Elevated Blood Lead Levels in Infants and Mothers in Benin and Potential Sources of Exposure
Abstract
:1. Introduction
2. Material and Methods
Ethics
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lanphear, B.P.; Hornung, R.; Khoury, J.; Yolton, K.; Baghurstl, P.; Bellinger, D.C.; Canfield, R.L.; Dietrich, K.N.; Bornschein, R.; Greene, T.; et al. (2005) Low-level environmental lead exposure and children’s intellectual function: An international pooled analysis. Environ. Health Perspect. 2005, 113, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Chiodo, L.M.; Jacobson, S.W.; Jacobson, J.L. Neurodevelopmental effects of postnatal lead exposure at very low levels. Neurotoxicol. Teratol. 2004, 26, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Plusquellec, P.; Muckle, G.; Dewailly, E.; Ayotte, P.; Begin, G.; Desrosiers, C.; Despres, C.; Saint-Amour, D.; Poitras, K. The relation of environmental contaminants exposure to behavioral indicators in Inuit preschoolers in Arctic Quebec. Neurotoxicology 2010, 31, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Landrigan, P.J. Neurobehavioural effects of developmental toxicity. Lancet Neurol. 2014, 13, 330–338. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Update on Blood Lead Levels in Children. Available online: http://wwwcdcgov/nceh/lead/ACCLPP/blood_lead_levelshtm (accessed on 3 March 2016).
- Ngueta, G.; Ndjaboue, R. Blood lead concentrations in sub-Saharan African children below 6 years: Systematic review. Trop Med. Int. Health 2013, 18, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Nriagu, J.O.; Blankson, M.L.; Ocran, K. Childhood lead poisoning in Africa: A growing public health problem. Sci. Total Environ. 1996, 181, 93–100. [Google Scholar] [CrossRef]
- Mathee, A. Towards the prevention of lead exposure in South Africa: Contemporary and emerging challenges. Neurotoxicology 2014, 45, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Kordas, K. Iron, Lead, and Children’s Behavior and Cognition. In Annual Review of Nutrition Cousins. Annu. Rev. Nutr. 2010, 30, 123–148. [Google Scholar] [CrossRef] [PubMed]
