Phosphate Adsorption from Membrane Bioreactor Effluent Using Dowex 21K XLT and Recovery as Struvite and Hydroxyapatite
Abstract
:1. Introduction
2. Material and Methods
2.1. Feed Water
2.2. Ion Exchange Resin
2.3. Chemical Analysis
2.4. Characteristics of Dowex 21K XLT
2.5. Effect of pH
2.6. Batch Mode Adsorption Experiment
2.7. Column Mode Adsorption Experiment
2.8. Desorption of Phosphate
2.9. Struvite Precipitation
2.10. Hydroxyapatite Precipitation
2.11. Phosphorus Content in Struvite and Hydroxyapatite
3. Results and Discussion
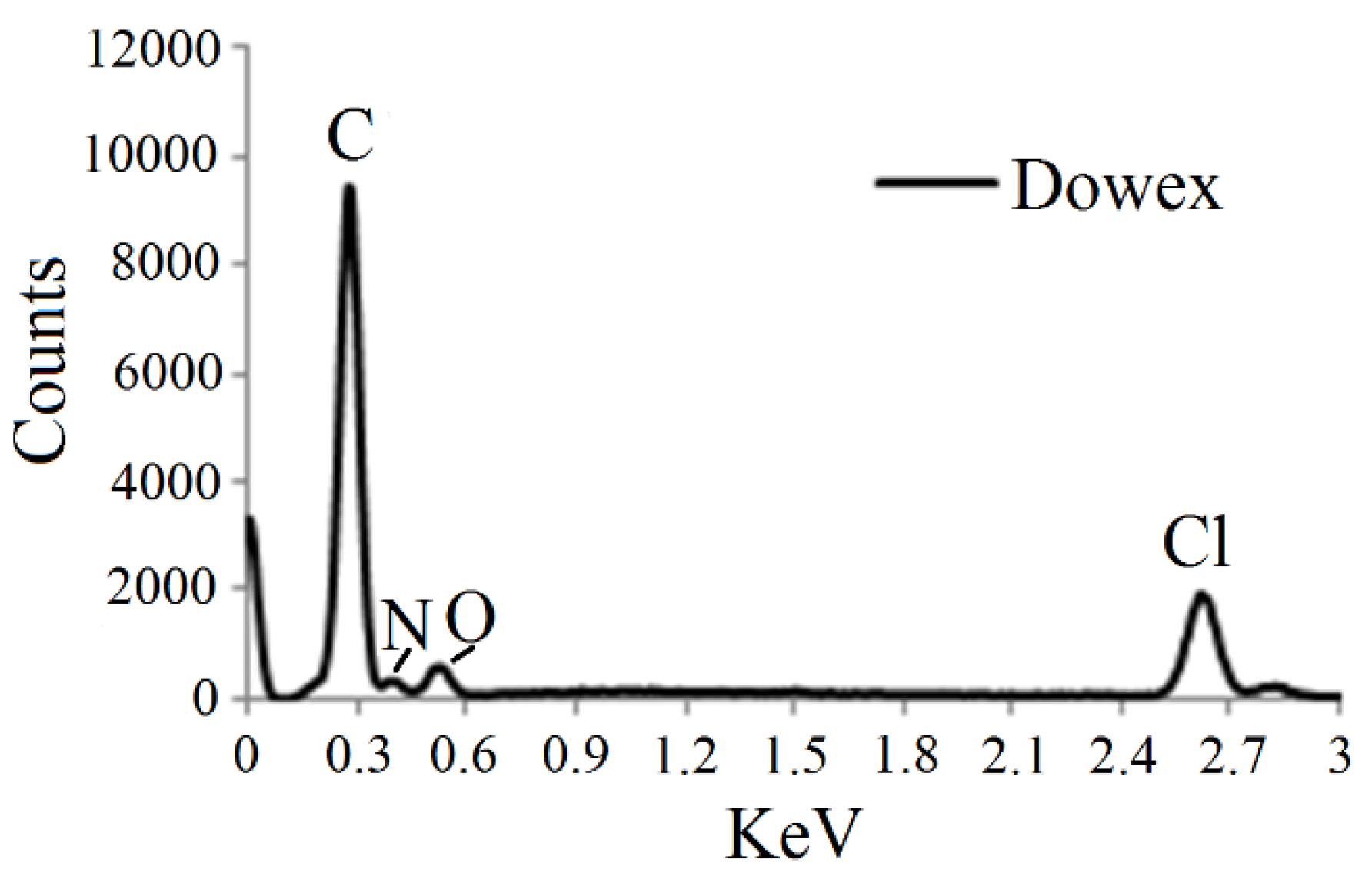
3.1. EDS Analysis
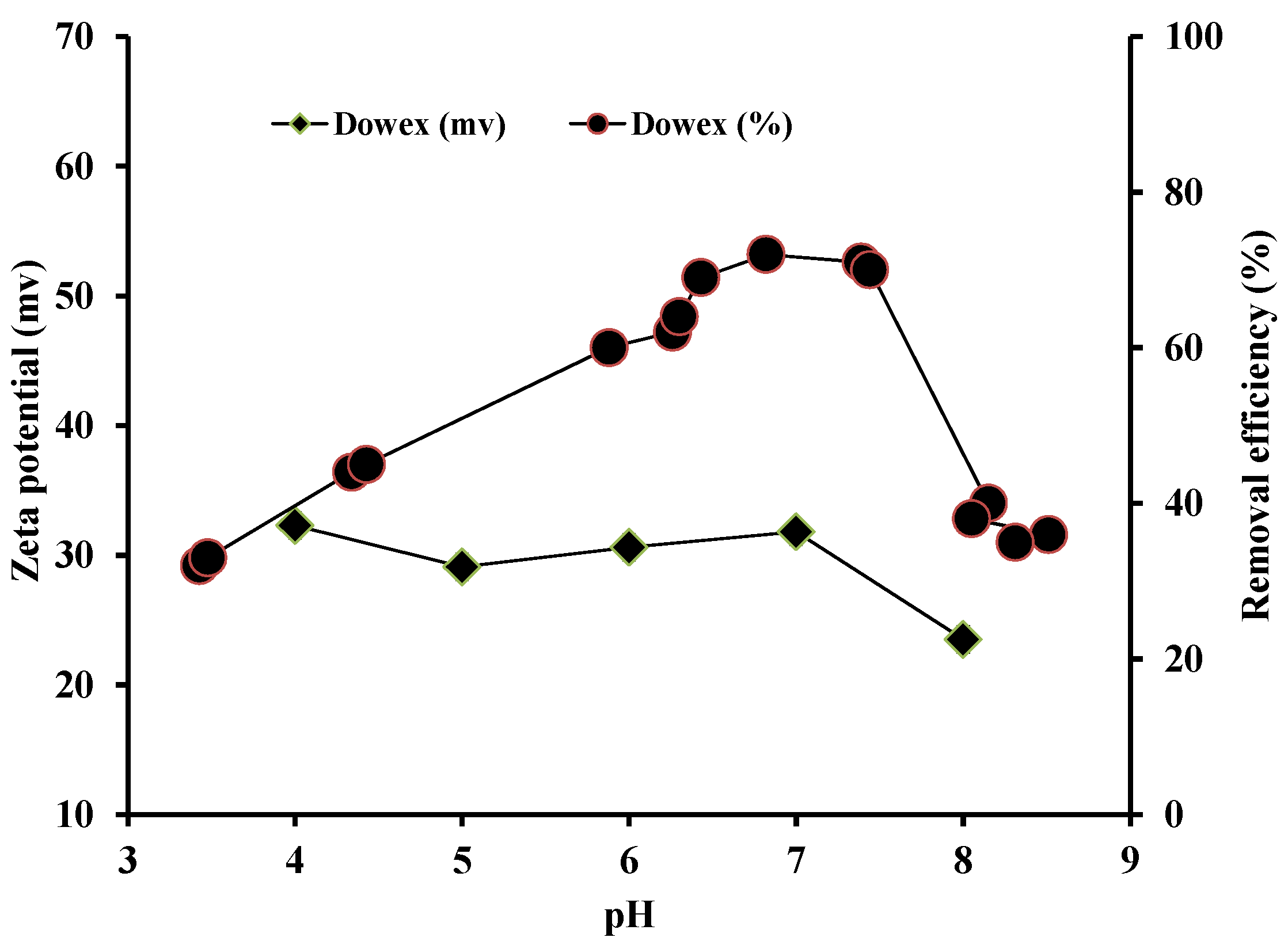
3.2. Zeta Potential and Effect of pH on P Adsorption
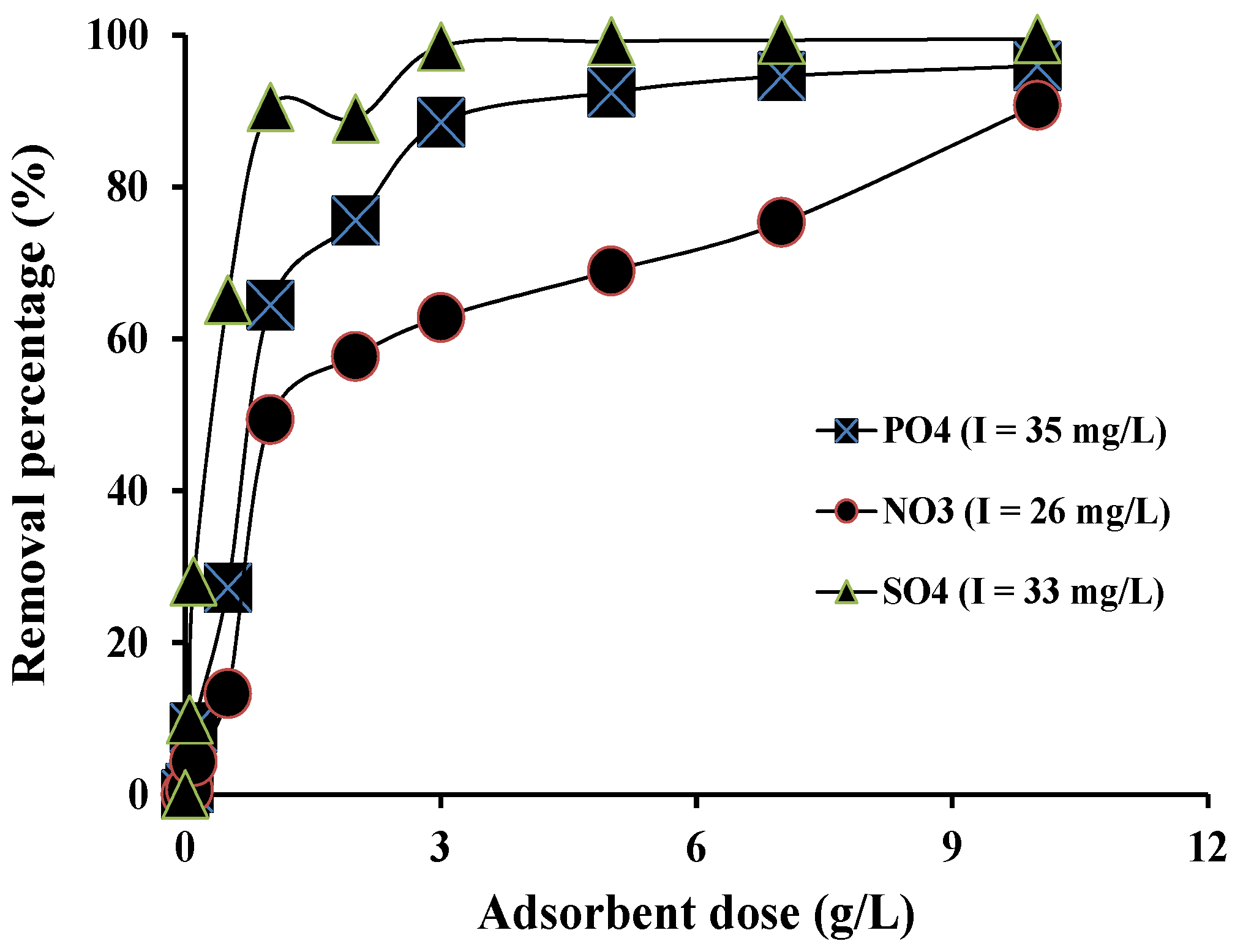
3.3. Batch Adsorption Experiment
3.4. Batch Adsorption Capacity
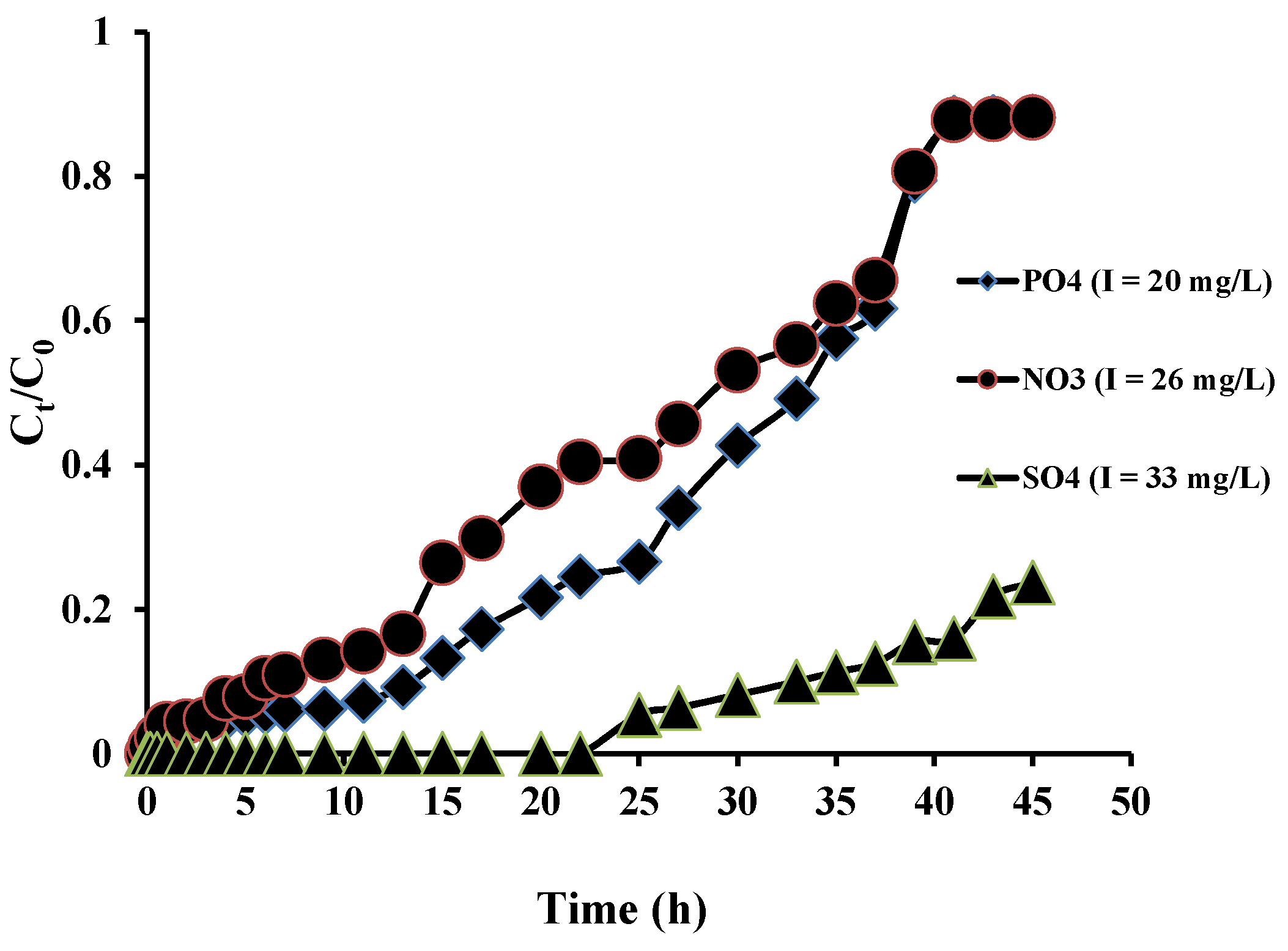
3.5. Fixed-Bed Column Adsorption Studies
3.6. Desorption of Phosphate
3.7. Phosphate Recovery as Struvite
3.8. Phosphate Recovery as Hydroxyapatite
4. Conclusions
Acknowledgment
Author Contributions
Conflicts of Interest
References
- Jorgensen, S.E. Water Quality: The Impact of Eutrophication, Lakes and Resevoirs, 1st ed.; The UNEP-International Environment Tecnology Centre (IETC) and the International Lake Environment Comitte (ILEC): Otsu, Japan, 2001. [Google Scholar]
