Health and Quality of Life Perception in Older Adults: The Joint Role of Cognitive Efficiency and Functional Mobility
Abstract
:1. Introduction
2. Experimental Section
2.1. Recruitment and Participants
2.2. Testing
- (1)
- Motor-motor dual-task: participants walking at maximal speed stepped with their preferred foot over two plastic hurdles of 15 and 45 cm height placed in succession on the mid-line of the course (hurdles WS).
- (2)
- Motor-cognitive dual-task: participants were asked to talk while walking, naming as many animals as they could beginning with either letter B or C (talk WS).
2.3. Statistical Analysis
- (1)
- Interaction variables were computed by multiplying each executive function by each functional mobility variable.
- (2)
- Three separate hierarchical multiple regression analyses with forced entry method were run for the prediction of MCS and PCS, one for each main executive function predictor. Maximal walking speed and index of sarcopenia were statistically controlled for by entering them in a first block, while the individual predictors (one executive function variable and the two dual-task WS variables) were entered in a second block and the interaction terms of the executive function with the dual-task WSs in a third block.
- (3)
- In case interaction terms significantly predicted MCS or PCS, posthoc analysis through a simple slope test was performed [40]. The statistical significance was set at p < 0.05.
3. Results
3.1. Descriptive Statistics and Preliminary Analyses
| All | Men | Women | p | |
|---|---|---|---|---|
| Age (years) | 69.6 ± 3.2 | 70.0 ± 3.3 | 69.4 ± 3.2 | n.s. |
| Height (cm) | 1.7 ± 0.1 | 172.9 ± 5.0 | 162.4 ± 7.0 | <0.001 |
| Weight (kg) | 72.8 ± 11.5 | 81.2 ± 9.6 | 66.7 ± 8.4 | <0.001 |
| Body fat (%) | 33.8 ± 7.9 | 28.7 ± 5.2 | 37.5 ± 7.4 | <0.001 |
| Sarcopenia (index) | 7.6 ± 1.3 | 8.8 ± 0.8 | 6.8 ± 0.8 | <0.001 |
| Maximal WS (m/s) | 1.9 ± 0.2 | 1.91 ± 0.2 | 1.8 ± 0.2 | n.s. |
| Talking WS (m/s) | 1.6 ± 0.3 | 1.6 ± 0.3 | 1.6 ± 0.2 | n.s. |
| Hurdles WS (m/s) | 1.6 ± 0.2 | 1.7 ± 0.2 | 1.6 ± 0.2 | <0.001 |
| Inhibition (std index) | 0.098 ± 0.8 | −0.08 ± 0.9 | 0.05 ± 1.0 | n.s. |
| Working memory (std index) | −0.013 ± 0.8 | −0.33 ± 1.07 | 0.22 ± 0.5 | 0.020 |
| Δ Trail (s) | 43.0 ± 29.1 | 52.9 ± 34.7 | 39.1 ± 29.5 | n.s. |
| MCS (score) | 83.8 ± 13.2 | 84.1 ± 12.6 | 82.9 ± 14.3 | n.s. |
| PCS (score) | 71.0 ± 13.4 | 71.4 ± 15.4 | 70.1 ± 12.3 | n.s. |
