovascular mortality: The SUN Project. Nutrients 2015, 7, 9116–9126. [Google Scholar] [CrossRef] [PubMed]
- Ofori, S.N.; Odia, O.J. Serum uric acid and target organ damage in essential hypertension. Vasc. Health Risk Manag. 2014, 10, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Nacak, H.; van Diepen, M.; de Goeij, M.C.; Rotmans, J.I.; Dekker, F.W. Uric acid: Association with rate of renal function decline and time until start of dialysis in incident pre-dialysis patients. BMC Nephrol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Sakurai, M.; Miura, K.; Morikawa, Y.; Yoshita, K.; Ishizaki, M.; Nakagawa, H. Alcohol intake and the risk of hyperuricaemia: A 6-year prospective study in Japanese men. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lu, J.; Wu, X.; Yang, C. Serum uric acid concentration and asymptomatic hyperuricemia with subclinical organ damage in general population. Angiology 2014, 65, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Hak, A.E.; Choi, H.K. Menopause, postmenopausal hormone use and serum uric acid levels in US women—The Third National Health and Nutrition Examination Survey. Arthritis Res. Ther. 2008. [Google Scholar] [CrossRef] [PubMed]
- Cook, L.J. Educating women about the hidden dangers of alcohol. J. Psychosoc. Nurs. Ment. Health Serv. 2004, 42, 24–31. [Google Scholar] [PubMed]
- World Health Organization. Diet, Nutrition and the Prevention of Chronic Diseases; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Lee, S.J.; Cho, Y.J.; Kim, J.G.; Ko, Y.; Hong, K.S.; Park, J.M.; Cha, J.K. Moderate alcohol intake reduces risk of ischemic stroke in Korea. Neurology 2015, 85, 1950–1956. [Google Scholar] [CrossRef] [PubMed]
- Tektonidis, T.G.; Åkesson, A.; Gigante, B.; Wolk, A.; Larsson, S.C. A Mediterranean diet and risk of myocardial infarction, heart failure and stroke: A population-based cohort study. Atherosclerosis 2015, 243, 93–98. [Google Scholar] [CrossRef] [PubMed]
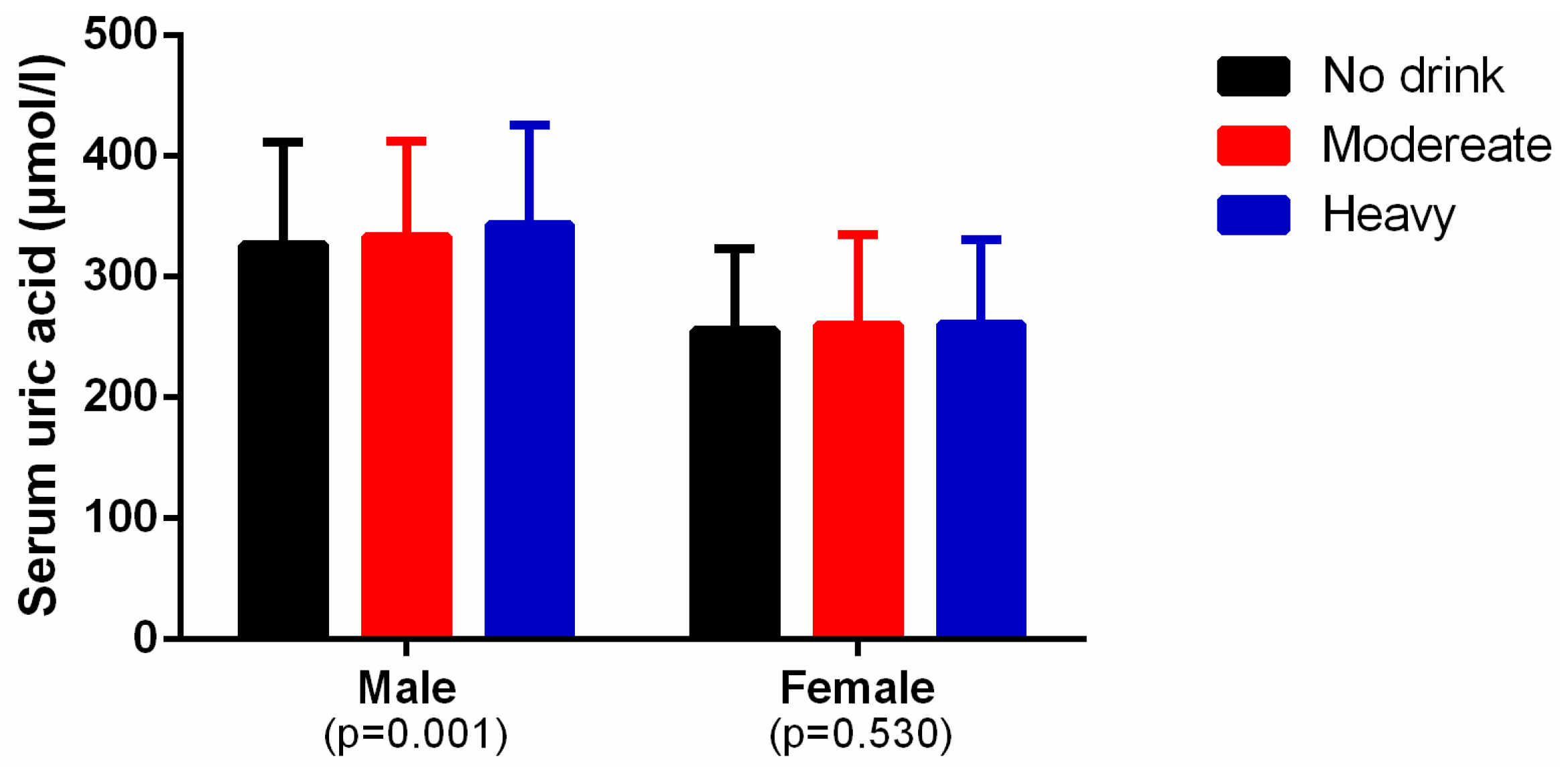

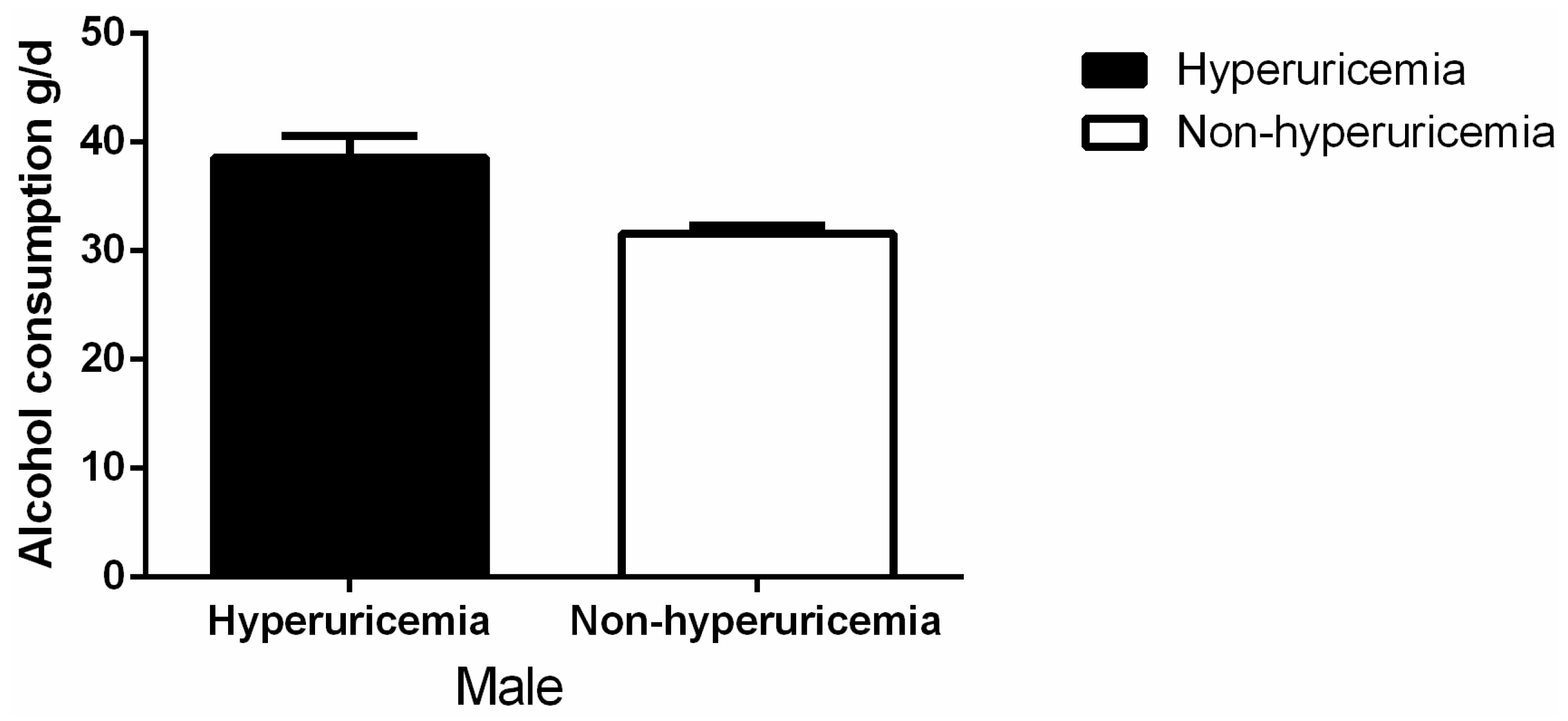
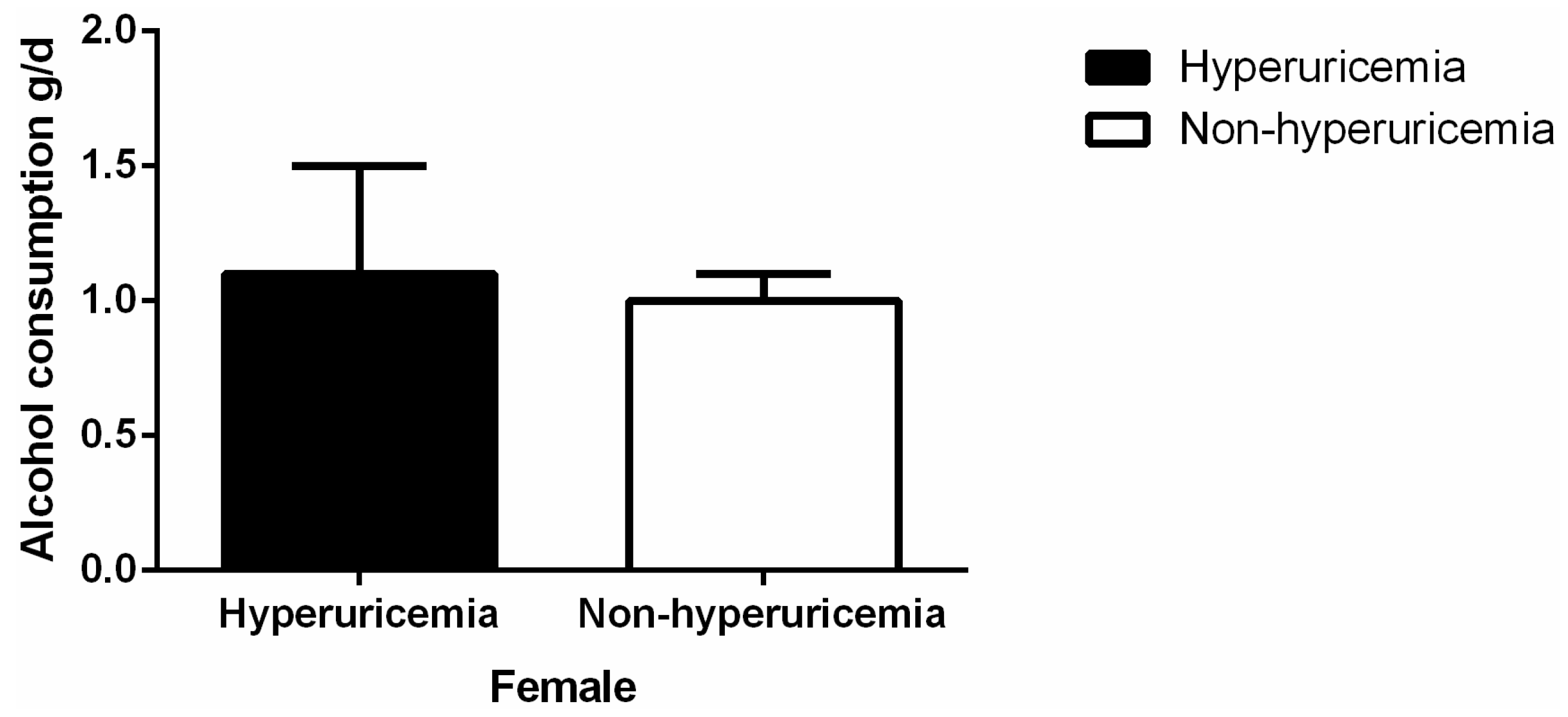

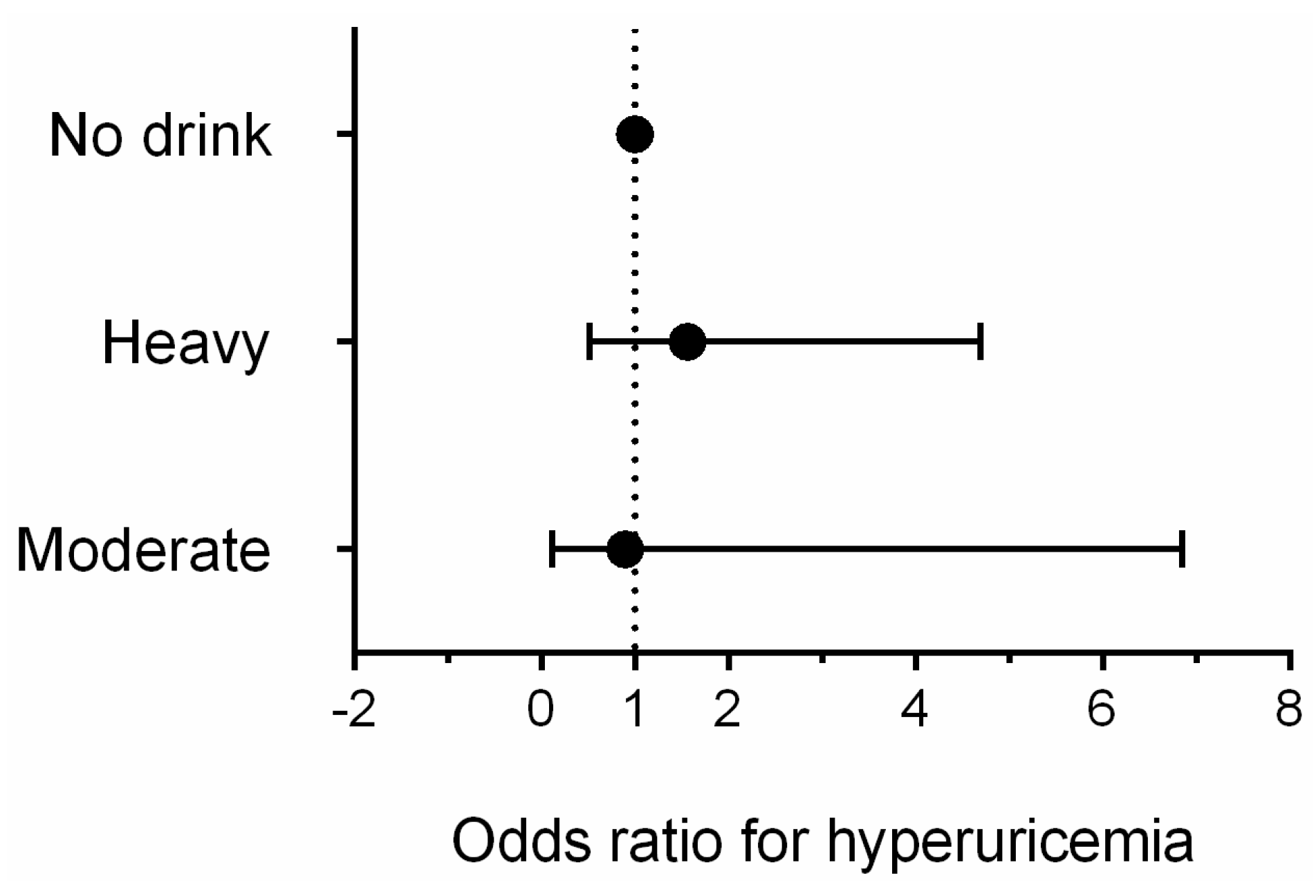
| Variables | Male (4997) | Female (6042) | p-value |
|---|---|---|---|
| Age (year) | 54.4 ± 10.8 | 53.4 ± 10.3 | <0.001 |
| Current drinking | 2244 (44.9%) | 173 (2.9%) | <0.001 |
| Total alcohol consumption (g/d) | 32.6 ± 49.6 | 1.0 ± 8.3 | <0.001 |
| Alcohol consumption from beer (g/d) | 3.9 ± 12.8 | 0.1 ± 2.0 | <0.001 |
