Green Infrastructure, Ecosystem Services, and Human Health
Abstract
:1. Introduction
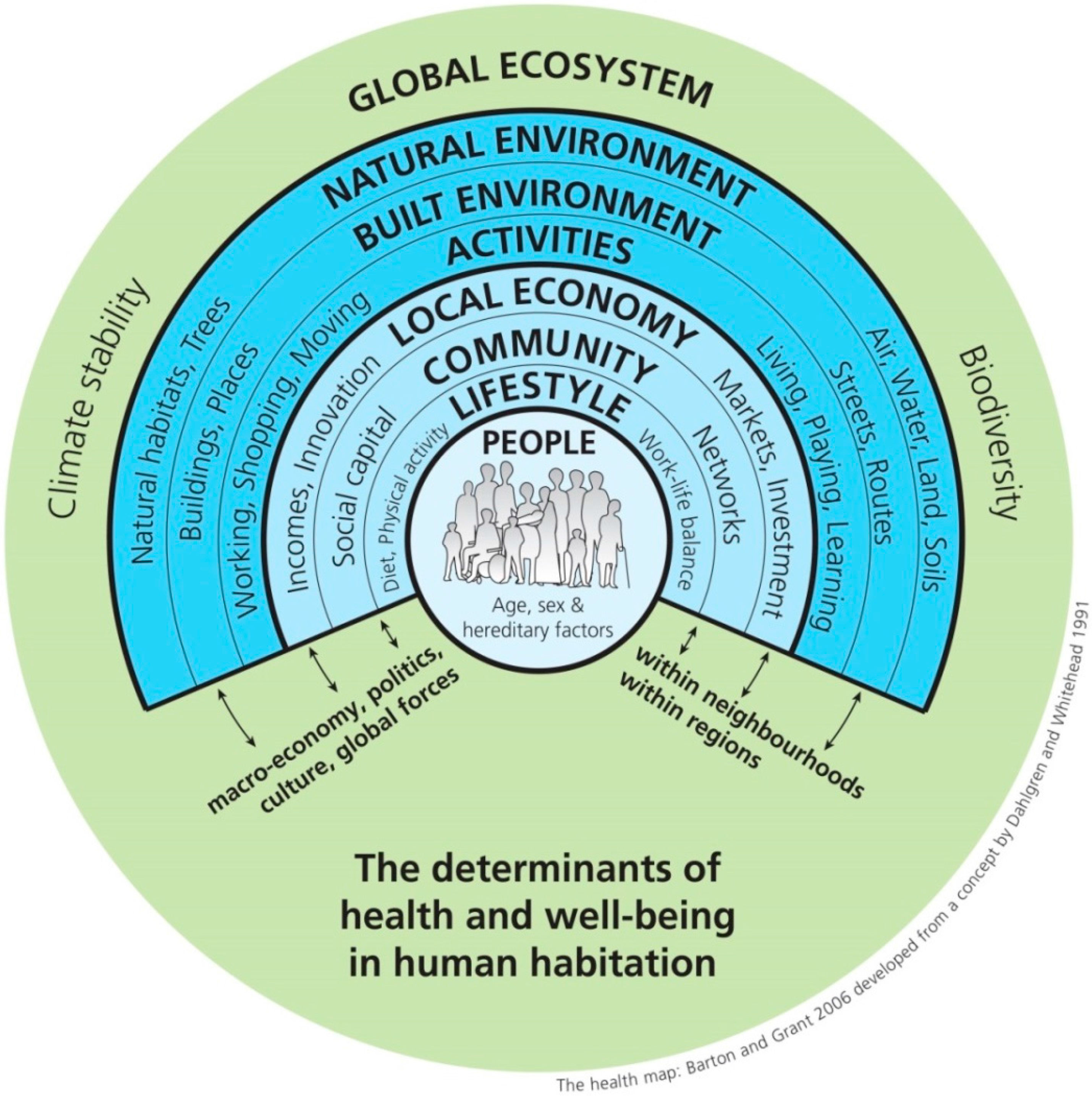
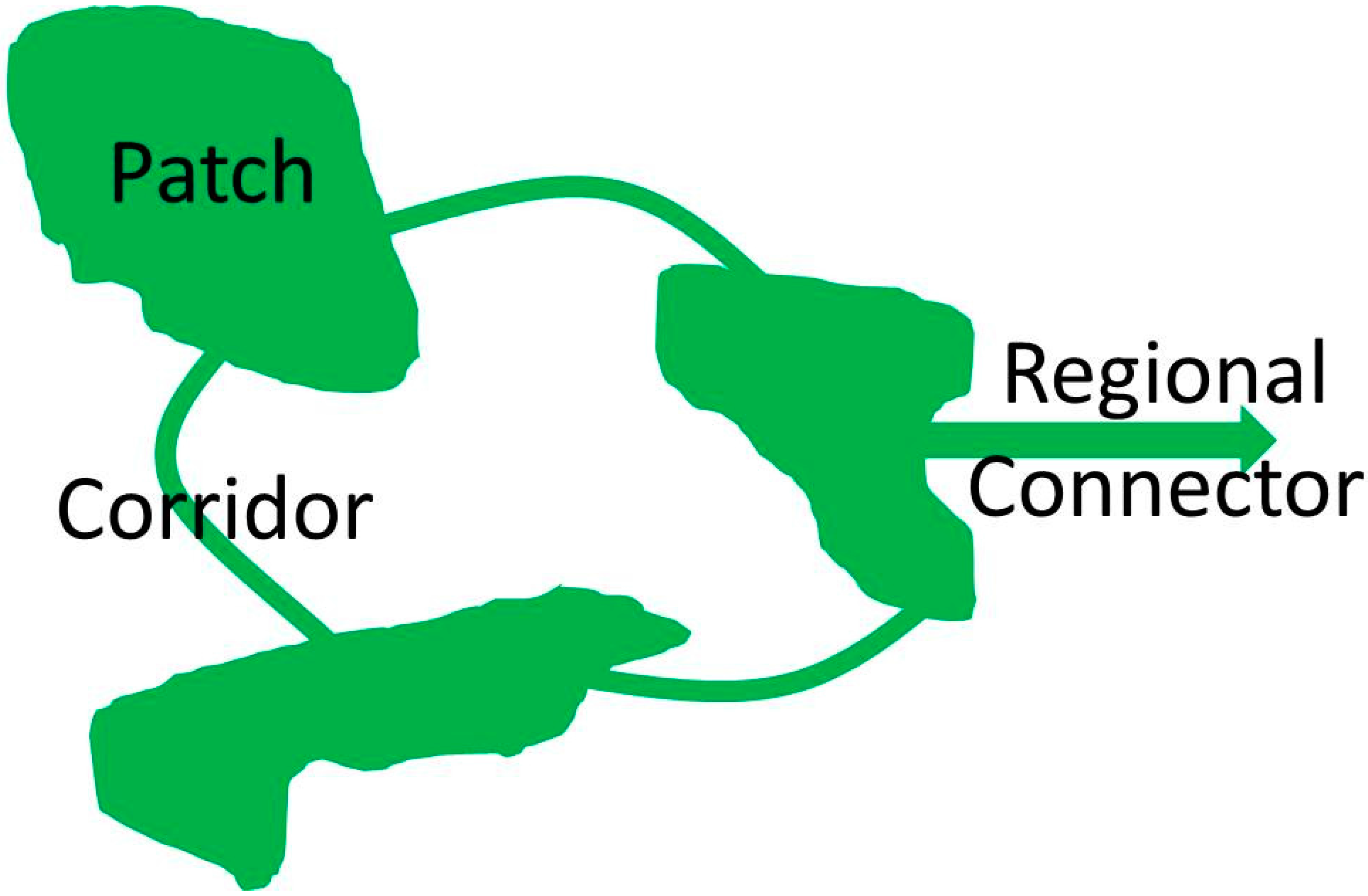
2. Green Infrastructure and Ecosystem Services
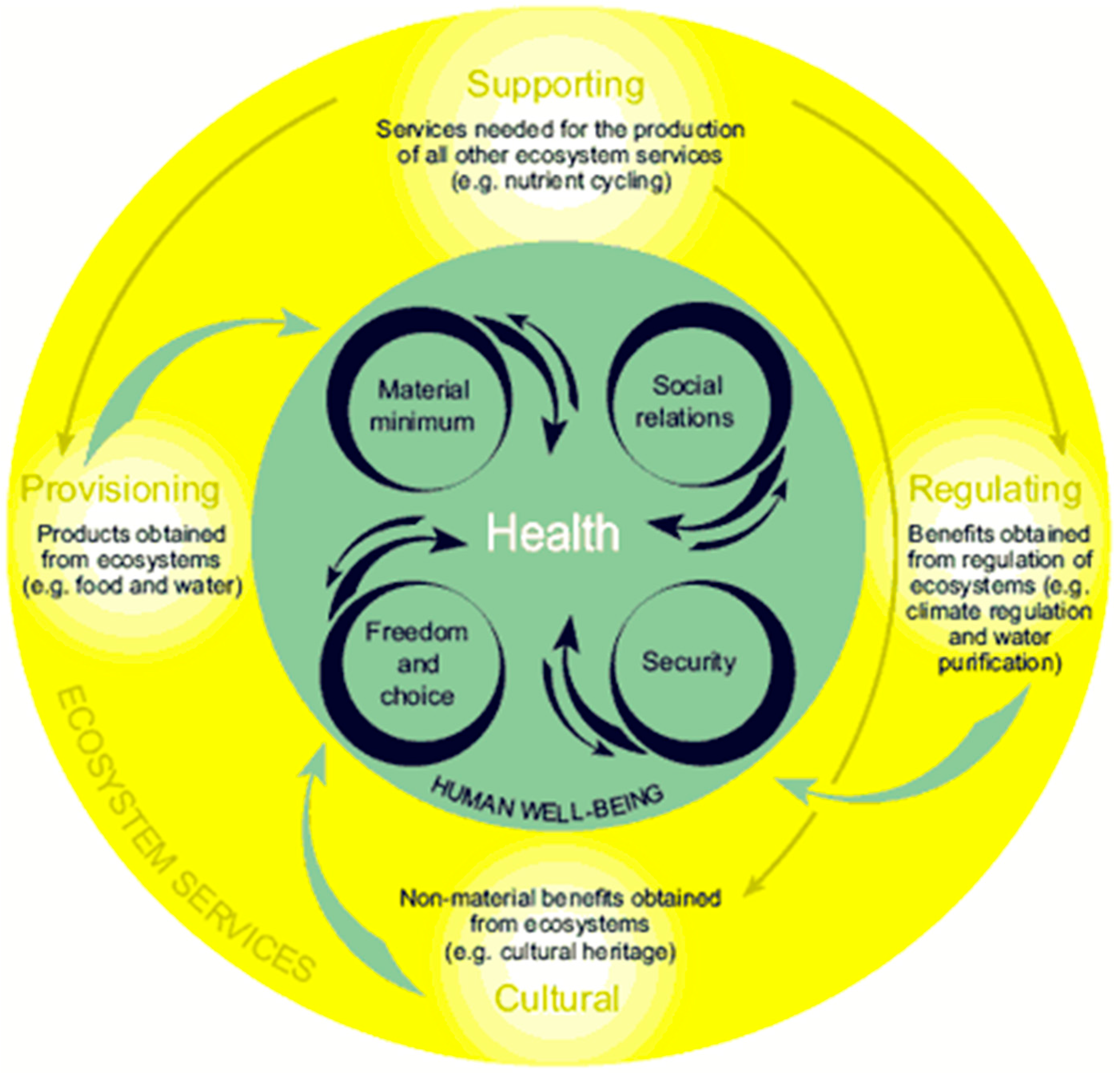
| Category of Service | Ecosystem Service |
|---|---|
| Provisioning | Water quantity and quality |
| Food quantity and quality | |
| Medicine | |
| Regulating | Air quality |
| Infectious disease modulation | |
| Climate regulation | |
| Cultural | Physical activity |
| Mental health | |
| Social capital |
3. Water
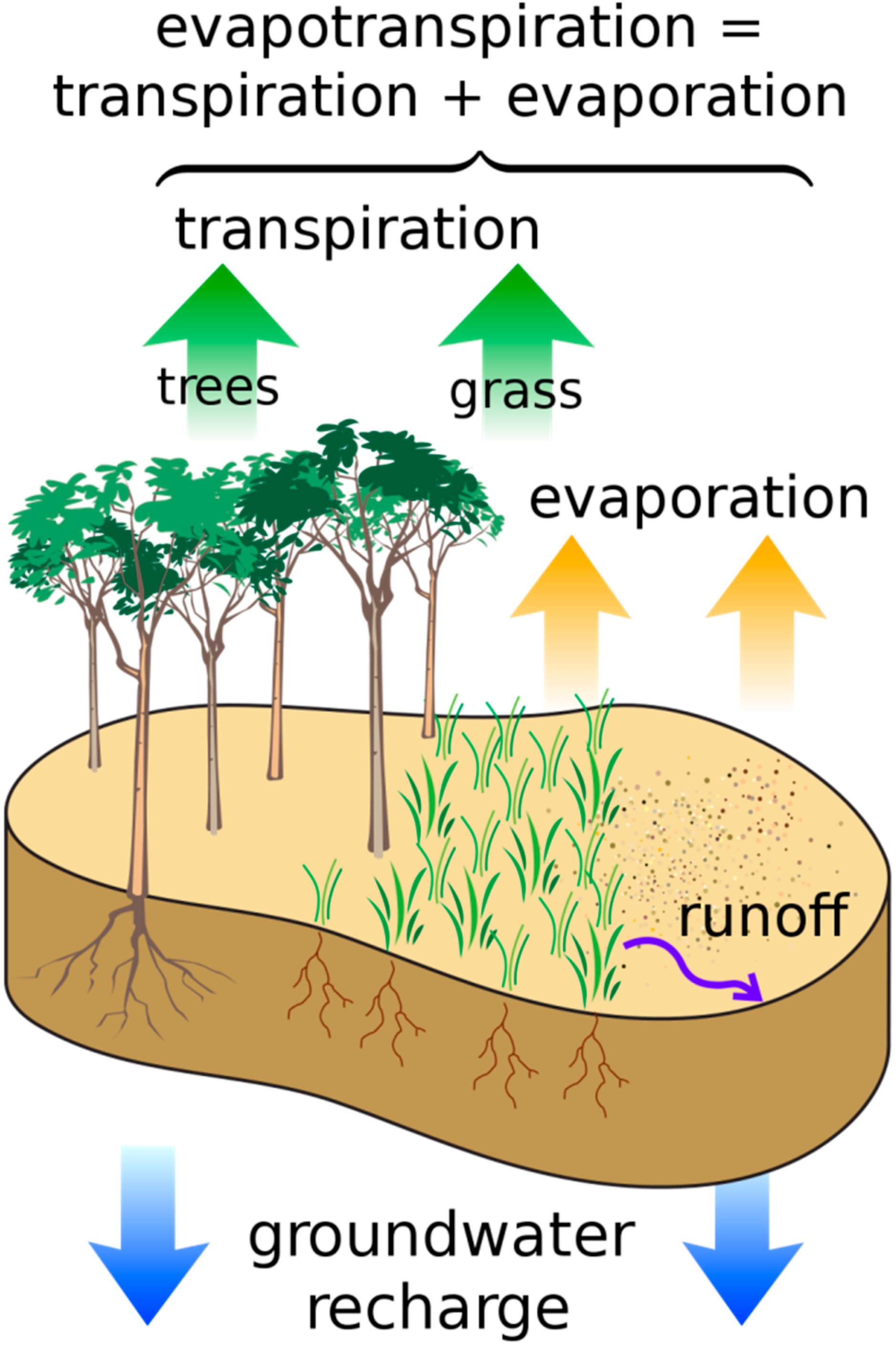
4. Food
5. Medicine
6. Air
7. Infectious Disease Modulation
7.1. Zoonotic Disease
7.2. Vector-Borne Disease
8. Climate Regulation
| Climatic Event | Intermediary | Health Outcome |
|---|---|---|
| Heat waves | direct to → | Heat stress, stroke |
| Increased ground-level ozone, pollen | Respiratory disease exacerbation | |
| Increased mean temperature | direct to → | Positive: Less hypothermia |
| More hospitable to disease vectors (e.g., mosquito, ticks) | Vector-borne diseases (e.g., Lyme, malaria, dengue) | |
| More hospitable to infectious disease agents (e.g., bacteria) | Food-poisoning, infectious disease (e.g., cholera) | |
