Distributions, Early Diagenesis, and Spatial Characteristics of Amino Acids in Sediments of Multi-Polluted Rivers: A Case Study in the Haihe River Basin, China
Abstract
:1. Introduction
2. Material and Methods
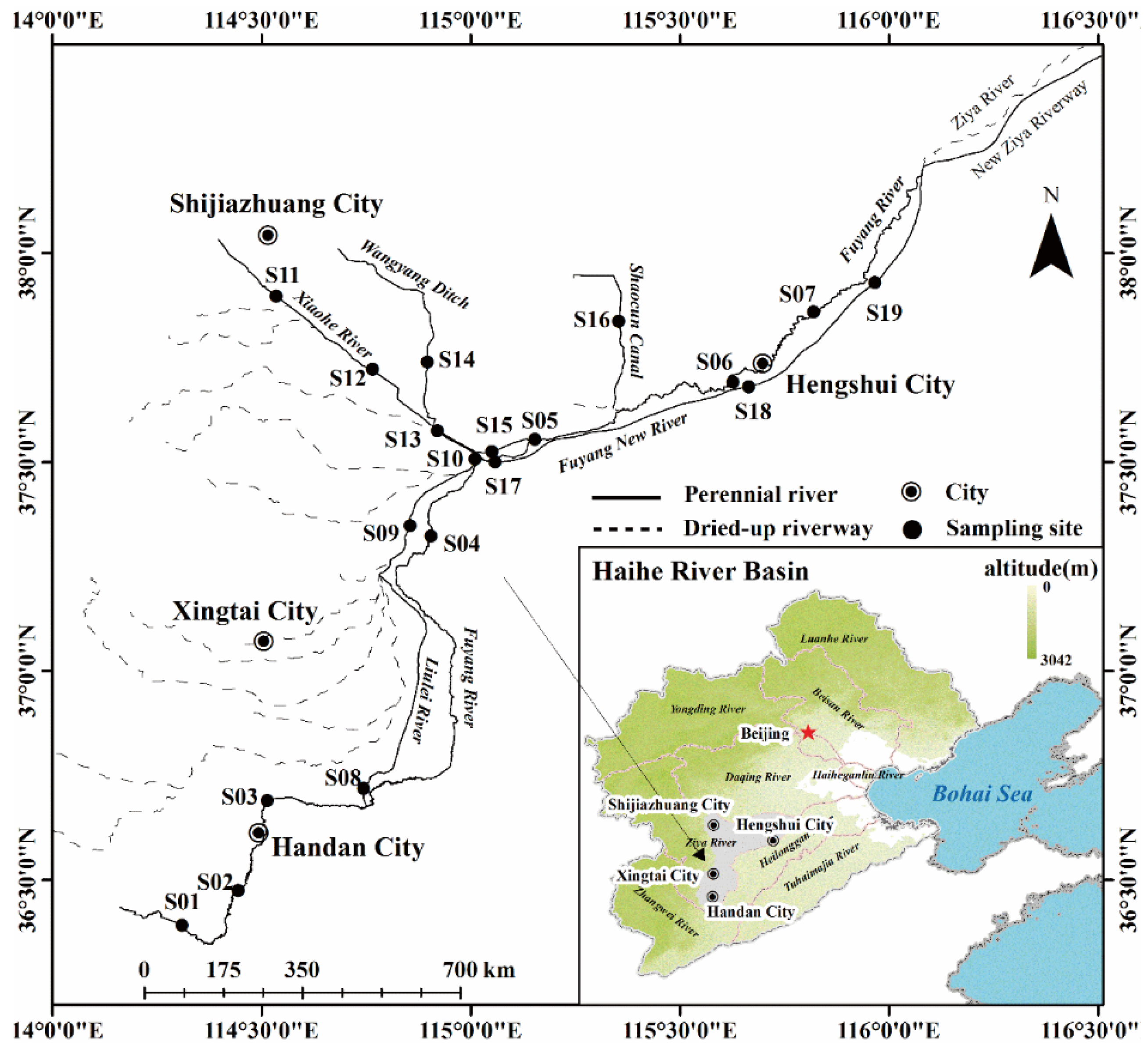
2.1. Study Area
2.2. Ethics Statement
2.3. Sample Collection and Analysis
2.4. Amino Acids Analysis
2.5. Statistical Analysis
3. Results
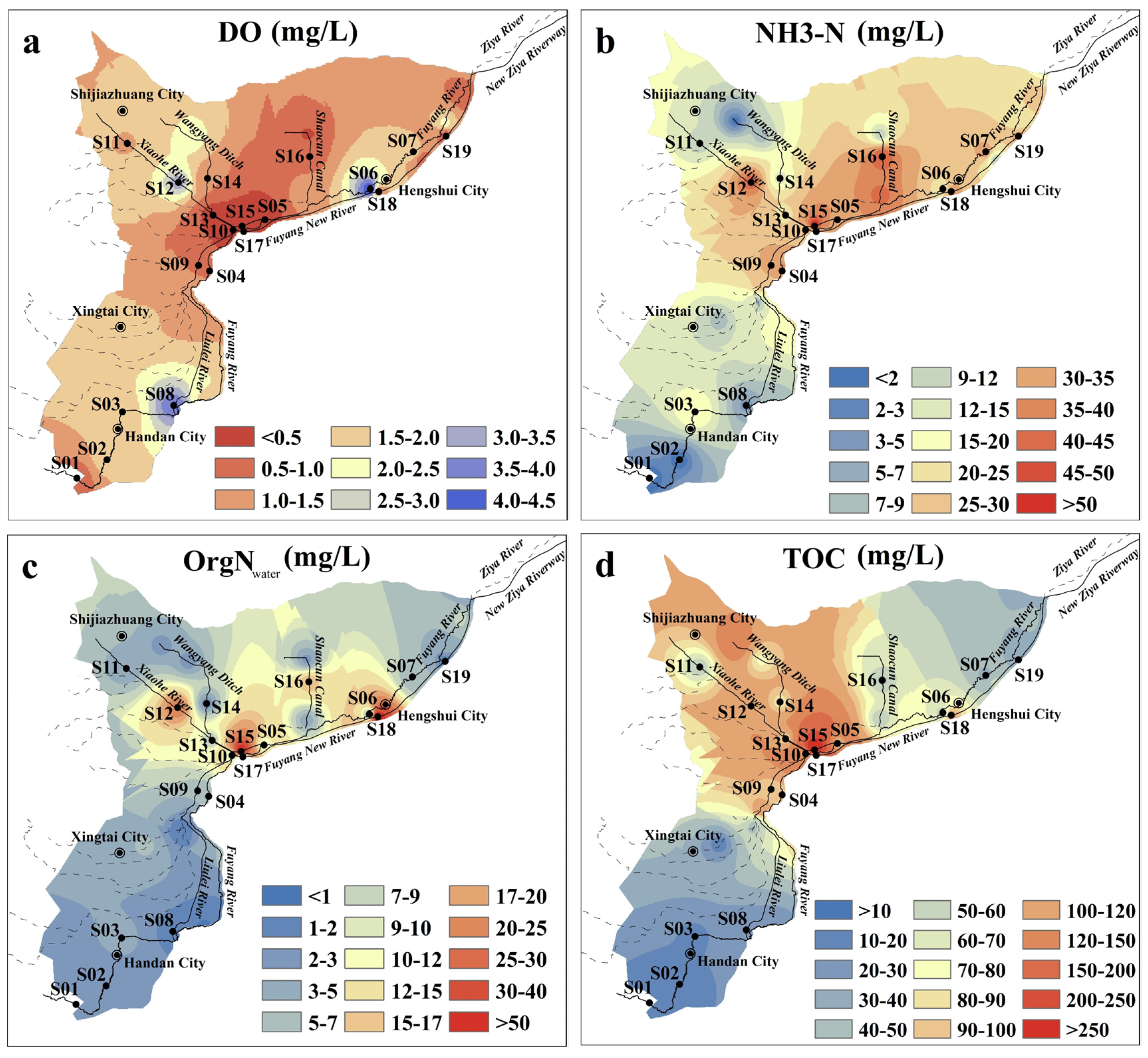
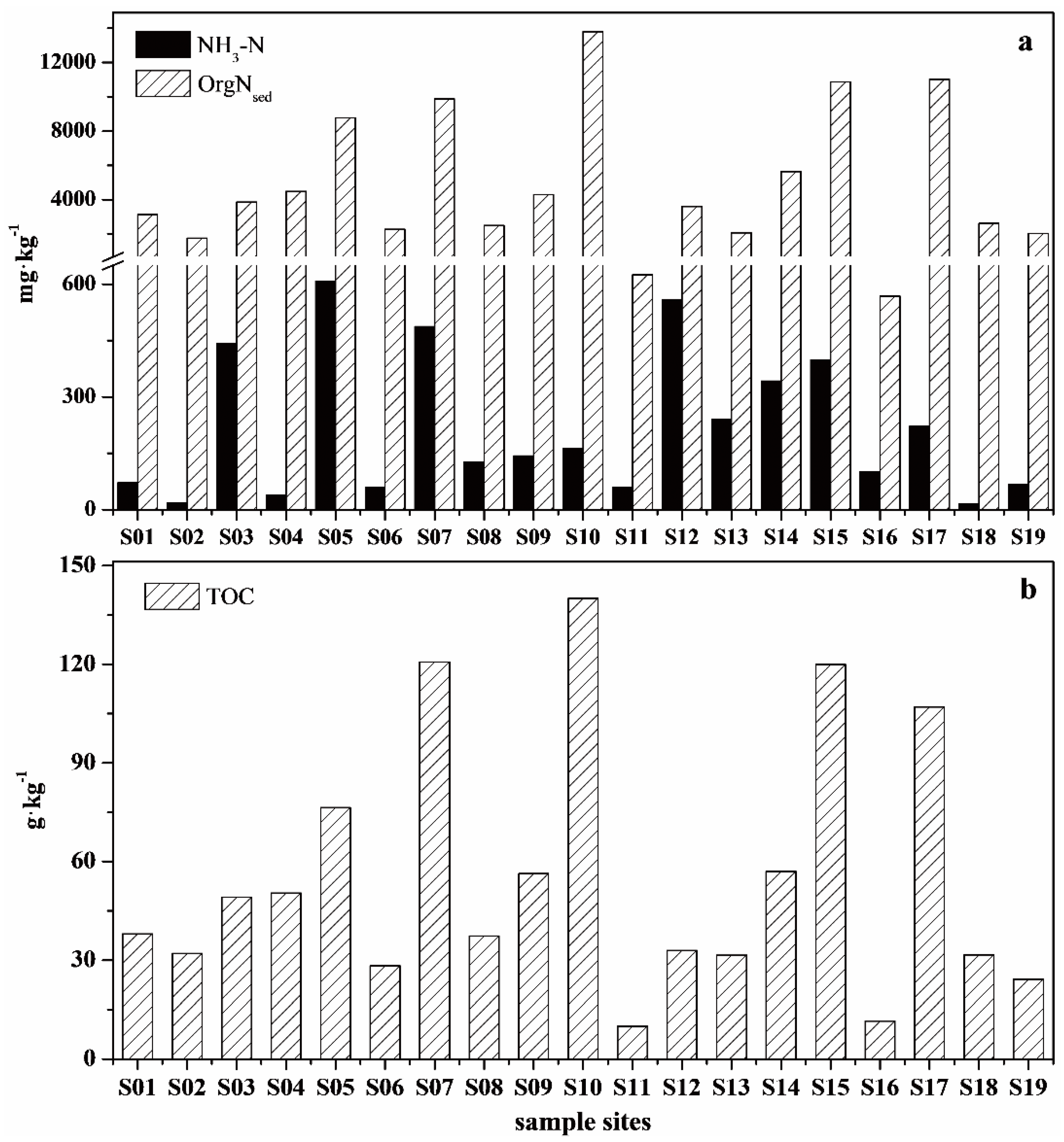
3.1. Physicochemical Characteristics of Surface Water and Surface Sediment Samples
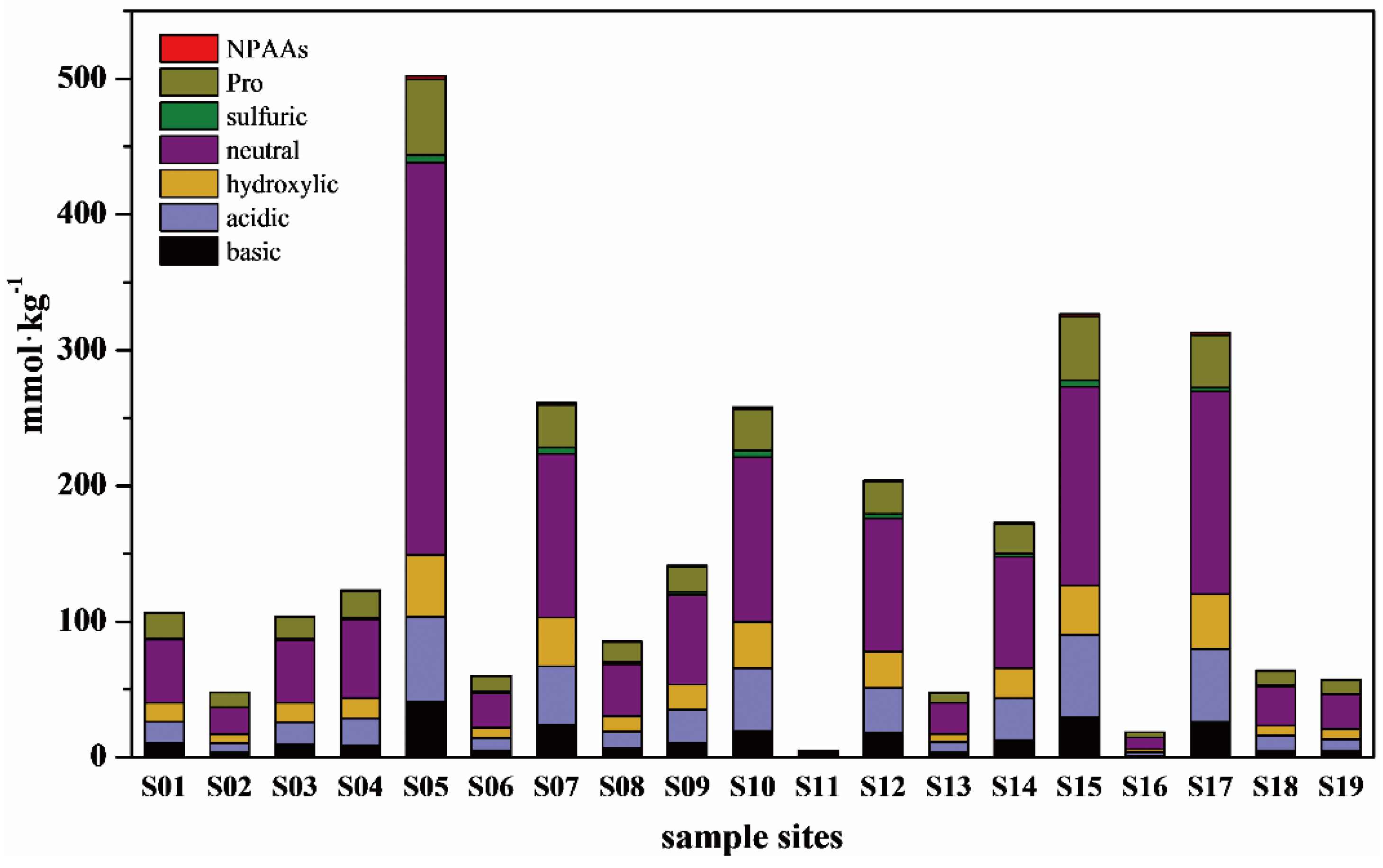
3.2. Amino Acids Concentration and Composition in Surface Sediments
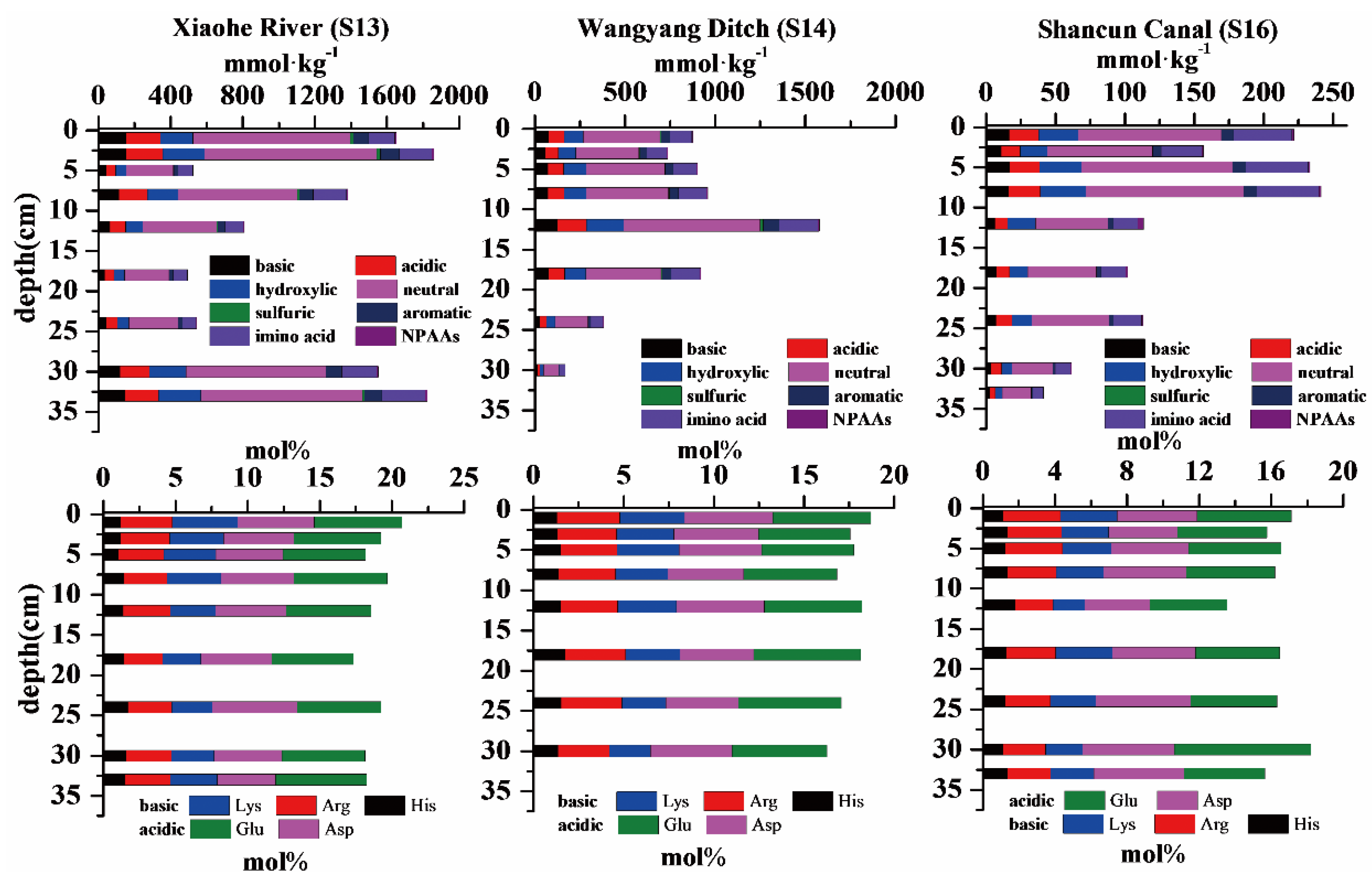
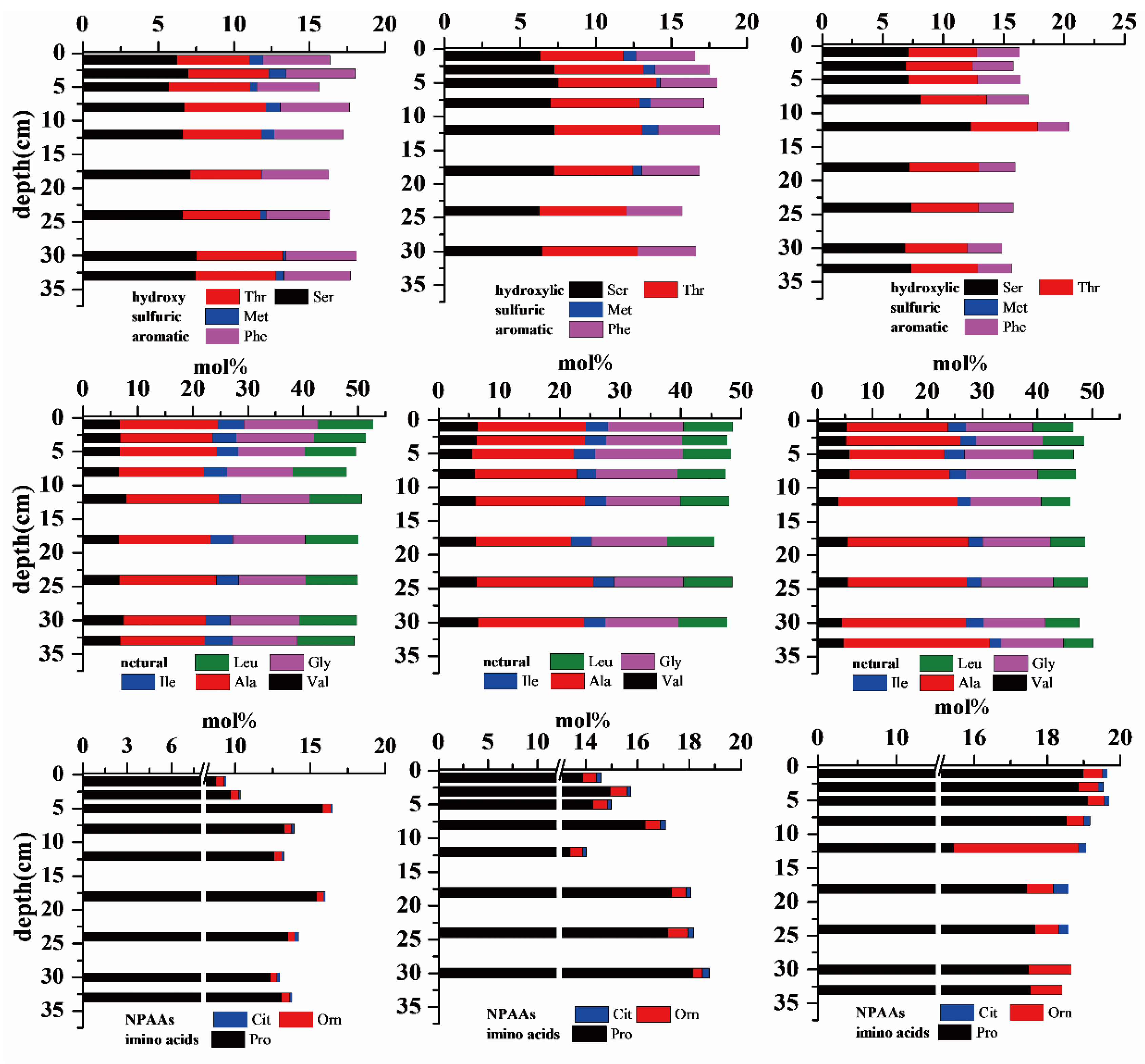
3.3. Amino Acids in Sediment Cores
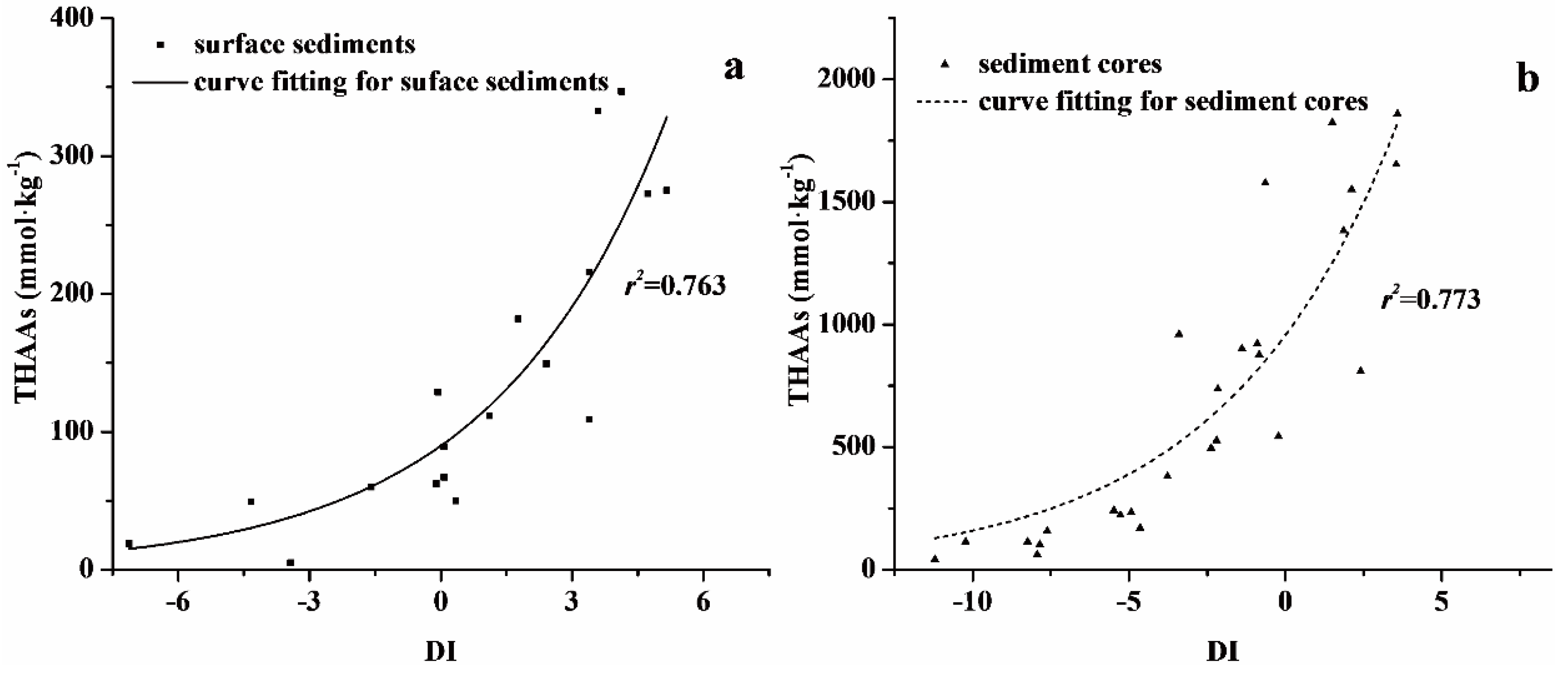
4. Discussion
4.1. Contribution of THAAs to Organic Matter in Surface Sediments
4.2. Degradation State in Surface Sediments and Sediment Cores
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hedges, J.I.; Keil, R.G. Sedimentary organic matter preservation: An assessment and speculative synthesis. Mar. Chem. 1995, 49, 81–115. [Google Scholar] [CrossRef]
- Hedges, J.I. Global biogeochemical cycles: Progress and problems. Mar. Chem. 1992, 39, 67–93. [Google Scholar] [CrossRef]
- Alkhatib, M.; Schubert, C.J.; Del Giorgio, P.A.; Gelinas, Y.; Lehmann, M.F. Organic matter reactivity indicators in sediments of the St. Lawrence Estuary. Estuar. Coast Shelf Sci. 2012, 102, 36–47. [Google Scholar] [CrossRef]