| Maximal WS | Hurdles WS | Talking WS | Inhibition | Working Memory | Δ Trail | |
|---|---|---|---|---|---|---|
| MCS | −0.057 | 0.178 | −0.082 | 0.359** | −0.030 | −0.063 |
| PCS | −0.083 | 0.103 | −0.114 | 0.113 | −0.111 | −0.112 |
3.2. Moderated Prediction Analyses
| Exploratory Prediction Model | PCS | R2 = 0.14 | F3,52 = 2.78, p = 0.050 |
| β | p | ||
| Inhibition | 0.337 | 0.016 | |
| Working Memory | −0.084 | 0.548 | |
| Cognitive Flexibility | −0.092 | 0.528 | |
| PCS | R2 = 0.08 | F3,52 = 1.49, p = 0.230 | |
| β | p | ||
| Inhibition | 0.029 | 0.834 | |
| Working Memory | −0.205 | 0.160 | |
| Cognitive Flexibility | −0.271 | 0.077 |
| Moderated Prediction Model | MCS | R2 = 0.25 | F5,54 = 3.25, p = 0.013 |
| β | p | ||
| Maximal WS | −0.171 | 0.302 | |
| Index of Sarcopenia | −0.169 | 0.197 | |
| Inhibition | 0.211 | 0.133 | |
| Talking WS | −0.119 | 0.476 | |
| Talking WS × Inhibition | 0.343 | 0.031 | |
| PCS | R2 = 0.18 | F5,55 = 2.17, p = 0.72 | |
| β | p | ||
| Maximal WS | −0.350 | 0.039 | |
| Index of Sarcopenia | −0.048 | 0.723 | |
| Cognitive Flexibility (Δ Trail) | −0.041 | 0.728 | |
| Hurdles WS | 0.325 | 0.053 | |
| Hurdles WS × Δ Trail | −0.360 | 0.020 |
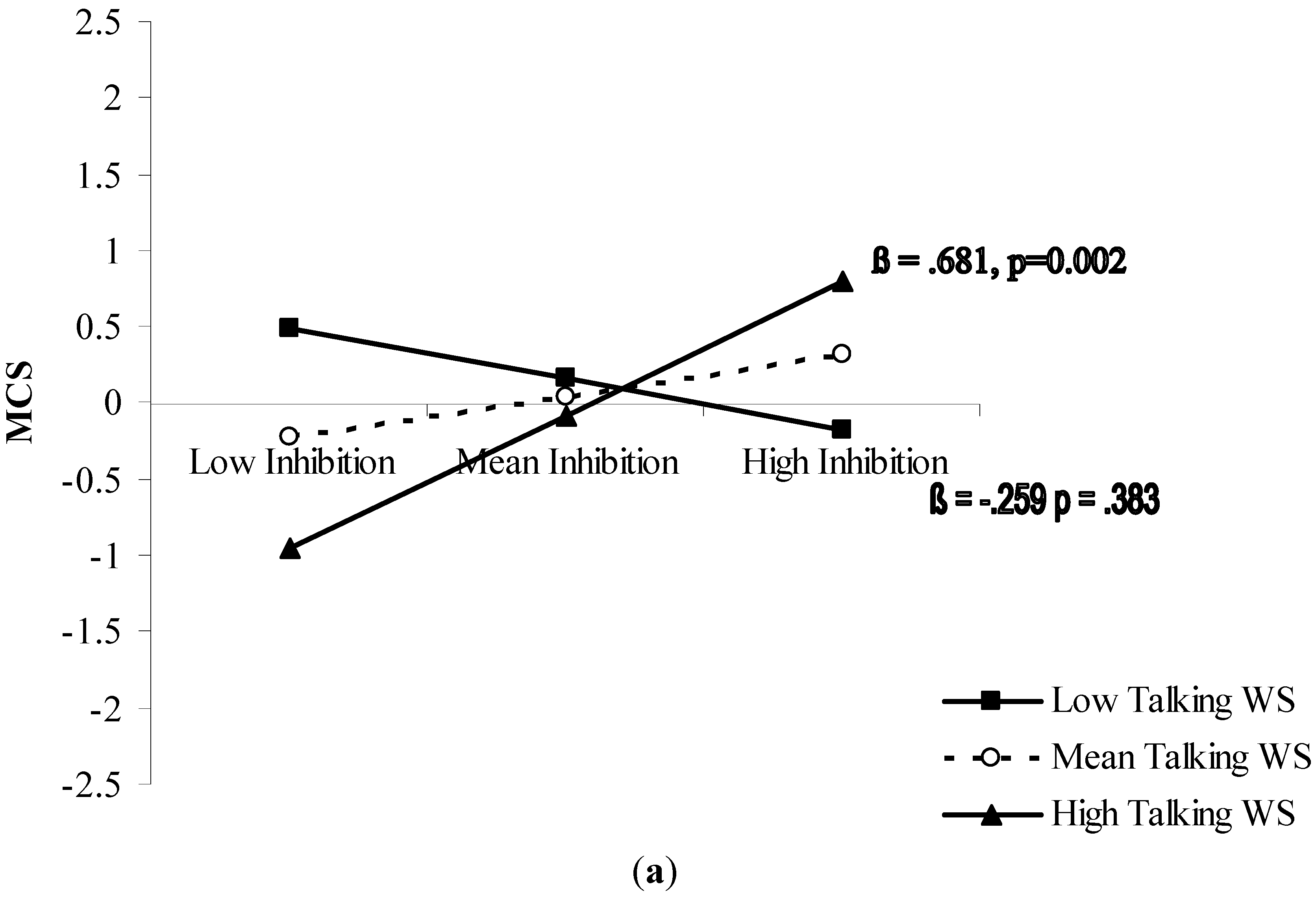
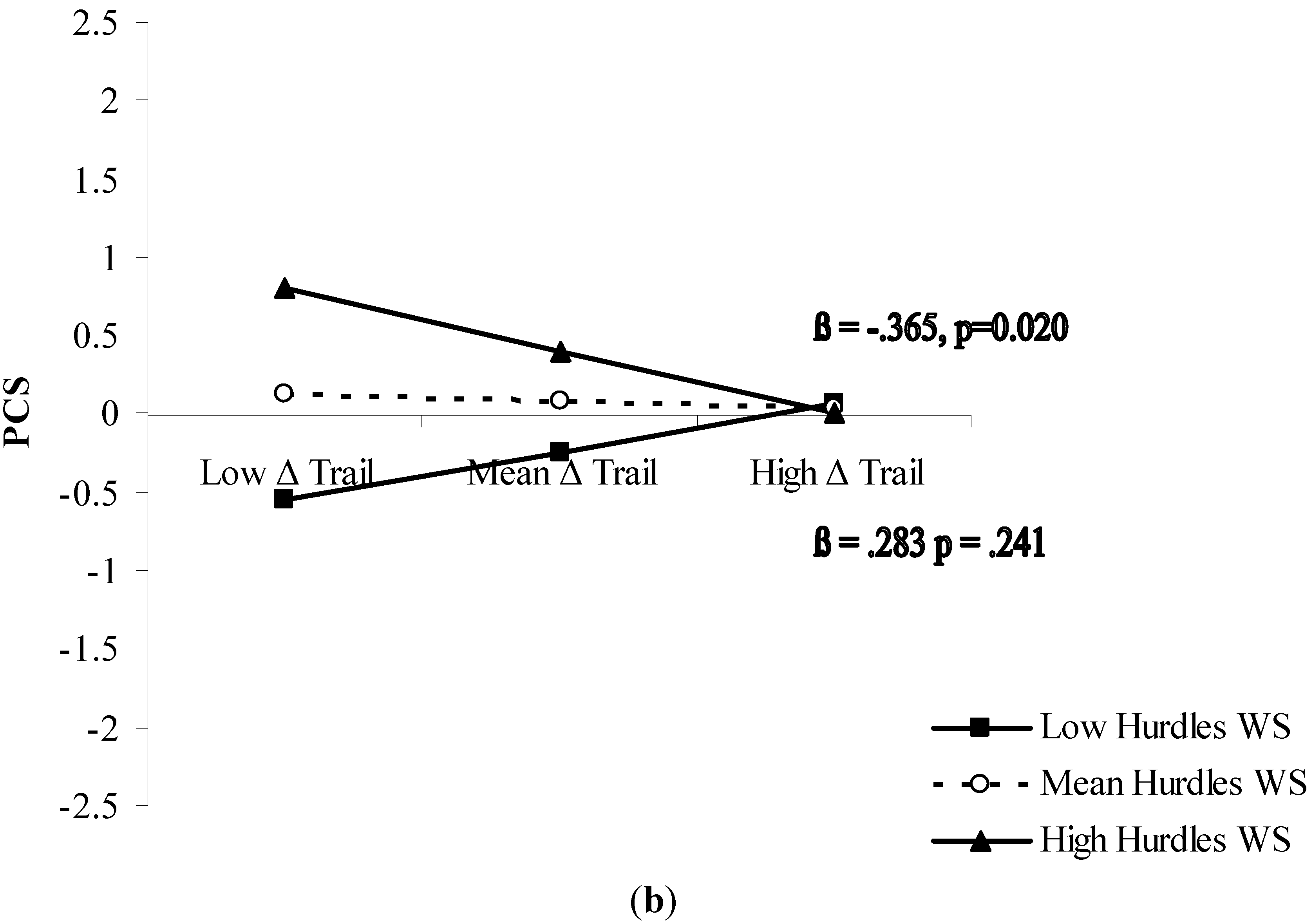
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Spirduso, W.W.; Francis, K.L.; MacRae, P.G. Health-related quality of life. In Physical Dimensions of Aging, 2nd ed.; Spirduso, W.W., Francis, K.L., MacRae, P.G., Eds.; Human Kinetics: Champaign, IL, USA, 2005; pp. 233–257. [Google Scholar]
- Werngren-Elgstrom, M.; Carlsson, C.; Iwarsson, S.A. 10-year follow-up study on subjective well-being and relationships to person-environment (P-E) fit and activity of daily living (ADL) dependence of older Swedish adults. Arch. Gerontol. Geriatr. 2009, 49, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, Z.; Bowling, A. Quality of life from the perspectives of older people. Ageing Soc. 2004, 24, 675–691. [Google Scholar] [CrossRef]
- Borg, C.; Fagerström, C.; Balducci, C.; Burholt, V.; Ferring, D.; Weber, G.; Wenger, C.; Holst, G.; Hallberg, I.R. Life satisfaction in 6 European countries: The relationship to health, self-esteem, and social and financial resources among people (aged 65–89) with reduced functional capacity. Geriatr. Nurs. 2008, 29, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Fagerstrom, C.; Borglin, G. Mobility, functional