Sugar-Sweetened Beverage Consumption Is Adversely Associated with Childhood Attention Deficit/Hyperactivity Disorder
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants and Recruitment
2.2. Dietary Habits, Consumption of SSBs and Intake of Sugar and Calories
2.3. Measurement of BLLs and Gene Polymorphism Analysis
2.4. Covariates
2.5. Statistical and Probabilistic Analysis
3. Results
3.1. Demographic Characteristics of Participants
3.2. Dietary Habits
3.3. Sugar Consumption and Caloric Intake
3.4. The Association between SSB Consumption and ADHD
3.5. Gene Polymorphisms
3.6. Lead Exposure
4. Discussion
4.1. The Association Between SSB Consumption and ADHD
4.2. Effect of Sugar in SSBs on ADHD
4.3. Effects of Artificial Food Colorings (AFCs) in SSBs on ADHD
4.4. Effect of Preservatives in SSBs on ADHD
4.5. The Relationship between Dietary Habits and SSBs
4.6. Children’s Exposure to Lead in Taiwan
4.7. Strengths and Limitations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ADHD | Attention deficit/hyperactivity disorder |
| SSB | Sugar-sweetened beverage |
| DRD4 | D4 dopamine receptor gene |
| DAT1 | Dopamine transporter gene |
| AFC | Artificial food coloring |
| DSM-IV-TR | Diagnostic and statistical manual of mental disorders, 4th edition, revised criteria |
| BLL | Blood lead levels |
| SNAP-IV | Swanson, Nolan and Pelham, fourth revision |
| MOHW | Ministry of Health and Welfare |
| TFDA | Taiwan Food and Drug Administration |
| LOD | Limit of detection |
| MC | Monte Carlo |
| OR | Odds ratio |
| CI | Confidence interval |
| ADI | Acceptable daily intake |
| MOS | Margin of safety |
| BA | Benzoic acid |
| SA | Sorbic acid |
| WHO | World Health Organization |
References
- Popkin, B.M.; Nielsen, S.J. The sweetening of the world’s diet. Obes. Res. 2003, 11, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Zhang, Z.; Gregg, E.W.; Flanders, W.D.; Merritt, R.; Hu, F.B. Added sugar intake and cardiovascular diseases mortality among U.S. adults. JAMA Intern. Med. 2014, 174, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Dykman, K.D.; Dykman, R.A. Effect of nutritional supplements on attentional-deficit hyperactivity disorder. Integr. Physiol. Behav. Sci. 1998, 33, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Arnold, L.E.; Lofthouse, N.; Hurt, E. Artificial food colors and attention-deficit/hyperactivity symptoms: Conclusions to dye for. Neurotherapeutics 2012, 9, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Bateman, B.; Warner, J.O.; Hutchinson, E.; Dean, T.; Rowlandson, P.; Gant, C.; Grundy, J.; Fitzgerald, C.; Stevenson, J. The effects of a double blind, placebo controlled, artificial food colourings and benzoate preservative challenge on hyperactivity in a general population sample of preschool children. Arch. Dis. Child. 2004, 89, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Schab, D.W.; Trinh, N.-H.T. Do artificial food colors promote hyperactivity in children with hyperactive syndromes? A meta-analysis of double-blind placebo-controlled trials. J. Dev. Behav. Pediatr. 2004, 25, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.D.; Kim, D.W.; Hong, Y.S.; Kim, Y.M.; Seo, J.H.; Choe, B.M.; Park, J.H.; Kang, J.W.; Yoo, J.H.; Chueh, H.W.; et al. Dietary patterns in children with attention deficit/hyperactivity disorder (ADHD). Nutrients 2014, 6, 1539–1553. [Google Scholar] [CrossRef] [PubMed]
- Kendig, M.D. Cognitive and behavioural effects of sugar consumption in rodents. A review. Appetite 2014, 80, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, R.; Gomez-Pinilla, F. “Metabolic syndrome” in the brain: Deficiency in omega-3 fatty acid exacerbates dysfunctions in insulin receptor signalling and cognition. J. Physiol. 2012, 590, 2485–2499. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Lu, H.; Lewis, T.L.; Li, L. Intake of sucrose-sweetened water induces insulin resistance and exacerbates memory deficits and amyloidosis in a transgenic mouse model of Alzheimer disease. J. Biol. Chem. 2007, 282, 36275–36282. [Google Scholar] [CrossRef] [PubMed]
- Kendig, M.D.; Boakes, R.A.; Rooney, K.B.; Corbit, L.H. Chronic restricted access to 10% sucrose solution in adolescent and young adult rats impairs spatial memory and alters sensitivity to outcome devaluation. Physiol. Behav. 2013, 120, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Soares, E.; Prediger, R.D.; Nunes, S.; Castro, A.A.; Viana, S.D.; Lemos, C.; De Souza, C.M.; Agostinho, P.; Cunha, R.A.; Carvalho, E.; et al. Spatial memory impairments in a prediabetic rat model. Neuroscience 2013, 250, 565–577. [Google Scholar] [CrossRef] [PubMed]
