Dose-Response Relationship between Cumulative Occupational Lead Exposure and the Associated Health Damages: A 20-Year Cohort Study of a Smelter in China
Abstract
:1. Introduction
2. Methods
2.1. Research Subjects
2.2. Data Collection
2.3. Health Care and Monitoring
2.4. Laboratory Examinations
2.5. Calculation of Cumulative Exposure
2.6. Grouping Method
2.7. Data Processing and Analyses
3. Results
3.1. Basic Occupational Health
3.2. Correlation among Cumulative Lead Exposure, Lead Poisoning, and Biomarkers
3.3. Dose-Response Relationships between Cumulative Lead Exposure, Lead Poisoning and Biomarker ZPP
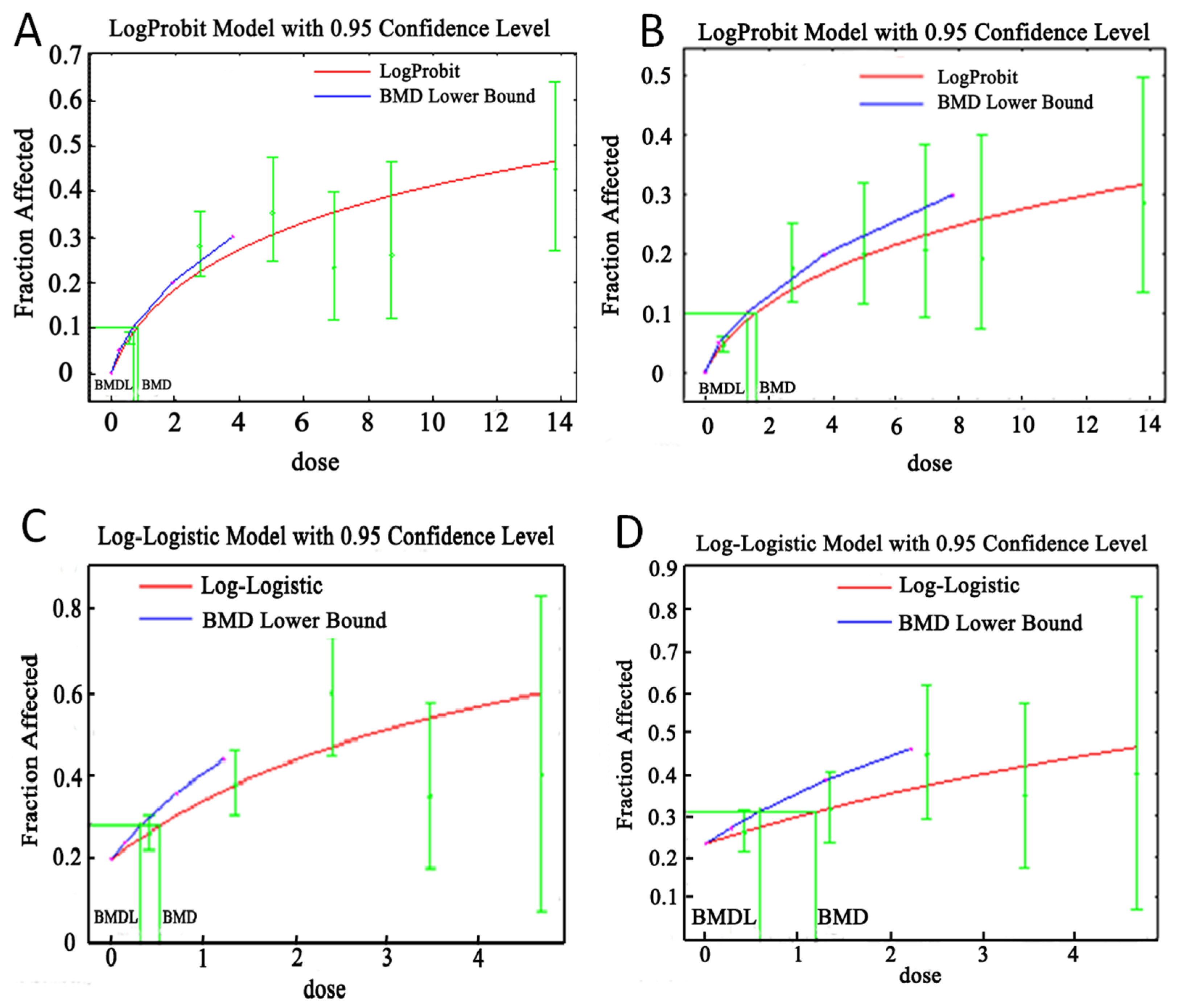
3.4. BMD and BMDLs for Lead Dust/Fume Exposure in Lead Poisoning and Other Biomarkers
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ALA | urinary δ-aminolevulinic acid |
| BMD | Benchmark dose |
| BMDLs | 95% lower confidence limit of the benchmark dose |
| CP | coproporphyrin |
| FEP | free erythrocyte protoporphyrin |
| OCELs | occupational cumulative exposure limits |
| PC-TWA | permissible concentration-time weighted average |
| T1/2 | half-life |
| TWA | time-weighted average |
| ZPP | erythrocyte zinc protoporphyrin |
References
- Nemsadze, K.; Sanikidze, T.; Ratiani, L.; Gabunia, L.; Sharashenidze, T. Mechanisms of lead-induced poisoning. Georgian Med. News 2009, 172–173, 92–96. [Google Scholar]
- Yilmaz, H.; Keten, A.; Karacaoglu, E.; Tutkun, E.; Akcan, R. Analysis of the hematological and biochemical parameters related to lead intoxication. J. Forensic Legal Med. 2012, 19, 452–454. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.J.; McWeeney, G.; Kim, R.; Tahirukaj, A.; Bulat, P.; Syla, S.; Savic, Z.; Amitai, Y.; Dignam, T.; Kaluski, D.N. Lead poisoning among internally displaced Roma, Ashkali and Egyptian children in the United Nations-Administered Province of Kosovo. Eur. J. Public Health 2010, 20, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, L.; Silvestroni, A.; Martino, M.G.; Gagliardi, T.; Corfiati, M.; Soleo, L. Evaluation of peripheral blood neutrophil leucocytes in lead-exposed workers. Int. Arch. Occup. Environ. Health 2006, 79, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Banner, W.; Kahn, C.M. Low blood lead level effects on intelligence: Can a dose-response curve be determined from the epidemiological data? Clin. Toxicol. 2014, 52, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Jakubowski, M. Low-level environmental lead exposure and intellectual impairment in children—The current concepts of risk assessment. Int. J. Occup. Med. Environ. Health 2011, 24, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Silveira, E.A.; Siman, F.D.; de Oliveira Faria, T.; Vescovi, M.V.; Furieri, L.B.; Lizardo, J.H.; Stefanon, I.; Padilha, A.S.; Vassallo, D.V. Low-dose chronic lead exposure increases systolic arterial pressure and vascular reactivity of rat aortas. Free Radic. Biol. Med. 2014, 67, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Sakai, T. Biomarkers of lead exposure. Ind. Health 2000, 38, 127–142. [Google Scholar] [CrossRef] [PubMed]
- Masci, O.; Carelli, G.; Vinci, F.; Castellino, N. Blood lead concentration and biological effects in workers exposed to very low lead levels. J. Occup. Environ. Med. 1998, 40, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Kosnett, M.J.; Wedeen, R.P.; Rothenberg, S.J.; Hipkins, K.L.; Materna, B.L.; Schwartz, B.S.; Hu, H.; Woolf, A. Recommendations for medical management of adult lead exposure. Environ. Health Perspect. 2007, 115, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Kosnett, M.J. Lead. In Poisoning and Drug Overdose: A Lange Clinical Manual, 2nd ed.; Olson, K.R., Ed.; Appleton and Lange: Norwalk, CT, USA, 1994; pp. 196–200. [Google Scholar]
- U.S. Department of Health and Human Services. Healthy People 2010, 2nd ed.Government Printing Office: Washington, DC, USA, 2000.
