A Multiscale Mapping Assessment of Lake Champlain Cyanobacterial Harmful Algal Blooms
Abstract
:1. Introduction
2. Methods
2.1. Lake Champlain
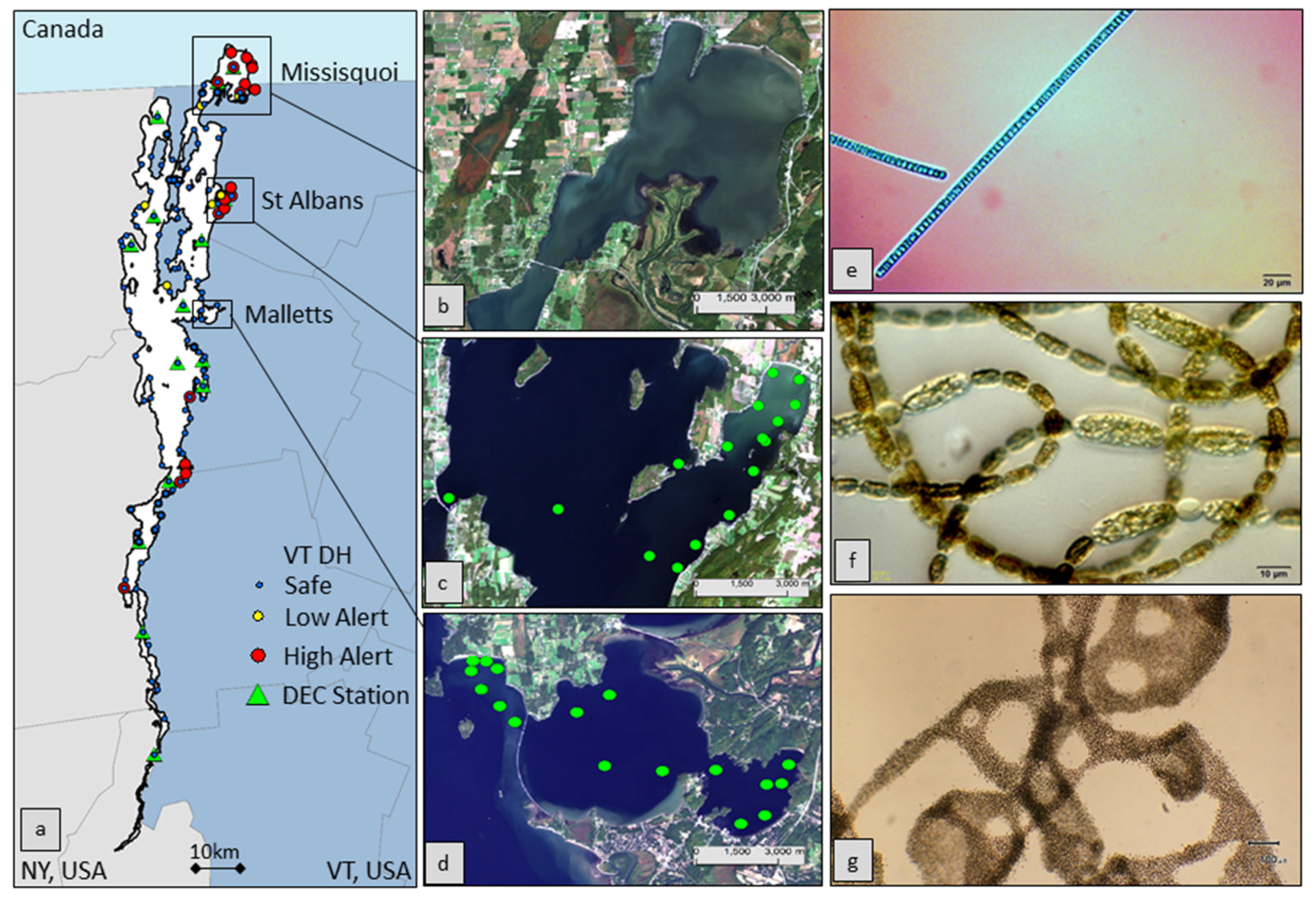
2.2. Field Data Collection
| Sensor | Spatial Resolution | Bands | Spectral | Footprint | Overpass Day Of Year |
|---|---|---|---|---|---|
| Landsat 8 OLI | 30 | 11 | VNIR, MIR, Thermal | 185 × 185 km | 267 |
| Proba-1 CHRIS | 18 | 19 | VNIR | 13 × 13 km | 265 |
| RapidEye | 5 | 5 | VNIR | 25 × 25 km | 260 |
2.3. Landsat 8 OLI
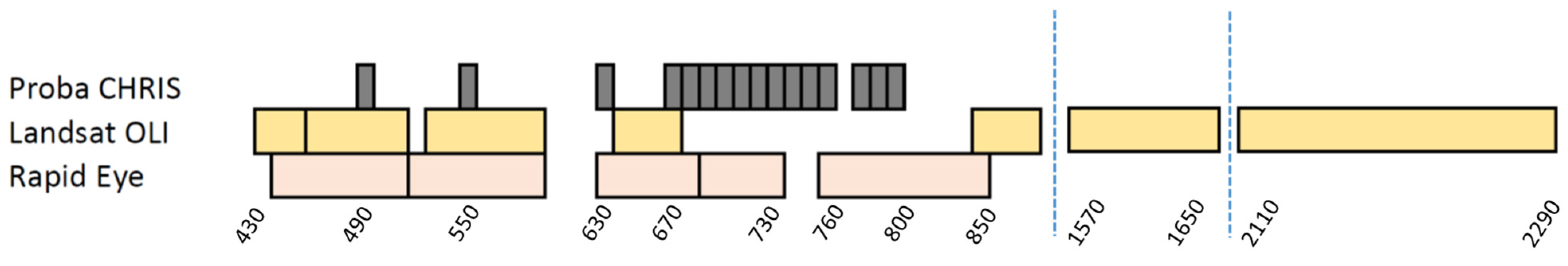
2.4. RapidEye
2.5. PROBA-1 CHRIS
2.6. Analytical Approach
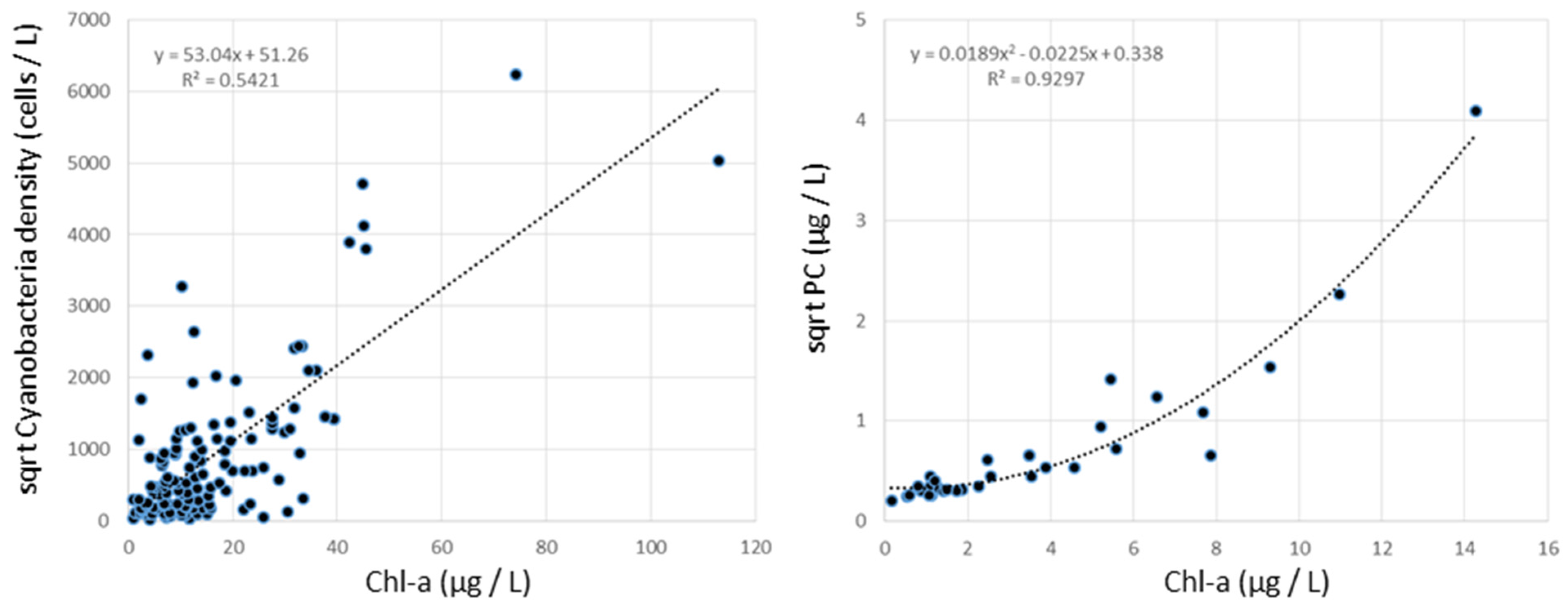
3. Results and Discussion
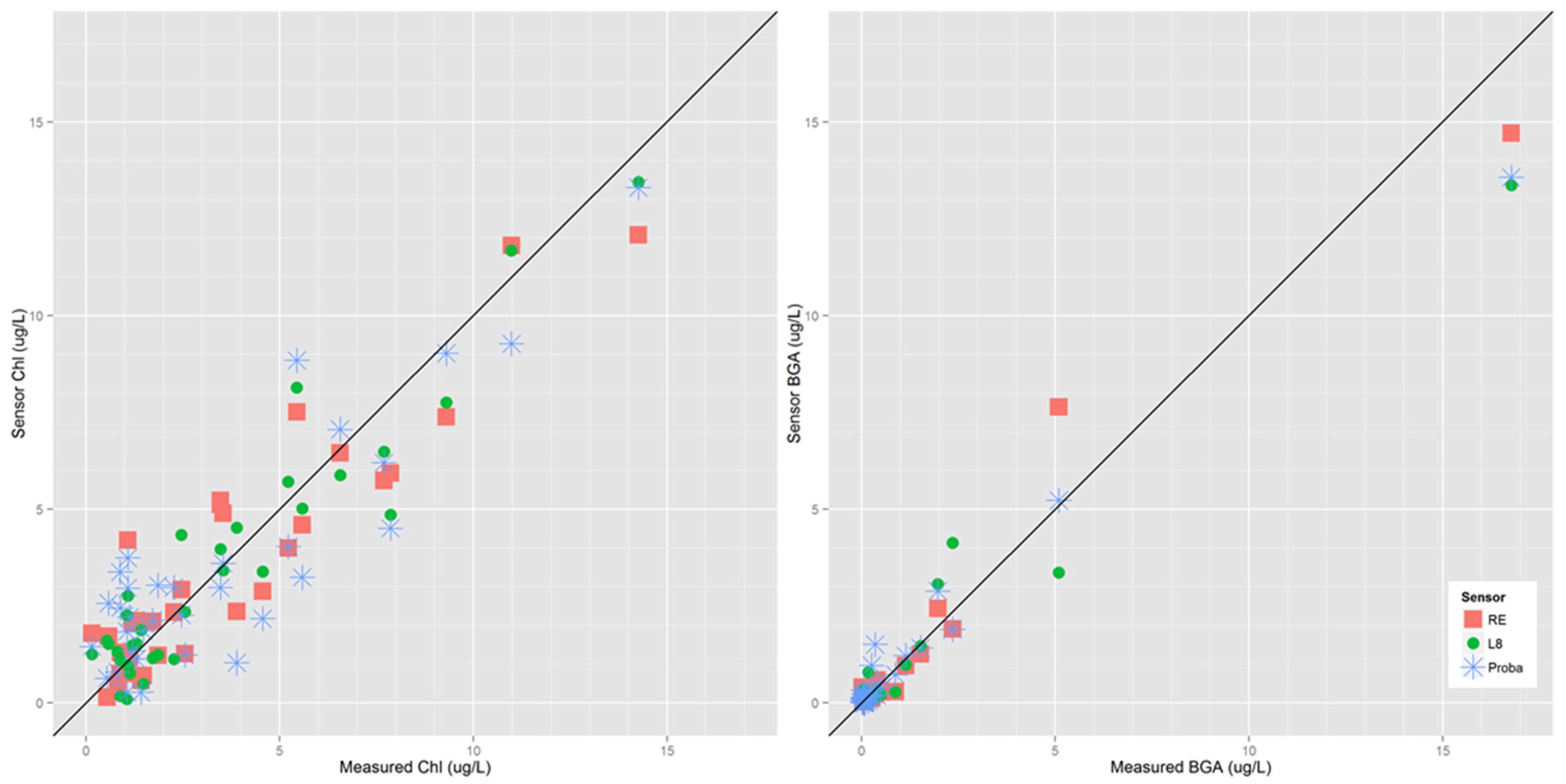
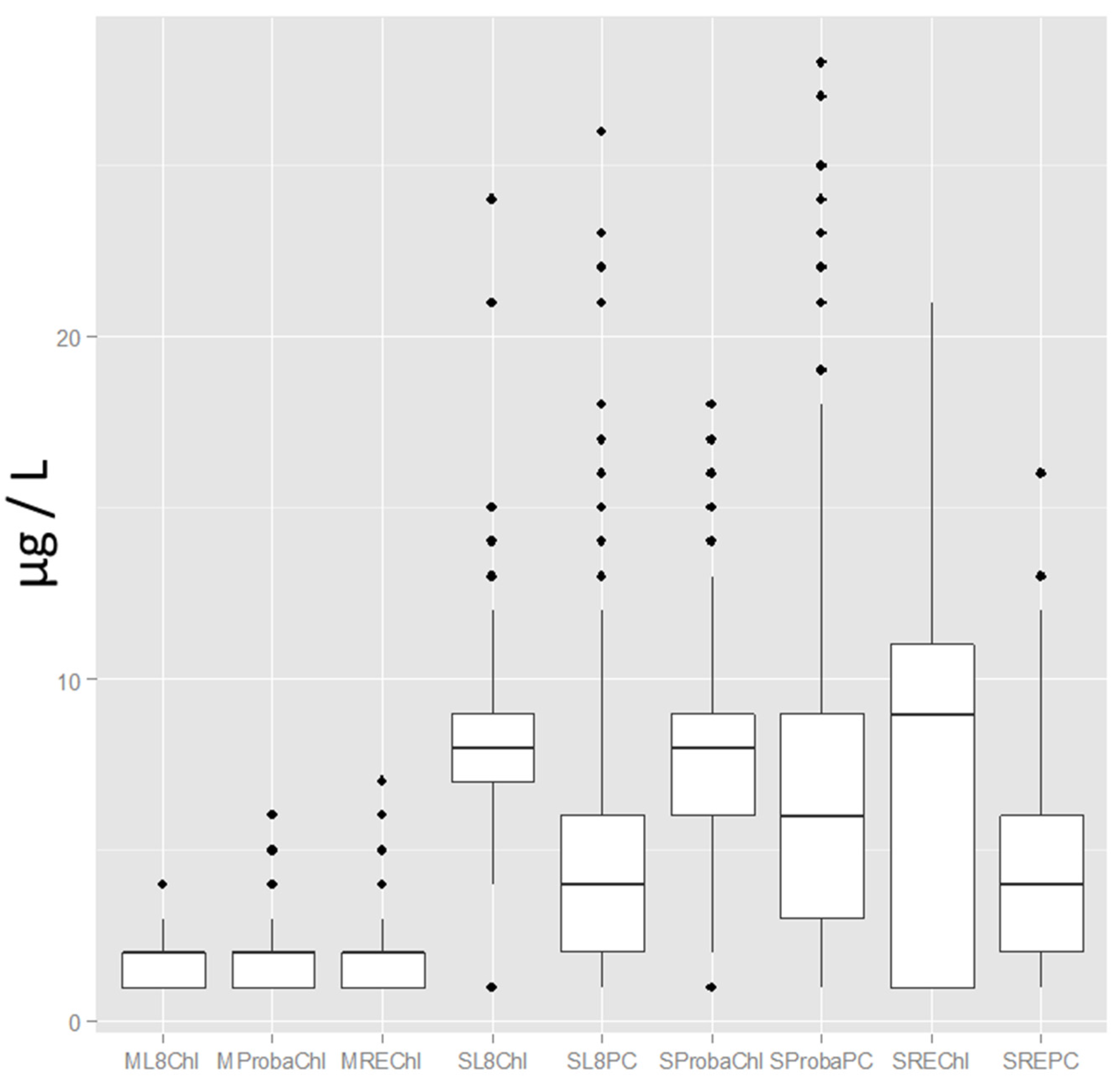
3.1. Sensor Evaluation
| Metric | Sensor | Model | Adj R2 | RMSE |
|---|---|---|---|---|
| chl-a | Landsat 8 OLI | − 59.33 + B4/B2 (34.7) + B5 (0.006) | 0.77 | 0.41 |
| chl-a | Proba-1 CHRIS | −4.26 + B1 (−338.6) + B2/B1 (−0.9) + B2 (682.9) + B15 (−939.1) | 0.88 | 0.54 |
| chl-a | RapidEye | −2.84 + B1 (−0.05) + B3 (0.08) | 0.81 | 1.46 |
| PC | Landsat 8 OLI | − 2.85 + B1 (0.013) + B3 (−0.43) + B4 (0.76) | 0.83 | 1.33 |
| PC | Proba-1 CHRIS | 6.2 + B2 (334.8) + B6 (− 1644.3) + B8 (2031.6) + B11 (−709.4) + B14 (−1324.3) | 0.88 | 1.02 |