| Alcohol consumption from liquor (g/d) | 28.6 ± 46.9 | 0.9 ± 7.7 | <0.001 |
| Alcohol consumption from wine (g/d) | 0.0 ± 1.8 | 0.0 ± 0.0 | <0.001 |
| Alcohol consumption | <0.001 | ||
| No drink | 2524 (50.5%) | 584 7 (96.8%) | |
| Moderate | 751 (15.0%) | 74 (1.2%) | |
| Heavy | 1722 (34.5%) | 121 (2.0%) |
| Variables | Female | Male | ||||
|---|---|---|---|---|---|---|
| Non-Hyperuricemia | Hyperuricemia | p-value | Non-Hyperuricemia | Hyperuricemia | p-value | |
| Age (year) | 53.1 ± 10.2 | 57.8 ± 10.6 | <0.001 | 54.6 ± 10.8 | 53.2 ± 11.1 | 0.002 |
| SBP (mmHg) | 139.7 ± 2.8 | 146.4 ± 25.0 | <0.001 | 143.2 ± 22.6 | 145.6 ± 22.9 | 0.010 |
| DBP(mmHg) | 80.3 ± 11.4 | 83.9 ± 12.7 | <0.001 | 83.1 ± 11.6 | 87.2 ± 12.7 | <0.001 |
| Serum creatinine (mmol/L) | 64.3 ± 11.8 | 82.7 ± 63.1 | <0.001 | 78.1 ± 16.2 | 88.7 ± 30.3 | <0.001 |
| eGFR(mL/min) | 95.2 ± 14.3 | 88.0 ± 19.4 | <0.001 | 92.9 ± 15.4 | 75.6 ± 19.1 | <0.001 |
| Serum uric acid (μmol/L) | 245.1 ± 53.7 | 413.8 ± 54.7 | <0.001 | 309.9 ± 58.3 | 482.9 ± 62.6 | <0.001 |
| FPG (mmol/L) | 5.8 ± 1.6 | 6.2 ± 1.6 | <0.001 | 5.9 ± 1.7 | 6.0 ± 1.5 | 0.705 |
| TC (mmol/L) | 5.3 ± 1.1 | 5.8 ± 1.3 | <0.001 | 5.1 ± 1.0 | 5.5 ± 1.2 | <0.001 |