- Levine, S.N.; Schindler, D.W. Phosphorus, nitrogen and carbon dynamics of experimental lake 303 during recovery from eutrophication. Can. J. Fish. Aquat. Sci. 1989, 46, 2–10. [Google Scholar] [CrossRef]
- Awual, R.; Jyo, A.; El-Safty, S.A.; Tamada, M.; Seka, N.A. A weak-fibrous anion exchanger effective for rapid phosphate removal from water. J. Hazard. Mater. 2011, 188, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Long, F.; Gong, J.; Zeng, G.; Chen, L.; Wang, X.; Deng, J.; Niu, Q.; Zhang, H.; Zhang, X. Removal of phosphate from aqueous solution by magnetic Fe-Zr binary oxide. Chem. Eng. J. 2011, 171, 448–455. [Google Scholar] [CrossRef]
- Collingwood, R.W. A Survey of Eutrophication in BRITAIN and its Effects on Water Supplies; Water Research Centre: Hertfordshire, UK, 1977. [Google Scholar]
- Davies, A.W. Scientific Investigations into the Eutrophication of the Norfolk Broads; Report No. 8; Freshwater Biological Association: Ambleside, UK, 1980. [Google Scholar]
- U.S. Environmental Protection Agency. Nutrient Control Design Manual: State of Technology Review Report; Office of Research and Development, National Risk Management Research Laboratory: Cincinnati, OH, USA, 2009.
- Liu, J.; Zhang, M.; Li, J.J. MBR Process Lisinopril Enalapril Pharmaceutical Wastewater. J. Eng. 2007, 5, 27–28. [Google Scholar]
