Abstract
Strongyloidiasis is a disease caused by soil transmitted helminths of the Strongyloides genus. Currently, it is predominately described as a neglected tropical disease. However, this description is misleading as it focuses on the geographical location of the disease and not the primary consideration, which is the socioeconomic conditions and poor infrastructure found within endemic regions. This classification may result in misdiagnosis and mistreatment by physicians, but more importantly, it influences how the disease is fundamentally viewed. Strongyloidiasis must be first and foremost considered as a disease of disadvantage, to ensure the correct strategies and control measures are used to prevent infection. Changing how strongyloidiasis is perceived from a geographic and clinical issue to an environmental health issue represents the first step in identifying appropriate long term control measures. This includes emphasis on environmental health controls, such as better infrastructure, sanitation and living conditions. This review explores the global prevalence of strongyloidiasis in relation to its presence in subtropical, tropical and temperate climate zones with mild and cold winters, but also explores the corresponding socioeconomic conditions of these regions. The evidence shows that strongyloidiasis is primarily determined by the socioeconomic status of the communities rather than geographic or climatic conditions. It demonstrates that strongyloidiasis should no longer be referred to as a “tropical” disease but rather a disease of disadvantage. This philosophical shift will promote the development of correct control strategies for preventing this disease of disadvantage.
1. Introduction
Strongyloidiasis is an underestimated disease caused by Strongyloides stercoralis and Strongyloides fuelleborni, two species of soil-transmitted helminths of the genus Strongyloides [1,2]. While Strongyloides fuelleborni is found sporadically in Africa and Papua New Guinea, Strongyloides stercoralis is distributed worldwide and clinically important [1]. Rhabditiform larvae of S. stercoralis are excreted in human feces, from where they develop into infected filariform larvae and can either repenetrate the intestinal mucosa and remain in the human organism, or distribute environmentally to new human hosts. A new host becomes infected with filariform larvae through intact skin penetration [3]. Strongyloidiasis can cause gastrointestinal symptoms, including abdominal pain, diarrhea, nausea and vomiting, skin problems including pruritus and dermatitis or respiratory symptoms such as cough, asthma and dyspnea [4,5,6]. Hyperinfection or disseminated strongyloidiasis can affect several organs, leading to fatal outcomes [1,7]. Chronic asymptomatic strongyloidiasis is another significant concern, as when coupled with immunosuppressive treatment, it has potential to develop into disseminated infection [8].
Currently, strongyloidiasis is predominately described as a neglected tropical disease, found in tropical and subtropical areas (Southeast Asia, Africa, Central and South America) [9,10,11,12,13,14]. Whilst these papers do not often include clear climate-area classifications, it seems inappropriate that the primary disease descriptor focuses on geographic and climate conditions. Recent studies have included countries of the temperate zones in the endemic areas for strongyloidiasis [15,16,17,18,19]. There are also cases of strongyloidiasis in some parts of the same climatic zone but not in others [20,21,22,23]. This indicates that climatic conditions are not the primary factors determining the disease presence. Few studies mentioned low sanitation and socioeconomic status of communities as a risk factor for strongyloidiasis, and those that did not examine socioeconomic and sanitation conditions in any depth [21,24].
This review explores the global prevalence of strongyloidiasis in relation to its presence in subtropical, tropical and temperate climate zones with mild and cold winters, but also explores the corresponding socioeconomic conditions of these regions. The review demonstrates that classifying the disease as “tropical” is misleading and runs the risk that physicians in other countries may not recognize this pathogen, resulting in misdiagnosis or mistreatment of the disease [15,25], but most importantly it influences how the disease is fundamentally viewed. Correct classification and perception of the strongyloidiasis is crucial, as it determines what strategies and control measures are used to prevent the infection. Considering the disease as an environmental health issue than a clinical issue based on geography would provoke a shift from drug administration to environmental health controls. Clinical treatment of strongyloidiasis will not always be effective [13,26]. Anthelminthic drugs do not prevent reinfection, and can also cause adverse health effects [27,28]. Additionally, resistance to ivermectin (the primary drug used to treat strongyloidiasis) has already been found in Strongyloides spp. infecting ruminants [29], suggesting that resistance to ivermectin in S. stercoralis is likely in the future. Therefore, environmental health interventions represent a safer and more effective way of infection treatment. It was more than twenty years ago that Grove [30] noted that controlling Strongyloides in the environment is the most effective way to reduce infection. He pointed out that installation of adequate waste disposal systems was the most effective method to control the nematode [30], although this has not become the primary approach to addressing the disease. A major step towards reducing Strongyloides infection is to change the global perception of strongyloidiasis as a neglected tropical disease to recognition that it is primarily a disease of disadvantage and poor sanitation.
The aim of the current review was to assess the global prevalence of S. stercoralis to determine prevalence in geographic locations or climate zones, and compare these with socioeconomic status and poor infrastructure of the communities. The review demonstrates that strongyloidiasis should no longer be referred to as a “tropical” disease but rather a disease of disadvantage. This philosophical shift will promote the development of correct control strategies for preventing the disease.
Studies that collectively demonstrate the global distribution of S. stercoralis are presented in Table 1. Studies were collated using the Google Scholar and PubMed journal databases and the key words Strongyloides, S. stercoralis, strongyloidiasis, global, socioeconomic, status. Only studies from 1990–2016, written in English or Russian, with S. stercoralis prevalence percentage and details on population studied were included in the review. Reference lists of the collected studies were also examined and relevant articles have been reviewed.

Table 1.
Global Strongyloides stercoralis prevalence distribution.