ability and health-related quality of life among people of 60 years and over. Aging Clin. Exp. Res. 2010, 22, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.D.; Mansour, E.; Valentijn, S.; Jolles, J.; van Boxtel, M. The SF-36 as a precursory measure of adaptive functioning in normal aging: The Maastricht aging study. Aging Clin. Exp. Res. 2010, 22, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.C.; Marra, C.A.; Najafzadeh, M.; Liu-Ambrose, T. The independent contribution of executive functions to health related quality of life in older women. BMC Geriatr. 2010, 1, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Royall, D.R.; Lauterbach, E.C.; Cummings, J.L.; Reeve, A.; Rummans, T.A.; Kaufer, D.I.; la France, W.C., Jr.; Coffey, C.E. Executive control function: A review of its promise and challenges for clinical research. A report from the Committee on Research of the American Neuropsychiatric Association. J. Neuropsychiatry Clin. Neurosci. 2002, 14, 377–405. [Google Scholar] [CrossRef] [PubMed]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A. The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: A latent variable analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef] [PubMed]
- Hickey, A.; Barker, M.; McGee, H.; O’Boyle, C. Measuring health-related quality of life in older patient populations. A review of current approaches. Pharmacoeconomics 2005, 23, 971–993. [Google Scholar] [CrossRef] [PubMed]
- Layte, R.; Sexton, E.; Savva, G. Quality of life in older age: Evidence from an Irish cohort study. J. Am. Geriatr. Soc. 2013, 61, S299–S305. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.H.; Faulkner, G. Inaugural editorial. Mental Health Phys. Act. 2008, 1, 1–8. [Google Scholar] [CrossRef]
- Yogev-Seligmann, G.; Hausdorff, J.M.; Giladi, N. The role of executive function and attention in gait. Mov. Disords 2008, 23, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Forte, R.; Pesce, C.; Leite, J.C.; de Vito, G.; Gibney, E.R.; Tomporowski, P.D.; Boreham, C.A. Executive function moderates the role of muscular fitness in determining functional mobility in older adults. Aging Clin. Exp. Res. 2013, 25, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Van Iersel, M.B.; Kessels, R.P.; Bloem, B.R.; Verbeek, A.L.; Rikkert, M.G.O. Executive functions are associated with gait and balance in community-living elderly people. J. Gerontol. 2008, 63, 1344–1349. [Google Scholar] [CrossRef]