- Johir, M.A.H.; George, J.; Vigneswaran, S.; Kandasamy, J.; Grasmick, A. Removal and recovery of nutrients by ion exchange from high rate membrane bio-reactor (MBR) effluent. Desalination 2011, 275, 197–202. [Google Scholar] [CrossRef]
- Yoshida, H.; Galinada, W.A. Equilibria for adsorption of phosphates on OH-Type strongly basic ion exchanger. AIChE J. 2002, 48, 2193–2202. [Google Scholar] [CrossRef]
- Cordell, D.; Drangert, J.-O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Karunanithi, R.; Szogi, A.A.; Bolan, N.; Naidu, R.; Loganathan, P.; Hunt, P.G.; Vanotti, M.B.; Saint, C.P.; Ok, Y.S.; Krishnamoorthy, S. Phosphorus Recovery and Reuse from Waste Streams. Adv. Agron. 2015, 131, 173–250. [Google Scholar]
- Radjenović, J.; Petrović, M.; Venturac, F.; Barceló, D. Rejection of pharmaceuticals in nanofiltration and reverse osmosis membrane drinking water treatment. Water Res. 2008, 42, 3601–3610. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Suna, P.; Hana, J.; Songa, Y.; Huc, Z.; Haiqing Fanb, H.; Lv, S. Inhibitory factors affecting the process of enhanced biological phosphorus removal (EBPR)—A mini-review. Process. Biochem. 2014, 49, 2207–2213. [Google Scholar] [CrossRef]