| PC | RapidEye | − 56.13 + B3 (0.12) + B1/B3 (9.49) | 0.77 | 1.52 |
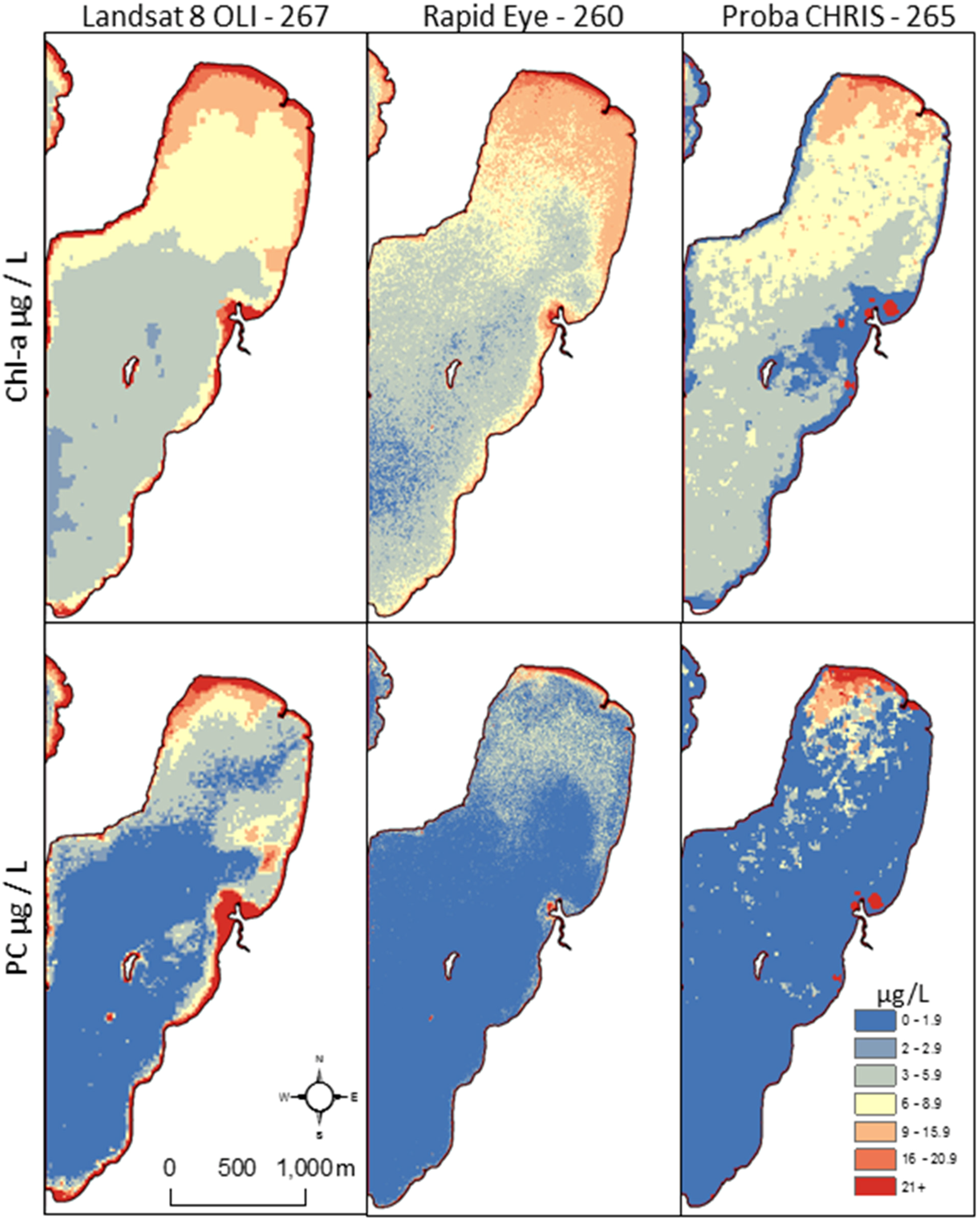
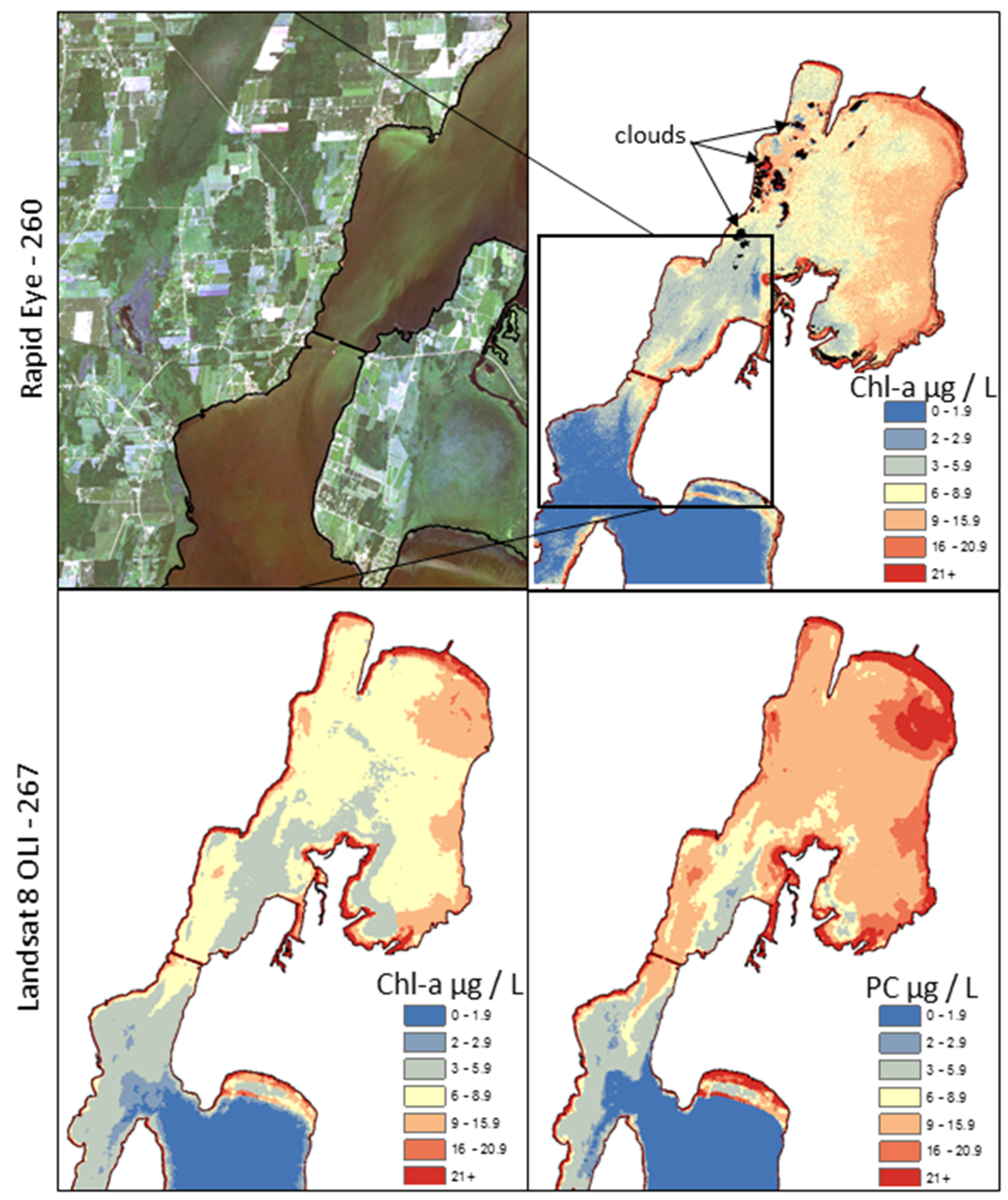
3.2. Alert Status Mapping
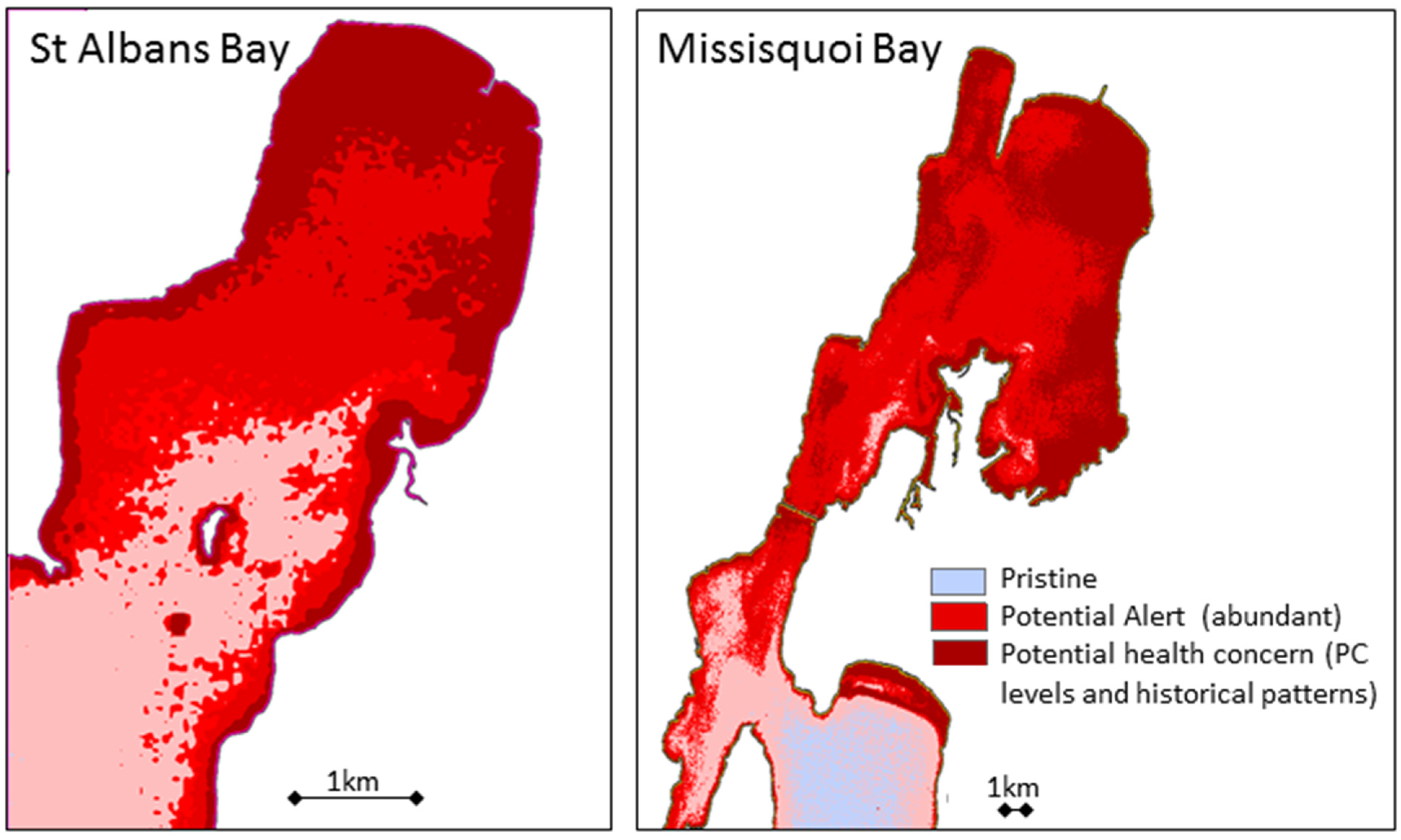
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rao, P.V.; Gupta, N.; Bhaskar, A.S.; Jayaraj, R. Toxins and bioactive compounds from cyanobacteria and their implications on human health. J. Environ. Biol. 2002, 23, 215–224. [Google Scholar] [PubMed]
- Codd, G.A.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Lévesque, B.; Gervais, M.C.; Chevalier, P.; Gauvin, D.; Anassour-Laouan-Sidi, E.; Gingras, S.; Fortin, N.; Brisson, G.; Greer, C.; Bird, D. Prospective study of acute health effects in relation to exposure to cyanobacteria. Sci. Total. Environ. 2014, 466–467, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Caller, T.A.; Doolin, J.W.; Haney, J.F.; Murby, A.J.; West, K.G.; Farrar, H.E.; Ball, A.; Harris, B.T.; Stommel, E.W. A cluster of amyotrophic lateral sclerosis in New Hampshire: A possible role for toxic cyanobacteria blooms. Amyotroph. Lateral Sc. 2009, 10, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Bradley, W.G.; Borenstein, A.R.; Nelson, L.M.; Codd, G.A.; Rosen, B.H.; Stommel, E.W.; Cox, P.A. Is