The Epidemiologic, Microbiologic and Clinical Picture of Bacteremia among Febrile Infants and Young Children Managed as Outpatients at the Emergency Room, before and after Initiation of the Routine Anti-Pneumococcal Immunization
Abstract
:1. Introduction
2. Patients and Methods
- Fever without source;
- Suspected occult bacteremia;
- Fever only.
2.1. Study Population
2.2. Treatment Protocol
2.3. Study Conduct
2.4. Microbiology
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Slonim, A.D.; Kurtines, H.C.; Sprague, B.M.; Singh, N. The cost associated with nosocomial blood stream infections in the pediatric intensive care unit. Pediatr. Crit. Care Med. 2001, 2, 170–174. [Google Scholar] [CrossRef] [PubMed]
- See, L.L.C. Bloodstream infection in children. Pediatr. Crit. Care Med. 2005, 6, S42–S44. [Google Scholar] [CrossRef] [PubMed]
- Baraff, L.J.; Bass, J.W.; Fleisher, G.R.; Klein, J.O.; McCracken, G.H.; Powell, K.R.; Schriger, D.L. Practice guideline for the management of infants and children 0 to 36 months of age with fever without source. Pediatrics 1993, 92, 1–12. [Google Scholar] [CrossRef]
- Baraff, L.J. Management of fever without source in infants and children. Ann. Emerg. Med. 2000, 36, 602–614. [Google Scholar] [CrossRef] [PubMed]
- Klein, J.O. Management of the febrile child without a focus of infection in the era of universal pneumococcal immunization. Pediatr. Infect. Dis. J. 2002, 21, 584–588. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, P. Fever without apparent source on clinical examination. Curr. Opin. Pediatr. 2005, 17, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Arora, R.; Mahajan, P. Evaluation of child with fever without source: Review of literature and update. Pediatr. Clin. N. Am. 2013, 60, 1049–1062. [Google Scholar] [CrossRef] [PubMed]
- Mekitarian Filho, E.; de Carvalho, W.B. Current management of occult bacteremia in infants. J. Pediatr. (Rio J.) 2015. [Google Scholar] [CrossRef]
- Bachur, R.; Perry, H.; Harper, M.B. Occult pneumonias: Empiric chest radiographs in febrile children with leukocytosis. Ann. Emerg. Med. 1999, 33, 166–173. [Google Scholar] [CrossRef]
- Marshall, R.; Teele, D.W.; Klein, J.O. Unsuspected bacteremia due to Haemophilus influenzae: Outcome in children not initially admitted to hospital. J. Pediatr. 1979, 95, 690–695. [Google Scholar] [CrossRef]
- Alario, A.J.; Nelson, E.W.; Shapiro, E.D. Blood cultures in the management of febrile outpatients later found to have bacteremia. J. Pediatr. 1989, 115, 195–199. [Google Scholar] [CrossRef]
- Ladhani, S.; Slack, M.P.; Heath, P.T.; von Gottberg, A.; Chandra, M.; Ramsay, M.E. Invasive Haemophilus influenzae disease, Europe, 1996–2006. Emerg. Infect. Dis. 2010, 16, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Black, S.; Shinefield, H.; Fireman, B.; Lewis, E.; Ray, P.; Hansen, J.R.; Elvin, L.; Ensor, K.M.; Hackell, J.; Malinovski, F.; et al. Efficacy, safety and immunogenicity of heptavalent pneumococcal conjugate vaccine in children. Pediatr. Infect. Dis. J. 2000, 19, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Pelton, S.I.; Loughlin, A.M.; Marchant, C.D. Seven valent pneumococcal conjugate vaccine immunization in two Boston communities—Changes in serotypes and antimicrobial susceptibility among Streptococcus pneumoniae isolates. Pediatr. Infect. Dis. J. 2004, 23, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Herz, A.M.; Greenhow, T.L.; Alcantara, J.; Hansen, J.; Baxter, R.P.; Black, S.B.; Shinefield, H.R. Changing epidemiology of outpatient bacteremia in 3- to 36-month-old children after the introduction of the heptavalent-conjugated pneumococcal vaccine. Pediatr. Infect. Dis. J. 2006, 25, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Baraff, L.J. Management of infants and young children with fever without source. Pediatr. Ann. 2008, 37, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shimol, S.; Greenberg, D.; Givon-Lavi, N.; Elias, N.; Glikman, D.; Rubinstein, U.; Dagan, R. Rapid reduction in invasive pneumococcal disease after introduction of PCV7 into the National Immunization Plan in Israel. Vaccine 2012, 30, 6600–6607. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shimol, S.; Greenberg, D.; Givon-Lavi, N.; Schlesinger, Y.; Somekh, E.; Aviner, S.; Miron, D.; Dagan, R. Early impact of sequential introduction of 7-valent and 13-valent pneumococcal conjugate vaccine on IPD in Israeli children <5 years: An active prospective nationwide surveillance. Vaccine 2014, 32, 3452–3459. [Google Scholar] [PubMed]