- Randlett, M.E.; Sollberger, S.; del Sontro, T.; Müller, B.; Corella, J.P.; Wehrli, B.; Schubert, C.J. Mineralization pathways of organic matter deposited in a river–lake transition of the Rhone River Delta, Lake Geneva. Environ. Sci. Process. Impacts 2015, 17, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Schubert, C.J.; Niggemann, J.; Klockgether, G.; Ferdelman, T.G. Chlorin index: A new parameter for organic matter freshness in sediments. Geochem. Geophys. Geosyst. 2005, 6, 153–156. [Google Scholar] [CrossRef]
- Lee, C.; Wakeham, S.G.; Hedges, J.I. Composition and flux of particulate amino acids and chloropigments in equatorial Pacific seawater and sediments. Deep Sea Res. Part I Oceanogr. Res. Pap. 2000, 47, 1535–1568. [Google Scholar] [CrossRef]
- Haake, B.; Ittekkot, V.; Honjo, S.; Manganini, S. Amino acid, hexosamine and carbohydrate fluxes to the deep subarctic Pacific (Station P). Deep Sea Res. Part I Oceanogr. Res. Pap. 1993, 40, 547–560. [Google Scholar] [CrossRef]
- Cowie, G.L.; Hedges, J.I. Sources and reactivities of amino acids in a coastal marine environment. Limnol. Oceanogr. 1992, 37, 703–724. [Google Scholar] [CrossRef]
- Meckler, A.N.; Schubert, C.J.; Cowie, G.L.; Peiffer, S.; Dittrich, M. New organic matter degradation proxies: Valid in lake systems? Limnol. Oceanogr. 2004, 49, 2023–2033. [Google Scholar] [CrossRef]
- Dauwe, B.; Middelburg, J.J. Amino acids and hexosamines as indicators of organic matter degradation state in North Sea sediments. Limnol. Oceanogr. 1998, 43, 782–798. [Google Scholar] [CrossRef]
- Liu, X.B.; Li, G.F.; Liu, Z.G.; Guo, W.H.; Gao, N.Y. Water pollution characteristics and assessment of lower reaches in Haihe River basin. Procedia Environ. Sci. 2010, 2, 199–206. [Google Scholar] [CrossRef]
- Tang, W.Z.; Cui, J.G.; Shan, B.Q.; Wang, C.; Zhang, W.Q. Heavy metal accumulation by periphyton is related to eutrophication in the Hai River Basin, Northern China. PloS ONE 2014, 9, e86458. [Google Scholar] [CrossRef] [PubMed]