- Loganathan, P.; Vigneswaran, S.; Kandasamy, J.; Bolan, N.S. Removal and recovery of phosphate from water using sorption. Crit. Rev. Environ. Sci. Tech. 2014, 44, 847–907. [Google Scholar] [CrossRef]
- Morse, G.K.; Brett, S.W.; Guy, J.A.; Lester, J.N. Review: Phosphorus removal and recovery technologies. Sci. Total Environ. 1998, 212, 69–81. [Google Scholar] [CrossRef]
- Nur, T.; Johir, M.A.H.; Loganathan, P.; Vigneswaran, S.; Kandasamy, J. Effectiveness of purolite A500PS and A520E ion exchange resins on the removal of nitrate and phosphate from synthetic water. Desalin. Water Treat. 2012, 47, 50–58. [Google Scholar] [CrossRef]
- Nur, T.; Johir, M.A.H.; Loganathan, P.; Nguyen, T.; Vigneswaran, S.; Kandasamy, J. Phosphate removal from water using an iron oxide impregnated strong base anion exchange resin. J. Ind. Eng. Chem. 2014, 20, 1301–1307. [Google Scholar] [CrossRef]
- Chen, J.P.; Chua, M.-L.; Zhang, B. Effects of competitive ions, humic acid, and pH on removal of ammonium and phosphorous from the synthetic industrial effluent by ion exchange resins. Waste Manag. 2002, 22, 711–719. [Google Scholar] [CrossRef]
- Midorikawa, I.; Aoki, H.; Omori, A.; Shimizu, T.; Kawaguchi, Y.; Katsusai, K.; Murakami, T. Recovery of high purity phosphorus from municipal wastewater secondary effluent by a high-speed adsorbent. Water Sci. Technol. 