- Forte, R.; Boreham, C.A.; Leite, J.C.; de Vito, G.; Brennan, L.; Gibney, E.R.; Pesce, C. Enhancing cognitive functioning in the elderly: Multicomponent vs. resistance training. Clin. Interv. Aging 2013, 8, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, L.; Giovanello, K. Executive function in daily life: Age-related influences of executive processes on instrumental activities of daily living. Psychol. Aging 2010, 25, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Von Hippel, W. Aging, executive functioning, and social control. Curr. Dir. Psychol. Sci. 2007, 16, 240–244. [Google Scholar] [CrossRef]
- Langlois, F.; Vu, T.T.; Kergoat, M.J.; Chassé, K.; Dupuis, G.; Bherer, L.; Enkvist, Å. The multiple dimensions of frailty: Physical capacity, cognition, and quality of life. Int. Psychogeriatr. 2012, 24, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Ekström, H.; Elmståhl, S. Associations between functional ability and life satisfaction in the oldest old: Results from the longitudinal population study Good Aging in Skåne. Clin. Interv. Aging 2012, 7, 313–320. [Google Scholar]
- Bentley, J.P.; Brown, C.J.; McGwin, G.; Sawyer, P.; Allman, R.M.; Roth, D.L. Functional status, life-space mobility, and quality of life: A longitudinal mediation analysis. Qual. Life Res. 2013, 22, 1621–1632. [Google Scholar] [CrossRef] [PubMed]
- Forhan, M.; Gill, S.V. Obesity, functional mobility and quality of life. Best Pract. Res. Clin. Endocr. Metab. 2013, 27, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Telenius, E.W.; Engedal, K.A.; Bergland, A. Physical performance and quality of life of nursing-home residents with mild and moderate dementia. Int. J. Environ. Res. Public Health 2013, 10, 6672–6686. [Google Scholar] [CrossRef] [PubMed]
- Vest, M.T.; Murphy, T.E.; Araujo, K.L.B.; Pisani, M.A. Disability in activities of daily living, depression, and quality of life among older medical ICU survivors: A prospective cohort study. Health Qual. Life Outcomes 2011, 9. [Google Scholar] [CrossRef] [PubMed]
- Király, E.; Gondos, T. The effect of functional movement ability on the quality of life after total hip replacement. J. Clin. Nurs. 2014, 23, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Eggenberger, P.; Schumacher, V.; Angst, M.; Theill, N.; de Bruin, E.D. Does multicomponent physical exercise with simultaneous cognitive training boost cognitive performance in older adults? A 6-month randomized controlled trial with a 1-year follow-up. Clin. Interv. Aging 2015, 10, 1335–1349. [Google Scholar] [PubMed]