- Bodeau-Livinec, F.; Briand, V.; Berger, J.; Xiong, X.; Massougbodji, A.; Day, K.P.; Cot, M. Maternal anemia in Benin: Prevalence, risk factors, and association with low birth weight. Am. J. Trop Med. Hyg. 2011, 85, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.; Mombo-Ngoma, G.; Ouedraogo, S.; Kakolwa, M.A.; Abdulla, S.; Accrombessi, M.; Aponte, J.J.; Akerey-Diop, D.; Basra, A.; Briand, V.; et al. Intermittent preventive treatment of malaria in pregnancy with mefloquine in HIV-negative women: A multicenter randomized controlled trial. PLOS Med. 2014, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Koura, K.G.; Boivin, M.J.; Davidson, L.L.; Ouedraogo, S.; Zoumenou, R.; Alao, M.J.; Garcia, A.; Massougbodji, A.; Cot, M.; Bodeau-Livinec, F. Usefulness of child development assessments for low-resource settings in francophone Africa. J. Develop Behav. Pediat. 2013, 34, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Etchevers, A.; Bretin, P.; Lecoffre, C.; Bidondo, M.-L.; Le Strat, Y.; Glorennec, P.; Le Tertre, A. Blood lead levels and risk factors in young children in France, 2008–2009. Int. J. Hyg. Environ. Health 2014, 217, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Etchevers, A.; le Tertre, A.; Lucas, J.-P.; Bretin, P.; Oulhote, Y.; Le Bot, B.; Glorennec, P. Environmental determinants of different blood lead levels in children: A quantile analysis from a nationwide survey. Environ. Int. 2015, 74, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Elburg, M.; Vroon, P.; van der Wagt, B.; Tchalikian, A. Sr and Pb isotopic composition of five USGS glasses (BHVO-2G, BIR-1G, BCR-2G, TB-1G, NKT-1G). Chem. Geol. 2005, 223, 196–207. [Google Scholar] [CrossRef]
- Glorennec, P.; Peyr, C.; Poupon, J.; Oulhote, Y.; le Bot, B. Identifying sources of lead exposure for children, with lead concentrations and isotope ratios. J. Occup. Environ. Hyg. 2010, 7, 253–260. [Google Scholar] [CrossRef] [PubMed]
- CDC. Recommendations to prevent and control iron deficiency in the United States. MMWR 1998, 47, 1–29. [Google Scholar]