exposure to cyanobacteria an environmental risk factor for amyotrophic lateral sclerosis and other neurodegenerative diseases? Amyotroph. Lateral Scler. Frontotemporal Degener. 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Torbick, N.; Hession, S.; Stommel, E.; Caller, T. Mapping amyotrophic lateral sclerosis lake risk factors across northern New England. Int. J. Health Geogr. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Caller, T.; Henegan, P.; Haney, J.; Murby, A.; Metcalf, J.S.; Powell, J.; Cox, P.A.; Stommel, E. Detection of cyanotoxins, β-N-methylamino-L-alanine and microcystins from a lake surrounded by cases of Amyotrophic Lateral Sclerosis. Toxins 2015, 7, 322–336. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, V.K.; Chakrabartty, A. Protein misfolding in the late-onset neurodegenerative diseases. Proteins 2013, 81, 1285–1303. [Google Scholar] [CrossRef] [PubMed]
- Grad, L.; Cashman, N.R. Prion-like activity of Cu/Zn superoxide dismutase: Implications for ALS. Prion 2014, 8, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Furukawa, K.; Okajima, T. Extracellular O-linked β-N-acetylglucosamine: Its biology and relationship to human disease. World J. Biol. Chem. 2014, 5, 224–230. [Google Scholar] [PubMed]