2. Global Prevalence of Strongyloidiasis and Climate Classification
Table 1 summarizes the information available on infection prevalence, population studied, country as the most likely infective source, climate and socioeconomic status of the country, type of the infection detection, presence of symptoms and the study reference details. The table indicates that all cases of strongyloidiasis occur in the following communities: poor communities, former war veterans, immigrants and travelers, immunocompromised populations, or groups occupationally exposed to soil.
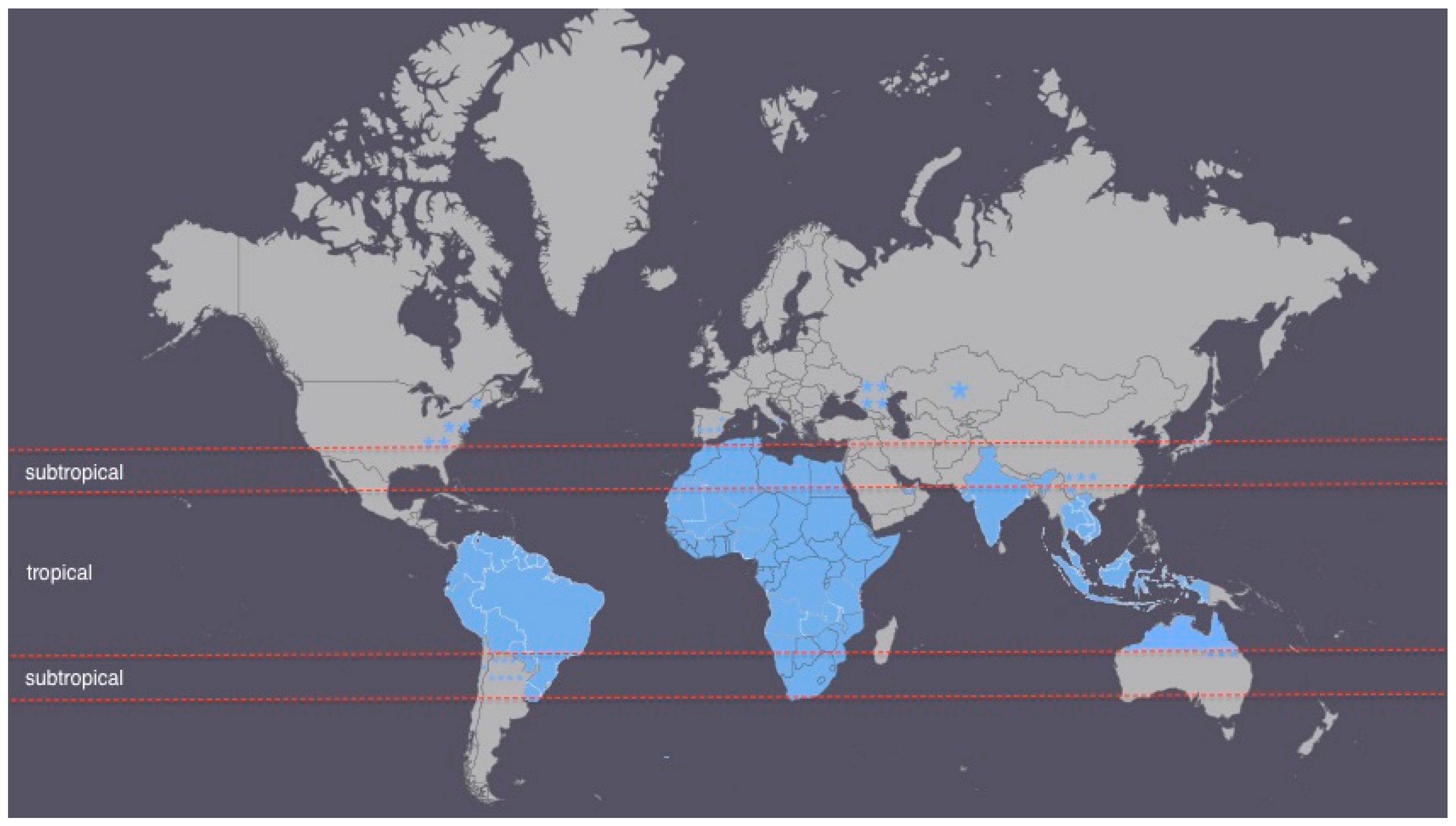
The climate classification used in this review is the Koppen climate classification system, which divides the world’s climate into six major climate groups each containing several subgroups [57]. Using the complete range of Koppen climate categories, about 80% of all the world areas falls into either tropical or subtropical zones [57]. This justifies the classification of strongyloidiasis as a “tropical” or “subtropical” disease, but lacks any meaning or association. Based on the major Koppen climate categories, the infection is still prevalent in other climate zones apart from tropical or subtropical ones (Figure 1). Certain areas of countries with Strongyloides stercoralis cases are shown as a “star” sign on the map.
Figure 1.
Countries with Strongyloides stercoralis cases (colored blue or marked as a “star” sign) on a world map divided into tropical and subtropical zones.
Figure 1 presents a world map divided into tropical and subtropical zones with the strongyloidiasis case countries/areas colored in blue. It can be seen that strongyloidiasis is highly prevalent in subtropical and tropical regions representing mostly developing countries with low socioeconomic status. Cases outside the tropical or subtropical areas correspond to more economically developed countries, but socioeconomically depressed communities (e.g., the Appalachia region population in the U.S., former USSR countries). This emphasizes that socioeconomic factors are more important than climatic conditions in defining the disease. The remaining cases presented are in risk groups of developed economy countries such as former war veterans, refugees, immigrants and travelers, immunosuppressed people or current or ex-farmers and their families, also identified by Schär et al. [24].