- Mathee, A.; Rollin, H.; von Schirnding, Y.; Levin, J.; Naik, I. Reductions in blood lead levels among school children following the introduction of unleaded petrol in South Africa. Environ. Res. 2006, 100, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, M.; Mathee, A. A preliminary study of residential paint lead concentrations in Johannesburg. Environ. Res. 2005, 98, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Graber, L.K.; Asher, D.; Anandaraja, N.; Bopp, R.F.; Merrill, K.; Cullen, M.R.; Luboga, S.; Trasande, L. Childhood lead exposure after the phaseout of leaded gasoline: An ecological study of school-age children in Kampala, Uganda. Environ. Health Perspect. 2010, 118, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Leggett, R.W. An age-specific kinetic-model of lead metabolism in humans. Environ. Health Perspect. 1993, 101, 598–616. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, A.S.; Roy, A.; Amarasiriwardena, C.J.; Smith, D.; Lupoli, N.; Mercado-Garcia, A.; Lamadrid-Figueroa, H.; Tellez-Rojo, M.M.; Hu, H.; Hernandez-Avila, M. Maternal blood, plasma, and breast milk lead: Lactational transfer and contribution to infant exposure. Environ. Health Perspect. 2014, 122, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, E.E.; Edwards, B.B.; Jensen, R.L.; Mahaffey, K.R.; Fomon, S.J. Absorption and retention of lead by infants. Pediat. Res. 1978, 12, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Levesque, B.; Duchesne, J.F.; Gariepy, C.; Rhainds, M.; Dumas, P.; Scheuhammer, A.M.; Proulx, J.F.; Dery, S.; Muckle, G.; Dallaire, F. Monitoring of umbilical cord blood lead levels and sources assessment among the Inuit. Occup. Environ. Med. 2003, 60, 693–695. [Google Scholar] [CrossRef] [PubMed]
- Bressler, J.P.; Olivi, L.; Cheong, J.H.; Kim, Y.; Bannon, D. Divalent metal transporter 1 in lead and cadmium transport. Ann. N Y Acad. Sci. 2004, 1012, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Worldwide Prevalence of Anaemia 1993–2005. Available online: http://www.who.int/vmnis/publications/anaemia_prevalence/en/ (accessed on 24 March 2014).
- Nriagu, J.; Afeiche, M.; Linder, A.; Arowolo, T.; Ana, G.; Sridhar, M.K.C.; Oloruntoba, E.O.; Obi, E.; Ebenebe, J.C.; Orisakwe, O.E.; et al. Lead poisoning associated with malaria in children of urban areas of Nigeria. Int. J. Hyg. Environ. Health 2008, 211, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.S.; Doniger, A.S.; Painting, S.; Houston, L.; Slaunwhite, M.; Mirabella, F.; Felsen, J.; Hunt, P.; Hyde, D.; Stich, E. Declines in elevated blood lead levels among children, 1997–2011. Am. J. Prev. Med. 2014, 46, 259–264. [Google Scholar] [CrossRef] [PubMed]