| TG (mmol/L) | 1.6 ± 1.2 | 2.5 ± 2.2 | <0.001 | 1.5 ± 1.4 | 2.5 ± 2.7 | <0.001 |
| HDL-C (mmol/L) | 1.4 ± 0.3 | 1.3 ± 0.3 | <0.001 | 1.4 ± 0.4 | 1.3 ± 0.4 | <0.001 |
| LDL-C (mmol/L) | 2.9 ± 0.8 | 3.3 ± 0.9 | <0.001 | 2.8 ± 0.8 | 3.0 ± 0.8 | <0.001 |
| Dietscore | 2.1 ± 1.1 | 2.0 ± 1.1 | 0.009 | 2.5 ± 1.1 | 2.6 ± 1.1 | 0.101 |
| BMI(kg/m2) | 24.7 ± 3.7 | 26.7 ± 3.8 | <0.001 | 24.5 ± 3.4 | 26.4 ± 3.9 | <0.001 |
| Race group (han) | 5361 (94.8%) | 374 (97.4%) | 0.010 | 4092 (94.7%) | 646 (95.6%) | 0.200 |
| Education | <0.001 | 0.535 | ||||
| Primary school or below | 3173 (56.1%) | 258 (67.2%) | 1811 (41.9%) | 278 (41.1%) | ||
| Middle school | 2047 (36.2%) | 97 (25.3%) | 2036 (47.1%) | 314 (46.4%) | ||
| High school or above | 438 (7.7%) | 29 (7.6%) | 474 (11.0%) | 84 (12.4%) | ||
| Physical activity | 0.010 | 0.001 | ||||
| Low | 3173 (56.1%) | 374 (97.4%) | 913 (21.1%) | 180 (26.6%) | ||
| Moderate | 2047 (36.2%) | 10 (2.6%) | 3160 (73.1%) | 471 (69.7%) | ||
| High | 438 (7.7%) | 384 (100.0%) | 248 (5.7%) | 25 (3.7%) | ||
| Currently smoking | 929 (16.4%) | 66 (17.2%) | 0.694 | 2504 (57.9%) | 351 (51.9%) | 0.002 |
| Family income (CNY/year) | 0.360 | 0.482 | ||||
| ≤5000 | 646 (11.4%) | 53 (13.8%) | 582 (13.5%) | 90 (13.3%) | ||