- De Ruyter, J.C.; Olthof, M.R.; Seidell, J.C.; Katan, M.B. A trial of sugar-free or sugar-sweetened beverages and body weight in children. N. Engl. J. Med. 2012, 367, 1397–1406. [Google Scholar] [CrossRef] [PubMed]
- Campos, V.; Despland, C.; Brandejsky, V.; Kreis, R.; Schneiter, P.; Chiolero, A.; Boesch, C.; Tappy, L. Sugar- and artificially sweetened beverages and intrahepatic fat: A randomized controlled trial. Obesity 2015, 23, 2335–2339. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Yuzbashian, E.; Asghari, G.; Hosseinpour-Niazi, S.; Azizi, F. Consumption of sugar sweetened beverage is associated with incidence of metabolic syndrome in Tehranian children and adolescents. Nutr. Metab. 2015, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shang, L.; Light, K.; O’Loughlin, J.; Paradis, G.; Gray-Donald, K. Associations between added sugar (solid vs. liquid) intakes, diet quality, and adiposity indicators in Canadian children. Appl. Physiol. Nutr. Metab. 2015, 40, 835–841. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.; Bucher Della Torre, S. Sugar-sweetened beverages and obesity among children and adolescents: A review of systematic literature reviews. Child. Obes. 2015, 11, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Wolraich, M.L.; Wilson, D.B.; White, J.W. The effect of sugar on behavior or cognition in children: A meta-analysis. JAMA 1995, 274, 1617–1621. [Google Scholar] [CrossRef] [PubMed]
- Ptacek, R.; Kuzelova, H.; Stefano, G.B.; Raboch, J.; Sadkova, T.; Goetz, M.; Kream, R.M. Disruptive patterns of eating behaviors and associated lifestyles in males with ADHD. Med. Sci. Monit. 2014, 20, 608. [Google Scholar] [PubMed]
- Lien, L.; Lien, N.; Heyerdahl, S.; Thoresen, M.; Bjertness, E. Consumption of soft drinks and hyperactivity, mental distress, and conduct problems among adolescents in Oslo, Norway. Am. J. Public Health 2006, 96, 1815–1820. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.L.; Gilstad-Hayden, K.; Carroll-Scott, A.; Grilo, S.A.; McCaslin, C.; Schwartz, M.; Ickovics, J.R. Energy drinks and youth self-reported hyperactivity/inattention symptoms. Acad. Pediatr. 2015, 15, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Millichap, J.G.; Yee, M.M. The diet factor in attention-deficit/hyperactivity disorder. Pediatrics 2012, 129, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Schnoll, R.; Burshteyn, D.; Cea-Aravena, J. Nutrition in the treatment of attention-deficit hyperactivity disorder: A neglected but important aspect. Appl. Psychophysiol. Biofeedback 2003, 28, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Curtis, L.T.; Patel, K. Nutritional and environmental approaches to preventing and treating autism and attention deficit hyperactivity disorder (ADHD): A review. J. Altern. Complement. Med. 2008, 14, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Chang, H. Correlation between attention deficit hyperactivity disorder and sugar consumption, quality of diet, and dietary behavior in school children. Nutr. Res. Pract. 2011, 5, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-T.; Huang, H.-L.; Huang, M.-C.; Chan, T.-F.; Ciou, S.-Y.; Lee, C.-Y.; Chiu, Y.-W.; Duh, T.; Lin, P.-L.; Wang, T.-N. Effects on uric acid, body mass index and blood pressure in adolescents of consuming beverages sweetened with high-fructose corn syrup. Int. J. Obes. 2013, 37, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-T.; Chan, T.-F.; Huang, H.-L.; Lee, C.-Y.; Tsai, S.; Wu, P.-W.; Yang, Y.-C.; Wang, T.-N.; Lee, C.-H. Fructose-rich beverage intake and central adiposity, uric acid, and pediatric insulin resistance. J. Pediatr. 2016, 171. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.-K. Taiwan Chain Store Almanac; Taiwan Chain Stores and Franchise Association: Taipei, Taiwan, 2015. [Google Scholar]
- Townsend, L. The Four-Year Spread of Bubble Tea across the UK, in BBC Magazine Monitor. 2014. Available online: http://www.bbc.com/news/magazine-28640188 (accessed on 15 March 2016).
- Trleu, R. Three Taiwanese Bubble Tea Chains Race To California, in Forbes Business. 2014. Available online: http://www.forbes.com/sites/rosatrieu/2014/01/01/three-taiwanese-bubble-tea-chains-racing-for-california/#3c3967d2645e (accessed on 15 March 2016).