- Philips, S.; Laanbroek, H.J.; Verstraete, W. Origin, causes and effects of increased nitrite concentrations in aquatic environments. Rev. Environ. Sci. Biotechnol. 2002, 1, 115–141. [Google Scholar] [CrossRef]
- Huo, S.L.; Zhang, J.T.; Xi, B.D.; Zan, F.Y.; Su, J.; Yu, H. Distribution of nitrogen forms in surface sediments of lakes from different regions, China. Environ. Earth Sci. 2014, 71, 2167–2175. [Google Scholar] [CrossRef]
- Zhao, Y.; Shan, B.Q.; Tang, W.Z.; Zhang, H. Nitrogen mineralization and geochemical characteristics of amino acids in surface sediments of a typical polluted area in the Haihe River Basin, China. Environ. Sci. Pollut. Res. Int. 2015, 22, 17975–17986. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.C.; Liu, G.P.; Zhu, Y.Q. Determination of water-soluble ammonium ion in soil by spectrophotometry. Analyst 1987, 112, 909–911. [Google Scholar] [CrossRef]
- Ming, T.Y.; Li, J.J.; Huo, P.; Wei, Y.; Chen, X. Analysis of free amino acids in Russula griseocarnosa harvested at different stages of maturity using iTRAQ®-LC-MS/MS. Food Anal. Methods 2014, 7, 1816–1823. [Google Scholar] [CrossRef]
- Dauwe, B.; Middelburg, J.J.; Herman, P.M.; Heip, C.H. Linking diagenetic alteration of amino acids and bulk organic matter reactivity. Limnol. Oceanogr. 1999, 44, 1809–1814. [Google Scholar] [CrossRef]
- Hansen, L.S.; Blackburn, T.H. Aerobic and anaerobic mineralization of organic material in marine sediment microcosms. Mar. Ecol. Prog. Ser. 1991, 75, 283–291. [Google Scholar] [CrossRef]
- Stockenberg, A.; Johnstone, R.W. Benthic denitrification in the Gulf of Bothnia. Estuar. Coast. Shelf Sci. 1997, 45, 835–843. [Google Scholar] [CrossRef]