- Kaplan, S.L.; Barson, W.J.; Lin, P.L.; Romero, J.R.; Bradley, J.S.; Tan, T.Q.; Hoffman, J.A.; Givner, L.B.; Mason, E.O., Jr. Early trends for invasive pneumococcal infections in children after the introduction of the 13-valent pneumococcal conjugate vaccine. Pediatr. Infect. Dis. J. 2013, 32, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Misty, R.D.; Wedin, T.; Balamuth, F.; McGowan, K.L.; Ellison, A.M.; Nelson, K.A.; McLoughlin, R.J.; Alpern, E.R. Emergency department epidemiology of pneumococcal bacteremia in children since the institution of widespread PCV7 vaccination. J. Emerg. Med. 2013, 45, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, J.; Dagan, R.; Press, J.; Sofer, S. Differences in the epidemiology of childhood community-acquired bacterial meningitis between two ethnic populations cohabiting in one geographic area. Pediatr. Infect. Dis. J. 1988, 7, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Fraser, D.; Givon-Lavi, N.; Bilenko, N.; Dagan, R. A decade (1989–1998) of pediatric invasive pneumococcal disease in 2 populations residing in 1 geographic location: Implications for vaccine choice. Clin. Infect. Dis. 2001, 33, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Novack, V.; Avnon, L.S.; Etzion, O.; Riesenberg, K.; Elbaz, G.; Schlaeffer, F. Differences between Bedouin and Jewish populations in incidence and characteristics of patients hospitalized with community-acquired pneumonia. Ethn. Dis. 2007, 17, 441–446. [Google Scholar] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibilioty Testing: 18th Informational Supplement; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- Stoll, M.L.; Rubin, L.G. Incidence of occult bacteremia among highly febrile young children in the era of the pneumococcal conjugate vaccine. Arch. Pediatr. Adolesc. Med. 2004, 158, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Cairstairs, K.L.; Tanen, D.A.; Johnson, A.S.; Kailes, S.B.; Riffenburgh, R.H. Pneumococcal bacteremia in febrile infants presenting to the emergency department before and after the introduction of the heptavalent pneumococcal vaccine. Ann. Emerg. Med. 2007, 49, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, M.; Bulloch, B.; Smith, M. Prevalence of occult bacteremia in children aged 3 to 36 months presenting to the emergency department with fever in the post pneumococcal conjugate vaccine era. Acad. Emerg. Med. 2009, 16, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Waddle, E.; Jhaveri, R. Outcomes of febrile children without localizing signs after pneumococcal conjugate vaccine. Arch. Dis. Child 2009, 94, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Bressan, S.; Berlese, P.; Mion, T.; Masiero, S.; Cavallaro, A.; Da Dalt, L. Bacteremia in feverish children presenting to the emergency department: A retrospective study and literature review. Acta Paediatr. 2012, 101, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Benito-Fernandez, J.; Mintegi, R.S.; Pocheville-Gurutzeta, I.; Sanchez Etxaniz, J.; Gomez Cortes, B.; Hernandez Almaraz, J.L. Pneumococcal bacteremia in febrile infants presenting to the emergency department 8 years after the introduction of pneumococcal conjugate vaccine in the Basque Country of Spain. Pediatr. Infect. Dis. J. 2010, 39, 1142–1144. [Google Scholar] [CrossRef] [PubMed]
- Ribitzky-Eisner, H.; Minuhin, Y.; Greenberg, D.; Greenberg, N.; Chodick, G.; Craiu, M.; Leibovitz, E. Epidemiologic and microbiologic characteristics of occult bacteremia among febrile children in Southern Israel, before and after initiation of the routine antipneumococcal immunization (2005–2012). Pediatr. Neonatol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gomez, B.; Hernandez-Bou, S. Bacteremia in previously healthy children in Emergency Departments: Clinical and microbiological characteristics and outcome. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Bou, S.; Trenchs, V.; Battle, A.; Gene, A.; Luaces, C. Occult bacteraemia is uncommon in febrile infants who appear well, and close clinical follow-up is more appropriate than blood tests. Acta Paediatr. 2015, 104, e76–e81. [Google Scholar] [CrossRef] [PubMed]
- NICE. Feverish Illness in Children: Assessment and Initial Management in Children Younger than 5 Years (CG160). Available online: http://www.nice.org.uk/guidance/cg160 (access on 28 July 2014).