- Ravits, J. Focality, stochasticity and neuroanatomic propagation in ALS pathogenesis. Exp. Neurol. 2014, 262, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, R.; Cox, P.A.; Banack, S.A.; Rodgers, K.J. The non-protein amino acid BMAA is misincorporated into human proteins in place of L-serine causing protein misfolding and aggregation. PLoS ONE 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Robberecht, W.; Philips, T. The changing scene of amyotrophic lateral sclerosis. Nature Rev. Neurosci. 2013, 14, 248–264. [Google Scholar] [CrossRef] [PubMed]
- John, D.; Whitton, B.; Brook, A. The Freshwater Algal Flora of the British Isles: An Identification Guide to Freshwater and Terrestrial Algae; Cambridge University Press: New York, NY, USA, 2002. [Google Scholar]
- Joosten, A. Flora of the Blue-Green Algae of the Netherlands. The Non-Filamentous Species of Inland Waters; KNNV Publishing: Utrect, The Netherlands, 2006. [Google Scholar]
- Prescott, G. Algae of the Western Great Lakes Area; Otto Koeltz Science Publishers: Koenigstein, Germany, 1982. [Google Scholar]
- Becker, R.H.; Sultan, M.I.; Boyer, G.L.; Twiss, M.R.; Konopko, E. Mapping cyanobacterial blooms in the Great Lakes using MODIS. J. Great Lakes Res. 2009, 35, 447–453. [Google Scholar] [CrossRef]