| Ozone depletion | UV radiation | Skin and eye maladies |
| Drought | Water/food shortage | Dehydration, malnutrition |
| Lack of water safety | Water-borne disease | |
| Extreme weather event (e.g., flooding, tornado, hurricane) | direct to → | Injuries, drowning |
| Population movement | Conflicts | |
| Lack of food/water safety | Water-borne disease, malnutrition | |
| Sea-level rise | direct to → | Injuries, drowning |
| Population movement | Conflicts | |
| Water/soil salinization | Dehydration, malnutrition | |
| Climate change generally | Stress | Mental health |
8.1. Infectious Disease and Climate
8.2. Green Infrastructure and Carbon Sequestration: Mitigation and Primary Prevention
8.3. Green Infrastructure and Extreme Weather and Climatic Events: Adaptation and Secondary Prevention
9. Physical Activity
10. Mental Health
10.1. Stress
10.2. Affect
10.3. Cognition and Attention
11. Social Capital
…precisely the context where social ecosystem health is at greatest risk and where urban trees are least present. While poverty is not synonymous with alienation and risk of crime, too many poor urban neighborhoods are characterized by high levels of mistrust, isolation, graffiti, property crime, and violent crime. It may be that the greatest benefits of urban forestry accrue to some of its historically most underserved constituencies. (p. 153)
12. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Coutts, C.; Forkink, A.; Weiner, J. The portrayal of nature in the evolution of the ecological public health paradigm. Int. J. Environ. Res. Public Health 2014, 11, 1005–1019. [Google Scholar] [CrossRef] [PubMed]
- Barton, H.; Grant, M. A health map for the local human habitat. J. R. Soc. Promot. Health 2006, 126, 252–253. [Google Scholar] [CrossRef] [PubMed]
- Dahlgren, G.; Whitehead, M. Policies and Strategies to Promote Social Equity in Health; WHO Regional Office for Europe: Copenhagen, Denmark, 1991. [Google Scholar]
- Hartig, T.; Mitchell, R.; de Vries, S.; Frumkin, H. Nature and health. Annu. Rev. Public Health 2014, 35, 21.1–21.22. [Google Scholar] [CrossRef] [PubMed]
- Benedict, M.A.; McMahon, E.T. Green infrastructure: Smart conservation for the 21st century. Renew. Resour. J. 2002, 20, 12–17. [Google Scholar]
- Benedict, M.A.; McMahon, E.T. Green Infrastructure: Linking Landscapes and Communities; Island Press: Washington, DC, USA, 2006. [Google Scholar]
- Ecosystem. Available online: http://oxforddictionaries.com/us/definition/american_english/ecosystem (accessed on 11 August 2015).