- Johnson, R.J.; Segal, M.S.; Sautin, Y.; Nakagawa, T.; Feig, D.I.; Kang, D.-H.; Gersch, M.S.; Benner, S.; Sánchez-Lozada, L.G. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 2007, 86, 899–906. [Google Scholar] [PubMed]
- Malik, V.S.; Popkin, B.M.; Bray, G.A.; Després, J.-P.; Willett, W.C.; Hu, F.B. Sugar-sweetened beverages and risk of metabolic syndrome and type 2 diabetes a meta-analysis. Diabetes Care 2010, 33, 2477–2483. [Google Scholar] [CrossRef] [PubMed]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P.; Garg, R. Metabolic syndrome a comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 2005, 111, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Pliszka, S.; AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J. Am. Acad. Child Adolesc. Psychiatry 2007, 46, 894–921. [Google Scholar] [CrossRef] [PubMed]
- Saadi, H.R.; Shamsuddin, K.; Sutan, R.; Alshaham, S.A. Socio-maternal risk factors of ADHD among Iraqi children: A case-control study. Open J. Prev. Med. 2013, 3, 251–257. [Google Scholar] [CrossRef]
- Huang, H.L.; Chao, C.C.; Tu, C.C.; Yang, P.C. Behavioral parent training for Taiwanese parents of children with attention-deficit/hyperactivity disorder. Psychiatry Clin. Neurosci. 2003, 57, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Willcutt, E.G. The prevalence of DSM-IV attention-deficit/hyperactivity disorder: A meta-analytic review. Neurotherapeutics 2012, 9, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Harpin, V.A. The effect of ADHD on the life of an individual, their family, and community from preschool to adult life. Arch. Dis. Child. 2005, 90, i2–i7. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.; Trampush, J.W.; Rindskopf, D.; Marks, D.J.; O’Neill, S.; Halperin, J.M. Association between variation in neuropsychological development and trajectory of ADHD severity in early childhood. Am. J. Psychiatry 2013, 170, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.V.; Perlis, R.H.; Doyle, A.E.; Smoller, J.W.; Goralnick, J.J.; Holmgren, M.A.; Sklar, P. Molecular genetics of attention-deficit/hyperactivity disorder. Biol. Psychiatry 2005, 57, 1313–1323. [Google Scholar] [CrossRef] [PubMed]
- Wallis, D.; Russell, H.F.; Muenke, M. Review: Genetics of attention deficit/hyperactivity disorder. J. Pediatr. Psychol. 2008, 33, 1085–1099. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-J.; Du, J.-C.; Chiou, H.-C.; Chung, M.-Y.; Yang, W.; Chen, Y.-S.; Fuh, M.-R.; Chien, L.-C.; Hwang, B.; Chen, M.-L. Increased risk of attention-deficit/hyperactivity disorder associated with exposure to organophosphate pesticide in Taiwanese children. Andrology 2016. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual-Text. Revision (DSM-IV-TRim, 2000); American Psychiatric Association: Washington, DC, USA, 2000. [Google Scholar]
- Swanson, J. SNAP-IV Scale; University of California Child Development Center: Irvine, CA, USA, 1995. [Google Scholar]
- Bussing, R.; Fernandez, M.; Harwood, M.; Hou, W.; Garvan, C.W.; Eyberg, S.M.; Swanson, J.M. Parent and teacher SNAP-IV ratings of attention deficit hyperactivity disorder symptoms psychometric properties and normative ratings from a school district sample. Assessment 2008, 15, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Gau, S.S.F.; Shang, C.Y.; Liu, S.K.; Lin, C.H.; Swanson, J.M.; Liu, Y.C.; Tu, C.L. Psychometric properties of the Chinese version of the Swanson, Nolan, and Pelham, Version IV Scale-Parent Form. Int. J. Method Psych. Res. 2008, 17, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Gau, S.S.-F.; Lin, C.-H.; Hu, F.-C.; Shang, C.-Y.; Swanson, J.M.; Liu, Y.-C.; Liu, S.-K. Psychometric properties of the Chinese version of the Swanson, Nolan, and Pelham, Version IV Scale-Teacher Form. J. Pediatr. Psychol. 2009, 34, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Swanson, J.M.; Kraemer, H.C.; Hinshaw, S.P.; Arnold, L.E.; Conners, C.K.; Abikoff, H.B.; Clevenger, W.; Davies, M.; Elliott, G.R.; Greenhill, L.L. Clinical relevance of the primary findings of the MTA: Success rates based on severity of ADHD and ODD symptoms at the end of treatment. J. Am. Acad. Child Psychiatry 2001, 40, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Min, J.E.; Green, D.B.; Kim, L. Calories and sugars in boba milk tea: Implications for obesity risk in Asian Pacific Islanders. Food Sci. Nutr. 2016. [Google Scholar] [CrossRef]