- Kalvelage, T.; Lavik, G.; Lam, P.; Contreras, S.; Arteaga, L.; Löscher, C.R.; Oschlies, A.; Paulmier, A.; Stramma, L.; Kuypers, M.M.M. Nitrogen cycling driven by organic matter export in the South Pacific oxygen minimum zone. Nat. Geosci. 2013, 6, 228–234. [Google Scholar] [CrossRef]
- Pietroski, J.P.; White, J.R.; DeLaune, R.D. Effects of dispersant used for oil spill remediation on nitrogen cycling in Louisiana coastal salt marsh soil. Chemosphere 2015, 119, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Carter, M.R. Soil Sampling and Methods of Analysis; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Aufdenkampe, A.K.; Hedges, J.I.; Richey, J.E.; Krusche, A.V.; Llerena, C.A. Sorptive fractionation of dissolved organic nitrogen and amino acids onto fine sediments within the Amazon Basin. Limnol. Oceanogr. 2001, 46, 1921–1935. [Google Scholar] [CrossRef]
- Lomstein, B.A.; Jørgensen, B.B.; Schubert, C.J.; Niggemann, J. Amino acid biogeo-and stereochemistry in coastal Chilean sediments. Geochim. Cosmochim. Acta 2006, 70, 2970–2989. [Google Scholar] [CrossRef]
- Vandewiele, S.; Cowie, G.; Soetaert, K.; Middelburg, J.J. Amino acid biogeochemistry and organic matter degradation state across the Pakistan margin oxygen minimum zone. Deep Sea Res. Part II Top. Stud. Oceanogr. 2009, 56, 376–392. [Google Scholar] [CrossRef]
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Hare, P.D.; Cress, W.A. Metabolic implications of stress-induced proline accumulation in plants. Plant Growth Regul. 1997, 21, 79–102. [Google Scholar] [CrossRef]