| 5000–20,000 | 3135 (55.4%) | 209 (54.4%) | 2349 (54.4%) | 353 (52.2%) | ||
| >20,000 | 1877 (33.2%) | 122 (31.8%) | 1390 (32.2%) | 233 (34.5%) | ||
| Medication used a | 0.004 | <0.001 | ||||
| Yes | 2277 (87.8%) | 317 (12.2%) | 2418 (96.6%) | 85 (3.4%) | ||
| No | 2008 (85.0%) | 355 (15.0%) | 3196 (91.5%) | 295 (8.5%) | ||
| Menopause status | <0.001 | |||||
| Yes | – | – | 3233 (91.9%) | 285 (8.1%) | ||
| No | – | – | 2385 (96.2%) | 94 (3.8%) | ||
| Variables | Male | Female | ||
|---|---|---|---|---|
| p-value | OR (95% CI) | p-value | OR (95% CI) | |
| Race (han) | 0.204 | 0.770 (0.515–1.152) | 0.069 | 0.457 (0.196–1.061) |
| Current smoking | 0.002 | 0.756 (0.634–0.902) | 0.038 | 0.656 (0.440–0.977) |
| Medication use a | 0.022 | 1.224 (1.030–1.454) | <0.001 | 1.734 (1.273–2.362) |
| Age (years) | <0.001 | 0.978 (0.969–0.988) | 0.308 | 1.009 (0.992–1.026) |
| Diabetes | 0.058 | 0.760 (0.572–1.009) | 0.012 | 1.563 (1.105–2.210) |
| Dyslipidemia | <0.001 | 2.167 (1.822–2.577) | <0.001 | 2.630 (1.995–3.468) |
| Obesity | <0.001 | 2.066 (1.569–2.719) | 0.001 | 1.886 (1.303–2.730) |
| Hypertension | 0.004 | 1.315 (1.094–1.581) | 0.023 | 1.422 (1.051–1.924) |
| decreased eGFR | <0.001 | 7.400 (4.581–11.953) | <0.001 | 11.136 (6.873–18.042) |