- Chan, T.-F.; Lin, W.-T.; Chen, Y.-L.; Huang, H.-L.; Yang, W.-Z.; Lee, C.-Y.; Chen, M.-H.; Wang, T.-N.; Huang, M.-C.; Chiu, Y.-W. Elevated serum triglyceride and retinol-binding protein 4 levels associated with fructose-sweetened beverages in adolescents. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Cats, A.; Kuipers, E.; Bosschaert, M.; Pot, R.; Vandenbroucke-Grauls, C.; Kusters, J. Effect of frequent consumption of a Lactobacillus casei-containing milk drink in Helicobacter pylori-colonized subjects. Aliment. Pharmacol. Ther. 2003, 17, 429–435. [Google Scholar] [CrossRef] [PubMed]
- MOHW. Drink Water and Get Away from Sugar-Sweetened Beverage. 2015. Available online: http://www.mohw.gov.tw/news/531551269 (accessed on 20 March 2016). (In Chinese) [Google Scholar]
- Taiwan Food and Drug Administration. Manual of Food Additives Application. 2015. Available online: http://www.fda.gov.tw/TC/siteContent.aspx?sid=4321#.VxeWP01JlMs (accessed on 15 March 2016). [Google Scholar]
- Yu, C.-J.; Du, J.-C.; Chiou, H.-C.; Yang, S.-H.; Liao, K.-W.; Yang, W.; Chung, M.-Y.; Chien, L.-C.; Hwang, B.; Chen, M.-L. Attention deficit/hyperactivity disorder and urinary nonylphenol levels: A case-control study in Taiwanese Children. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Verner, M.A.; Hart, J.E.; Sagiv, S.K.; Bellinger, D.C.; Altshul, L.M.; Korrick, S.A. Measured prenatal and estimated postnatal levels of Polychlorinated Biphenyls (PCBs) and ADHD-related behaviors in 8-year-old children. Environ. Health Perspect. 2015, 123, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Leventakou, V.; Micali, N.; Georgiou, V.; Sarri, K.; Koutra, K.; Koinaki, S.; Vassilaki, M.; Kogevinas, M.; Chatzi, L. Is there an association between eating behaviour and attention-deficit/hyperactivity disorder symptoms in preschool children? J. Child Psychol. Psychiatry 2015. [Google Scholar] [CrossRef] [PubMed]
- Skoglund, C.; Chen, Q.; Lichtenstein, P.; Larsson, H. Familial confounding of the association between maternal smoking during pregnancy and ADHD in offspring. J. Child Psychol. Psychiatry 2014, 55, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Biederman, J.; Martelon, M.; Woodworth, K.Y.; Spencer, T.J.; Faraone, S.V. Is maternal smoking during pregnancy a risk factor for cigarette smoking in offspring? A longitudinal controlled study of ADHD children grown up. J. Atten. Disord. 2014. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, A.K.; Skogen, J.C.; Ystrom, E.; Sivertsen, B.; Tell, G.S.; Torgersen, L. Maternal pre-pregnancy risk drinking and toddler behavior problems: The Norwegian Mother and Child. Cohort Study. Eur. Child Adolesc. Psychiatry 2014, 23, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Han, J.-Y.; Kwon, H.-J.; Ha, M.; Paik, K.-C.; Lim, M.-H.; Lee, S.G.; Yoo, S.-J.; Kim, E.-J. The effects of prenatal exposure to alcohol and environmental tobacco smoke on risk for ADHD: A large population-based study. Psychiatry Res. 2015, 225, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Mundry, R.; Fischer, J. Use of statistical programs for nonparametric tests of small samples often leads to incorrect p values: Examples from animal behaviour. Anim. Behav. 1998, 56, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-J.; Yeh, N.-H. Dietary nutrient intake and major food sources: The Nutrition and Health Survey of Taiwan Elementary School Children 2001–2002. Asia Pac. J. Clin. Nutr. 2007, 16, 518. [Google Scholar] [PubMed]