3. Countries of Strongyloidiasis Prevalence and Socioeconomic Status
3.1. Socioeconomic Status of the Strongyloidiasis Case Communities in Subtropical and Tropical Zones (Hyperendemic)
The socioeconomic status of the countries are presented in Table 1, based on their economy status and the income using World Bank data and the United Nations “World’s Economic Situation and Prospects 2016”report [58,59]. It is globally accepted that an area with S. stercoralis prevalence of more than 5% is considered hyperendemic [60]. From Table 1 it can be seen that almost all the reported countries are shown to be hyperendemic for strongyloidiasis, with exception of the Appalachia region in the U.S., Okinawa in Japan and North Caucasus in the former USSR. The reported endemic areas for strongyloidiasis (Southeast Asia, Africa, Central and South America) are mostly countries with developing economies, as can be seen in Table 1. Socioeconomic inequalities result in poor sanitation and hygienic conditions, which act as a triggering factor for the pathogen infection [17]. The lifecycle of S. stercoralis and a mode of infection transmission justifies the notion that improper sanitation conditions are risk factors for infection [3]. Increased urbanization processes happening in such countries cause inappropriate living conditions for the population such as 5–6 people living in one room and the use of one cubicle shower and a toilet [32]. It has been frequently shown that low socioeconomic status communities present higher mortality and morbidity rates compared to higher socioeconomic class population [61,62].
3.2. Socioeconomic Status of the Strongyloidiasis Case Communities in Temperate Zones
Apart from high prevalence strongyloidiasis cases detected in most of the subtropical and tropical countries in the world, cases with strongyloidiasis prevalence were also shown in some continental climate regions (Appalachia, North Caucasus, Kazakhstan). Although the study conducted in the North Caucasus does not meet the current review’s criterion for the year of publication of papers, it is still included as not many studies from that area are available. North Caucasus has a continental climate and the study findings highlight that strongyloidiasis is not dependent only on climatic conditions [55].
While moist and warm soil, enriched with nutrients are favourable conditions for the survival of free-living S. stercoralis larvae with further potential to infect a human host, the factors influencing direct or indirect development of infective filariform larvae (L3) are poorly understood [1,60]. Previous reports have indicated that larvae cannot survive temperatures below 8 °C or above 40 °C [63]. However, studies have demonstrated S. stercoralis larvae surviving at lower temperatures infecting a human [55]. Considering the parthenogenesis and autoinfection features of this nematode, the likelihood of the larvae remaining and reproducing within the host is high. In conditions of inadequate sanitary and hygiene environment there is then a high risk of rhabditiform larvae excreted in stools passing to other human hosts.
As seen in Table 1, these regions belong to countries or a country with transitional or developed economies with the strongyloidiasis cases identified only in disadvantaged communities [21,22,55]. For example, rural Appalachia regions in Kentucky, West Virginia, Georgia and Tennessee in the United States are identified as areas with high infection prevalence among low socioeconomic status populations [21,64]. The Strongyloides infection case reported in Kazakhstan children were adopted children from orphanages, who probably were exposed to poor sanitary environments [22]. The study in the North Caucasus reported different levels of strongyloidiasis prevalence (0.1%–1.4%) in different areas with different temperatures (the lowest being 4 °C). Poor sanitary conditions were however reported in almost all the communities studied [55]. These single Strongyloides infection cases occurred in areas of continental climate, where the precipitation level is low and temperatures go below zero, demonstrating that strongyloidiasis is not primarily influenced by climate conditions but rather sanitary and hygiene factors.
Australia is known to have tropical and subtropical climates, however, strongyloidiasis there is frequently found among indigenous communities and not the general population [20,23]. Indigenous communities (Aborigines and Torres Strait Islanders) are identified as of a low socioeconomic status populations and are generally reported to live in poorer housing, sanitary and infrastructure conditions, which results in numerous worse health outcomes compared with non-indigenous Australians [65].
3.3. Clinical Treatment of Strongyloidiasis and Infrastructure, Housing, and Environmental Health
Currently, anthelminthic drugs (albendazole, mebendazole, and ivermectin) and nemiticides are used to treat the strongyloidiasis in humans [66]. Treatment of soil-transmitted helminthiasis is difficult due to the development of resistance and facile reinfection from the environment. Among soil-transmitted helminth infections, strongyloidiasis is the most challenging to treat and clinically important because of a parasite’s rhabditiform larvae unique ability of autoinfection [2,13,26,32]. Moreover, parthenogenesis allows for a single female parasite remaining in a host to reproduce reinfecting that person [1]. The drug treatment efficacy depends on number of factors including an individual’s immune system status, co-infection with HTLV-1, history of drug use, and bowel ileus [51,67,68,69]. Furthermore, monitoring treatment efficacy has some difficulties associated with the low sensitivity of fecal examination [13]. Additionally, the drugs can cause adverse effects, including liver disfunction, gastrointestinal symptoms (nausea, vomiting, loose stool, abdominal distension or pain), chest tightness or pain, itching, fever, cough and wheezing, dizziness, and neurological effects [27,28,70,71].
New anthelminthic drugs and nematicides have to be frequently introduced to the market due to quick resistance development in nematodes and great toxicity they produce to humans [26]. Resistance in nematodes to different drugs has been studied and demonstrated frequently in the veterinary field in the last decades [72,73,74]. This suggests that human-infecting nematodes are also likely, at some stage in the future, to become resistant to the available drugs. Indeed, studies on some drugs used against human nematodes have already reported low drug treatment efficacy, calling for great attention and warnings of possible resistance development [75,76]. Although it is more difficult to study and confirm anthelminthic resistance in human parasites due to number of factors, the potential for resistance is mostly overlooked and should be more carefully examined in drug treatment application [77].