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Health Synthesis; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- Melillo, J.; Sala, O. Ecosystem services. In Sustaining Life: How Human Health Depends on Biodiversity; Chivian, E., Bernstein, A., Eds.; Oxford University Press: New York, NY, USA, 2008; pp. 75–115. [Google Scholar]
- The Economics of Ecosystems and Biodiversity Ecosystem Services. Available online: http://www.teebweb.org/resources/ecosystem-services/ (accessed on 22 May 2015).
- De Groot, R.S.; Alkemade, R.; Braat, L.; Hein, L.; Willemen, L. Challenges in integrating the concept of ecosystem services and values in landscape planning, management and decision making. Ecol. Complex. 2010, 7, 260–272. [Google Scholar] [CrossRef]
- De Groot, R.S.; Fisher, B.; Christie, M. Integrating the ecological and economic dimensions in biodiversity and ecosystem service valuation. In The Economics of Ecosystems and Biodiversity Ecological and Economic Foundations; Kumar, P., Ed.; Earthscan: London, UK, 2010; pp. 1–40. [Google Scholar]
- Northridge, M.E.; Sclar, E.D.; Biswas, P. Sorting out the connections between the built environment and health: A conceptual framework for navigating pathways and planning healthy cities. J. Urban Heal. 2003, 80, 556–568. [Google Scholar] [CrossRef] [PubMed]
- Schulz, A.; Northridge, M.E. Social determinants of health: Implications for environmental health promotion. Heal. Educ. Behav. 2004, 31, 455–471. [Google Scholar] [CrossRef] [PubMed]
- Tyrväinen, L.; Pauleit, S.; Seeland, K.; de Vries, S. Benefits and uses of urban forests and trees. In Urban Forests and Trees: A Reference Book; Konijnendijk, C., Nilsson, K., Randrup, T., Schipperijn, J., Eds.; Springer: Berlin, Germany, 2005; pp. 81–114. [Google Scholar]
- US Geological Service. The Water Cylcle: Evapotranspiration. Available online: http://ga.water.usgs.gov/edu/watercycleevapotranspiration.html (accessed on 11 April 2013).
- Kling, G. The Flow of Energy: Primary Production to Higher Trophic Levels. Available online: http://www.globalchange.umich.edu/globalchange1/current/lectures/kling/energyflow/energyflow.html (accessed on 26 August 2013).
- Vitousek, P.; Ehrlich, P.; Ehrlich, A.; Matson, P. Human appropriation of the products of photosynthesis. Bioscience 1986, 36, 368–373. [Google Scholar] [CrossRef]
- Krausmann, F.; Erb, K.-H.; Gingrich, S.; Haberl, H.; Bondeau, A.; Gaube, V.; Lauk, C.; Plutzar, C.; Searchinger, T.D. Global human appropriation of net primary production doubled in the 20th century. Proc. Natl. Acad. Sci. USA 2013, 110, 10324–10329. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Hillel, D.; Rosenzweig, C. Biodiversity and food production. In Sustaining Life: How Human Health Depends on Biodiversity; Chivian, E., Bernstein, A., Eds.; Oxford University Press: New York, NY, USA, 2008; pp. 325–381. [Google Scholar]
- Morse, R.A.; Calderone, N.W. The value of honey bees as pollinators of US crops in 2000. Bee Culture 2000, 128, 1–15. [Google Scholar]
- Pimentel, D.; Wilson, C.; McCullum, C.; Huang, R.; Dwen, P.; Flack, J.; Tran, Q.; Saltman, T.; Cliff, B. Economic and Environmental Benefits of Biodiversity. Bioscience 1997, 47, 747–757. [Google Scholar] [CrossRef]
- Kearns, C.; Inouye, D.; Waser, N. Endangered mutualisms: The conservation of plant-pollinator interactions. Annu. Rev. Ecol. Syst. 1998, 29, 83–112. [Google Scholar] [CrossRef]
- Karjalainen, E.; Sarjala, T.; Raitio, H. Promoting human health through forests: Overview and major challenges. Environ. Health Prev. Med. 2010, 15, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Grifo, F.; Newman, D.; Fairfield, A.; Bhattacharya, B.; Grupenhoff, J. The origin of prescription drugs. In Biodiversity of Human Health; Grifo, F., Rosenthal, J., Eds.; Island Press: Washington, DC, USA, 1997; pp. 131–163. [Google Scholar]
- United Nations. Trade in Medicinal Plants; Food and Agriculture Organization of the United Nations: Rome, Italy, 2004. [Google Scholar]
- Kris-Etherton, P.; Hecker, K. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113, 71S–88S. [Google Scholar] [CrossRef]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar] [PubMed]
- Patisaul, H.; Jefferson, W. The pros and cons of phytoestrogens. Front. Neuroendocrinol. 2010, 31, 400–419. [Google Scholar] [CrossRef] [PubMed]
- Bealey, W.J.; McDonald, A.G.; Nemitz, E.; Donovan, R.; Dragosits, U.; Duffy, T.R.; Fowler, D. Estimating the reduction of urban PM10 concentrations by trees within an environmental information system for planners. J. Environ. Manag. 2007, 85, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Beckett, K.P.; Freer-Smith, P.H.; Taylor, G. Urban woodlands: Their role in reducing the effects of particulate pollution. Environ. Pollut. 1998, 99, 347–360. [Google Scholar] [CrossRef]
- Hedin, L. Deposition of nutrients and pollutants to ecosystems. In Methods in Ecosystem Science; Sala, O.E., Jackson, R.B., Mooney, H.A., Howarth, R.W., Eds.; Springer-Verlag: New York, NY, 2000; pp. 265–276. [Google Scholar]
- Wolf, K. Trees, Parking and Green Law: Strategies for Sustainability; Georgia Forestry Commission, Urban and Community Forestry: Stone Mountain, GA, USA, 2004. [Google Scholar]
- Nowak, D.J.; Crane, D.E.; Stevens, J.C. Air pollution removal by urban trees and shrubs in the United States. Urban For. Urban Green. 2006, 4, 115–123. [Google Scholar] [CrossRef]