- Verbruggen, N.; Hermans, C. Proline accumulation in plants: A review. Amino Acids 2008, 35, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Yoshiba, Y.; Kiyosue, T.; Katagiri, T.; Ueda, H.; Mizoguchi, T.; Yamaguchi-Shinozaki, K.; Wada, K.; Harada, Y.; Shinizaki, K. Correlation between the induction of a gene for Δ1-pyrroline-5-carboxylate synthetase and the accumulation of proline in Arabidopsis thaliana under osmotic stress. Plant J. 1995, 7, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Siripornadulsil, S.; Traina, S.; Verma, D.P.S.; Sayre, R.T. Molecular mechanisms of proline-mediated tolerance to toxic heavy metals in transgenic microalgae. Plant Cell 2002, 14, 2837–2847. [Google Scholar] [CrossRef] [PubMed]
- Maggio, A.; Miyazaki, S.; Veronese, P.; Fujita, T.; Ibeas, J.I.; Damsz, B.; Narasimhan, M.L.; Hasegawa, P.M.; Joly, R.J.; Bressan, R.A. Does proline accumulation play an active role in stress-induced growth reduction? Plant J. 2002, 31, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Q.; Shan, B.Q.; Zhang, H.; Tang, W.Z. Assessment of preparation methods for organic phosphorus analysis in phosphorus-polluted Fe/Al-rich Haihe River Sediments using solution 31P-NMR. PLoS ONE 2013, 8, e76525. [Google Scholar] [CrossRef] [PubMed]
- Ruklisha, M.; Paegle, L.; Denina, I. L-valine biosynthesis during batch and fed-batch cultivations of Corynebacterium. glutamicum: Relationship between changes in bacterial growth rate and intracellular metabolism. Process Biochem. 2007, 42, 634–640. [Google Scholar] [CrossRef]
- Woulds, C.; Middelburg, J.J.; Cowie, G.L. Alteration of organic matter during infaunal polychaete gut passage and links to sediment organic geochemistry. Part I: Amino acids. Geochim. Cosmochim. Acta 2012, 77, 396–414. [Google Scholar] [CrossRef]