It is well established that sanitary conditions, including housing and infrastructure, play the most vital role in determining health outcomes [78,79]. Overcrowding, poor ventilation, bad living conditions and inadequate sewerage systems create higher risks for infectious and parasitic diseases such as skin infections, respiratory infections and diarrheal diseases [80]. Thus, environmental health approaches such as ensuring better infrastructure and sanitation should be the primary approach to controlling Strongyloides in the environment. Only this approach will provide the most effective way of infection reduction.
Strongyloidiasis has been also reported in certain groups such as former war veterans, refugees, immigrants and travelers, immunocompromised people and people occupationally exposed to soil (Table 1). Poor sanitary and hygiene living conditions are common during times of war, which could explain cases of strongyloidiasis in former war veterans [16,40]. The Okinawa Prefecture area of Japan was reported to have a high prevalence of S. stercoralis infections during World War II, which decreased to about 0.5%–1.5% after the war years. This was associated with improved sanitary conditions and systematic monitoring for parasitic diseases after the war [81].
Studies of refugees and immigrants with high S. stercoralis infection prevalence have demonstrated an association with inadequate sanitary and hygienic conditions in their home countries, including lack of an access to shower and toilet facilities [4,8,19,31,33,36,38,41,50].
Individual health condition (immunosuppressed or immunocompromised status) is another risk factor influencing the disease [5]. S. stercoralis is especially life-threatening to immunocompromised people due to possible development of the disseminated disease form [5,8] which approaches a 90% mortality rate [5,82]. The study by Zaha et al. [81] demonstrated that there was a high prevalence of S. stercoralis among Human T-Lymphotropic Virus type I (HTLV-1) positive patients (17.5%) compared to HTLV-1 negative patients (6.7%). Schar et al. [24] found an association between strongyloidiasis and HIV infection (OR: 2.17 BCI: 1.18–4.01) and alcoholism (OR: 6.69; BCI: 1.47–33.8). HTLV-1 and HIV infections and alcoholism have been associated with poverty [61,83,84,85].
High prevalence of strongyloidiasis in subtropical South China has been reported by Wang et al. [86]. While the cases reported are within subtropical areas, they are mostly associated with the farming lifestyle in those regions and/or poor hygiene practices. The infection rates in these areas are as high as 11%–14% [86]. The studies’ findings are not included in Table 1, as the original papers are only available in Chinese. Similarly, studies in France and Spain [37,42,87] reported strongyloidiasis cases in local current or ex-farmworkers and their family members who have never travelled to endemic areas. While there is no available information on the income of the studied population, ingestion of non-potable water and possible infection transmission to family members due to unhygienic behavior is reported in one of the studies [42]. This might indicate either inappropriate living conditions due to the depressed socioeconomic status in the area or population unawareness of proper hygienic and sanitary standards. On the other hand, in another study by Roman-Sanchez et al. [37] the assessed area (Gandia, Valencia), is reported to have the highest per capita income compared to other European Union regions, adequate hygiene-sanitary conditions and high prevalence of the strongyloidiasis. Whether the use of a more sensitive detection method, the agar-plate culture technique, compared to other studies impacted on this result cannot be known until several studies using the same detection tests are conducted. It can, however, be concluded that occupation is likely to contribute to acquiring the infection in this case.
Currently, it is estimated that between 30–100 million people are infected by Strongyloides worldwide [2,88]. There is however a general consensus amongst the scientific community that the prevalence is underestimated due to inadequate diagnostic techniques [88], and the lack of sensitivity in tests for S. stercoralis and the similarity of its symptoms to other diseases result in great underestimation of the infection and 300 million people infected globally is probably a more accurate estimate [47,89]. Misclassification of the disease may also be contributing to the underestimation of its prevalence. Diagnostic test methods are presented in Table 1 for completeness.
4. Conclusions
It is well established that strongyloidiasis is mainly restricted to tropical and subtropical areas throughout the world. However, within these regions, exposure to infection with the helminth is strongly associated with poor sanitary and living conditions. Thus, immigrants, refugees, travelers, war veterans, immunocompromised and occupationally soil-exposed groups—and their family members—are at especially high risk of strongyloidiasis. This review emphasizes that strongyloidiasis is a disease of disadvantage, and suggests that control measures to prevent the infection should focus as much, or more, on changing the environmental conditions that increase overall risks of the disease, as on the medical treatment of infected persons, especially since the latter is ineffective in preventing reinfection and has the potential for the development of drug resistance.
Acknowledgments
We have received no funds to publish in open access.
