Health Risk Assessment of Heavy Metals in Soils from Witwatersrand Gold Mining Basin, South Africa
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling and Sample Preparation
2.3. Soil Sample Analysis
3. Health Risk Assessment
3.1. Theory of Risk Assessment
3.1.1. Ingestion of Heavy Metals through Soil
3.1.2. Inhalation of Heavy Metals via Soil Particulates
3.1.3. Dermal Contact with Soil
3.2. Non-Carcinogenic Risk Assessment
3.3. Carcinogenic Risk Assessment
4. Results and Discussion
4.1. Concentrations of Heavy Metals in Soil from the Gold Mining Area
4.2. Non-Carcinogenic Risk of Heavy Metals for Adults and Children
4.3. Carcinogenic Risk Assessment of Heavy Metals for Adults and Children
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Duffus, J.H. “Heavy metals”—A meaningless term? Clinical chemistry section, commission on toxicology. Pure Appl. Chem. 2002, 74, 793–807. [Google Scholar]
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils, 14th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2007. [Google Scholar]
- Lane, T.W.; Morel, F.M. A biological function for cadmium in marine diatoms. Proc. Natl. Acad. Sci. USA 2009, 9, 462–431. [Google Scholar] [CrossRef] [PubMed]
- Kabata-Pendias, A. Trace Elements in Soil and Plants, 4th ed.; Taylor & Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- Agency for Toxic Substances and Disease Registry. Guidance for the Preparation of a Twenty First Set Toxicological Profile. 2007. Available online: http://www.atsdr.cdc.gov/toxprofiles/guidance/set_21_guidance.pdf (accessed on 12 May 2015). [Google Scholar]
- Agency for Toxic Substances and Disease Registry. Lead: Toxicological Profiles; Centers for Disease Control and Prevention: Atlanta, GA, USA, 1999. [Google Scholar]
- National Research Council. Arsenic in Drinking Water; National Research Council: Washington, DC, USA, 1999; pp. 251–257. [Google Scholar]
- United Nations Environmental Programme. Global Mercury Assessment; United Nations: Geneva, Switzerland, 2002.
- Podsiki, C. Chart of Heavy Metals, Their Salts and Other Compounds. 2008. Available online: http://www.conservation-us.org/docs/default-source/resource-guides/chart-of-heavy-metals-their-salts-and-other-compounds-nbsp-.pdf (accessed on 15 May 2015).
- Ogwuegbu, M.O.C.; Muhanga, W. Investigation of lead concentration in the blood of people in the copper belt province of Zambia. J. Environ. 2005, 1, 66–75. [Google Scholar]
- Khan, K.; Lu, Y.; Khan, H. Heavy metals in agricultural soils and crops and their health risks in Swat District, northern Pakistan. Food Chem. Toxicol. 2013, 58, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Chen, J.; Zhang, J. Heavy metals in rice and garden vegetables and their potential health risks to inhabitants in the vicinity of an industrial zone in Jiangsu, China. J. Environ. Sci. 2010, 22, 1792–1799. [Google Scholar] [CrossRef]
- Nolan, K. Copper toxicity syndrome. J. Orthomol. Psychiat. 2003, 12, 270–282. [Google Scholar]
- Winde, F.; Stoch, E.J. Part III: Planning and uncertainty—Lessons from history. In Threats and Opportunities for Post-Closure Development in Dolomitic Gold Mining Areas of the West Rand and Far West Rand (South Africa)—A Hydraulic View; Water SA: Pretoria, South Africa, 2010. [Google Scholar]
- Cairncross, E.; Kisting, S.; Liefferink, M.; van Wyk, D. Case Study on Extractive Industries Prepared for the Lancet Commission on Global Governance, South Africa. 2013. Available online: http://www.thejournalist.org.za/wp-content/uploads/2014/09/Lancet-Study-Gold-Platinum-Mines-in-SA.pdf (accessed on 19 May 2016).
- Liefferink, M. Assessing the Past and the Present Role of the National Nuclear Regulator as a Public Protector against Potential Health Injuries: The West and Far West Rand as Case Study. 2011. Available online: http://dspace.nwu.ac.za/handle/10394/6612?mode=full (accessed on 4 October 2015).