| Annual income (CNY/year) | ||||
| ≤5000 | 0.864 | 1.000 (reference) | 1.000 (reference) | |
| 5000–20,000 | 0.589 | 0.929 (0.711–1.214) | 0.753 | 1.071 (0.698–1.643) |
| >20,000 | 0.679 | 0.940 (0.703–1.258) | 0.495 | 1.179 (0.735–1.890) |
| Dietscore | 0.354 | 1.038 (0.959–1.123) | 0.619 | 0.970 (0.859–1.095) |
| Physical activity | ||||
| Low | 0.013 | 1.000 (reference) | 1.000 (reference) | |
| Moderate | 0.047 | 0.811 (0.659–0.997) | 0.050 | 1.340 (0.999–1.795) |
| High | 0.007 | 0.535 (0.339–0.845) | 0.991 | 0.996 (0.523–1.899) |
| Education level | ||||
| Primary school or below | 0.733 | 1.000 (reference) | 0.659 | 1.000 (reference) |
| Middle school | 0.442 | 0.927 (0.764–1.124) | 0.425 | 0.874 (0.628–1.217) |
| High school or above | 0.880 | 0.978 (0.732–1.306) | 0.813 | 1.068 (0.621–1.835) |
| Beer (bottles/weeks) | 0.222 | 1.010 (0.994–1.027) | 0.088 | 1.135 (0.981–1.312) |
| Alcohol consumption group | ||||
| No | <0.001 | 1.000 (reference) | 0.979 | 1.000 (reference) |
| Moderate | 0.168 | 1.201 (0.926–1.558 ) | 0.843 | 0.814 (0.106–6.238) |
| Heavy | <0.001 | 1.573 (1.285–1.926) | 0.951 | 0.956 (0.228–4.001) |