- Mikkilä, V.; Räsänen, L.; Raitakari, O.; Pietinen, P.; Viikari, J. Consistent dietary patterns identified from childhood to adulthood: The cardiovascular risk in Young Finns Study. Br. J. Nutr. 2005, 93, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Gulati, S.; Misra, A.; Colles, S.L.; Kondal, D.; Gupta, N.; Goel, K.; Bansal, S.; Mishra, M.; Madkaikar, V.; Bhardwaj, S. Dietary intakes and familial correlates of overweight/obesity: A four-cities study in India. Ann. Nutr. Metab. 2013, 62, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Kubik, M.Y.; Lytle, L.A.; Hannan, P.J.; Perry, C.L.; Story, M. The association of the school food environment with dietary behaviors of young adolescents. Am. J. Public Health 2003, 93, 1168–1173. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.-H.; Kwon, K.-I.; Kim, J.-Y.; Lee, J.-S.; Kim, J.-W.; Park, H.-K.; Kim, M.-C.; Kim, G.-H. Monitoring of total sugar contents in processed foods and noncommercial foodservice foods. Korean J. Food Sci. Technol. 2008, 40, 337–342. [Google Scholar]
- Øverby, N.; Høigaard, R. Diet and behavioral problems at school in Norwegian adolescents. Food Nutr. Res. 2012, 56, 17231. [Google Scholar] [CrossRef] [PubMed]
- Thapar, A.; Cooper, M.; Eyre, O.; Langley, K. Practitioner review: What have we learnt about the causes of ADHD? J. Child Psychol. Psychiatry 2013, 54, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Benton, D. Sucrose and behavioral problems. Crit. Rev. Food Sci. Nutr. 2008, 48, 385–401. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F. Effects of diet on behaviour and cognition in children. Br. J. Nutr. 2004, 92, S227–S232. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.S.; Kim, P.; Park, J.H.; Gonzales, E.L.; Kim, K.C.; Cho, K.S.; Ko, M.J.; Yang, S.M.; Seung, H.; Han, S.H.; et al. High sucrose consumption during pregnancy induced ADHD-like behavioral phenotypes in mice offspring. J. Nutr. Biochem. 2015, 26, 1520–1526. [Google Scholar] [CrossRef] [PubMed]
- Ventura, E.E.; Davis, J.N.; Goran, M.I. Sugar content of popular sweetened beverages based on objective laboratory analysis: Focus on fructose content. Obesity 2011, 19, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Armstrong, L.E.; Bray, G.M.; Caballero, B.; Frei, B.; Willett, W.C. A new proposed guidance system for beverage consumption in the United States. Am. J. Clin. Nutr. 2006, 83, 529–542. [Google Scholar] [PubMed]
- Chan, T.-F.; Lin, W.-T.; Huang, H.-L.; Lee, C.-Y.; Wu, P.-W.; Chiu, Y.-W.; Huang, C.-C.; Tsai, S.; Lin, C.-L.; Lee, C.-H. Consumption of sugar-sweetened beverages is associated with components of the metabolic syndrome in adolescents. Nutrients 2014, 6, 2088–2103. [Google Scholar] [CrossRef] [PubMed]
- Stevens, L.J.; Burgess, J.R.; Stochelski, M.A.; Kuczek, T. Amounts of artificial food dyes and added sugars in foods and sweets commonly consumed by children. Clin. Pediatr. 2015, 54, 309–321. [Google Scholar] [CrossRef] [PubMed]
- McCann, D.; Barrett, A.; Cooper, A.; Crumpler, D.; Dalen, L.; Grimshaw, K.; Kitchin, E.; Lok, K.; Porteous, L.; Prince, E.; et al. Food additives and hyperactive behaviour in 3-year-old and 8/9-year-old children in the community: A randomised, double-blinded, placebo-controlled trial. Lancet 2007, 370, 1560–1567. [Google Scholar] [CrossRef]
- Connolly, A.; Hearty, A.; Nugent, A.; McKevitt, A.; Boylan, E.; Flynn, A.; Gibney, M. Pattern of intake of food additives associated with hyperactivity in Irish children and teenagers. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2010, 27, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Kobylewski, S.; Jacobson, M.F. Toxicology of food dyes. Int. J. Occup. Environ. Health 2012, 18, 220–246. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, China. National Food Safety Standard for Uses of Food Additives; Ministry of Health: Beijing, China, 2011. (In Chinese)