2008, 58, 1601–1607. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, K.; Midorikawa, I.; Aoki, H.; Omori, A.; Shimizu, T.; Hashimoto, T. Pilot testing of a high efficiency adsorbent system for phosphorus removal and recovery from municipal wastewater secondary effluent. In Proceedings of the Water Environment Federation, Nutrient Recovery and Management, Alexandria, VA, USA, 9–12 January 2011; pp. 1205–1213.
- Nur, T. Nitrate, Phosphate and Fluoride Removal from Water Using Adsorption Process, Ph.D. Thesis, University of Technology Sydney, Sydney, Australia, 30 June 2014. [Google Scholar]
- Ok, Y.S.; Jeon, C. Selective adsorption of the gold–cyanide complex from waste rinse water using Dowex 21K XLT resin. J. Ind. Eng. Chem. 2014, 20, 1308–1312. [Google Scholar] [CrossRef]
- Dow Chemical Company: Dowex 21K XLT. Avaliable online: http://msdssearch.dow.com/PublishedLiteratureDOWCOM/dh_0061/0901b803800611ab.pdf?filepath=liquidseps/pdfs/noreg/177-01893.pdf&fromPage=GetDoc. (accessed on 29 December 2015).
- Le Corre, K.S.; Valsami-Jones, E.; Hobbs, P.; Parsons, S.A. Phosphorus recovery from wastewater by struvite crystallization: A review. Crit. Rev. Environ. Sci. 2009, 39, 433–477. [Google Scholar] [CrossRef]