- Kamunda, C.; Mathuthu, M.; Madhuku, M. Assessment of radiological hazards from gold mine tailings in Gauteng province, South Africa. Int. J. Environ. Res. Public Health 2016, 13, 138. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Energy Agency. Measurement of Radionuclides in Food and the Environment—Technical Report Series No. 295; International Atomic Energy Agency: Vienna, Austria, 1989. [Google Scholar]
- U.S. Environmental Protection Agency. Framework for Determining a Mutagenic Mode of Action for Carcinogenicity: Review Draft. 2007. Available online: http://www.epa.gov/osa/mmoaframework/pdfs/MMOA-ERD-FINAL-83007.pdf (accessed on 3 October 2015). [Google Scholar]
- U.S. Environmental Protection Agency. Toxics Release Inventory: Public Data Release Report. 2001. Available online: www.epa.gov/tri/tridata/tri01 (accessed on 24 February 2015). [Google Scholar]
- Wang, X.; Sato, T.; Xing, B. Health risks of heavy metals to the general public in Tianjin, China via consumption of vegetables and fish. Sci. Total Environ. 2005, 350, 28–37. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. Risk Assessment Guidance for Superfund Volume 1: Human Health Evaluation Manual (Part A); Office of Emergency and Remedial Response: Washington, DC, USA, 1989. [Google Scholar]
- U.S. Environmental Protection Agency. Risk Assessment Guidance for Superfund Volume I: Human Health Evaluation Manual (Part E, Supplemental Guidance for Dermal Risk Assessment); USEPA: Washington, DC, USA, 2004. [Google Scholar]
- Department of Environmental Affairs. The Framework for the Management of Contaminated Land, South Africa. 2010. Available online: http://sawic.environment.gov.za/documents/562.pdf (accessed on 5 February 2016). [Google Scholar]
- U.S. Environmental Protection Agency. Human Health Evaluation Manual, Supplemental Guidance: Standard Default Exposure Factors; USEPA: Washington, DC, USA, 1991. [Google Scholar]
- Luo, X.S.; Ding, J.; Xu, B. Incorporating bioaccessibility into human health risk assessments of heavy metals in urban park soils. Sci. Total Environ. 2012, 424, 88–96. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. Recommended Use of BW3/4 as the Default Method in Derivation of the Oral Reference Dose. Available online: http://www.epa.gov/raf/publications/pdfs/recommended-use-of-bw34.pdf (accessed on 12 October 2015).
- Lee, D.-Y.; Lee, C.-H. Regulatory Standards of Heavy Metal Pollutants in Soil and Groundwater in Taiwan; National Taiwan University: Taipei, Taiwan, 2011. [Google Scholar]
- Mtunzi, F.M.; Dikio, E.D.; Moja, S.J. Evaluation of heavy metal pollution on soil in Vaderbijlpark, South Africa. Int. J. Environ. Monit. Anal. 2015, 3, 44–49. [Google Scholar] [CrossRef]
- Contaminated Land Exposure Assessment—Heavy Metal Guidelines in Soil: Assessment of Potentially Toxic Elements. Available online: www.yara.co.uk/images/6_Heavy_Metals_tcm430-99440.pdf (accessed on 18 February 2016).
- Environment Protection Authority of Australia. Classification and Management of Contaminated Soil for Disposal. Available online: http://epa.tas.gov.au/regulation/document?docid=55 (accessed on 7 March 2016).