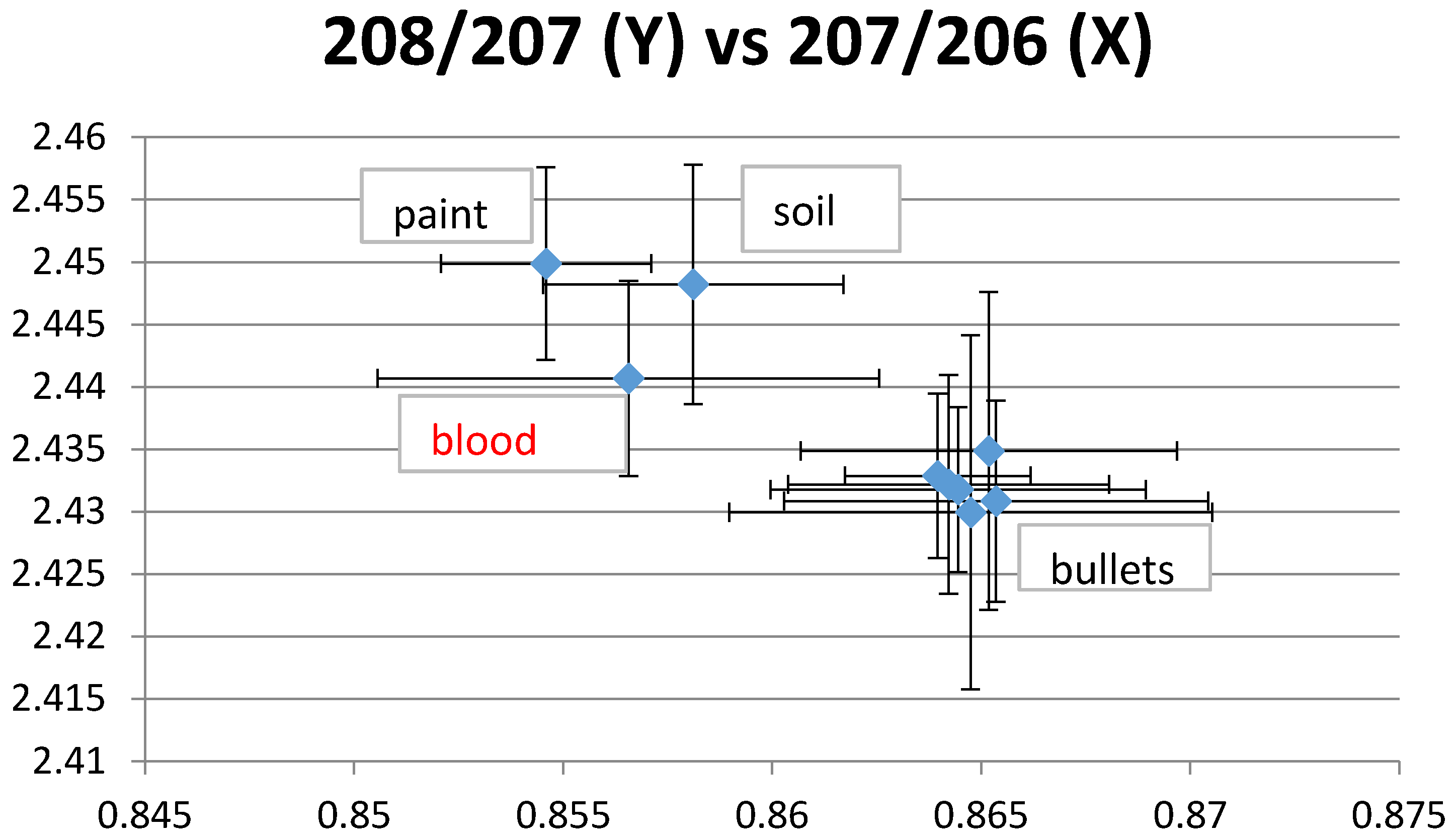
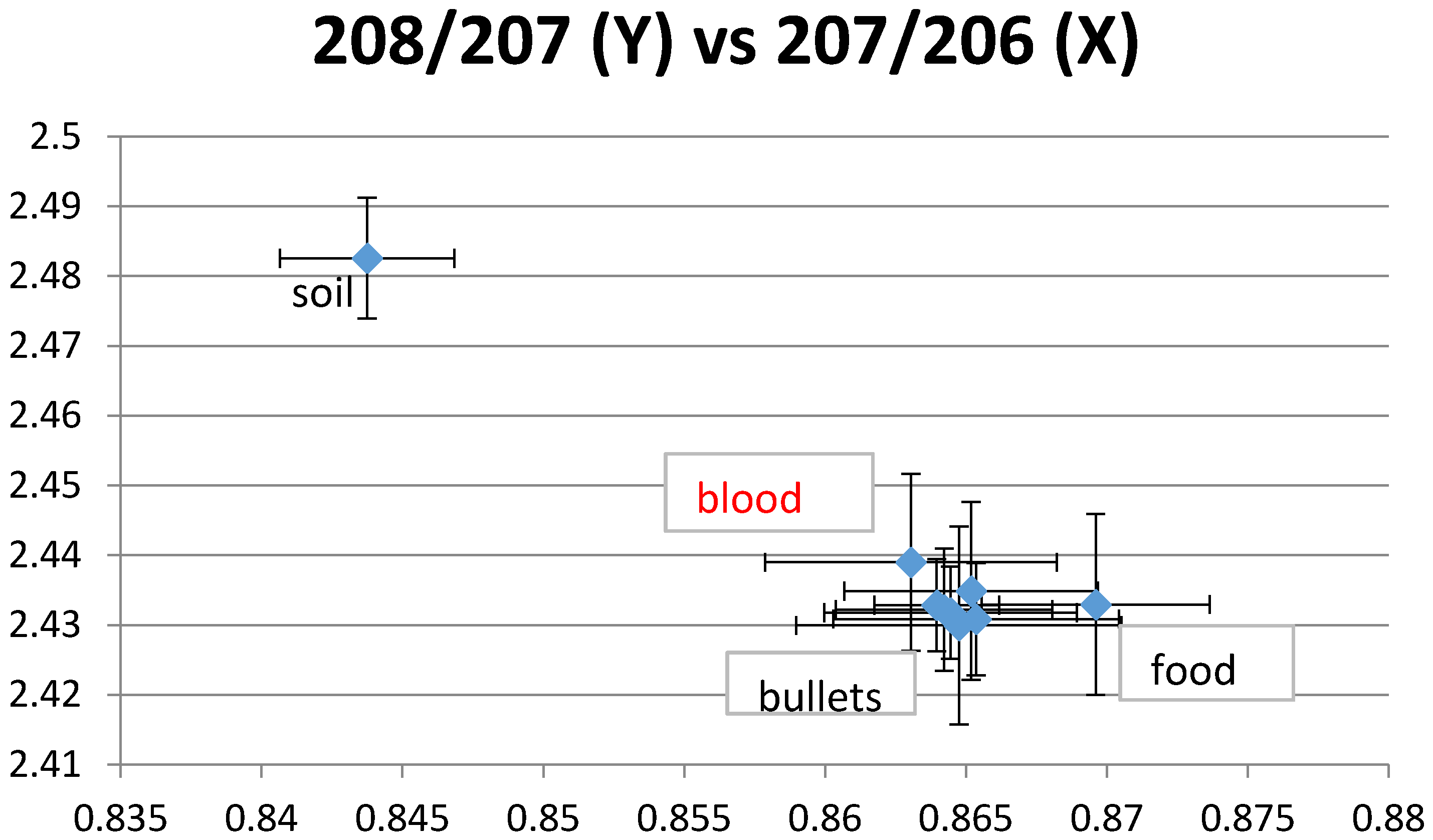
| Blood Lead Level | Children N = 685 | Mothers N = 227 |
|---|---|---|
| Mean ± SD | 72.2 (60.8) | 51.4 (22.3) |
| Median | 55.1 | 46.6 |
| Minimum | 8.3 | 22.8 |
| Maximum | 630.0 | 202.0 |
| 1st quartile | 39.2 | 36.5 |
| 3rd quartile | 85.0 | 60.1 |
| % over guideline value, % (n) | ||
| BLL>100 (µg/L) | 16.4 (112) | 2.6 (6) |
| BLL>50 (µg/L) * | 57.8 (396) | 43.6 (99) |
| Potential Sources of Pb | In Children | In Mothers | |||
|---|---|---|---|---|---|
| n (%) | Increment in LogBLL Compared to Referent Group (95% CI) | Crude OR for BLL>50 µg/L (95% CI) | Increment in LogBLL Compared to Referent Group (95% CI) | Crude OR for BLL>50 µg/L (95% CI) | |
| Child has ever eaten soil | 338 (56.1) | 0.01 (−0.08,0.11) | 0.78 (0.56,1.08) | 0.05 (−0.06,0.15) | |
| Child drinking any piped water | 499 (82.8) | 0.08 (−0.04,0.20) | 0.95 (0.62,1.47) | 0.16 (0.04,0.28) * | 2.42 (1.19,4.89) * |
| Child drinking any well water | 333 (55.2) | 0.07 (−0.03,0.16) | 1.08 (0.78,1.49) | −0.10 (−0.20, −0.01) * | 0.59 (0.34,1.02) |
| House in mud | 413 (61.0) | 0.07 (−0.03,0.16) | 1.25 (0.92,1.71) | 0.03 (−0.08,0.13) | 1.28 (0.73,2.25) |
| Painted walls in household | 96 (14.1) | 0.07 (−0.05,0.20) | 1.14 (0.73,1.77) | −0.03 (−0.17,0.12) | 0.68 (0.30,1.56) |
| Presence of paint chips in household | 35 (5.8) | 0.23 (0.03,0.42) * | 1.26 (0.62,2.54) | −0.04 (−0.25,0.18) | 0.95 (0.29,3.10) |
| Gasoline stored at home | 375 (62.2) | −0.11 (−0.20,-0.01) | 0.70 (0.50,0.97) * | −0.07 (−0.17,0.03) | 0.94 (0.53,1.66) |
| Cooking utensils with recycled material | 603 (100.0) | − | − | − | − |
| Metal cooking utensils | 360 (59.7) | −0.05 (−0.15,0.04) | 0.76 (0.54,1.06) | −0.05 (−0.15,0.05) | 0.94 (0.55,1.62) |
| Mud cooking utensils | 272 (45.1) | 0.04 (−0.05,0.13) | 1.00 (0.72,1.38) | −0.11 (−0.11,0.9) | 0.86 (0.50,1.49) |
| Enamel eating utensils | 581 (96.4) | ||||
| Pottery eating utensils | 55 (9.1) | 0.06 (−0.10,0.22) | 1.21 (0.68,2.13) | −0.05 (−0.22,0.12) | 1.23 (0.48,3.15) |