- Chipman, J.; Lillesand, T.; Schmaltz, J.; Leale, J.; Nordheim, M. Mapping lake water clarity with Landsat images in Wisconsin, USA. Can. J. Remote Sens. 2004, 30, 1–7. [Google Scholar] [CrossRef]
- Dekker, A.; Peters, S. A TM study of eutrophic lakes in the Netherlands. Int. J. Remote Sens. 1993, 14, 799–821. [Google Scholar] [CrossRef]
- Kloiber, S.; Brezonik, P.; Bauer, M. Application of Landsat imagery to regional-scale assessments of lake clarity. Water Res. 2002, 36, 4330–4340. [Google Scholar] [CrossRef]
- Lathrop, R. Landsat thematic mapper monitoring of turbid inland water quality. Photogramm. Eng. Remote Sens. 1992, 58, 465–470. [Google Scholar]
- Gitelson, A.; Zhou, J.; Gurlin, A.; Verma, S.; Moses, W.; Ioannou, I.; Ahmed, S. Algorithms for remote estimation of chlorophyll-a in coastal and inland waters using red and near infrared bands. Opt. Express 2010, 18, 24109–24125. [Google Scholar]
- Gons, H.; Rijkeboer, M.; Ruddick, K. A chlorophyll-retrieval algorithm for satellite imagery (Medium Resolution Imaging Spectrometer) of inland and coastal waters. J. Plankton Res. 2002, 24, 947–951. [Google Scholar] [CrossRef]