- Diouf, F.; Berg, K.; Ptok, S.; Lindtner, O.; Heinemeyer, G.; Heseker, H. German database on the occurrence of food additives: Application for intake estimation of five food colours for toddlers and children. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2014, 31, 197–206. [Google Scholar] [CrossRef] [PubMed]
- FSANZ (Food Standards Australia New Zealand). Survey of Added Colours in Foods Available in Australia: Study of Concentrations in Foods Including Dietary Exposure Assessment and Risk Characterisation. 2008. Available online: https://www.foodstandards.gov.au/science/surveillance/documents/Colours%20Survey_Final%20Report%2022%20Oct%2008%20(2).pdf (accessed on 19 March 2016). [Google Scholar]
- Beezhold, B.; Johnston, C.S. Sodium benzoate intake in beverages may contribute to ADHD symptoms in college students. FASEB J. 2012, 26, 632–635. [Google Scholar]
- Beezhold, B.L.; Johnston, C.S.; Nochta, K.A. Sodium benzoate-rich beverage consumption is associated with increased reporting of ADHD symptoms in college students: A pilot investigation. J. Atten. Disord. 2014, 18, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Wang, W. Headspace solid-phase microextraction applied to the simultaneous determination of sorbic and benzoic acids in beverages. Anal. Chim. Acta 2006, 562, 23–29. [Google Scholar] [CrossRef]
- Lino, C.; Pena, A. Occurrence of caffeine, saccharin, benzoic acid and sorbic acid in soft drinks and nectars in Portugal and subsequent exposure assessment. Food Chem. 2010, 121, 503–508. [Google Scholar] [CrossRef]
- Sauver, J.L.S.; Barbaresi, W.J.; Katusic, S.K.; Colligan, R.C.; Weaver, A.L.; Jacobsen, S.J. Early life risk factors for attention-deficit/hyperactivity disorder: A population-based cohort study. Mayo Clin. Proc. 2004, 79, 1124–1131. [Google Scholar] [CrossRef]
- Millichap, J.G. Etiologic classification of attention-deficit/hyperactivity disorder. Pediatrics 2008, 121, e358–e365. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-L.; Chen, X.-T.; Yang, B.; Ma, F.-L.; Wang, S.; Tang, M.-L.; Hao, M.-G.; Ruan, D.-Y. Case-control study of blood lead levels and attention deficit hyperactivity disorder in Chinese children. Environ. Health Perspect. 2008, 116, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Winkleby, M.A.; Jatulis, D.E.; Frank, E.; Fortmann, S.P. Socioeconomic status and health: How education, income, and occupation contribute to risk factors for cardiovascular disease. Am. J. Public Health 1992, 82, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Gracia, A.; de Magistris, T. Organic food product purchase behaviour: A pilot study for urban consumers in the south of Italy. Span. J. Agric. Res. 2013, 5, 439–451. [Google Scholar] [CrossRef]
- Howard, A.L.; Robinson, M.; Smith, G.J.; Ambrosini, G.L.; Piek, J.P.; Oddy, W.H. ADHD is associated with a “Western” dietary pattern in adolescents. J. Atten. Disord. 2011, 15, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.; Fischer, N.; Fritzenwanger, M.; Thude, H.; Barz, D.; Figulla, H.-R. Social and behavioural aspects and their consequences in obese teenagers: Importance of family’s history. Nutr. Hosp. 2009, 24, 693–700. [Google Scholar] [PubMed]
- Mahoney, J.L.; Larson, R.W.; Eccles, J.S. Organized Activities as Contexts of Development: Extracurricular Activities, after-School and Community Programs; Psychology Press: Mahwah, NJ, USA, 2005; pp. 3–22. [Google Scholar]
- TFDA. Nutrition Labeling Requirements. 2015. Available online: http://www.fda.gov.tw/TC/site.aspx?sid=3462 (accessed on 16 March 2016). (In Chinese) [Google Scholar]