- Durrant, A.E.; Scrimshaw, M.D.; Stratful, I.; Lester, J.N. Review of the feasibility of recovering phosphate from wastewater for use as a raw material by phosphate industry. Environ. Technol. 1999, 20, 749–758. [Google Scholar] [CrossRef]
- Marshall, W.E.; Wartelle, L.H. An anion exchange resin from soybean hulls. J. Chem. Technol. Biot. 2004, 79, 1286–1292. [Google Scholar] [CrossRef]
- Hamoudi, S.; Saad, R.; Belkacemi, K. Adsorptive removal of phosphate and nitrate anions from aqueous solutions using ammonium-functionalized mesoporous silica. Ind. Eng. Chem. Res. 2007, 46, 8806–8812. [Google Scholar] [CrossRef]
- Gupta, M.D.; Loganathan, P.; Vigneswaran, S. Adsorptive removal of nitrate and phosphate from water by a Purolite ion exchange resin and hydrous ferric oxide columns in series. Separ. Sci. Technol. 2012, 47, 1785–1792. [Google Scholar] [CrossRef]
- Loganathan, P.; Hedley, M.J.; Grace, N.D.; Lee, J.; Cronin, S.J.; Bolan, N.S.; Zanders, J.M. Fertiliser contaminants in New Zealand grazed pasture with special reference to cadmium and fluorine—A review. Aust. J. Soil Res. 2003, 41, 501–532. [Google Scholar] [CrossRef]
- Leech, F. Cycling of Phosphorus in Grazing Systems. Avaliable online: http://www.dpi.nsw.gov.au/__data/assets/pdf_file/0010/289774/cycling-of-phosphorus-in-grazing-systems.pdf (accessed on 29 December 2015).