| Child eats animals killed by ammunition | 234 (38.8) | 0.11 (0.01,0.20) * | 1.50 (1.07,2.09) * | 0.09 (−0.00,0.19) | 1.81 (1.04,3.14) * |
| Maternal consumption of clay during pregnancy | 180 (26.3) | ||||
| Green clay (kalaba) | 142 (23.5) | −0.00 (−0.11,0.11) | 1.08 (0.74,1.58) | 0.07 (−0.05,0.19) | 1.15 (0.59,2.23) |
| White clay (kaolin) | 39 (10.9) | −0.05 (−0.25,0.15) | 0.76 (0.39,1.49) | −0.10 (−0.27,0.07) | 0.65 (0.24,1.79) |
| Maternal use of eye cosmetics | 113 (18.7) | 0.01 (−0.11,0.13) | 0.95 (0.63,1.43) | −0.11 (−0.25,0.04) | 0.63 (0.28,1.42) |
| Breastfeeding at 12 months old | 668 (98.1) | ||||
| Paternal high or moderate risk of occupational Pb exposure | 132 (19.7) | −0.01 (−0.13,0.10) | 0.81 (0.55,1.19) | 0.10 (−0.03,0.23) | 1.26 (0.62,2.56) |
| Activity in child’s house or neighborhood: | |||||
| Metal smelter | 17 (2.8) | ||||
| Battery recycling/storage | 3 (0.5) | ||||
| Radiator repair | 3 (0.5) | ||||
| Metal recycling/storage | 4 (0.7) | ||||
| Lead solder | 3 (0.5) | ||||
| Vehicle repair (car, motorcycle) | 32 (5.3) | ||||
| Manufacturing of ammunition, metal fish baits or metal objects | 1 (0.2) | ||||
| Sociodemographic characteristics: | |||||
| Child’s age at blood sampling (≥17 months) | 74 (10.9) | 0.17 (0.03,0.32) * | 1.37 (0.85,2.20) | ||
| Child’s gender (male) | 348 (51.1) | –0.02 (–0.10,0.07) | 1.08 (0.80,1.47) | ||
| Time playing outside: | |||||
| <2 h/day | 27 (4.48) | 0 | 1 | ||
| 2–6 h/day | 254 (42.1) | 0.05 (−0.18,0.28) | 1.48 (0.67,3.28) | ||
| >6 h/day | 322 (53.4) | 0.09 (−0.14,0.32) | 1.51 (0.69,3.32) | ||
| Wealth score (median ± SD) | 5 (2.76) | ||||
| Collective housing | 100 (14.8) | −0.15 (−0.28,−0.02) * | 1.39 (0.91,2.12) | −0.09 (−0.23,0.04) | 1.65 (0.76,3.59) |
| Maternal age (years) | −0.05 (−0.13,−0.03) | 0.87 (0.67,1.14) | −0.01 (−0.10,0.07) | 1.13 (0.71,1.80) | |
| Maternal education ≥ high school | 53 (7.8) | 0.11 (−0.06,0.27) | 1.22 (0.68,2.17) | −0.04 (−0.21,0.13) | 1.07 (0.43,2.70) |
| Working mother | 628 (92.2) | −0.18 (−0.35,−0.02) * | 0.75 (0.42,1.34) | −0.17 (−0.39,0.04) | 0.37 (0.11,1.26) |
| Married | 673 (99.4) | ||||
| Polygamous | 247 (36.7) | 0.01 (−0.08,0.11) | 1.20 (0.87,1.65) | 0.02 (−0.08,0.12) | 1.46 (0.85,2.53) |
| Maternity ward | |||||
| Sekou | 421 (61.8) | 0 | 1 | ||