- Wollesen, B.; Voeleker-Rehage, C. Training effects on motor-cognitive dual-task performance in older adults. Eur. Rev. Aging Phys. Act. 2014, 11, 5–24. [Google Scholar] [CrossRef]
- Holtzer, R.; Verghese, J.; Xue, X.; Lipton, R.B. Cognitive processes related to gait velocity: Results from the Einstein aging study. Neuropsychology 2006, 20, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Greig, C.A.; Young, A.; Skelton, D.A.; Pippet, E.; Butler, F.M.; Mahmud, S.M. Exercise studies with elderly volunteers. Age Ageing 1994, 23, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, R.; Koehler, K.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Lindeman, R.D. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Towse, J.; Neil, D. Analyzing human random generation behavior: A review of methods used and a computer program for describing performance. Behav. Res. Methods Instrum. Comput. 1998, 30, 583–591. [Google Scholar] [CrossRef]
- Strauss, E.; Sherman, E.M.; Spreen, O. Trail making test. In A Compendium of Neuropsychological Tests. Administration, Norms, and Commentary, 3rd ed.; Strauss, E., Sherman, E.M.S., Spreen, O., Eds.; Oxford University Press: Oxford, UK, 2006; pp. 655–677. [Google Scholar]
- Ware, J.E.; Kosinski, M.; Gandek, B. SF-36® Health Survey: Manual & Interpretation Guide; Quality Metric Incorporated: Lincoln, RI, USA, 2005. [Google Scholar]
- Towse, J.; Cheshire, A. Random number generation and working memory. Eur. J. Cogn. Psychol. 2007, 19, 374–394. [Google Scholar] [CrossRef]
- Audiffren, M.; Tomporowski, P.D.; Zagrodnik, J. Acute aerobic exercise and information processing: Modulation of executive control in a Random Number Generation task. Acta Psychol. 2009, 132, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Shumway-Cook, A.; Guralnik, J.M.; Phillips, C.L.; Coppin, A.K.; Ciol, M.A.; Bandinelli, S.; Ferrucci, L. Age-associated declines in complex walking task performance: The Walking In CHIANTI toolkit. J. Am. Geriatr. Soc. 2007, 55, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Shumway-Cook, A.; Patla, A.E.; Stewart, A.; Ferrucci, L.; Ciol, M.A.; Guralnik, J.M. Environmental demands associated with community mobility in older adults with and without mobility disabilities. Phys. Ther. 2002, 82, 670–681. [Google Scholar] [PubMed]