- Yagupsky, P.; Dagan, R.; Howard, C.B.; Einhorn, M.; Kassis, I.; Simu, A. Clinical features and epidemiology of invasive Kingella kingae infections in southern Israel. Pediatrics 1993, 92, 800–804. [Google Scholar] [PubMed]
- Yagupsky, P.; Dagan, R. Kingella kingae bacteremia in children. Pediatr. Infect. Dis. J. 1994, 13, 1148–1149. [Google Scholar] [CrossRef] [PubMed]
- Yagupsky, P.; Dagan, R. Kingella kingae: An emerging cause of invasive infections in young children. Clin. Infect. Dis. 1997, 24, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Yagupsky, P.; Peled, N.; Katz, O. Epidemiological features of invasive Kingella kingae infections and respiratory carriage of the organism. J. Clin. Microbiol. 2002, 40, 4180–4184. [Google Scholar] [CrossRef] [PubMed]
- Yagupsky, P.; Weiss-Salz, I.; Fluss, R.; Freedman, L.; Peled, N.; Trefler, R.; Porat, N.; Dagan, R. Dissemination of Kingella kingae in the community and long-term persistence of invasive clones. Pediatr. Infect. Dis. J. 2009, 28, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Amit, U.; Dagan, R.; Porat, N.; Trefler, R.; Yagupsky, P. Epidemiology of invasive Kingella kingae infections in 2 distinct pediatric populations cohabiting in one geographic area. Pediatr. Infect. Dis. J. 2012, 31, 415–417. [Google Scholar] [CrossRef] [PubMed]
- Gene, A.; Garcia-Garcia, J.J.; Sala, P.; Sierra, M.; Huguet, R. Enhanced culture detection of Kingella kingae, a pathogen of increasing clinical importance in pediatrics. Pediatr. Infect. Dis. J. 2004, 23, 886–888. [Google Scholar] [CrossRef] [PubMed]
- Yagupsky, P.; Porsch, E.; St Geme, J.W., III. Kingella kingae: An emerging pathogen in young children. Pediatrics 2011, 127, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Anis, E.; Levental, A.; Grotto, I.; Gandacu, D.; Warshavsky, B.; Shimshony, A.; Israeli, A. Recent trends in human brucellosis in Israel. Isr. Med. Assoc. J. 2011, 13, 359–362. [Google Scholar] [PubMed]