- Chiodo, L.M.; Jacobson, S.W.; Jacobson, J.L. Neurodevelopmental effects of postnatal lead exposure at very low levels. Neurotoxicol. Teratol. 2004, 26, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Eubig, P.A.; Aguiar, A.; Schantz, S.L. Lead and PCBs as risk factors for attention deficit/hyperactivity disorder. Environ. Health Perspect. 2010, 118, 1654–1667. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, A.; Eubig, P.A.; Schantz, S.L. Attention deficit/hyperactivity disorder: A focused overview for children’s environmental health researchers. Environ. Health Perspect. 2010, 118, 1646–1653. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-L.; Chuang, H.-Y.; Ho, C.-K.; Yang, C.-Y.; Tsai, J.-L.; Wu, T.-S.; Wu, T.-N. Relationship between blood lead concentrations and learning achievement among primary school children in Taiwan. Environ. Res. 2002, 89, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.-C.; Su, P.-H.; Chen, H.-Y.; Huang, H.-B.; Tsai, J.-L.; Huang, H.-I.; Wang, S.-L. Childhood blood lead levels and intellectual development after ban of leaded gasoline in Taiwan: A 9-year prospective study. Environ. Int. 2012, 40, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Joshi, K.; Lad, S.; Kale, M.; Patwardhan, B.; Mahadik, S.P.; Patni, B.; Chaudhary, A.; Bhave, S.; Pandit, A. Supplementation with flax oil and vitamin C improves the outcome of Attention Deficit Hyperactivity Disorder (ADHD). Prostaglandins Leukot. Essent. Fat. Acids 2006, 74, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Goksugur, S.B.; Tufan, A.E.; Semiz, M.; Gunes, C.; Bekdas, M.; Tosun, M.; Demircioglu, F. Vitamin D status in children with attention-deficit-hyperactivity disorder. Pediatr. Int. 2014, 56, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.A.; Patel, D.R. Complementary and alternative medicine in developmental disabilities. Indian J. Pediatr. 2005, 72, 949–952. [Google Scholar] [CrossRef] [PubMed]
| Variables | Controls | ADHD | p-Value |
|---|---|---|---|
| n = 159 | n = 173 | ||
| Demographic characteristics | |||
| Gender (%) | <0.01 * | ||
| Female | 68 (42.8%) | 25 (14.5%) | |
| Male | 91 (57.2%) | 148 (85.5%) | |
| Body weight | 32.1 ± 11.6 | 33.8 ± 12.3 | 0.20 |
| Age (years) | 8.9 ± 2.0 | 9.2 ± 2.7 | 0.31 |
| Gestational age at birth (%) | 0.34 | ||
| <37 weeks | 15 (9.4%) | 22 (12.7%) | |
| ≥37 weeks | 144 (90.6%) | 151 (87.3%) | |
| Parity (%) | 0.80 | ||
| Primiparous | 87 (54.7%) | 97 (56.1%) | |
| Multiparous | 72 (45.3%) | 76 (43.9%) | |
| Birth Order (%) | 0.87 | ||
| 1st | 87 (54.7%) | 93 (54.7%) | |
| 2nd | 56 (35.2%) | 57 (33.5%) | |
| 3rd and above | 16 (10.1%) | 20 (11.8%) | |
| Paternal education level (%) | <0.01 * | ||
| High school or below | 39 (24.5%) | 98 (56.6%) | |
| College or advanced training | 120 (75.5%) | 75 (43.4%) | |
| Maternal education level (%) | <0.01 * | ||
| High school or below | 46 (28.9%) | 93 (53.8%) | |
| College or advanced training | 113 (71.1%) | 80 (46.2%) | |
| Family history of nervous system diseases (%) | 0.02 * | ||
| No | 130 (81.8%) | 123 (71.1%) | |
| Yes | 29 (18.2%) | 50 (28.9%) | |
| Maternal age at birth | 30.3 ± 4.4 | 30.1 ± 5.0 | 0.80 |
| History of still or dead birth (%) | 0.17 | ||
| No | 117 (73.6%) | 115 (66.5%) | |
| Yes | 42 (26.4%) | 58 (33.5%) | |
| Smoking during pregnancy (%) | 0.50 | ||
| No | 150 (94.3%) | 160 (92.5%) | |
| Yes | 9 (5.7%) | 13 (7.5%) | |
| Alcohol consumption during pregnancy (%) | <0.01 * | ||
| No | 152 (95.6%) | 151 (87.3%) | |
| Yes | 7 (4.4%) | 22 (12.7%) | |
| Chronic disease during pregnancy (%) | 0.16 | ||
| No | 147 (92.5%) | 152 (87.9%) | |
| Yes | 12 (7.5%) | 21 (12.1%) | |
| Variables | Controls | ADHD | p-Value |
|---|---|---|---|
| n = 159 | n = 101 | ||
| Dietary habits | |||
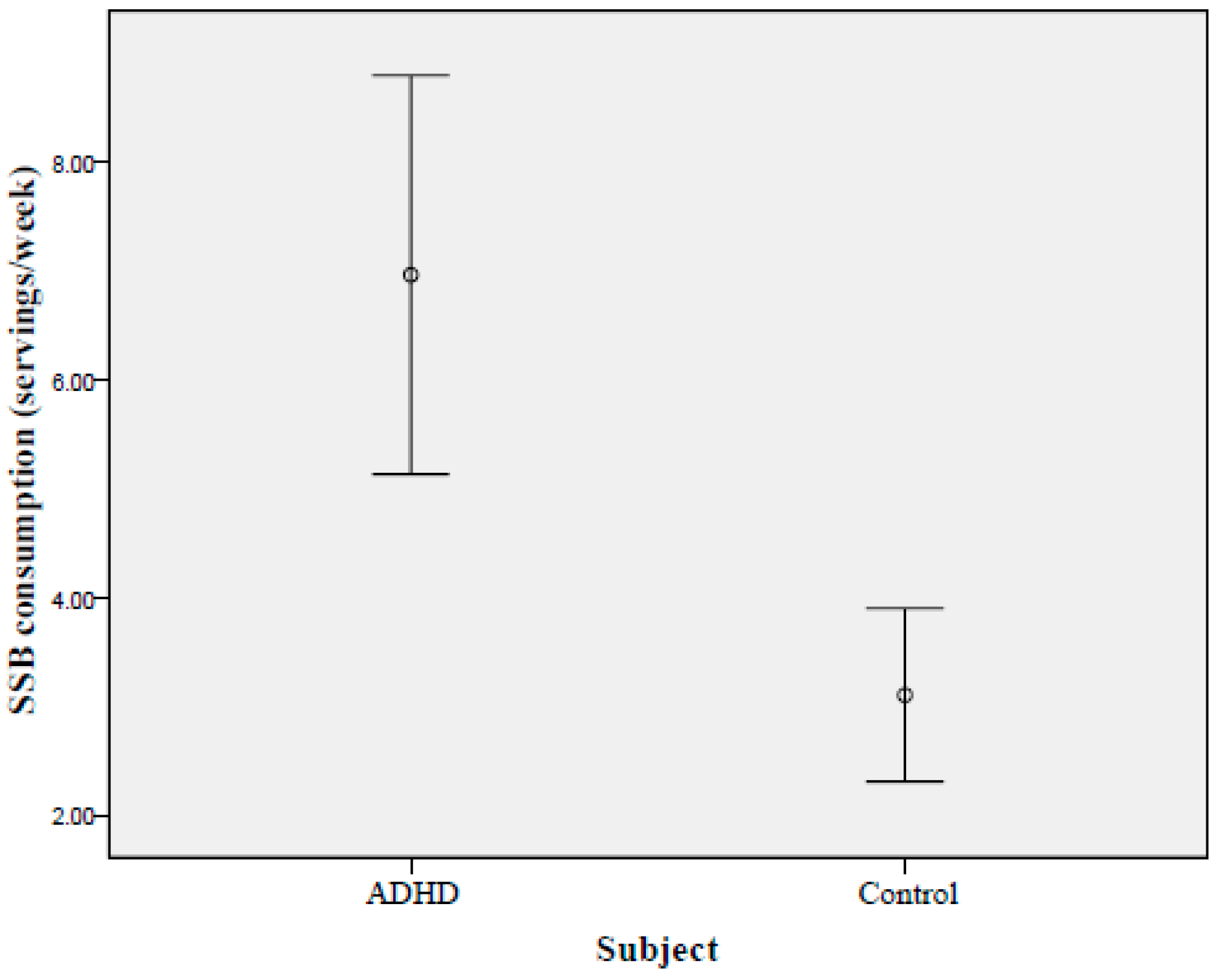
| Total SSBs consumed (servings/week) | <0.01 * | ||
| 3.10 ± 5.08 | 6.96 ± 9.27 | ||
| Categorized SSB consumption (servings/week) | <0.01 * | ||
| 0 | 51 (32.1%) | 19 (18.8%) | |
| 1–6 | 89 (56.0%) | 49 (48.5%) | |