- Nowak, D.J.; Hirabayashi, S.; Bodine, A.; Greenfield, E. Tree and forest effects on air quality and human health in the United States. Environ. Pollut. 2014, 193, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; McBride, J.; Zhou, J.; Sun, Z. The urban forest in Beijing and its role in air pollution reduction. Urban For. Urban Green. 2005, 3, 65–78. [Google Scholar] [CrossRef]
- Jim, C.Y.; Chen, W.Y. Assessing the ecosystem service of air pollutant removal by urban trees in Guangzhou (China). J. Environ. Manag. 2008, 88, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Lovasi, G.S.; Quinn, J.W.; Neckerman, K.M.; Perzanowski, M.S.; Rundle, A. Children living in areas with more street trees have lower prevalence of asthma. J. Epidemiol. Community Health 2008, 62, 647–649. [Google Scholar] [CrossRef] [PubMed]
- Lovasi, G.S.; O’Neil-Dunne, J.P.M.; Lu, J.W.T.; Sheehan, D.; Perzanowski, M.S.; Macfaden, S.W.; King, K.L.; Matte, T.; Miller, R.L.; Hoepner, L.A.; et al. Urban tree canopy and asthma, wheeze, rhinitis, and allergic sensitization to tree pollen in a New York city birth cohort. Environ. Health Perspect. 2013, 121, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Donovan, G.H.; Butry, D.T.; Michael, Y.L.; Prestemon, J.P.; Liebhold, A.M.; Gatziolis, D.; Mao, M.Y. The relationship between trees and human health: Evidence from the spread of the emerald ash borer. Am. J. Prev. Med. 2013, 44, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Tomalak, M.; Rossi, E.; Ferrini, F.; Moro, P. Negative aspects and hazardous effects of forest environment on human health. In Forests, Trees and Human Health; Nilsson, K., Sangster, M., Gallis, C., Hartig, T., de Vries, S., Seeland, K., Schipperijn, J., Eds.; Springer Verlag: New York, NY, USA, 2011; pp. 77–124. [Google Scholar]
- Pawankar, R.; Canonica, G.W.; Holgate, S.T.; Lockey, R.F. WAO White Book on Allergy 2011–2012: Executive Summary; World Allergy Organization: Tokyo, Japan, 2011. [Google Scholar]
- Lucadamo, K. City’s plan to plant million trees is pitting allergy sufferers versus aesthetics. New York Daily News, 24 April 2011. [Google Scholar]
- Ostfeld, R.S.; Keesing, F.; Eviner, V. Infectious Disease Ecology: Effects of Ecosystems on Disease and of Disease on Ecosystems; Princeton University Press: Princeton, NJ, USA, 2008. [Google Scholar]
- Defries, R.S.; Foley, J.; Asner, G. Land-use choices: Balancing human needs and ecosystem function. Front. Ecol. Environ. 2004, 2, 249–257. [Google Scholar] [CrossRef]
- Gottdenker, N.L.; Streicker, D.G.; Faust, C.L.; Carroll, C.R. Anthropogenic land use change and infectious diseases: A review of the evidence. Ecohealth 2014, 11, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Karesh, W.B.; Dobson, A.; Lloyd-Smith, J.O.; Lubroth, J.; Dixon, M.A; Bennett, M.; Aldrich, S.; Harrington, T.; Formenty, P.; Loh, E.H.; et al. Ecology of zoonoses: Natural and unnatural histories. Lancet 2012, 380, 1936–1945. [Google Scholar] [CrossRef]
- Nasi, R.; Taber, A.; van Vliet, N. Empty forests, empty stomachs? Bushmeat and livelihoods in the Congo and Amazon Basins. Int. For. Rev. 2011, 13, 355–368. [Google Scholar] [CrossRef]
- LeBreton, M.; Pike, B.; Saylors, K.; le Doux Diffo, J.; Fair, J.N.; Rimoin, A.W.; Ortiz, N.; Djoko, C.; Tamoufe, U.; Wolfe, N.D. Bushmeat and infectious disease emergence. In New Directions in Conservation Medicine: Applied Cases in Ecological Health; Aguirre, A.A., Ostfeld, R.S., Daszak, P., Eds.; Oxford University Press: New York, NY, USA, 2012; pp. 164–178. [Google Scholar]
- Goldberg, T.; Paige, S.; Chapman, C. The Kibale EcoHealth Project: Exploring connections among human health, animal health, and landscape dynamics in Western Uganda. In New Directions in Conservation Medicine: Applied Cases in Ecological Health; Aguirre, A.A., Ostfeld, R.S., Daszak, P., Eds.; Oxford University Press: New York, NY, USA, 2012; pp. 452–465. [Google Scholar]
- Goldberg, T.L.; Gillespie, T.; Rwego, I.; Estoff, E.; Chapman, C. Forest fragmentation as cause of bacterial transmission among nonhuman primates, humans, and livestock, Uganda. Emerg. Infect. Dis. 2008, 14, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Patz, J.A.; Olson, S.H.; Uejio, C.K.; Gibbs, H.K. Disease emergence from global climate and land use change. Med. Clin. North Am. 2008, 92, 1473–1491. [Google Scholar] [CrossRef] [PubMed]
- Reiter, P. Climate change and mosquito-borne disease. Environ. Health Perspect. 2001, 109, 141–161. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.K.; Ruesch, A.S.; Achard, F.; Clayton, M.K.; Holmgren, P.; Ramankutty, N.; Foley, J. A Tropical forests were the primary sources of new agricultural land in the 1980s and 1990s. PNAS 2010, 107, 16732–16737. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.; Molyneux, D.; BIrley, M. Deforestation: Effects on vector-borne disease. Parasitology 1993, 30, 55–75. [Google Scholar] [CrossRef]
- Afrane, Y.; Lawson, B.W.; Githeko, A.K.; Yan, G. Effects of microclimatic changes caused by land use and land cover on duration of gonotrophic cycles of Anopheles gambiae (Diptera: Culicidae) in Western Kenya Highlands. J. Med. Entomol. 2005, 42, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Confalonieri, U.; Menne, B.; Akhtar, R.; Ebi, K.; Hauengue, M.; Kovats, R.; Revich, B.; Woodward, A. Human health. In Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Parry, M., Canziani, O., Palutikof, J., van der Linden, P., Hanson, C., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 391–431. [Google Scholar]