- Fruchtman, Y.; Wilkof-Segev, R.; Asher-Golan, A.; Dalem, Y.; Abu Tailakh, M.; Novak, V.; Peled, N.; Craiu, M.; Leibovitz, E. Epidemiological, diagnostic, clinical and therapeutic aspects of Brucella bacteremia in children in southern Israel: A 7-year retrospective study (2005–2011). Vector Borne Zoon. Dis. 2015, 15, 195–201. [Google Scholar] [CrossRef] [PubMed]
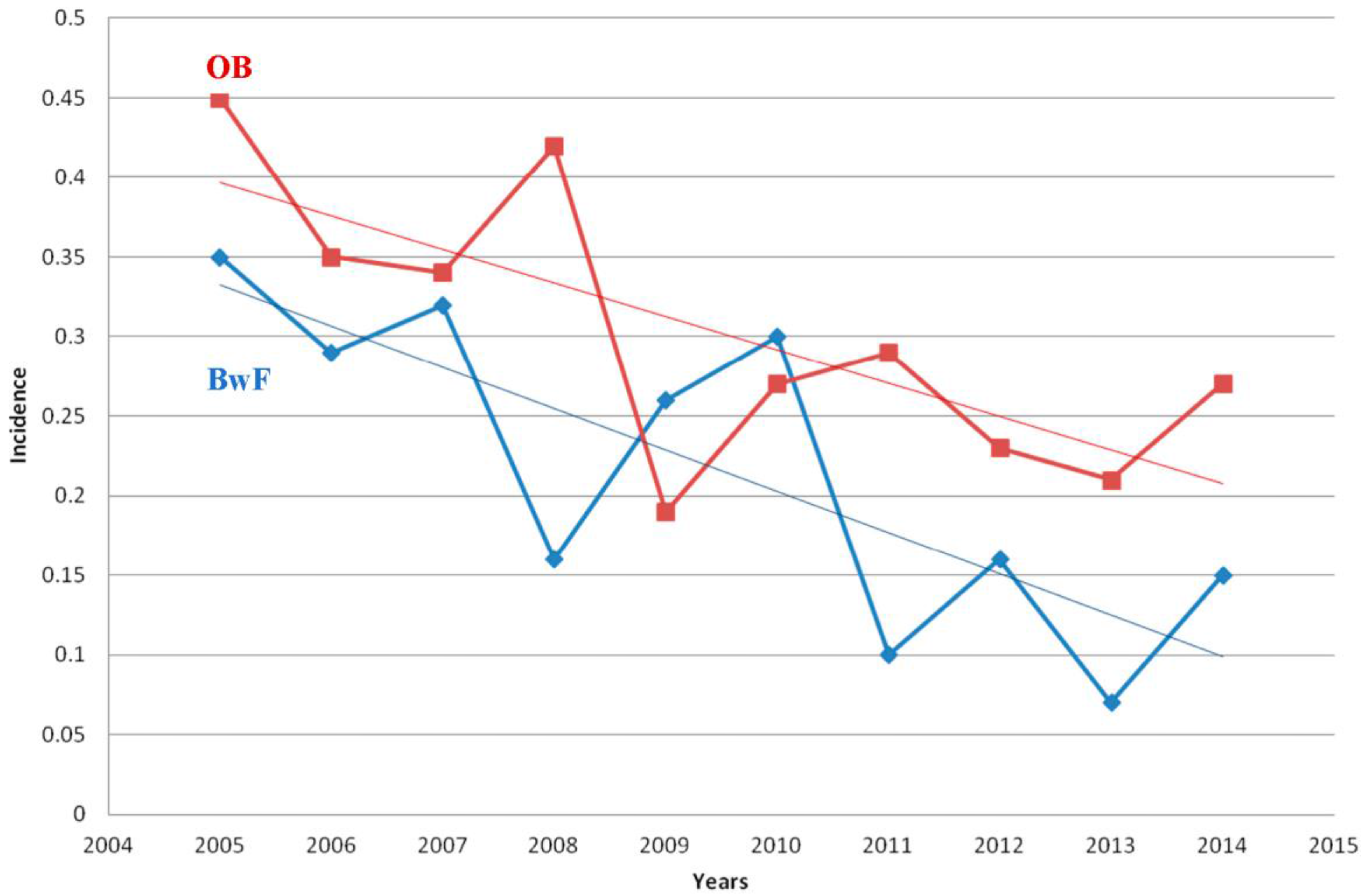
| Year | No. Children with Fever Discharged from PER | OB | BwF | ||
|---|---|---|---|---|---|
| N | (%) | N | (%) | ||
| 2005 | 4895 | 17 | 0.35 | 22 | 0.45 |
| 2006 | 4809 | 14 | 0.29 | 17 | 0.35 |
| 2007 | 4941 | 16 | 0.32 | 17 | 0.34 |
| 2008 | 4515 | 7 | 0.16 | 19 | 0.42 |
| 2009 | 4217 | 11 | 0.26 | 8 | 0.19 |
| 2010 | 4370 | 13 | 0.30 | 12 | 0.27 |
| 2011 | 4088 | 4 | 0.10 | 12 | 0.29 |
| 2012 | 3840 | 6 | 0.16 | 9 | 0.23 |
| 2013 | 4209 | 3 | 0.07 | 9 | 0.21 |
| 2014 | 3363 | 5 | 15.0 | 9 | 27.0 |
| Total | 43,247 | 96 | 134 | ||
| Mean ± SD | 4324.7 ± 496.5 | 9.6 ± 5.21 | 0.216 | 13.4 ± 4.97 | 0.302 |
| Total N (%) | OB N (%) | BwF N (%) | p Value | |
|---|---|---|---|---|
| Streptococcus pneumoniae | 76 (33.0) | 36 (37.50) | 40 (29.8) | 0.2 |
| Kingella kingae | 23 (10.0) | 11 (11.40) | 12 (8.95) | 0.5 |
| Streptococcus viridans spp. | 25 (10.87) | 7 (7.30) | 18 (13.40) | 0.14 |
| Salmonella spp. * | 10 (4.34) | 7 (7.30) | 3 (2.20) | 0.1 |
| Gram negative bacilli | 8 (3.50) | 5 (5.21) | 3 (2.20) | |
| Acinetobacter spp. | 9 (3.90) | 5 (5.21) | 4 (3.0) | |
| Staphylococcus aureus | 7 (3.0) | 5 (5.21) | 2 (1.50) | |
| Neisseria spp. ** | 5 (2.17) | 3 (3.12) | 2 (1.50) | |
| Brucella spp. | 20 (8.70) | 3 (3.12) | 17 (12.70) | 0.01 |
| Escherichia coli * | 10 (4.34) | 2 (2.10) | 8 (6.0) | 0.20 |
| Enterococcus spp. | 2 (0.87) | 2 (2.10) | 0 (0) | |
| Pseudomonas spp. | 9 (3.90) | 2 (2.10) | 7 (5.20) | 0.31 |
| Enterobacter cloacae * | 1 (0.42) | 1 (1) | 0 (0) | |
| Gram positive cocci | 2 (0.87) | 2 (2.10) | 0 (0) | |
| Haemophilus influenzae non-typable | 6 (2.60) | 1 (1) | 5 (3.70) | 0.40 |
| Klebsiella pneumoniae * | 1 (0.43) | 1 (1) | 0 (0) | |
| Pantoea agglomerans | 1 (0.43) | 1 (1) | 0 (0) | |
| Pasteurella multocida | 1 (0.43) | 1 (1) | 0 (0) | |
| Streptococcus pyogenes | 5 (2.17) | 1 (1) | 4 (3.0) | |
| OTHER | 9 (4.90) | 0 (0) | 9 (6.70) | |