Author Contributions
Meruyert Beknazarova, Harriet Whiley and Kirstin Ross conceived and participated in review design and coordination. Meruyert Beknazarova drafted manuscript, and Harriet Whiley and Kirstin Ross provided academic input and all authors approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Grove, D.I. Human strongyloidiasis. Adv. Parasitol. 1995, 38, 251–309. [Google Scholar]
- Olsen, A.; van Lieshout, L.; Marti, H.; Polderman, T.; Polman, K.; Steinmann, P.; Stothard, R.; Thybo, S.; Verweij, J.J.; Magnussen, P. Strongyloidiasis—The most neglected of the neglected tropical diseases? Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Ericsson, C.D.; Steffen, R.; Siddiqui, A.A.; Berk, S.L. Diagnosis of Strongyloides stercoralis infection. Clin. Infect. Dis. 2001, 33, 1040–1047. [Google Scholar]
- Caruana, S.R.; Kelly, H.A.; Ngeow, J.Y.; Ryan, N.J.; Bennett, C.M.; Chea, L.; Nuon, S.; Bak, N.; Skull, S.A.; Biggs, B.A. Undiagnosed and potentially lethal parasite infections among immigrants and refugees in australia. J. Travel Med. 2006, 13, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, N.S.; Moro, R.N.; Sheth, A.N.; Montgomery, S.P.; Steurer, F.; McAuliffe, I.T.; Wang, Y.F.; Armstrong, W.; Rivera, H.N.; Lennox, J.L. High prevalence of persistent parasitic infections in foreign-born, hiv-infected persons in the United States. PLoS Negl. Trop. Dis. 2011, 5, e1034. [Google Scholar] [CrossRef] [PubMed]
- Mascarello, M.; Gobbi, F.; Angheben, A.; Gobbo, M.; Gaiera, G.; Pegoraro, M.; Lanzafame, M.; Buonfrate, D.; Concia, E.; Bisoffi, Z. Prevalence of Strongyloides stercoralis infection among HIV-positive immigrants attending two Italian hospitals, from 2000 to 2009. Ann. Trop. Med. Parasitol. 2011, 105, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Croker, C.; Reporter, R.; Redelings, M.; Mascola, L. Strongyloidiasis-related deaths in the United States, 1991–2006. Am. J. Trop. Med. Hyg. 2010, 83, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Seybolt, L.M.; Christiansen, D.; Barnett, E.D. Diagnostic evaluation of newly arrived asymptomatic refugees with Eosinophilia. Clin. Infect. Dis. 2006, 42, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Genta, R.M. Global prevalence of strongyloidiasis: Critical review with epidemiologic insights into the prevention of disseminated disease. Rev. Infect. Dis. 1989, 11, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Katz, K.; Krajden, S.; Fuksa, M.; Keystone, J.S.; Kain, K.C. Complicated and fatal strongyloides infection in canadians: Risk factors, diagnosis and management. Can. Med. Assoc. J. 2004, 171, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.F.; Hadip, F.; Ngui, R.; Lim, Y.A.; Mahmud, R. Serological and molecular detection of Strongyloides stercoralis infection among an orang asli community in Malaysia. Parasitol. Res. 2013, 112, 2811–2816. [Google Scholar] [CrossRef] [PubMed]
- Repetto, S.; Soto, C.A.; Cazorla, S.; Tayeldin, M.; Cuello, S.; Lasala, M.; Tekiel, V.; Cappa, S.G. An improved DNA isolation technique for PCR detection of Strongyloides stercoralis in stool samples. Acta Trop. 2013, 126, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Toma, H.; Sato, Y.; Shiroma, Y.; Kobayashi, J.; Shimabukuro, I.; Takara, M. Comparative studies on the efficacy of three anthelmintics on treatment of human strongyloidiasis in Okinawa, Japan. Southeast Asian J. Trop. Med. Public Health 2000, 31, 147–151. [Google Scholar] [PubMed]
- Uparanukraw, P.; Phongsri, S.; Morakote, N. Fluctuations of larval excretion in Strongyloides stercoralis infection. Am. J. Trop. Med. Hyg. 1999, 60, 967–973. [Google Scholar] [PubMed]
- Buonfrate, D.; Angheben, A.; Gobbi, F.; Muñoz, J.; Requena-Mendez, A.; Gotuzzo, E.; Mena, M.A.; Bisoffi, Z. Imported strongyloidiasis: Epidemiology, presentations, and treatment. Curr. Infect. Dis. Rep. 2012, 14, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Einsiedel, L.; Spelman, D. Strongyloides stercoralis: Risks posed to immigrant patients in an Australian tertiary referral centre. Int. Med. J. 2006, 36, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Steinmann, P.; Zhou, X.-N.; Du, Z.-W.; Jiang, J.-Y.; Wang, L.-B.; Wang, X.-Z.; Li, L.-H.; Marti, H.; Utzinger, J. Occurrence of Strongyloides stercoralis in Yunnan Province, China, and comparison of diagnostic methods. PLoS Negl. Trop. Dis. 2007, 1, e75. [Google Scholar] [CrossRef] [PubMed]
- Cabezas-Fernández, M.T.; Salas-Coronas, J.; Lozano-Serrano, A.B.; Vazquez-Villegas, J.; Cabeza-Barrera, M.I.; Cobo, F. Strongyloidiasis in immigrants in Southern Spain. Enferm. Infecc. Microbiol. Clín. 2015, 33, 37–39. [Google Scholar] [CrossRef] [PubMed]
- Valerio, L.; Roure, S.; Fernández-Rivas, G.; Basile, L.; Martínez-Cuevas, O.; Ballesteros, Á.-L.; Ramos, X.; Sabrià, M.; North Metropolitan Working Group on Imported Diseases. Strongyloides stercoralis, the hidden worm. Epidemiological and clinical characteristics of 70 cases diagnosed in the north metropolitan area of Barcelona, Spain, 2003–2012. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 465–470. [Google Scholar] [PubMed]