- Amati, M.; Taylor, L. From green belts to green infrastructure. Plan. Pract. Res. 2010, 25, 143–155. [Google Scholar] [CrossRef]
- Patz, J.A.; Campbell-Lendrum, D.; Holloway, T.; Foley, J.A. Impact of regional climate change on human health. Nature 2005, 438, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Ebi, K.L. The ABCD of adaptation. In Climate Briefing Series; American Meterological Society: Washington, DC, USA, 2010. [Google Scholar]
- English, P.B.; Sinclair, A.H.; Ross, Z.; Anderson, H.; Boothe, V.; Davis, C.; Ebi, K.; Kagey, B.; Malecki, K.; Shultz, R.; et al. Environmental health indicators of climate change for the United States: Findings from the State Environmental Health Indicator Collaborative. Environ. Health Perspect. 2009, 117, 1673–1681. [Google Scholar] [CrossRef] [PubMed]
- Coutts, C.; Berke, T. The extent and context of human health considerations in London’s spatial development and climate action strategy. J. Urban Plan. Dev. 2013, 139, 322–330. [Google Scholar] [CrossRef]
- Frumkin, H.; Hess, J.; Luber, G.; Malilay, J.; McGeehin, M. Climate change: The public health response. Am. J. Public Health 2008, 98, 435–445. [Google Scholar] [CrossRef] [PubMed]
- McMichael, A.J.; Woodruff, R.; Hales, S. Climate change and human health: Present and future risks. Lancet 2006, 367, 859–869. [Google Scholar] [CrossRef]
- Costello, A.; Abbas, M.; Allen, A.; Ball, S.; Bell, S.; Bellamy, R.; Friel, S.; Groce, N.; Johnson, A.; Kett, M.; et al. Managing the health effects of climate change. Lancet 2009, 373, 1693–1733. [Google Scholar] [CrossRef]
- McMichael, A.J.; Campbell-Lendrum, D.; Corvalan, C.; Ebi, K.; Githeko, A.; Scheraga, J.; Woodward, A. Climate Change and Human Health: Risks and Responses; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- McMichael, A.J.; Campbell-Lendrum, D.; Kovats, S.; Edwards, S.; Wilkinson, P.; Wilson, T.; Nicholls, R.; Hales, S.; Tanser, F.; le Sueur, D.; et al. Global climate change. In Comparative Quantification of Health Risks: Global and Regional Burden of Disease Due to Selected Major Risk Factors; Ezzati, M., Lopez, A., Rodgers, A., Murray, C., Eds.; WHO: Geneva, Switzerland, 2004; pp. 1543–1649. [Google Scholar]
- Goklany, I.M. Death and Death Rates Due to Extreme Weather Events: Global and U.S. Trends, 1900–2006; International Policy Network: London, UK, 2007. [Google Scholar]
- Altizer, S.; Ostfeld, R.S.; Johnson, P.T. J.; Kutz, S.; Harvell, C.D. Climate change and infectious diseases: From evidence to a predictive framework. Science 2013, 341, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Mills, J.N.; Gage, K.L.; Khan, A.S. Potential influence of climate change on vector-borne and zoonotic diseases: A review and proposed research plan. Environ. Health Perspect. 2010, 118, 1507–1514. [Google Scholar] [CrossRef] [PubMed]
- Gage, K.L.; Burkot, T.R.; Eisen, R.J.; Hayes, E.B. Climate and vectorborne diseases. Am. J. Prev. Med. 2008, 35, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Rohr, J.R.; Dobson, A.P.; Johnson, P.T.J.; Kilpatrick, A.M.; Paull, S.H.; Raffel, T.R.; Ruiz-Moreno, D.; Thomas, M.B. Frontiers in climate change-disease research. Trends Ecol. Evol. 2011, 26, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Kovats, R.S.; Campbell-Lendrum, D.H.; McMichael, A.J.; Woodward, A.; Cox, J.S. Early effects of climate change: Do they include changes in vector-borne disease? Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2001, 356, 1057–1068. [Google Scholar] [CrossRef]
- Gray, J.S. Ixodes ricinus seasonal activity: Implications of global warming indicated by revisiting tick and weather data. Int. J. Med. Microbiol. 2008, 298, 19–24. [Google Scholar] [CrossRef]
- Welbergen, J.A.; Klose, S.M.; Markus, N.; Eby, P. Climate change and the effects of temperature extremes on Australian flying-foxes. Proc. R. Soc. B Biol. Sci. 2008, 275, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Yates, T.L.; Mills, J.N.; Parmenter, C.A.; Ksiazek, T.G.; Parmenter, R.R.; Vande Castle, J.R.; Calisher, C.H.; Nichol, S.T.; Abbott, K.D.; Young, J.C.; et al. The ecology and evolutionary history of an emergent disease: Hantavirus pulmonary syndrome. Bioscience 2002, 52, 989–998. [Google Scholar] [CrossRef]
- Watts, D.M.; Burke, D.S.; Harrison, B.A.; Whitmire, R.E.; Nisalak, A. Effect of temperature on the vector efficiency of Aedes aegypti for dengue 2 virus. Am. J. Trop. Med. Hyg. 1987, 36, 143–152. [Google Scholar] [PubMed]
- Reisen, W.K.; Meyer, R.P.; Presser, S.B.; Hardy, J.L. Effect of temperature on the transmission of western equine encephalomyelitis and St. Louis encephalitis viruses by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2000, 30, 151–160. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y.; Martinez, V.M. Effects of temperature on the transmission of west nile virus by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2006, 43, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Noden, B.H.; Kent, M.D.; Beier, J.C. The impact of variations in temperature on early Plasmodium falciparum development in Anopheles stephensi. Parasitology 1995, 111, 539–545. [Google Scholar] [CrossRef] [PubMed]
- US EPA Inventory of U.S. greenhouse gas emissions and sinks: 1990–2011. J. ICRU 2013, 9, ES1–ES26.