- Kalaruban, M.; Loganathan, P.; Shim, W.G.; Kandasamy, J.; Naidu, G.; Nguyen, T.V.; Vigneswaran, S. Removing nitrate from water using iron-modified Dowex 21K XLT ion exchange resin: Batch and fluidised-bed adsorption studies. Sep. Purif. Technol. 2016, 158, 62–70. [Google Scholar] [CrossRef]
| Parameters | Unit | MBR Treated Water |
|---|---|---|
| Phosphate | mg/L | 20.0, 35.0 * |
| Nitrate | mg/L | 26.0 |
| Sulphate | mg/L | 33.0 |
| TOC | mg/L | 8.9 |
| pH | 7.0–7.5 |
| Desorption Time (min) | Phosphate Concentration of Desorbed Solution (M) | Chemical Composition, Weight (%) | ||
|---|---|---|---|---|
| Phosphate | Ammonium | Magnesium | ||
| 30 | 0.007 | 41.7 ± 0.29 | 10.9 ± 0.68 | 12.6 ± 0.55 |
| 60 | 0.004 | 21.7 ± 0.49 | 7.9 ± 0.68 | 8.6 ± 0.55 |
| Desorption Time (min) | Phosphate Concentration of Desorbed Solution (M) | Molar Ratio Phosphate: Calcium | Chemical Composition, Weight (%) | |
|---|---|---|---|---|
| Phosphate | Calcium | |||
| 30 | 0.007 | 1.0:0.5 | 25.1 ± 0.28 | 35.1 ± 0.27 |
| 1.0:2.0 | 38.5 ± 0.28 | 32.8 ± 0.45 | ||
| 60 | 0.004 | 1.0:0.5 | 26.4 ± 0.19 | 28.4 ± 0.49 |
| 1.0:2.0 | 29.9 ± 0.58 | 33.7 ± 0.75 | ||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nur, T.; Loganathan, P.; Kandasamy, J.; Vigneswaran, S. Phosphate Adsorption from Membrane Bioreactor Effluent Using Dowex 21K XLT and Recovery as Struvite and Hydroxyapatite. Int. J. Environ. Res. Public Health 2016, 13, 277. https://doi.org/10.3390/ijerph13030277
Nur T, Loganathan P, Kandasamy J, Vigneswaran S. Phosphate Adsorption from Membrane Bioreactor Effluent Using Dowex 21K XLT and Recovery as Struvite and Hydroxyapatite. International Journal of Environmental Research and Public Health. 2016; 13(3):277. https://doi.org/10.3390/ijerph13030277
Chicago/Turabian StyleNur, Tanjina, Paripurnanda Loganathan, Jaya Kandasamy, and Saravanamuthu Vigneswaran. 2016. "Phosphate Adsorption from Membrane Bioreactor Effluent Using Dowex 21K XLT and Recovery as Struvite and Hydroxyapatite" International Journal of Environmental Research and Public Health 13, no. 3: 277. https://doi.org/10.3390/ijerph13030277
APA StyleNur, T., Loganathan, P., Kandasamy, J., & Vigneswaran, S. (2016). Phosphate Adsorption from Membrane Bioreactor Effluent Using Dowex 21K XLT and Recovery as Struvite and Hydroxyapatite. International Journal of Environmental Research and Public Health, 13(3), 277. https://doi.org/10.3390/ijerph13030277