| ≥7 | 19 (11.9%) | 33 (32.7%) | |
| Milk tea (%) | <0.01 * | ||
| 0 | 104 (65.4%) | 52 (51.5%) | |
| 1–6 | 51 (32.1%) | 37 (36.6%) | |
| ≥7 | 4 (2.5%) | 12 (11.9%) | |
| Juice or fruit-flavored drinks | <0.01 * | ||
| 0 | 109 (68.6%) | 50 (49.5%) | |
| 1–6 | 49 (30.8%) | 45 (44.6%) | |
| ≥7 | 1 (0.6%) | 6 (5.9%) | |
| Yakult drinks | <0.01 * | ||
| 0 | 108 (67.9%) | 49 (48.5%) | |
| 1–6 | 50 (31.4%) | 47 (46.5%) | |
| ≥7 | 1 (0.6%) | 5 (5.0%) | |
| Other SSBs | 0.11 | ||
| 0 | 91 (57.2%) | 45 (44.6%) | |
| 1–6 | 63 (39.6%) | 50 (49.5%) | |
| ≥7 | 5 (3.1%) | 6 (5.9%) | |
| Rice/Noodle (servings/week) | 20.53 ± 5.30 | 20.42 ± 4.62 | 0.34 |
| Meat (servings/week) | 14.02 ± 9.98 | 18.14 ± 13.89 | 0.04 * |
| Milk (servings/week) | 3.33 ± 2.93 | 5.01 ± 5.02 | 0.02 * |
| Eggs (servings/week) | 4.32 ± 2.78 | 4.43 ± 3.01 | 0.77 |
| Vegetables (servings/week) | 16.61 ± 12.64 | 11.97 ± 9.04 | <0.01 * |
| Fruits (servings/week) | 5.70 ± 3.61 | 4.78 ± 3.68 | 0.02 * |
| Fish (servings/week) | 5.92 ± 7.74 | 6.66 ± 8.22 | 0.65 |
| Shellfish (servings/week) | 1.98 ± 3.24 | 1.92 ± 3.08 | 0.79 |
| Other types of seafood (servings/week) | 1.12 ± 1.86 | 1.36 ± 2.32 | 0.94 |
| Caloric intake (day) | |||
| All | |||
| From foods other than SSBs | 1660.2 ± 352.2 | 1733.9 ± 386.6 | 0.07 |
| From SSBs | 141.4 ± 225.2 | 327.2 ± 440.0 | <0.01 * |
| Total calories | 1801.6 ± 448.3 | 2061.1 ± 689.7 | <0.01 * |
| Girls | |||
| From foods other than SSBs | 1679.2 ± 392.8 | 1776.1 ± 415.5 | 0.27 |
| From SSBs | 118.6 ± 208.9 | 488.9 ± 592.9 | <0.01 * |
| Total calories | 1797.7 ± 482.0 | 2265.0 ± 897.5 | 0.04 * |
| Boys | |||
| From foods other than SSBs | 1646.1 ± 320.1 | 1724.7 ± 382.1 | 0.16 |
| From SSBs | 158.4 ± 236.9 | 292.1 ± 395.2 | 0.02 * |
| Total calories | 1804.5 ± 424.1 | 2016.9 ± 634.1 | 0.01 * |
| Ingested sugar from SSBs (g/week) | |||
| 117.3 ± 198.1 | 296.7 ± 419.5 | <0.01 * | |
| Variables | Crude | Adjusted 1 | ||
|---|---|---|---|---|
| OR (95% CI) | p Value | OR (95% CI) | p Value | |
| (A) All subjects | <0.01 * | 0.02 * | ||
| 0 | Reference | Reference | ||
| 1–6 | 1.48 (0.79–2.78) | 1.36 (0.61–3.05) | ||
| ≥7 | 4.66 (2.15–10.09) | 3.69 (1.291–10.60) | ||
| (B) Boys only | 0.01 * | 0.06 | ||
| 0 | Reference | Reference | ||
| 1–6 | 1.33 (0.65–2.73) | 1.05 (0.43–2.59) | ||
| ≥7 | 3.61 (1.47–8.88) | 3.54 (1.057–11.95) | ||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, C.-J.; Du, J.-C.; Chiou, H.-C.; Feng, C.-C.; Chung, M.-Y.; Yang, W.; Chen, Y.-S.; Chien, L.-C.; Hwang, B.; Chen, M.-L. Sugar-Sweetened Beverage Consumption Is Adversely Associated with Childhood Attention Deficit/Hyperactivity Disorder. Int. J. Environ. Res. Public Health 2016, 13, 678. https://doi.org/10.3390/ijerph13070678
Yu C-J, Du J-C, Chiou H-C, Feng C-C, Chung M-Y, Yang W, Chen Y-S, Chien L-C, Hwang B, Chen M-L. Sugar-Sweetened Beverage Consumption Is Adversely Associated with Childhood Attention Deficit/Hyperactivity Disorder. International Journal of Environmental Research and Public Health. 2016; 13(7):678. https://doi.org/10.3390/ijerph13070678
Chicago/Turabian StyleYu, Ching-Jung, Jung-Chieh Du, Hsien-Chih Chiou, Chun-Cheng Feng, Ming-Yi Chung, Winnie Yang, Ying-Sheue Chen, Ling-Chu Chien, Betau Hwang, and Mei-Lien Chen. 2016. "Sugar-Sweetened Beverage Consumption Is Adversely Associated with Childhood Attention Deficit/Hyperactivity Disorder" International Journal of Environmental Research and Public Health 13, no. 7: 678. https://doi.org/10.3390/ijerph13070678
APA StyleYu, C.-J., Du, J.-C., Chiou, H.-C., Feng, C.-C., Chung, M.-Y., Yang, W., Chen, Y.-S., Chien, L.-C., Hwang, B., & Chen, M.-L. (2016). Sugar-Sweetened Beverage Consumption Is Adversely Associated with Childhood Attention Deficit/Hyperactivity Disorder. International Journal of Environmental Research and Public Health, 13(7), 678. https://doi.org/10.3390/ijerph13070678