- Ortega, F.; Counter, S.A.; Buchanan, L.H.; Parra, A.M.; Collaguaso, M.A.; Jacobs, A.B. Tracking blood lead and zinc protoporphyrin levels in Andean adults working in a lead contaminated environment. J. Toxicol. Environ. Health. Part A 2013, 76, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Sun, D.; Zhou, Z.; Zhu, G.; Lei, L.; Zhang, H.; Chang, X.; Jin, T. Estimation of benchmark dose for bone damage and renal dysfunction in a Chinese male population occupationally exposed to lead. Ann. Occup. Hyg. 2008, 52, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.G.; Daston, G.P.; Evans, J.S.; Jarabek, A.M.; Kavlock, R.J.; Kimmel, C.A.; Park, C.; Spitzer, H.L. Benchmark Dose Workshop: Criteria for use of benchmark dose to estimate a reference dose. Regul. Toxicol. Pharmacol. 1995, 21, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Maxim, L.D.; Yu, C.P.; Oberdorster, G.; Utell, M.J. Quantitative risk analyses for RCF: Survey and synthesis. Regul. Toxicol. Pharmacol. 2003, 38, 400–416. [Google Scholar]
- Murata, K.; Iwata, T.; Dakeishi, M.; Karita, K. Lead toxicity: Does the critical level of lead resulting in adverse effects differ between adults and children? J. Occup. Health 2009, 51, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Tai-Yi, J. Benchmark dose approach for renal dysfunction in workers exposed to lead. Environ. Toxicol. 2007, 22, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Shao, B.; Jin, T.Y.; Wu, X.W.; Kong, Q.H.; Ye, T.T. Application of benchmark dose (BMD) in estimating biological exposure limit (BEL) to cadmium. Biomed. Environ. Sci. 2007, 20, 460–464. [Google Scholar] [PubMed]
- Wilker, E.; Korrick, S.; Nie, L.H.; Sparrow, D.; Vokonas, P.; Coull, B.; Wright, R.O.; Schwartz, J.; Hu, H. Longitudinal changes in bone lead levels: The VA Normative Aging Study. J. Occup. Environ. Med. 2011, 53, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.; Parsons, P.J.; Slavin, W. Transferability of blood lead determinations by furnace atomic absorption spectrophotometry and continuum background correction. Clin. Chem. 1995, 41, 1451–1454. [Google Scholar] [PubMed]
- U.S. EPA. Benchmark Dose Technical Guidance. EPA/100/R-12/001; 2012. Available online: https://www.epa.gov/sites/production/files/2015-01/documents/benchmark_dose_guidance.pdf (accessed on 14 March 2016). [Google Scholar]
- Wang, Q.; Ye, L.X.; Zhao, H.H.; Chen, J.W.; Zhou, Y.K. Benchmark dose approach for low-level lead induced haematogenesis inhibition and associations of childhood intelligences with ALAD activity and ALA levels. Sci. Total Environ. 2011, 409, 1806–1810. [Google Scholar] [CrossRef] [PubMed]
- Riva, M.A.; Lafranconi, A.; D'Orso, M.I.; Cesana, G. Lead poisoning: Historical aspects of a paradigmatic “occupational and environmental disease”. Saf. Health Work 2012, 3, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Kianoush, S.; Balali-Mood, M.; Mousavi, S.R.; Shakeri, M.T.; Dadpour, B.; Moradi, V.; Sadeghi, M. Clinical, toxicological, biochemical, and hematologic parameters in lead exposed workers of a car battery industry. Iran. J. Med. Sci. 2013, 38, 30–37. [Google Scholar] [PubMed]