- Hulme, M.; Jenkins, G.; Lu, X.; Turnpenny, J.; Mitchell, T.; Jones, R.; Lowe, J.; Murphy, J.; Hassell, D.; Boorman, P.; McDonald, R.; Hill, S. Climate Change Scenarios for the United Kingdom: The UKCIP02 Scientific Report; University of East Anglia: Norwich, UK, 2002. [Google Scholar]
- Nowak, D.J.; Crane, D.E. Carbon storage and sequestration by urban trees in the USA. Environ. Pollut. 2002, 116, 381–389. [Google Scholar] [CrossRef]
- Woodbury, P.B.; Smith, J.E.; Heath, L.S. Carbon sequestration in the U.S. forest sector from 1990 to 2010. For. Ecol. Manag. 2007, 241, 14–27. [Google Scholar] [CrossRef]
- Bellassen, V.; Luyssaert, S. Carbon sequestration: Managing forests in uncertain times. Nature 2014, 506, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, A.; Clair, J.S.; Silverstein, K. The politics of “natural” disaster: Who made Mitch so bad? Int. J. Heal. Serv. 1999, 29, 459–462. [Google Scholar] [CrossRef]
- Barbier, E.B.; Koch, E.W.; Silliman, B.R.; Hacker, S.D.; Wolanski, E.; Primavera, J.; Granek, E.F.; Polasky, S.; Aswani, S.; Cramer, L.A.; et al. Coastal ecosystem-based management with nonlinear ecological functions and values. Science 2008, 319, 321–323. [Google Scholar] [CrossRef] [PubMed]
- World Meteorological Organization. WMO Statement on the Status of the Global Climate in 2013; World Meteorological Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Oke, T.R. The energetic basis of the urban heat island. Q. J. R. Meteorol. Soc. 1982, 108, 1–24. [Google Scholar] [CrossRef]
- Gill, S.; Handley, J.; Ennos, A.; Pauleit, S. Adapting cities for climate change: The role of the green infrastructure. Built Environ. 2007, 33, 115–133. [Google Scholar] [CrossRef]
- Souch, C.; Grimmond, S. Applied climatology: Urban climate. Prog. Phys. Geogr. 2006, 30, 270–279. [Google Scholar] [CrossRef]
- Arnfield, A.J. Two decades of urban climate research: A review of turbulence, exchanges of energy and water, and the urban heat island. Int. J. Climatol. 2003, 23, 1–26. [Google Scholar] [CrossRef]
- US EPA Heat Island Effect. Available online: http://www.epa.gov/hiri/ (accessed on 11 April 2013).
- Wilby, R. Past and projected trends in London’s urban heat island. Weather 2003, 58, 251–260. [Google Scholar] [CrossRef]
- Allen, A.; Segal-Gidan, F. Heat-related illness in the elderly. Clin. Geriatr. 2007, 15, 37–45. [Google Scholar]
- US EPA. Cooling Summertime Temperatures: Strategies to Reduce Urban Heat Islands; US EPA: Washington, DC, USA, 2003. [Google Scholar]
- US EPA Heat Island Effect: Heat Island Impacts. Available online: http://www.epa.gov/heatisland/impacts/index.htm (accessed on 11 April 2013).
- Akbari, H.; Pomerantz, M.; Taha, H. Cool surfaces and shade trees to reduce energy use and improve air quality in urban areas. Sol. Energy 2001, 70, 295–310. [Google Scholar] [CrossRef]
- Akbari, H. Energy Saving Potentials and Air Quality Benefits of Urban Heat Island Mitigation (Working Paper); Lawrence Berkeley National Laboratory: Berkeley, CA, USA, 2005. [Google Scholar]
- Akbari, H.; Kurn, D.; Bretz, S.; Hanford, J. Peak power and cooling energy savings of shade trees. Energy Build. 1997, 25, 139–148. [Google Scholar] [CrossRef]
- Smith, C.; Levermore, G. Designing urban spaces and buildings to improve sustainability and quality of life in a warmer world. Energy Policy 2008, 36, 4558–4562. [Google Scholar] [CrossRef]
- Spronken-Smith, R.; Oke, T. Scale modelling of nocturnal cooling in urban parks. Boundary Layer Meteorol. 1999, 93, 287–312. [Google Scholar] [CrossRef]
- Pauleit, S.; Duhme, F. Assessing the environmental performance of land cover types for urban planning. Landsc. Urban Plan. 2000, 52, 1–20. [Google Scholar] [CrossRef]
- Pauleit, S.; Duhme, F. GIS assessment of Munich’s urban forest structure for urban planning. J. Arboric. 2000, 26, 133–141. [Google Scholar]
- Heisler, G.; Grant, R.; Grimmond, S.; Souch, C. Urban forests—Cooling our communities? In Proceedings: 7th National Urban Forest Conference; Kollin, C., Barratt, M., Eds.; American Forests: Washington, DC, USA, 1995; pp. 31–34. [Google Scholar]
- Ferdinand, A.O.; Sen, B.; Rahurkar, S.; Engler, S.; Menachemi, N. The relationship between built environments and physical activity: A systematic review. Am. J. Public Health 2012, 102, e7–e13. [Google Scholar] [CrossRef] [PubMed]
- Kaczynski, A.T.; Henderson, K.A. Environmental correlates of physical activity: A review of evidence about parks and recreation. Leis. Sci. 2007, 29, 315–354. [Google Scholar] [CrossRef]
- Croucher, K.; Myers, L.; Bretherton, J. The Links between Greenspace and Health: A Critical Literature Review; Greenspace Scotland: Stirling, UK, 2007. [Google Scholar]
- Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of childhood and adult obesity in the United States, 2011–2012. J. Am. Med. Assoc. 2014, 311, 806–814. [Google Scholar] [CrossRef] [PubMed]
- WHO Obesity and Overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 20 January 2015).