- McCoullough, I.; Loftin, C.; Sader, S. High-frequency remote monitoring of large lakes with MODIS 500 m imagery. Remote Sens. Environ. 2012, 124, 234–241. [Google Scholar] [CrossRef]
- Moore, T.; Dowell, M.; Bradt, S.; Verdu, A. An optical water type framework for selecting and blending retrievals from bio-optical algorithms in lakes and coastal waters. Remote Sens. Environ. 2014, 143, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Olmanson, L.; Brezonik, P.; Bauer, M. Evaluation of medium to low resolution satellite imagery for regional lake water quality assessments. Water Resour. Res. 2011, 47, W09515. [Google Scholar] [CrossRef]
- Ruiz-Verdu, A.; Simis, S.; Hoyos, C.; Gon, H.; Pena-Martinez, R. An evaluation of algorithms for the remote sensing of cyanobacterial biomass. Remote Sens. Environ. 2008, 112, 3996–4008. [Google Scholar] [CrossRef]
- Simis, S.; Ruiz-Verdu, A.; Dominguez-Gomez, J.; Pena-Martinez, R.; Peters, S.; Gons, H. Influence of phytoplankton pigment composition on remote sensing of cyanobacterial biomass. Remote Sen. Environ. 2007, 28, 414–427. [Google Scholar] [CrossRef]
- Stumpf, R.; Wynne, T.; Baker, D.; Fahnenstiel, G. Interannual variability of cyanobacterial blooms in Lake Erie. PLoS ONE 2012, 8. [Google Scholar] [CrossRef] [PubMed]