- Bean, J.F.; Kiely, D.K.; Larose, S.; Goldstein, R.; Frontera, W.R.; Leveille, S.G. Are changes in leg power responsible for clinically meaningful improvement in mobility in older adults? J. Am. Geriatr. Soc. 2010, 58, 2363–2368. [Google Scholar] [CrossRef] [PubMed]
- Aiken, L.S.; West, S.G. Multiple Regression: Testing and Interpreting Interactions; Sage: Newbury Park, CA, USA, 1996. [Google Scholar]
- Matta Mello Portugal, E.; Cevada, T.; SobralMonteiro-Junior, R.; Teixeira Guimarães, T.; da Cruz Rubini, E.; Lattari, E.; Camaz Deslandes, A. Neuroscience of exercise: From neurobiology mechanisms to mental health. Neuropsychobiology 2013, 68, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.C.; Bryan, S.; McLeod, R.; Rogers, J.; Khan, K.; Liu-Ambrose, T. Exploration of the association between quality of life, assessed by the EQ-5D and ICECAP-O, and falls risk, cognitive function and daily function, in older adults with mobility impairments. BMC Geriatr. 2012, 24, 12–65. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Feeny, D.H.; Patrick, D.L. Measuring health-related quality of life. Ann. Internal Med. 1993, 118, 622–629. [Google Scholar] [CrossRef]
- Paw, M.J.M.C.A.; de Jong, N.; Schouten, E.G.; van Staveren, W.A.; Kok, F.J. Physical exercise or micronutrient supplementation for the wellbeing of the frail elderly? A randomised controlled trial. Brit. J. Sports Med. 2002, 3, 126–131. [Google Scholar] [CrossRef]
- Conradsson, M.; Littbrand, H.; Boström, G.; Lindelöf, N.; Gustafson, Y.; Rosendahl, E. Is a change in functional capacity or dependency in activities of daily living associated with a change in mental health among older people living in residential care facilities? Clin. Interv. Aging 2013, 8, 1561–1568. [Google Scholar] [CrossRef] [PubMed]
- Gokalsing, E.; Robert, P.H.; Lafont, V.; Medecin, I.; Baudu, C.; Boyer, P.; Darcourt, G. Evaluation of the supervisory system in elderly subjects with and without disinhibition. Eur. Psychiatry 2000, 15, 407–415. [Google Scholar] [CrossRef]
- Murtagh, E.M.; Murphy, M.H.; Murphy, N.; Woods, C.; Lane, A. Stay Active—The physical activity, ageing and health study. Final Report. Mary Immaculate College and Centre for Ageing Research and Development in Ireland (CARDI) 2014. Available online: http://www.cardi.ie/sites/default/files/publications/Murtagh%20Stay%20Active%20final%20report.pdf (accessed on 24 August 2015).