- Atanassov, I. New Bulgarian soil pollution standards. Bulgarian J. Agric. Sci. 2007, 14, 68–75. [Google Scholar]
- Canadian Ministry of the Environment. Soil, Ground Water and Sediment Standards for Use under Part XV.1 of the Environmental Protection Act; Canadian Ministry of the Environment: Toronto, ON, Canada, 2009. [Google Scholar]
- Environmental Protection Ministry of China. Standards of Soil Environmental Quality of Agricultural Land; Environmental Protection Ministry of China: Beijing, China, 2015. [Google Scholar]
- He, Z.; Shentu, J.; Yan, X.; Baligar, V.C.; Zhang, T.; Stoffella, P.J. Heavy metal contamination of soils: Sources, indicators, and assessment. J. Environ. Indic. 2015, 9, 17–18. [Google Scholar]
- Chiroma, T.M.; Ebewele, R.O.; Hymore, K. Comparative assessment of heavy metal levels in soil, vegetables and urban grey waste water used for irrigation in Yola and Kano. Int. Ref. J. Eng. Sci. 2014, 3, 1–9. [Google Scholar]
- European Commission on Environment. Heavy Metals in Wastes. 2002. Available online: http://c.ymcdn.com/sites/www.productstewardship.us/resource/resmgr/imported/Heavy%20Metals%20in%20Waste.pdf (accessed on 22 January 2016).
- Government of South Africa. Regulation Gazette: No. 8454, Vol 490, No. 28755; Government of South Africa: Pretoria, South Africa, 2006.
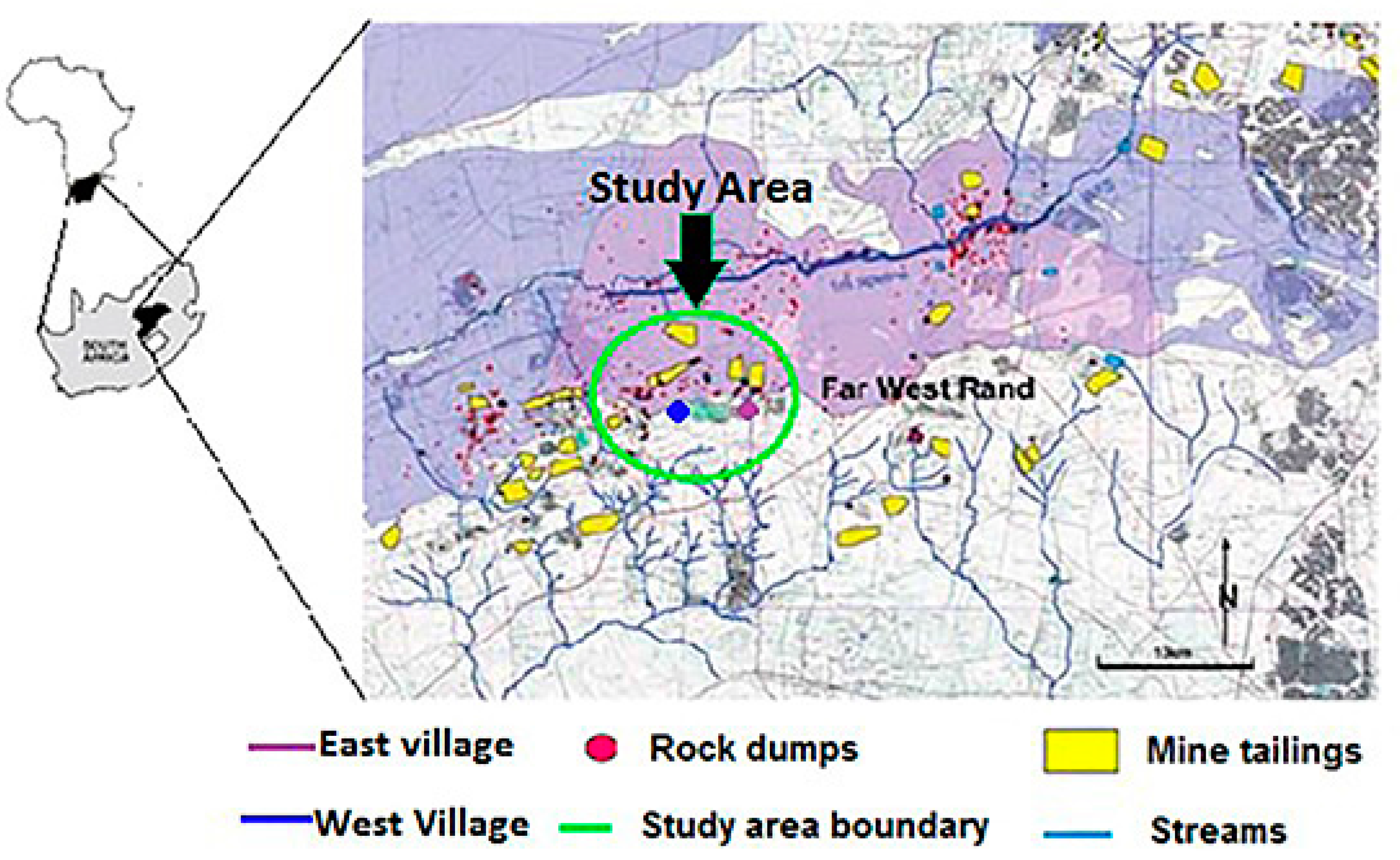
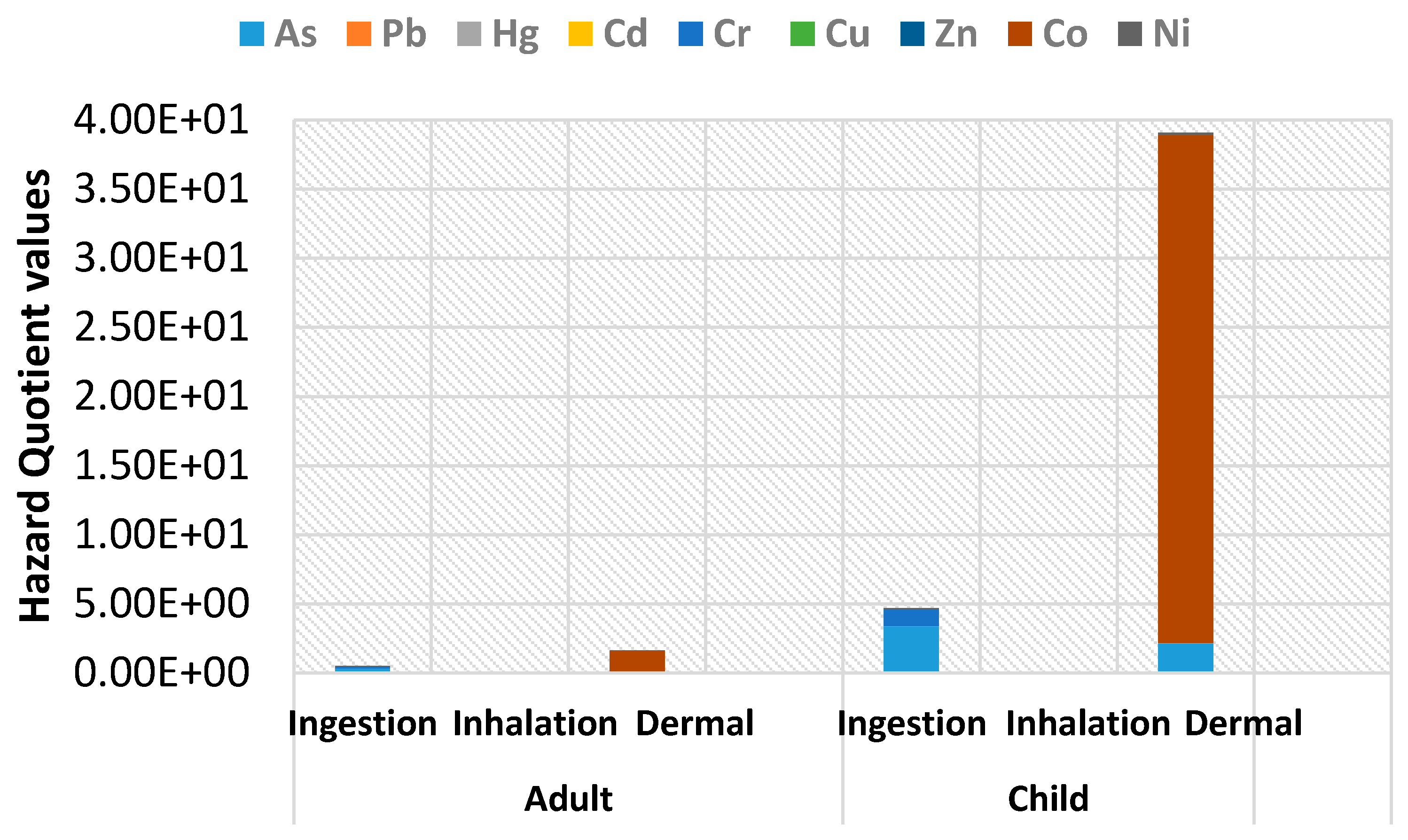
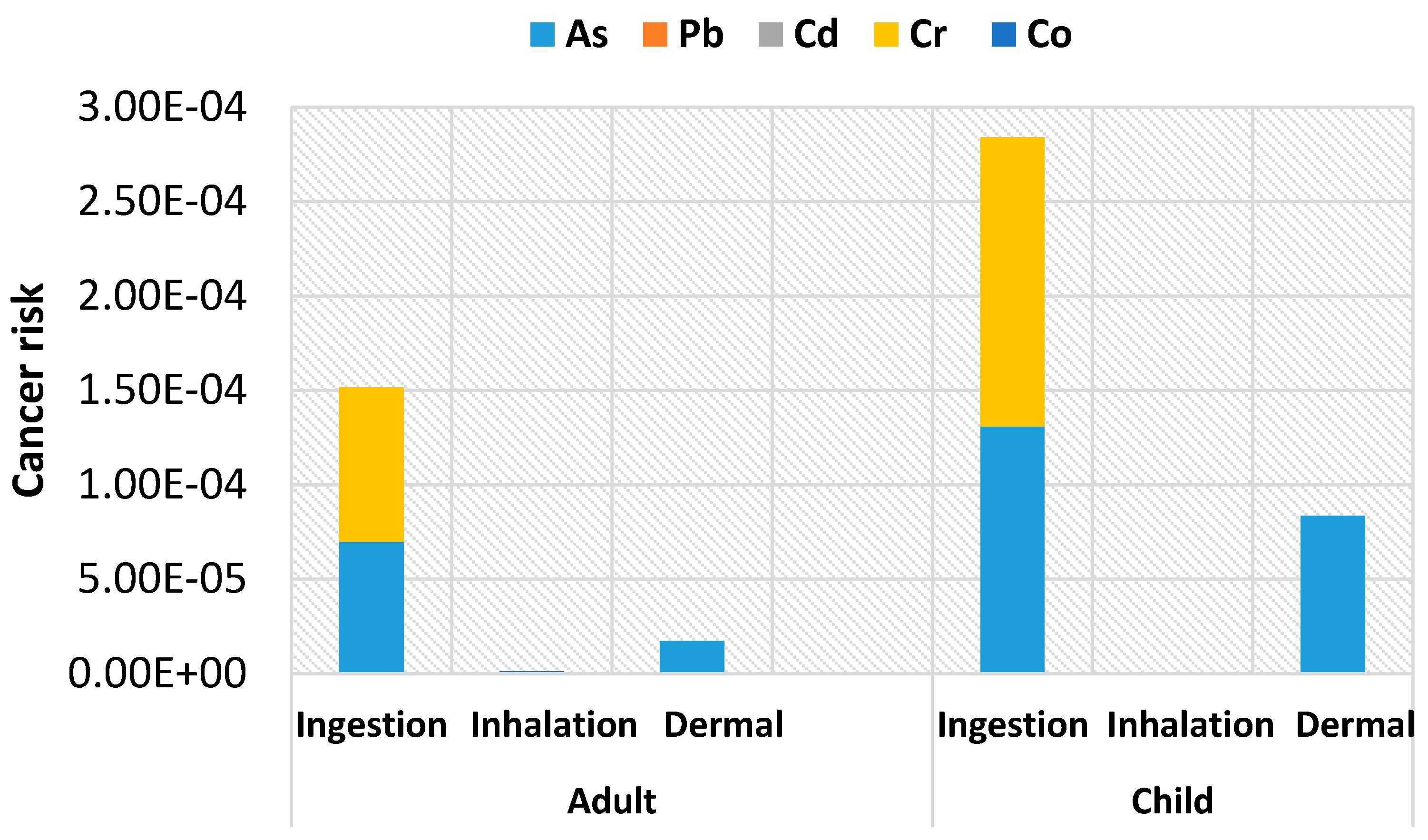
| Parameter | Unit | Child | Adult | References |