- Torbick, N.; Hu, F.; Zhang, J.; Qi, J.; Zhang, H.; Becker, B. Mapping Chlorophyll-a Concentrations in West Lake China using Landsat 7 ETM+. J. Great Lakes Res. 2008, 34, 559–565. [Google Scholar] [CrossRef]
- Torbick, N.; Hession, S.; Hagen, S.; Wiangwang, N.; Becker, B.; Qi, J. Mapping inland lake water quality across the Lower Peninsula of Michigan using Landsat TM imagery. Int. J. Remote Sens. 2013, 34, 7607–7624. [Google Scholar] [CrossRef]
- Vincent, R.; Qin, X.; McKay, R.; Miner, J.; Czajkowski, K.; Savino, J.; Bridgeman, T. Phycocyanin detection from Landsat TM data for mapping cyanobacterial blooms in Lake Erie. Remote Sens. Environ. 2004, 89, 381–392. [Google Scholar] [CrossRef]
- Mouw, C.B.; Greb, S.; Aurin, D.; DiGiacomo, P.; Lee, Z.; Twardowski, M.; Binding, C.; Hu, C.; Ma, R.; Moore, T.; et al. Aquatic color radiometry remote sensing of coastal and inland waters: Challenges and recommendations for future satellite missions. Remote Sens Environ 2015, 160, 15–30. [Google Scholar] [CrossRef]
- Trescott, A. Remote Sensing Models of Algal Blooms and Cyanobacteria in Lake Champlain. Available online: http://scholarworks.umass.edu/cgi/viewcontent.cgi?article=1045&context=cee_ewre (accessed on 2 January 2012).