- Townsend, N.; Wickramasinghe, K.; Williams, J.; Bhatnagar, P.; Rayner, M. Physical Activity Statistics 2015. British Heart Foundation: London 2015. Available online: https://www.bhf.org.uk/~/media/files/publications/research/bhf_physical-activity-statistics-2015feb.pdf (accessed on 24 August 2015).
- Blake, C.; Codd, M.B.; O’Meara, Y.M. The Short Form 36 (SF-36) Health Survey: Normative data for the Irish population. Ir. J. Med. Sci. 2000, 169, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Comfortable and maximum walking speed of adults aged 20–79 years: Reference values and determinants. Age Ageing 1997, 26, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, E.; Nyberg, L.; Backman, L.; Neely, A. Plasticity of executive functioning in young and older adults: Immediate training gains, transfer, and long-term maintenance. Psychol. Aging 2008, 23, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Weuve, J.; Kang, J.H.; Manson, J.E.; Breteler, M.M.; Ware, J.H.; Grodstein, F. Physical activity, including walking, and cognitive function in older women. JAMA 2004, 292, 1454–1461. [Google Scholar] [CrossRef] [PubMed]
- Paterson, D.H.; Warburton, D.E.R. Physical activity and functional limitations in older adults: A systematic review related to Canada’s Physical Activity Guidelines. Int. J. Behav. Nutr. Phys. Act. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Clinical Excellence. Promoting and Creating Built or Natural Environments that Encourage and Support Physical Activity (NICE Public Health Guidance 8); National Institute for Health and Clinical Excellence: London, UK, 2008. [Google Scholar]
- Saelens, B.E.; Handy, S.L. Built environment correlates of walking: A review. Med. Sci. Sports Exerc. 2008, 40, S550–S566. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forte, R.; Boreham, C.A.G.; De Vito, G.; Pesce, C. Health and Quality of Life Perception in Older Adults: The Joint Role of Cognitive Efficiency and Functional Mobility. Int. J. Environ. Res. Public Health 2015, 12, 11328-11344. https://doi.org/10.3390/ijerph120911328
Forte R, Boreham CAG, De Vito G, Pesce C. Health and Quality of Life Perception in Older Adults: The Joint Role of Cognitive Efficiency and Functional Mobility. International Journal of Environmental Research and Public Health. 2015; 12(9):11328-11344. https://doi.org/10.3390/ijerph120911328
Chicago/Turabian StyleForte, Roberta, Colin A.G. Boreham, Giuseppe De Vito, and Caterina Pesce. 2015. "Health and Quality of Life Perception in Older Adults: The Joint Role of Cognitive Efficiency and Functional Mobility" International Journal of Environmental Research and Public Health 12, no. 9: 11328-11344. https://doi.org/10.3390/ijerph120911328
APA StyleForte, R., Boreham, C. A. G., De Vito, G., & Pesce, C. (2015). Health and Quality of Life Perception in Older Adults: The Joint Role of Cognitive Efficiency and Functional Mobility. International Journal of Environmental Research and Public Health, 12(9), 11328-11344. https://doi.org/10.3390/ijerph120911328