| Total | 230 (100%) | 96 (100%) | 134 (100%) |
| Serotype | Total | OB | BwF | p Value |
|---|---|---|---|---|
| N = 76 | N = 36 | N = 40 | ||
| 19A | 9 | 8 | 1 | 0.01 |
| 1 | 8 | 3 | 5 | 0.7 |
| 5 | 2 | - | 2 | |
| 6B | 9 | 3 | 6 | 0.49 |
| 9V | 6 | 3 | 3 | 1.0 |
| 19F | 4 | 2 | 2 | 1.0 |
| 12F | 3 | 2 | 1 | 0.6 |
| 14 | 6 | 2 | 4 | 0.67 |
| 23F | 4 | 2 | 2 | 1.0 |
| 3 | 1 | 1 | - | |
| 6A | 4 | 1 | 3 | 0.62 |
| 6C | 1 | 1 | - | |
| 10A | 1 | 1 | - | |
| 15 | 1 | - | 1 | |
| 15A | 1 | - | 1 | |
| 15B | 2 | - | 2 | |
| 15B/C | 3 | 1 | 2 | 1.0 |
| 18A | 1 | 1 | - | |
| 21 | 1 | - | 1 | |
| 24F | 1 | 1 | - | |
| 28A | 1 | 1 | - | |
| 33A | 1 | - | 1 | |
| 33F | 2 | 1 | 1 | |
| 34 | 1 | - | 1 | |
| 35B | 2 | 1 | 1 | |
| 46 | 1 | 1 | - | |
| Total PCV7 | 29 (38.20%) | 12 (33.30%) | 17 (42.50%) | |
| Additional six seroytpes | 24 (31.60%) | 13 (36.10%) | 11 (27.50%) | |
| Total PCV13 | 53 (69.70%) | 25 (69.50%) | 28 (70.0%) | |
| Non-PCV13 serotypes | 23 (30.30%) | 11 (30.50%) | 12 (30.0%) | |
| Total | 76 (100%) | 36 (100.0%) | 40 (100%) |
| Serotype | 2005–2010 | 2011–2014 | p Value |
|---|---|---|---|
| OB (N = 36) | N = 32 | N = 4 | |
| PCV13 isolates | 25 | 0 | 0.05 |
| Non-PCV13 | 7 | 4 | 0.005 |
| (6C, 10A, 12F, 18A, 24F, 28A, 46) | (12F, 15B/C, 33F, 35B) | ||
| BwF (N = 40) | N = 31 | N = 9 | |
| PCV13 isolates | 27 | 1 | |
| Non-PCV13 isolates | 4 | 8 | |
| (15A, 33A, 33F, 34) | (12F, 15, 15B-2, 15B/C-2, 21, 35B) |
| Diagnosis | Total BwF Cases | Most Common Pathogen (No. of Cases Caused by the Pathogen, %) |
|---|---|---|
| Pneumonia | 40 | S. pneumoniae (25, 62.50 %) |
| S. viridans (8, 20.0%) | ||
| H. influenzae non-typable (3, 7.50%) | ||
| Acute Otitis Media | 33 | S. pneumoniae (11, 33.30%) |
| K. kingae (5, 15.20%) | ||
| Acute Gastroenteritis | 25 | S. viridans (7, 28.0%) |
| Gram negative bacilli (3, 12.0%) | ||
| Salmonella spp. (3, 12.0%) | ||
| Brucellosis ** | 15 | - |
| Periorbital Cellulitis | 5 | S. pneumoniae (3, 60.0%) |
| Urinary Tract Infection | 4 | E. coli (3, 75.0%) |
| Limping/Arthritis | 4 | Brucella spp. (2, 50.0%) |
| K. kingae (2, 50.0%) | ||
| Acute Tonsillitis | 3 | |