- Fisher, D.; McCarry, F.; Currie, B. Strongyloidiasis in the northern territory. Under-recognised and under-treated? Med. J. Aust. 1993, 159, 88–90. [Google Scholar] [PubMed]
- Russell, E.S.; Gray, E.B.; Marshall, R.E.; Davis, S.; Beaudoin, A.; Handali, S.; McAuliffe, I.; Davis, C.; Woodhall, D. Prevalence of Strongyloides stercoralis antibodies among a rural appalachian population—Kentucky, 2013. Am. J. Trop. Med. Hyg. 2014, 91, 1000–1001. [Google Scholar] [CrossRef] [PubMed]
- Peeters, E.; Meeus, M.; Ramet, J.; van Gompel, A.; Wojciechowski, M. 163 health status of children adopted from Kazakhstan in Belgium. Pediatr. Res. 2010, 68, 85–86. [Google Scholar] [CrossRef]
- Prociv, P.; Luke, R. Observations on strongyloidiasis in Queensland aboriginal communities. Med. J. Aust. 1993, 158, 160–163. [Google Scholar] [PubMed]
- Schär, F.; Trostdorf, U.; Giardina, F.; Khieu, V.; Muth, S.; Marti, H.; Vounatsou, P.; Odermatt, P. Strongyloides stercoralis: Global distribution and risk factors. PLoS Negl. Trop. Dis. 2013, 7, e2288. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.L.; Schneider, A.E.; Young, R.L.; Hinrichs, S.H.; Iwen, P.C. Strongyloides stercoralis infection in a non-endemic area. Lab. Med. 2013, 44, 339–343. [Google Scholar] [CrossRef]
- Molento, M.B. Parasite control in the age of drug resistance and changing agricultural practices. Vet. Parasitol. 2009, 163, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Zaha, O.; Hirata, T.; Kinjo, F.; Saito, A.; Fukuhara, H. Efficacy of ivermectin for chronic strongyloidiasis: Two single doses given 2 weeks apart. J. Infect. Chemother. 2002, 8, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Marti, H.; Haji, H.J.; Savioli, L.; Chwaya, H.M.; Mgeni, A.F.; Ameir, J.S.; Hatz, C. A comparative trial of a single-dose ivermectin versus three days of albendazole for treatment of Strongyloides stercoralis and other soil-transmitted helminth infections in children. Am. J. Trop. Med. Hyg. 1996, 55, 477–484. [Google Scholar] [PubMed]
- Maroto, R.; Jiménez, A.; Romero, J.; Alvarez, V.; de Oliveira, J.; Hernández, J. First report of anthelmintic resistance in gastrointestinal nematodes of sheep from Costa Rica. Vet. Med. Int. 2011, 2011, 145312. [Google Scholar] [CrossRef] [PubMed]
- Grove, D.I. A History of Human Helminthology; CAB International Wallingford: Oxford, UK, 1990. [Google Scholar]
- De Silva, S.; Saykao, P.; Kelly, H.; MacIntyre, C.; Ryan, N.; Leydon, J.; Biggs, B. Chronic Strongyloides stercoralis infection in laotian immigrants and refugees 7–20 years after resettlement in Australia. Epidemiol. Infect. 2002, 128, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Paula, F.; Costa-Cruz, J. Epidemiological aspects of strongyloidiasis in Brazil. Parasitol. Camb. 2011, 138, 1331. [Google Scholar] [CrossRef] [PubMed]
- Gibney, K.B.; Mihrshahi, S.; Torresi, J.; Marshall, C.; Leder, K.; Biggs, B.-A. The profile of health problems in African immigrants attending an infectious disease unit in Melbourne, Australia. Am. J. Trop. Med. Hyg. 2009, 80, 805–811. [Google Scholar] [PubMed]
- Yelifari, L.; Bloch, P.; Magnussen, P.; van Lieshout, L.; Dery, G.; Anemana, S.; Agongo, E.; Polderman, A.M. Distribution of human oesophagostomum bifurcum, hookworm and Strongyloides stercoralis infections in Northern Ghana. Trans. R. Soc. Trop. Med. Hyg. 2005, 99, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Nontasut, P.; Muennoo, C.; Sa-nguankiat, S.; Fongsri, S.; Vichit, A. Prevalence of strongyloides in Northern Thailand and treatment with ivermectin vs. albendazole. Southeast Asian J. Trop. Med. Public Health 2005, 36, 442. [Google Scholar] [PubMed]
- Joseph, L.; Gyorkos, T.W.; Coupal, L. Bayesian estimation of disease prevalence and the parameters of diagnostic tests in the absence of a gold standard. Am. J. Epidemiol. 1995, 141, 263–272. [Google Scholar] [PubMed]
- Roman-Sanchez, P.; Pastor-Guzman, A.; Moreno-Guillen, S.; Igual-Adell, R.; Er-Generoso, S.S.; Tornero-Estebanez, C. High prevalence of Strongyloides stercoralis among farm workers on the Mediterranean Coast of Spain: Analysis of the predictive factors of infection in developed countries. Am. J. Trop. Med. Hyg. 2003, 69, 336–340. [Google Scholar] [PubMed]
- Posey, D.L.; Blackburn, B.G.; Weinberg, M.; Flagg, E.W.; Ortega, L.; Wilson, M.; Secor, W.E.; Sanders-Lewis, K.; Won, K.; Maguire, J.H. High prevalence and presumptive treatment of schistosomiasis and strongyloidiasis among African refugees. Clin. Infect. Dis. 2007, 45, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.D.; Lindo, J.F.; Neva, F.A.; Gam, A.A.; Vogel, P.; Terry, S.I.; Cooper, E.S. Immunoepidemiologic studies of Strongyloides stercoralis and human T lymphotropic virus type I infections in Jamaica. J. Infect. Dis. 1994, 169, 692–696. [Google Scholar] [CrossRef] [PubMed]
- Gill, G.; Welch, E.; Bailey, J.; Bell, D.; Beeching, N. Chronic Strongyloides stercoralis infection in former British far east prisoners of war. QJM 2004, 97, 789–795. [Google Scholar] [CrossRef] [PubMed]
- Gyorkos, T.W.; Genta, R.M.; Viens, P.; Maclean, J.D. Seroepidemiology of strongyloides infection in the Southeast Asian refugee population in Canada. Am. J. Epidemiol. 1990, 132, 257–264. [Google Scholar] [PubMed]