|---|---|---|---|---|
| Body weight (BW) | kg | 15 | 70 | [24] |
| Exposure frequency (EF) | days/year | 350 | 350 | [24] |
| Exposure duration (ED) | years | 6 | 30 | [24] |
| Ingestion rate (IR) | mg/day | 200 | 100 | [24] |
| Inhalation rate (IRair) | m3/day | 10 | 20 | [24] |
| Skin surface area (SA) | cm2 | 2100 | 5800 | [24] |
| Soil adherence factor (AF) | mg/cm2 | 0.2 | 0.07 | [24] |
| Dermal Absorption factor (ABS) | none | 0.1 | 0.1 | [24] |
| Dermal exposure ratio (FE) | none | 0.61 | 0.61 | [24] |
| Particulate emission factor (PEF) | m3/kg | 1.3 × 109 | 1.3 × 109 | [24] |
| Conversion factor (CF) | kg/mg | 10−6 | 10−6 | [23] |
| Average time (AT) | days | [24] | ||
| For carcinogens | 365 × 70 | 365 × 70 | [24] | |
| For non-carcinogens | 365 × ED | 365 × ED | [24] |
| Heavy Metal | Oral RfD | Dermal RfD | Inhalation RfD | Oral CSF | Dermal CSF | Inhalation CSF | References |
|---|---|---|---|---|---|---|---|
| As | 3.00E−04 | 3.00E−04 | 3.00E−04 | 1.50E+00 | 1.50E+00 | 1.50E+01 | [24,25] |
| Pb | 3.60E−03 | - | - | 8.50E−03 | - | 4.20E−02 | [24,26] |
| Hg | 3.00E−04 | 3.00E−04 | 8.60E−05 | - | - | - | [24] |
| Cd | 5.00E−04 | 5.00E−04 | 5.70E−05 | - | - | 6.30E+00 | [24,25] |
| Cr (VI) | 3.00E−03 | - | 3.00E−05 | 5.00E−01 | - | 4.10E+01 | [19,24] |
| Co | 2.00E−02 | 5.70E−06 | 5.70E−06 | - | - | 9.80E+00 | [27] |
| Ni | 2.00E−02 | 5.60E−03 | - | - | - | - | [24] |
| Cu | 3.7.00E−02 | 2.40E−02 | - | - | - | - | [24,27] |