- Wheeler, S.; Morrissey, L.; Levine, S.; Livingston, G.; Vincent, W. Mapping cyanobacterial blooms in Lake Champlain’s Missisquoi Bay using QuickBird and MERIS satellite data. J. Great Lakes Res. 2012, 38, 68–75. [Google Scholar] [CrossRef]
- Gower, J.; King, S.; Goncalves, P. Global monitoring of plankton blooms using MERIS MCI. Int. J. Remote Sens. 2008, 29, 6209–6216. [Google Scholar] [CrossRef]
- Simis, S.; Peters, S.; Gons, H. Remote sensing of the cyanobacterial pigment phycocyanin in turbid inland water. Limnol. Oceanogr. 2005, 50, 237–245. [Google Scholar] [CrossRef]
- Wynne, T.; Stumpf, R.; Tomlinson, M.; Dyble, J. Characterizing a cyanobacterial blooms in western Lake Erie. J. Marine Syst. 2010, 83, 210–220. [Google Scholar]
- Lunetta, R.; Blake, A.; Schaeffer, B.; Stumpf, R.P.; Keith, D.; Jacobs, S.A.; Murphy, M.S. Evaluation of cyanobacteria cell count detection derived from MERIS imagery across the eastern USA. Remote Sens. Environ. 2015, 157, 24–34. [Google Scholar] [CrossRef]
- Wynne, T.T.; Stumpf, R.P.; Tomlinson, M.C.; Warner, R.A.; Tester, P.A.; Dyble, J.; Fahnenstiel, G.L. Relating spectral shape to cyanobacterial blooms in the Laurentian Great Lakes. Int. J. Remote Sens. 2008, 29, 3665–3672. [Google Scholar] [CrossRef]
- Masek, J.G.; Vermote, E.F.; Saleous, N.; Wolfe, R.; Hall, F.G.; Huemmrich, F.; Gao, F.; Kutler, J.; Lim, T.K. A Landsat Surface Reflectance Data Set for North America, 1990–2000. IEEE Geosci. Remote S. 2006, 3, 68–72. [Google Scholar] [CrossRef]
- Irish, R.; Barker, J.; Goward, S.; Arvidson, T. Characterization of the Landsat-7 ETM+ automated cloud cover assessment (ACCA) algorithm. Photogramm. Eng. Remote Sens. 2006, 72, 1179–1188. [Google Scholar] [CrossRef]
- Fomferra, N.; Brockmann, C. Beam, the ENVISAT MERIS and AASTR Toolbox. Available online: http://adsabs.harvard.edu/full/2005ESASP.597E..13F (accessed on 4 June 2015).
- Guanter, L.; Alonso, L.; Moreno, J. CHRIS Proba Atmospheric Correction Module. Algorithm Theoretical Basis Document. Available online: http://www.brockmann-consult.de/beam-wiki/download/attachments/32964611/chrisbox-atmospheric_correction_atbd-2.0.pdf (accessed on 4 June 2015).
- Casal, G.; Kutser, T.; Dominguez-Gomez, J.; Sanchez-Carnero, N.; Freire, J. Mapping benthic macroalgal communities in the coastal zone using CHRIS-PROBA mode 2 images. Estuar. Coast. Shelf S. 2011, 94, 281–290. [Google Scholar] [CrossRef]
- Gomez-Chova, L.; Alonso, L.; Gaunter, L.; Calpe, J.; Moreno, J. Algorithm Theoretical Basis Document. CHRIS Proba Noise Reduction Module. Available online: http://www.brockmann-consult.de/beam-wiki/download/attachments/32964611/chrisbox-noise_reduction_atbd-2.0.pdf?version=1&modificationDate=1251269690000 (accessed on 4 June 2015).
- Cernicharo, J.; Verger, A.; Camacho, F. Empirical and physical estimation of canopy water content from CHRIS/Proba data. Remote Sens. 2013, 5, 5265–5284. [Google Scholar] [CrossRef]
- Millan, V.; Sanchez-Azofeifa, A.; Garcia, G.; Rivard, B. Quantifying tropical dry forest succession in the Americas. Remote Sens. Environ. 2014, 144, 120–136. [Google Scholar] [CrossRef]
- Verrelst, J.; Romijn, E.; Kooistra, L. Mapping vegetation density in a heterogeneous river floodplain ecosystem using pointable CHRIS Proba Data. Remote Sens. 2012, 4, 2866–2889. [Google Scholar] [CrossRef]
- Daviesm, W.; North, P. Synergistic angular and spectral estimation of aerosol properties using CHRIS Proba-1 and simulated Sentinel-3 data. Atmos. Meas. Tech. 2015, 8, 1719–1731. [Google Scholar] [CrossRef]
- Dall’Olmo, G.; Gitelson, A.A. Effect of bio-optical parameter variability and uncertainties in reflectance measurements on the remote estimation of chlorophyll-a concentration in turbid productive waters: Modeling results. Appl. Opt. 2006, 45, 3577–3592. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Mishra, D.R. Normalized difference chlorophyll index: A novel model for remote estimation of chlorophyll aconcentration in turbid productive waters. Remote Sens. Environ. 2011, 117, 394–406. [Google Scholar] [CrossRef]
- Olmanson, L.; Bauer, M.; Brezonik, P. A 20-year Landsat Water Clarity Census of Minnesota’s 10,000 Lakes. Remote Sensing of Environment. Remote Sens. Environ. 2008, 112, 4086–4097. [Google Scholar] [CrossRef]
- Wang, F.; Han, L.; Kung, H.; van Arsdale, R. Applications of Landsat-5 TM imagery in assessing and mapping water quality in Reelfoot Lake, Tennessee. Int. J. Remote Sens. 2006, 27, 5269–5283. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torbick, N.; Corbiere, M. A Multiscale Mapping Assessment of Lake Champlain Cyanobacterial Harmful Algal Blooms. Int. J. Environ. Res. Public Health 2015, 12, 11560-11578. https://doi.org/10.3390/ijerph120911560
Torbick N, Corbiere M. A Multiscale Mapping Assessment of Lake Champlain Cyanobacterial Harmful Algal Blooms. International Journal of Environmental Research and Public Health. 2015; 12(9):11560-11578. https://doi.org/10.3390/ijerph120911560
Chicago/Turabian StyleTorbick, Nathan, and Megan Corbiere. 2015. "A Multiscale Mapping Assessment of Lake Champlain Cyanobacterial Harmful Algal Blooms" International Journal of Environmental Research and Public Health 12, no. 9: 11560-11578. https://doi.org/10.3390/ijerph120911560
APA StyleTorbick, N., & Corbiere, M. (2015). A Multiscale Mapping Assessment of Lake Champlain Cyanobacterial Harmful Algal Blooms. International Journal of Environmental Research and Public Health, 12(9), 11560-11578. https://doi.org/10.3390/ijerph120911560