- Sánchez, P.R.; Guzman, A.P.; Guillen, S.M.; Adell, R.I.; Estruch, A.M.; Gonzalo, I.N.; Olmos, C.R. Endemic strongyloidiasis on the Spanish Mediterranean Coast. QJM 2001, 94, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Jongsuksuntigul, P.; Intapan, P.; Wongsaroj, T.; Nilpan, S.; Singthong, S.; Veerakul, S.; Maleewong, W. Prevalence of Strongyloides stercoralis infection in Northeastern Thailand (agar plate culture detection). J. Med. Assoc.Thail. 2003, 86, 737–741. [Google Scholar]
- Devi, U.; Borkakoty, B.; Mahanta, J. Strongyloidiasis in Assam, India: A community-based study. Trop. Parasitol. 2011, 1, 30. [Google Scholar] [PubMed]
- Alzain, B.F. Study on the status of prevalence of Strongyloides stercoralis infection among children in agricultural areas in Beit Lahia, Gaza Strip. Islam Univ. J. 2006, 14, 67–73. [Google Scholar]
- Feitosa, G.; Bandeira, A.C.; Sampaio, D.P.; Badaró, R.; Brites, C. High prevalence of giardiasis and strongyloidiasis among HIV-infected patients in Bahia, Brazil. Braz. J. Infect. Dis. 2001, 5, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Krolewiecki, A.J.; Ramanathan, R.; Fink, V.; McAuliffe, I.; Cajal, S.P.; Won, K.; Juarez, M.; di Paolo, A.; Tapia, L.; Acosta, N. Improved diagnosis of Strongyloides stercoralis using recombinant antigen-based serologies in a community-wide study in Northern Argentina. Clin. Vaccine Immunol. 2010, 17, 1624–1630. [Google Scholar] [CrossRef] [PubMed]
- Safdar, A.; Malathum, K.; Rodriguez, S.J.; Husni, R.; Rolston, K.V. Strongyloidiasis in patients at a comprehensive cancer center in the United States. Cancer 2004, 100, 1531–1536. [Google Scholar] [CrossRef] [PubMed]
- Sithithaworn, P.; Srisawangwong, T.; Tesana, S.; Daenseekaew, W.; Sithithaworn, J.; Fujimaki, Y.; Ando, K. Epidemiology of Strongyloides stercoralis in North-East Thailand: Application of the agar plate culture technique compared with the enzyme-linked immunosorbent assay. Trans. R. Soc. Trop. Med. Hyg. 2003, 97, 398–402. [Google Scholar] [CrossRef]
- Sudarshi, S.; Stümpfle, R.; Armstrong, M.; Ellman, T.; Parton, S.; Krishnan, P.; Chiodini, P.L.; Whitty, C.J. Clinical presentation and diagnostic sensitivity of laboratory tests for Strongyloides stercoralis in travellers compared with immigrants in a non-endemic country. Trop. Med. Int. Health 2003, 8, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Sultana, Y.; Gilbert, G.L.; Ahmed, B.-N.; Lee, R. Strongyloidiasis in a high risk community of Dhaka, Bangladesh. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Babatunde, S.; Salami, A.; Fabiyi, J.; Agbede, O.; Desalu, O. Prevalence of intestinal parasitic infestation in HIV seropositive and seronegative patients in Ilorin, Nigeria. Ann. Afr. Med. 2010, 9, 3. [Google Scholar]
- Naves, M.M.; Costa-Cruz, J.M. High prevalence of Strongyloides stercoralis infection among the elderly in Brazil. Rev. Inst. Med. Trop. Sao Paulo 2013, 55, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Khieu, V.; Schär, F.; Forrer, A.; Hattendorf, J.; Marti, H.; Duong, S.; Vounatsou, P.; Muth, S.; Odermatt, P. High prevalence and spatial distribution of Strongyloides stercoralis in rural Cambodia. PLoS Negl. Trop. Dis. 2014, 8, e2854. [Google Scholar] [CrossRef] [PubMed]
- Prokhorov, A.; Golovan, T. Epidemiology of strongyloidiasis in Northern Caucasus, USSR. Med. Parazitol. Parazit. Bolezn. 1983, 61, 34–38. [Google Scholar]
- Hirata, T.; Nakamura, H.; Kinjo, N.; Hokama, A.; Kinjo, F.; Yamane, N.; Fujita, J. Prevalence of blastocystis hominis and Strongyloides stercoralis infection in Okinawa, Japan. Parasitol. Res. 2007, 101, 1717–1719. [Google Scholar] [CrossRef] [PubMed]
- Pidwirny, M. Köppen climate classification system. Retr. Jan. 2011, 6, 2015. [Google Scholar]
- United Nations. Worlds Economic Situation and Prospects 2016; United Nations: New York, NY, USA, 2016. [Google Scholar]
- Pasquali, V. Countries by Income Group. Available online: https://www.gfmag.com/global-data/economic-data/pagfgt-countries-by-income-group?page=1 (accessed on 17 May 2016).
- Grove, D.I. Strongyloidiasis: A Major Roundworm Infection of Man; Taylor and Francis Ltd.: London, UK, 1989. [Google Scholar]
- Adler, N.E.; Ostrove, J.M. Socioeconomic status and health: What we know and what we don’t. Ann. N. Y. Acad. Sci. 1999, 896, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Feinstein, J.S. The relationship between socioeconomic status and health: A review of the literature. Milbank Q. 1993, 279–322. [Google Scholar] [CrossRef]
- Farrar, J.; Hotez, P.; Junghanss, T.; Kang, G.; Lalloo, D.; White, N.J. Manson’s Tropical Diseases; Elsevier Health Sciences: London, UK, 2013. [Google Scholar]
- Walzer, P.D.; Milder, J.E.; Banwell, J.G.; Kilgore, G.; Klein, M.; Parker, R. Epidemiologic features of Strongyloides stercoralis infection in an endemic area of the United States. Am. J. Trop. Med. Hyg. 1982, 31, 313–319. [Google Scholar] [PubMed]
- Trewin, D.; Madden, R. The Health and Welfare of Australia’s Aboriginal and Torres Strait Islander Peoples. Canberra Aust. Bur. Stat. 2005. Available online: http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.156.2187&rep=rep1&type=pdf (accessed on 17 May 2016).