| Zn | 3.00E−01 | 7.50E−02 | - | - | - | - | [24,27] |
| Location & GPS Points | No. of Samples | Average Concentrations of Heavy Metals in Different Locations in mg·kg−1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| As | Pb | Hg | Cd | Cr | Cu | Zn | Co | Ni | ||
| Tailings one (26°22’ S:27°29’ E) | 11 | 94.17 | 8.85 | 0.13 | 0.05 | 441.52 | 46.78 | 46.15 | 33.68 | 131.04 |
| Tailings two (26°22’ S:27°30’ E) | 13 | 115.19 | 10.22 | 0.13 | 0.05 | 270.76 | 45.48 | 51.95 | 31.76 | 115.08 |
| Tailings three (26°22’ S:27°26’ E) | 8 | 71.33 | 2.31 | 0.06 | 0.05 | 77.50 | 46.25 | 82.50 | 30.00 | 152.50 |
| Tailings four (26°21’ S:27°27’ E) | 12 | 73.18 | 2.96 | 0.07 | 0.04 | 104.17 | 55.83 | 60.00 | 21.67 | 99.83 |
| Tailings five (26°23’ S:27°25’ E) | 12 | 67.08 | 3.31 | 0.06 | 0.05 | 97.50 | 47.50 | 48.33 | 21.67 | 125.83 |
| West village (26°23’ S:27°28’ E) | 6 | 65.17 | 1.58 | 0.10 | 0.05 | 861.67 | 36.67 | 48.33 | 28.33 | 68.33 |
| East village (26°22’ S:27°30’ E) | 11 | 69.69 | 4.32 | 0.06 | 0.05 | 98.18 | 19.09 | 21.82 | 11.82 | 91.82 |
| Average | 79.40 | 4.79 | 0.09 | 0.05 | 278.76 | 42.51 | 51.30 | 25.56 | 112.06 | |
| Minimum | 65.17 | 1.58 | 0.06 | 0.04 | 77.50 | 19.09 | 21.82 | 11.82 | 68.33 | |
| Maximum | 115.19 | 10.22 | 0.13 | 0.05 | 861.67 | 55.83 | 82.50 | 33.68 | 152.50 | |
| Country | Maximum Allowable Limit of Concentrations of Heavy Metals in Soil (mg × kg−1) for Different Countries | References | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| As | Pb | Hg | Cd | Cr | Cu | Zn | Co | Ni | ||