- Ravichandran, B.; Ravibabu, K.; Raghavan, S.; Krishnamurthy, V.; Rajan, B.K.; Rajmohan, H.R. Environmental and biological monitoring in a lead acid battery manufacturing unit in India. J. Occup. Health 2005, 47, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, E.G.; Virji, M.A.; McClean, M.D.; Weinberg, J.; Woskie, S.; Pepper, L.D. Personal exposure, behavior, and work site conditions as determinants of blood lead among bridge painters. J. Occup. Environ. Hyg. 2010, 7, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Hodgkins, D.G.; Hinkamp, D.L.; Robins, T.G.; Schork, M.A.; Krebs, W.H. Influence of high past lead-in-air exposures on the lead-in-blood levels of lead-acid battery workers with continuing exposure. J. Occup. Med. 1991, 33, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Bledsoe, M.L.; Pinkerton, L.E.; Silver, S.; Deddens, J.A.; Biagini, R.E. Thyroxine and free thyroxine levels in workers occupationally exposed to inorganic lead. Environ. Health Insights 2011, 5, 55–61. [Google Scholar] [PubMed]
- Weuve, J.; Press, D.Z.; Grodstein, F.; Wright, R.O.; Hu, H.; Weisskopf, M.G. Cumulative exposure to lead and cognition in persons with Parkinson’s disease. Mov. Disord. 2013, 28, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Shih, R.; Rothenberg, S.; Schwartz, B.S. The epidemiology of lead toxicity in adults: Measuring dose and consideration of other methodologic issues. Environ. Health Perspect. 2007, 115, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.M.; Friesen, M.C.; Xiang, Y.B.; Cai, H.; Koh, D.H.; Ji, B.T.; Yang, G.; Li, H.L.; Locke, S.J.; Rothman, N.; et al. Occupational lead exposure and associations with selected cancers: The Shanghai Men’s and Women’s Health Study Cohorts. Environ. Health Perspect. 2016, 124, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Jangid, A.P.; John, P.J.; Yadav, D.; Mishra, S.; Sharma, P. Impact of chronic lead exposure on selected biological markers. Indian J. Clin. Biochem. 2012, 27, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Kavlock, R.J. Recent advances in mathematical modeling of developmental abnormalities using mechanistic information. Reprod. Toxicol. 1997, 11, 423–434. [Google Scholar] [CrossRef]
- Slob, W. Benchmark dose and the three Rs. Part II. Consequences for study design and animal use. Crit. Rev. Toxicol. 2014, 44, 568–580. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.X.; Gang, B.Q.; Gu, X.Q. The development of occupational exposure limits for chemical substances in China. Regul. Toxicol. Pharmacol. 1995, 22, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Far, H.S.; Pin, N.T.; Kong, C.Y.; Fong, K.S.; Kian, C.W.; Yan, C.K. An evaluation of the significance of mouth and hand contamination for lead absorption in lead-acid battery workers. Int. Arch. Occup. Environ. Health 1993, 64, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Askin, D.P.; Volkmann, M. Effect of personal hygiene on blood lead levels of workers at a lead processing facility. Am. Ind. Hyg. Assoc. J. 1997, 58, 752–753. [Google Scholar] [CrossRef] [PubMed]
| Grades | Diagnostic Criteria |
|---|---|
| Observed object | Have a close history of lead exposure, no clinical manifestations of lead poisoning, have one of the following performance: |
| |
| |