- Geneva, W.; Organization, W.H. Preventive chemotherapy in human helminthiasis. In Coordinated Use of Anthelminthic Drugs in Control Interventions: A Manual for Health Professionals and Programme Managers; WHO Press: Geneva, Switzerland, 2006. [Google Scholar]
- Vadlamudi, R.S.; Chi, D.S.; Krishnaswamy, G. Intestinal strongyloidiasis and hyperinfection syndrome. Clin. Mol. Allergy 2006, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Scowden, E.B.; Schaffner, W.; Stone, W.J. Overwhelming strongyloidiasis: An unappreciated opportunistic infection. Medicine 1978, 57, 527–544. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, E.; da Fonseca Porto, A. Epidemiological and clinical interaction between htlv-1 and Strongyloides stercoralis. Parasite Immunol. 2004, 26, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Shikiya, K.; Zaha, O.; Niimura, S.; Uehara, T.; Ohshiro, J.; Kinjo, F.; Saito, A.; Asato, R. Clinical study on ivermectin against 125 strongyloidiasis patients. Kansenshogaku Zasshi. 1994, 68, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Lichtenberger, P.; Rosa-Cunha, I.; Morris, M.; Nishida, S.; Akpinar, E.; Gaitan, J.; Tzakis, A.; Doblecki-Lewis, S. Hyperinfection strongyloidiasis in a liver transplant recipient treated with parenteral ivermectin. Transpl. Infect. Dis. 2009, 11, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Prichard, R. Anthelmintic resistance. Vet. Parasitol. 1994, 54, 259–268. [Google Scholar] [CrossRef]
- Coles, G.; Jackson, F.; Pomroy, W.; Prichard, R.; von Samson-Himmelstjerna, G.; Silvestre, A.; Taylor, M.; Vercruysse, J. The detection of anthelmintic resistance in nematodes of veterinary importance. Vet. Parasitol. 2006, 136, 167–185. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.M. Drug resistance in nematodes of veterinary importance: A status report. Trends Parasitol. 2004, 20, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Albonico, M.; Engels, D.; Savioli, L. Monitoring drug efficacy and early detection of drug resistance in human soil-transmitted nematodes: A pressing public health agenda for helminth control. Int. J. Parasitol. 2004, 34, 1205–1210. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, D.; Sacko, M.; Behnke, J.; Gilbert, F.; Dorny, P.; Vercruysse, J. Failure of mebendazole in treatment of human hookworm infections in the southern region of Mali. Am. J. Trop. Med. Hyg. 1997, 57, 25–30. [Google Scholar] [PubMed]
- Geerts, S.; Gryseels, B. Drug resistance in human helminths: Current situation and lessons from livestock. Clin. Microbiol. Rev. 2000, 13, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Cassel, J. The contribution of the social environment to host resistance. Am. J. Epidemiol. 1976, 104, 107–123. [Google Scholar] [PubMed]
- Audy, J.R.; Dunn, F.L. Community health. In Human Ecology; Sargent, F., Ed.; North Holland Publ. Co.: Amsterdam, The Netherland, 1974; pp. 345–363. [Google Scholar]
- Waters, A.-M. Do Housing Conditions Impact on Health Inequalities between Australia’s Rich and Poor? Final Report; Australian Housing and Urban Research Institute: Victoria, Australia, 2001. [Google Scholar]
- Zaha, O.; Hirata, T.; Kinjo, F.; Saito, A. Strongyloidiasis-progress in diagnosis and treatment. Int. Med. 2000, 39, 695–700. [Google Scholar] [CrossRef]
- Igra-Siegman, Y.; Kapila, R.; Sen, P.; Kaminski, Z.C.; Louria, D.B. Syndrome of hyperinfection with Strongyloides stercoralis. Rev. Infect. Dis. 1981, 3, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, C.; Rajapakse, S. HIV, poverty and women. Int. Health 2010, 2, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Fenton, L. Preventing HIV/AIDS through poverty reduction: The only sustainable solution? Lancet 2004, 364, 1186–1187. [Google Scholar] [CrossRef]
- Adler, N.E.; Boyce, T.; Chesney, M.A.; Cohen, S.; Folkman, S.; Kahn, R.L.; Syme, S.L. Socioeconomic status and health: The challenge of the gradient. Am. Psychol. 1994, 49, 15. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xu, J.; Zhou, X.; Li, J.; Yan, G.; James, A.A.; Chen, X. Strongyloidiasis: An emerging infectious disease in China. Am. J. Trop. Med. Hyg. 2013, 88, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Magnaval, J.-F.; Mansuy, J.-M.; Villeneuve, L.; Cassaing, S. A retrospective study of autochthonous strongyloidiasis in region Midi-Pyrenees (Southwestern France). Eur. J. Epidemiol. 2000, 16, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Bethony, J.; Brooker, S.; Albonico, M.; Geiger, S.M.; Loukas, A.; Diemert, D.; Hotez, P.J. Soil-transmitted helminth infections: Ascariasis, trichuriasis, and hookworm. Lancet 2006, 367, 1521–1532. [Google Scholar] [CrossRef]
- Taylor, M.J.; Garrard, T.A.; O’Donahoo, F.J.; Ross, K.E. Human strongyloidiasis: Identifying knowledge gaps, with emphasis on environmental control. Res. Rep. Trop. Med. 2014. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).