| Germany | 50 | 70.0 | 0.5 | 1.0 | 60.0 | 40.0 | 150.0 | n.a. | 50.0 | [28] |
| Poland | n.a. | 100 | n.a. | 3 | 100 | 100 | 300 | 50 | 100 | [29] |
| UK | 32 | 450 | 10 | 10 | 130 | n.a. | n.a. | n.a. | 130 | [30] |
| Australia | 20 | 300 | 1 | 3 | 50 | 100 | 200 | n.a. | 60 | [31] |
| Taiwan | 60 | 300 | 2 | 5 | 250 | 200 | 600 | n.a. | 200 | [28] |
| Bulgaria | 10 | 26 | 0.03 | 0.4 | 65 | 34 | 88 | 20 | 46 | [32] |
| Canada | 20 | 200 | 0.8 | 3 | 250 | 150 | 500 | n.a. | 100 | [33] |
| China | 30 | 80 | 0.7 | 0.5 | 200 | 100 | 250 | n.a. | 50 | [34] |
| Tanzania | 1 | 200 | 2 | 1 | 100 | 200 | 150 | n.a. | 100 | [35] |
| FAO/WHO Guidelines | 20 | 100 | n.a. | 3 | 100 | 100 | 300 | 50 | 50 | [36] |
| EU Guidelines | n.a. | 300 | n.a. | 3 | 150 | 140 | 300 | n.a. | 75 | [37] |
| South Africa | 5.8 | 20 | 0.93 | 7.5 | 6.5 | 16 | 240 | 300 | 91 | [24] |
| Receptor | Pathway | Average Daily Intake (ADI) Values for Heavy Metals in mg/kg/day | Total | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| As | Pb | Hg | Cd | Cr | Cu | Zn | Co | Ni | |||
| Adult | Ingestion | 1.09E−04 | 6.57E−06 | 1.19E−07 | 6.61E−08 | 3.82E−04 | 5.82E−05 | 7.03E−05 | 3.50E−05 | 1.54E−04 | 8.14E−04 |
| Inhalation | 1.60E−08 | 9.66E−10 | 1.75E−11 | 9.72E−12 | 5.62E−08 | 8.56E−09 | 1.03E−08 | 5.15E−09 | 2.26E−08 | 1.20E−07 | |
| Dermal | 2.69E−05 | 1.63E−06 | 2.95E−08 | 1.64E−08 | 9.46E−05 | 1.44E−05 | 1.74E−05 | 8.67E−06 | 3.80E−05 | 2.02E−04 | |