| Aphtous Stomatitis | 2 | S. aureus (1, 50.0%) |
| Cellulitis | 1 | |
| Acute Otitis Externa | 1 | S. aureus (1) |
| Fever Convulsion | 1 | |
| Other | 6 | |
| Total | 143 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leibovitz, E.; David, N.; Ribitzky-Eisner, H.; Abo Madegam, M.; Abuabed, S.; Chodick, G.; Maimon, M.; Fruchtman, Y. The Epidemiologic, Microbiologic and Clinical Picture of Bacteremia among Febrile Infants and Young Children Managed as Outpatients at the Emergency Room, before and after Initiation of the Routine Anti-Pneumococcal Immunization. Int. J. Environ. Res. Public Health 2016, 13, 723. https://doi.org/10.3390/ijerph13070723
Leibovitz E, David N, Ribitzky-Eisner H, Abo Madegam M, Abuabed S, Chodick G, Maimon M, Fruchtman Y. The Epidemiologic, Microbiologic and Clinical Picture of Bacteremia among Febrile Infants and Young Children Managed as Outpatients at the Emergency Room, before and after Initiation of the Routine Anti-Pneumococcal Immunization. International Journal of Environmental Research and Public Health. 2016; 13(7):723. https://doi.org/10.3390/ijerph13070723
Chicago/Turabian StyleLeibovitz, Eugene, Nuphar David, Haya Ribitzky-Eisner, Mouner Abo Madegam, Said Abuabed, Gabriel Chodick, Michal Maimon, and Yariv Fruchtman. 2016. "The Epidemiologic, Microbiologic and Clinical Picture of Bacteremia among Febrile Infants and Young Children Managed as Outpatients at the Emergency Room, before and after Initiation of the Routine Anti-Pneumococcal Immunization" International Journal of Environmental Research and Public Health 13, no. 7: 723. https://doi.org/10.3390/ijerph13070723
APA StyleLeibovitz, E., David, N., Ribitzky-Eisner, H., Abo Madegam, M., Abuabed, S., Chodick, G., Maimon, M., & Fruchtman, Y. (2016). The Epidemiologic, Microbiologic and Clinical Picture of Bacteremia among Febrile Infants and Young Children Managed as Outpatients at the Emergency Room, before and after Initiation of the Routine Anti-Pneumococcal Immunization. International Journal of Environmental Research and Public Health, 13(7), 723. https://doi.org/10.3390/ijerph13070723