- Thomas, C.J.; Blair, N.E. Transport and digestive alteration of uniformly C-13-labeled diatoms in mudflat sediments. J. Mar. Res. 2002, 60, 517–535. [Google Scholar] [CrossRef]
- Tuominen, L. Comparison of leucine uptake methods and a thymidine incorporation method for measuring bacterial activity in sediment. J. Microbiol. Methods 1995, 24, 125–134. [Google Scholar] [CrossRef]
- Yao, X.; Zhu, G.W.; Cai, L.L.; Zhu, M.Y.; Zhao, L.L.; Gao, G.; Qin, B. Geochemical characteristics of amino acids in sediments of lake Taihu, a large, shallow, eutrophic freshwater lake of China. Aquat. Geochem. 2012, 18, 263–280. [Google Scholar] [CrossRef]
| Sample | mol% | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| sites | His | Arg | Lys | Asp | Glu | Ser | Thr | Val | Ala | Ile | Gly | Leu | Met | Tyr | Phe | Pro | Orn | Cit |
| S01 | 0.98 | 3.50 | 5.01 | 6.95 | 7.09 | 5.33 | 7.12 | 4.94 | 13.56 | 3.37 | 13.25 | 6.70 | 0.81 | 1.45 | 2.96 | 16.31 | 0.46 | 0.21 |
| S02 | 0.92 | 2.32 | 3.58 | 7.65 | 6.83 | 7.08 | 5.80 | 4.99 | 13.66 | 3.29 | 12.52 | 5.64 | nd | nd | 2.74 | 22.56 | 0.42 | nd |
| S03 | 1.23 | 3.26 | 4.71 | 7.53 | 7.01 | 6.76 | 6.44 | 5.56 | 13.81 | 3.19 | 12.98 | 7.01 | 1.18 | 0.92 | 3.22 | 14.73 | 0.46 | nd |
| S04 | 1.27 | 2.70 | 2.58 | 8.44 | 7.32 | 5.42 | 6.01 | 5.17 | 13.66 | 4.20 | 13.91 | 8.08 | 1.09 | 0.96 | 3.54 | 15.16 | 0.48 | nd |
| S05 | 1.68 | 2.84 | 3.30 | 5.74 | 6.33 | 4.38 | 4.35 | 3.97 | 9.43 | 3.10 | 8.95 | 29.96 | 1.06 | 1.42 | 2.35 | 10.66 | 0.41 | 0.07 |
| S06 | 0.77 | 2.73 | 4.35 | 7.77 | 7.54 | 4.98 | 6.80 | 5.76 | 12.82 | 3.74 | 11.93 | 7.36 | 1.16 | 0.67 | 3.11 | 18.02 | 0.50 | nd |
| S07 | 1.81 | 2.97 | 3.86 | 8.41 | 7.29 | 7.10 | 6.06 | 4.81 | 12.95 | 4.45 | 13.30 | 8.28 | 1.71 | 1.30 | 3.71 | 11.39 | 0.52 | 0.06 |