| Attogon | 200 (29.4) | 0.08 (−0.02,0.18) | 0.87 (0.50,1.52) | 0 | 1 |
| Allada | 60 (8.8) | −0.01 (−0.17,0.15) | 1.17 (0.84,1.65) | −0.01 (−0.13,0.10) | 0.87 (0.47,1.61) |
| In Children | In Mothers | |||
|---|---|---|---|---|
| Increment in LogBLL Compared to Referent Group (95% CI) | Adjusted OR for BLL>50 µg/L (95% CI) | Increment in LogBLL Compared to Referent Group (95% CI) | Adjusted OR for BLL>50 µg/L (95% CI) | |
| Model 1 1 | (n = 601) | (n = 601) | (n = 215) | (n = 215) |
| Presence of paint chips in household | 0.21 (0.01,0.41) * | 1.22 (0.60,2.50) | −0.09 (−0.31,0.12) | 0.69 (0.21,2.32) |
| Child eats animals killed by ammunition | 0.12 (0.02,0.21) * | 1.49 (1.06,2.09) * | 0.10 (0.01,0.20) * | 1.92 (1.09,3.38) * |
| Piped water | 0.08 (−0.05,0.20) | 0.98 (0.64,1.51) | 0.17 (0.05,0.29) * | 2.60 (1.27,5.36) * |
| Model 2 2 | (n = 598) | (n = 598) | (n = 212) | (n = 212) |
| Presence of paint chips in household | 0.21 (0.01,0.40) * | 1.29 (0.62,2.67) | −0.13 (−0.35,0.08) | 0.56 (0.16,1.97) |
| Child eats animals killed by ammunition | 0.11 (0.01,0.20) * | 1.45 (1.03,2.05) * | 0.10 (−0.00,0.19) | 1.86 (1.05,3.31) * |
| Piped water | 0.10 (−0.02,0.23) | 1.09 (0.69,1.72) | 0.19 (0.07,0.32) * | 2.56 (1.20,5.46) * |
| Child age at blood sampling (≥17 months) | 0.02 (−0.00,0.05) | 1.50 (0.87,2.59) | ||
| Type of housing | 0.12 (−0.02,0.25) | 1.07 (0.66,1.72) | 0.08 (−0.06,0.22) | 1.72 (0.74,3.99) |
| Gasoline stored at home | −0.09 (−0.20,0.03) | 0.77 (0.52,1.12) | −0.05 (−0.17,0.06) | 0.87 (0.44,1.70) |
| Wealth score | −0.01 (−0.03,0.01) | 0.97 (0.90,1.04) | −0.01 (−0.03,0.01) | 1.03 (0.91,1.16) |
| Maternal occupation | −0.21 (−0.39,−0.04) * | 0.67 (0.35,1.29) | −0.18 (−0.40,0.04) | 0.38 (0.10,1.43) |
| Model 3 3 | (n = 193) | (n = 193) | ||
| Presence of paint chips in household | 0.49 (0.13,0.86) * | 0.69 (0.18,2.57) | ||
| Child eats animals killed by ammunition | 0.04 (−0.13:0.21) | 1.27 (0.69,2.34) | ||
| Piped water | −0.01 (−0.21,0.20) | 0.88 (0.42,1.86) | ||
| Mother BLL | 0.01 (0.00,0.01) * | 1.02 (1.00,1.03) | ||
| Child age (≥17 months) | 0.03 (−0.00,0.06) | 1.32 (0.68,2.58) | ||
| Type of housing | −0.10 (−0.34,0.14) | 0.76 (0.32,1.79) | ||
| Gasoline stored at home | 0.00 (−0.21,0.21) | 0.75 (0.37,1.53) | ||