| Mild poisoning | Blood lead ≥ 60 μg/dL or urine lead ≥ 120 μg/L, and having one of the following performances: |
| |
| |
| |
| |
| Moderate poisoning | On the basis of mild poisoning, with one of the following symptoms: |
| |
| |
| |
| Severe poisoning | With one of the following manifestations: |
| |
|
| Characteristics | Lead Dust Exposure | Lead Fumes Exposure | Total | ||
|---|---|---|---|---|---|
| Men | Women | Men | Women | ||
| Number of subjects | 629 | 35 | 982 | 99 | 1745 |
| Age (years) | 30 (20–51) | 34 (22–49) | 30 (20–58) | 32 (20–52) | 30.5 (20–58) |
| Work seniority (years) | 7.9 (1.0–31.7) | 10.9 (1.8–26.6) | 8.7 (1.0–32.9) | 10.2 (1.0–28.5) | 9.0 (1.0–32.9) |
| Cumulative Dose (mg·y/m3) | N | Lead Poisoning (N) | Abnormal ZPP (N) |
|---|---|---|---|
| 0- | 913 | 68 | 41 |
| 2- | 79 | 22 | 14 |
| 4- | 37 | 13 | 7 |
| 6- | 25 | 6 | 6 |
| 8- | 27 | 9 | 8 |
| χ2 | 78.061 | 64.224 | |
| p-value | <0.001 | <0.001 | |
| Linear trend | |||
| χ2 | 56.319 | 58.191 | |
| p-value | <0.001 | <0.001 |
| Cumulative Dose (mg·y/m3) | N | Lead Poisoning (N) | Abnormal ZPP (N) |
|---|---|---|---|
| 0- | 441 | 114 | 83 |
| 1- | 148 | 56 | 37 |
| 2- | 47 | 28 | 17 |
| 3- | 28 | 10 | 10 |
| χ2 | 26.748 | 7.024 | |
| p-value | <0.001 | >0.05 | |
| Linear trend | |||
| χ2 | 17.249 | 5.284 | |
| p-value | <0.001 | <0.05 |
| Effect Indicator | Types of Airborne Lead | Model | BMD (mg·y/m3) | BMD (mg/m3) | BMDL (mg·y/m3) | BMDL (mg/m3) | χ2-Value | p-Value * |
|---|---|---|---|---|---|---|---|---|
| Lead poisoning | dust | Log-probit | 0.80 | 0.03 | 0.68 | 0.02 | 8.46 | 0.076 |
| fume | Log-logistic | 0.47 | 0.02 | 0.30 | 0.01 | 6.98 | 0.073 | |
| ZPP | dust | Log-probit | 1.62 | 0.05 | 1.30 | 0.04 | 2.39 | 0.665 |
| fume | Log-logistic | 0.65 | 0.02 | 0.44 | 0.02 | 3.06 | 0.383 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Y.; Gu, J.-M.; Huang, Y.; Duan, Y.-Y.; Huang, R.-X.; Hu, J.-A. Dose-Response Relationship between Cumulative Occupational Lead Exposure and the Associated Health Damages: A 20-Year Cohort Study of a Smelter in China. Int. J. Environ. Res. Public Health 2016, 13, 328. https://doi.org/10.3390/ijerph13030328
Wu Y, Gu J-M, Huang Y, Duan Y-Y, Huang R-X, Hu J-A. Dose-Response Relationship between Cumulative Occupational Lead Exposure and the Associated Health Damages: A 20-Year Cohort Study of a Smelter in China. International Journal of Environmental Research and Public Health. 2016; 13(3):328. https://doi.org/10.3390/ijerph13030328
Chicago/Turabian StyleWu, Yue, Jun-Ming Gu, Yun Huang, Yan-Ying Duan, Rui-Xue Huang, and Jian-An Hu. 2016. "Dose-Response Relationship between Cumulative Occupational Lead Exposure and the Associated Health Damages: A 20-Year Cohort Study of a Smelter in China" International Journal of Environmental Research and Public Health 13, no. 3: 328. https://doi.org/10.3390/ijerph13030328
APA StyleWu, Y., Gu, J.-M., Huang, Y., Duan, Y.-Y., Huang, R.-X., & Hu, J.-A. (2016). Dose-Response Relationship between Cumulative Occupational Lead Exposure and the Associated Health Damages: A 20-Year Cohort Study of a Smelter in China. International Journal of Environmental Research and Public Health, 13(3), 328. https://doi.org/10.3390/ijerph13030328