| S08 | 0.94 | 2.41 | 4.16 | 6.92 | 6.76 | 6.62 | 6.38 | 5.54 | 14.34 | 3.64 | 12.70 | 6.52 | 1.88 | 0.70 | 3.37 | 16.51 | 0.60 | nd |
| S09 | 1.09 | 2.77 | 3.39 | 8.90 | 7.26 | 5.29 | 7.26 | 5.93 | 13.73 | 4.57 | 11.77 | 8.20 | 1.43 | 1.50 | 3.80 | 12.60 | 0.53 | nd |
| S10 | 1.67 | 2.83 | 2.66 | 9.28 | 7.67 | 6.42 | 6.10 | 5.73 | 12.65 | 4.39 | 13.18 | 8.51 | 1.89 | 1.64 | 3.68 | 11.17 | 0.44 | 0.09 |
| S11 | 1.26 | 2.68 | 3.95 | 10.16 | 7.49 | 7.42 | 4.34 | 3.44 | 22.22 | nd | 14.69 | 5.05 | nd | nd | 2.23 | 15.06 | nd | nd |
| S12 | 1.84 | 2.86 | 3.66 | 8.36 | 6.91 | 5.87 | 6.64 | 5.52 | 14.45 | 4.47 | 12.82 | 8.28 | 1.58 | 1.75 | 3.49 | 11.10 | 0.41 | nd |
| S13 | 1.17 | 2.87 | 3.05 | 7.93 | 7.21 | 5.60 | 7.04 | 5.66 | 14.64 | 4.20 | 13.55 | 8.11 | nd | 0.70 | 3.41 | 14.54 | 0.30 | nd |
| S14 | 1.35 | 2.42 | 3.29 | 9.74 | 7.02 | 6.50 | 5.93 | 5.72 | 13.63 | 4.43 | 13.31 | 8.06 | 1.18 | 1.28 | 3.46 | 12.13 | 0.49 | 0.06 |
| S15 | 2.12 | 3.18 | 3.28 | 9.04 | 8.37 | 5.91 | 4.58 | 5.15 | 12.25 | 4.48 | 12.21 | 8.18 | 1.38 | 2.48 | 3.32 | 13.52 | 0.45 | 0.09 |
| S16 | 0.94 | 2.56 | 2.60 | 8.41 | 6.25 | 6.26 | 5.09 | 5.22 | 20.38 | 2.48 | 11.82 | 5.93 | nd | 0.22 | 2.56 | 19.28 | nd | nd |
| S17 | 1.82 | 3.11 | 2.89 | 9.33 | 6.81 | 6.70 | 5.47 | 5.81 | 12.78 | 4.78 | 12.88 | 8.62 | 0.95 | 2.14 | 3.81 | 11.52 | 0.54 | 0.04 |
| S18 | 0.93 | 2.57 | 3.27 | 9.23 | 7.66 | 5.57 | 5.71 | 5.00 | 12.96 | 4.34 | 12.77 | 7.88 | 1.37 | 0.49 | 3.49 | 15.88 | 0.70 | 0.17 |
| S19 | 1.20 | 3.06 | 3.27 | 7.52 | 6.66 | 6.17 | 6.97 | 4.70 | 12.79 | 4.00 | 14.23 | 7.15 | nd | 0.76 | 2.87 | 18.12 | 0.54 | nd |
| Chemical Index | NH3-N | TN | OrgNsed | TOC | THAAs-C | THAAs-N |