| Menopause | -- | 0.595 | 1.083 (0.806–1.456) | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Guo, X.; Liu, Y.; Chang, Y.; Sun, Y.; Zhu, G.; Abraham, M.R. The Relation of Moderate Alcohol Consumption to Hyperuricemia in a Rural General Population. Int. J. Environ. Res. Public Health 2016, 13, 732. https://doi.org/10.3390/ijerph13070732
Li Z, Guo X, Liu Y, Chang Y, Sun Y, Zhu G, Abraham MR. The Relation of Moderate Alcohol Consumption to Hyperuricemia in a Rural General Population. International Journal of Environmental Research and Public Health. 2016; 13(7):732. https://doi.org/10.3390/ijerph13070732
Chicago/Turabian StyleLi, Zhao, Xiaofan Guo, Yamin Liu, Ye Chang, Yingxian Sun, Guangshuo Zhu, and Maria Roselle Abraham. 2016. "The Relation of Moderate Alcohol Consumption to Hyperuricemia in a Rural General Population" International Journal of Environmental Research and Public Health 13, no. 7: 732. https://doi.org/10.3390/ijerph13070732
APA StyleLi, Z., Guo, X., Liu, Y., Chang, Y., Sun, Y., Zhu, G., & Abraham, M. R. (2016). The Relation of Moderate Alcohol Consumption to Hyperuricemia in a Rural General Population. International Journal of Environmental Research and Public Health, 13(7), 732. https://doi.org/10.3390/ijerph13070732