| Wealth score | 0.02 (−0.02,0.05) | 1.02 (0.90,1.16) | ||
| Maternal occupation | −0.21 (−0.57,0.14) | 0.61 (0.15,2.52) | ||
| Child No. | BLL in Infants (µg/L) | Dust (µg/m2) | Soil (mg/kg) | Drinking Water (µg/L) | Cooking Dish (µg/L) | Paint (mg/kg) | Food (µg/kg) | Child Eats Animals Killed by Ammunition | Paint Inside House | Still Breastfed |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 110 | 1735 | 25 | <LOQ * | 111 | Yes | No | Yes | ||
| 2 | 135 | 189 | 3 | <LOQ | Yes | No | Yes | |||
| 3 | 99 | 1640 | 34 | 1.1 | 411 | Yes | Yes | Yes | ||
| 4 | 109 | 9 | 4 | 2.3 | < LOQ | No | Yes | Yes | ||
| 5 | 209 | 107 | 33 | <LOQ | 7020 | 94 | Yes | No | Yes | |
| 6 | 136 | 19 | 4 | <LOQ | < LOQ | Yes | No | Yes | ||
| 7 | 105 | 294 | 48 | <LOQ | < LOQ | Yes | No | Yes | ||
| 8 | 113 | 32 | 3 | 5.4 | 4952 | No | No | Yes |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bodeau-Livinec, F.; Glorennec, P.; Cot, M.; Dumas, P.; Durand, S.; Massougbodji, A.; Ayotte, P.; Le Bot, B. Elevated Blood Lead Levels in Infants and Mothers in Benin and Potential Sources of Exposure. Int. J. Environ. Res. Public Health 2016, 13, 316. https://doi.org/10.3390/ijerph13030316
Bodeau-Livinec F, Glorennec P, Cot M, Dumas P, Durand S, Massougbodji A, Ayotte P, Le Bot B. Elevated Blood Lead Levels in Infants and Mothers in Benin and Potential Sources of Exposure. International Journal of Environmental Research and Public Health. 2016; 13(3):316. https://doi.org/10.3390/ijerph13030316
Chicago/Turabian StyleBodeau-Livinec, Florence, Philippe Glorennec, Michel Cot, Pierre Dumas, Séverine Durand, Achille Massougbodji, Pierre Ayotte, and Barbara Le Bot. 2016. "Elevated Blood Lead Levels in Infants and Mothers in Benin and Potential Sources of Exposure" International Journal of Environmental Research and Public Health 13, no. 3: 316. https://doi.org/10.3390/ijerph13030316
APA StyleBodeau-Livinec, F., Glorennec, P., Cot, M., Dumas, P., Durand, S., Massougbodji, A., Ayotte, P., & Le Bot, B. (2016). Elevated Blood Lead Levels in Infants and Mothers in Benin and Potential Sources of Exposure. International Journal of Environmental Research and Public Health, 13(3), 316. https://doi.org/10.3390/ijerph13030316