| Total | 1.36E−04 | 8.19E−06 | 1.49E−07 | 8.25E−08 | 4.76E−04 | 7.27E−05 | 8.77E−05 | 4.37E−05 | 1.92E−04 | 1.02E−03 | |
| Child | Ingestion | 1.02E−03 | 6.13E−05 | 1.11E−06 | 6.17E−07 | 3.56E−03 | 5.44E−04 | 6.56E−04 | 3.27E−04 | 1.43E−03 | 7.60E−03 |
| Inhalation | 4.07E−08 | 2.46E−09 | 4.46E−11 | 2.47E−11 | 1.43E−07 | 2.18E−08 | 2.63E−08 | 1.31E−08 | 5.74E−08 | 3.05E−07 | |
| Dermal | 6.50E−04 | 3.92E−05 | 7.13E−07 | 3.95E−07 | 2.28E−03 | 3.48E−04 | 4.20E−04 | 2.09E−04 | 9.18E−04 | 4.87E−03 | |
| Total | 1.67E−03 | 1.01E−04 | 1.83E−06 | 1.01E−06 | 5.85E−03 | 8.92E−04 | 1.08E−03 | 5.36E−04 | 2.35E−03 | 1.25E−02 | |
| Receptor | Pathway | Average Daily Intake (ADI) Values for Heavy Metals in mg/kg/day | Total | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| As | Pb | Hg | Cd | Cr | Cu | Zn | Co | Ni | |||
| Adult | Ingestion | 4.66E−05 | 2.81E−06 | 5.11E−08 | 2.83E−08 | 1.64E−04 | 2.50E−05 | 3.01E−05 | 1.50E−05 | 6.58E−05 | 3.49E−04 |
| Inhalation | 6.86E−09 | 4.14E−10 | 7.51E−12 | 4.17E−12 | 2.41E−08 | 3.67E−09 | 4.43E−09 | 2.21E−09 | 9.67E−09 | 5.13E−08 | |
| Dermal | 1.15E−05 | 6.97E−07 | 1.27E−08 | 7.02E−09 | 4.05E−05 | 6.18E−06 | 7.46E−06 | 3.72E−06 | 1.63E−05 | 8.64E−05 | |
| Total | 5.82E−05 | 3.51E−06 | 6.38E−08 | 3.54E−08 | 2.04E−04 | 3.11E−05 | 3.76E−05 | 1.87E−05 | 8.21E−05 | 4.36E−04 | |
| Child | Ingestion | 8.70E−05 | 5.25E−06 | 9.54E−08 | 5.29E−08 | 3.05E−04 | 4.66E−05 | 5.62E−05 | 2.80E−05 | 1.23E−04 | 6.52E−04 |