|---|---|---|---|---|---|---|
| NH3-N | 1 | 0.529 * | 0.475 * | 0.437 | 0.716 * | 0.679 * |
| TN | 1 | 0.998 ** | 0.978 ** | 0.839 ** | 0.872 ** | |
| OrgNsed | 1 | 0.982 ** | 0.817 ** | 0.854 ** | ||
| TOC | 1 | 0.744 ** | 0.791 ** | |||
| THAAs-C | 1 | 0.994 ** | ||||
| THAAs-N | 1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Shan, B.; Tang, W.; Zhang, H.; Rong, N.; Ding, Y. Distributions, Early Diagenesis, and Spatial Characteristics of Amino Acids in Sediments of Multi-Polluted Rivers: A Case Study in the Haihe River Basin, China. Int. J. Environ. Res. Public Health 2016, 13, 234. https://doi.org/10.3390/ijerph13020234
Zhao Y, Shan B, Tang W, Zhang H, Rong N, Ding Y. Distributions, Early Diagenesis, and Spatial Characteristics of Amino Acids in Sediments of Multi-Polluted Rivers: A Case Study in the Haihe River Basin, China. International Journal of Environmental Research and Public Health. 2016; 13(2):234. https://doi.org/10.3390/ijerph13020234
Chicago/Turabian StyleZhao, Yu, Baoqing Shan, Wenzhong Tang, Hong Zhang, Nan Rong, and Yuekui Ding. 2016. "Distributions, Early Diagenesis, and Spatial Characteristics of Amino Acids in Sediments of Multi-Polluted Rivers: A Case Study in the Haihe River Basin, China" International Journal of Environmental Research and Public Health 13, no. 2: 234. https://doi.org/10.3390/ijerph13020234
APA StyleZhao, Y., Shan, B., Tang, W., Zhang, H., Rong, N., & Ding, Y. (2016). Distributions, Early Diagenesis, and Spatial Characteristics of Amino Acids in Sediments of Multi-Polluted Rivers: A Case Study in the Haihe River Basin, China. International Journal of Environmental Research and Public Health, 13(2), 234. https://doi.org/10.3390/ijerph13020234