| Inhalation | 3.49E−09 | 2.10E−10 | 3.82E−12 | 2.12E−12 | 1.22E−08 | 1.87E−09 | 2.25E−09 | 1.12E−09 | 4.92E−09 | 2.61E−08 | |
| Dermal | 5.57E−05 | 3.36E−06 | 6.11E−08 | 3.39E−08 | 1.96E−04 | 2.98E−05 | 3.60E−05 | 1.79E−05 | 7.87E−05 | 4.17E−04 | |
| Total | 1.43E−04 | 8.62E−06 | 1.56E−07 | 8.68E−08 | 5.01E−04 | 7.64E−05 | 9.22E−05 | 4.60E−05 | 2.01E−04 | 1.07E−03 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamunda, C.; Mathuthu, M.; Madhuku, M. Health Risk Assessment of Heavy Metals in Soils from Witwatersrand Gold Mining Basin, South Africa. Int. J. Environ. Res. Public Health 2016, 13, 663. https://doi.org/10.3390/ijerph13070663
Kamunda C, Mathuthu M, Madhuku M. Health Risk Assessment of Heavy Metals in Soils from Witwatersrand Gold Mining Basin, South Africa. International Journal of Environmental Research and Public Health. 2016; 13(7):663. https://doi.org/10.3390/ijerph13070663
Chicago/Turabian StyleKamunda, Caspah, Manny Mathuthu, and Morgan Madhuku. 2016. "Health Risk Assessment of Heavy Metals in Soils from Witwatersrand Gold Mining Basin, South Africa" International Journal of Environmental Research and Public Health 13, no. 7: 663. https://doi.org/10.3390/ijerph13070663
APA StyleKamunda, C., Mathuthu, M., & Madhuku, M. (2016). Health Risk Assessment of Heavy Metals in Soils from Witwatersrand Gold Mining Basin, South Africa. International Journal of Environmental Research and Public Health, 13(7), 663. https://doi.org/10.3390/ijerph13070663