- Slentz, C.A.; Aiken, L.; Houmard, J.A.; Bales, C.W.; Johnson, J.L.; Tanner, C.J.; Duscha, B.D.; Kraus, W.E. Inactivity, exercise, and visceral fat. STRRIDE: A randomized, controlled study of exercise intensity and amount. J. Appl. Physiol. 2005, 99, 1613–1618. [Google Scholar] [CrossRef] [PubMed]
- Carroll, A.E. To lose weight, eating less is far more important than exercising more. New York Times, 15 June 2015. [Google Scholar]
- Lachowycz, K.; Jones, A.P. Greenspace and obesity: A systematic review of the evidence. Obes. Rev. 2011, 12, e183–e189. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.S.; Hansen, K.B. Do green areas affect health? Results from a Danish survey on the use of green areas and health indicators. Health Place 2007, 13, 839–850. [Google Scholar] [CrossRef] [PubMed]
- Ellaway, A.; Macintyre, S.; Bonnefoy, X. Graffiti, greenery, and obesity in adults: Secondary analysis of European cross sectional survey. BMJ 2005, 331, 611–612. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.F.; Wilson, J.S.; Liu, G.C. Neighborhood greenness and 2-year changes in body mass index of children and youth. Am. J. Prev. Med. 2008, 35, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, A.; Oakes, J.M.; Schmitz, K.H.; Hearst, M. Does residential density increase walking and other physical activity? Urban Stud. 2007, 44, 679–697. [Google Scholar] [CrossRef]
- Telama, R.; Yang, X.; Viikari, J.; Välimäki, I.; Wanne, O.; Raitakari, O. Physical activity from childhood to adulthood: A 21-year tracking study. Am. J. Prev. Med. 2005, 28, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Bird, W. Natural Thinking: Investigating the Links between the Natural Environment, Biodiversity and Mental Health; Royal Society for the Protection of Birds: Sandy, UK, 2007. [Google Scholar]
- Hartig, T.; Staats, H. Guest editors’ introduction: Restorative environments. J. Environ. Psychol. 2003, 23, 103–107. [Google Scholar] [CrossRef]
- Hartig, T. Three steps to understanding restorative environments as health resources. In Open Space: People Space2; Ward-Thompson, C., Travlou, P., Eds.; Routledge: Abingdon, UK, 2007; pp. 163–180. [Google Scholar]
- Kaplan, S. The restorative benefits of nature: Toward an integrative framework. J. Environ. Psychol. 1995, 15, 169–182. [Google Scholar] [CrossRef]
- Hartig, T.; Evans, G.W.; Jamner, L.D.; Davis, D.S.; Gärling, T. Tracking restoration in natural and urban field settings. J. Environ. Psychol. 2003, 23, 109–123. [Google Scholar] [CrossRef]
- Alcock, I.; White, M. Longitudinal effects on mental health of moving to greener and less green urban areas. Environ. Sci. Technol. 2014, 48, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Leslie, E.; Giles-Corti, B.; Owen, N. Associations of neighbourhood greenness with physical and mental health: Do walking, social coherence and local social interaction explain the relationships? J. Epidemiol. Community Health 2008, 62, e9–e9. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R.S. View through a window may influence recovery from surgery. Science 1984, 224, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R.S.; Simons, R.F.; Losito, B.D.; Fiorito, E.; Miles, M.A.; Zelson, M. Stress recovery during exposure to natural and urban environments. J. Environ. Psychol. 1991, 11, 201–230. [Google Scholar] [CrossRef]
- Grahn, P.; Stigsdotter, U.A. Landscape planning and stress. Urban For. Urban Green. 2003, 2, 1–18. [Google Scholar] [CrossRef]
- Stigsdotter, U.K.; Ekholm, O.; Schipperijn, J.; Toftager, M.; Kamper-Jørgensen, F.; Randrup, T.B. Health promoting outdoor environments—Associations between green space, and health, health-related quality of life and stress based on a Danish national representative survey. Scand. J. Public Health 2010, 38, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.E.; Maas, J.; Verheij, R.A.; Groenewegen, P.P. Green space as a buffer between stressful life events and health. Soc. Sci. Med. 2010, 70, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Chang, C.; Sullivan, W.C. A dose of nature: Tree cover, stress reduction, and gender differences. Landsc. Urban Plan. 2014, 132, 26–36. [Google Scholar] [CrossRef]
- Duncan, S.; Barrett, L.F. Affect is a form of cognition: A neurobiological analysis. Cogn. Emot. 2007, 21, 1184–1211. [Google Scholar] [CrossRef] [PubMed]
- Kelly, V.C. A primer of affect psychology. In The Art of Intimacy and the Hidden Challenge of Shame; Tomkins Press: Raleigh, NC, USA, 2012; pp. 158–191. [Google Scholar]
- Ulrich, R.S. Visual landscapes and psychological well-being. Landsc. Res. 1979, 4, 17–23. [Google Scholar] [CrossRef]
- Hartig, T.; Book, A.; Garvill, J.; Olsson, T.; Garling, T. Environmental influences on psychological restoration. Scand. J. Psychol. 1996, 37, 378–393. [Google Scholar] [CrossRef] [PubMed]
- Hartig, T.; Mang, M.; Evans, G.W. Restorative effects of natural environment experiences. Environ. Behav. 1991, 23, 3–26. [Google Scholar] [CrossRef]
- Aspinall, P.; Mavros, P.; Coyne, R.; Roe, J. The urban brain: Analysing outdoor physical activity with mobile EEG. Br. J. Sports Med. 2015, 49, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Malone, K.; Tranter, P. Children’s environmental learning and the use, design and management of schoolgrounds. Child. Youth Environ. 2003, 13, 87–137. [Google Scholar]
- Neisser, U. Cognitive Psychology; Prentice Hall: Englewood Cliffs, NJ, USA, 1967. [Google Scholar]
- Berman, M.G.; Jonides, J.; Kaplan, S. The cognitive benefits of interacting with nature. Psychol. Sci. 2008, 19, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Laumann, K.; Gärling, T.; Stormark, K.M. Selective attention and heart rate responses to natural and urban environments. J. Environ. Psychol. 2003, 23, 125–134. [Google Scholar] [CrossRef]
- Kuo, F.E.; Faber Taylor, A. A potential natural treatment for attention-deficit/hyperactivity disorder: Evidence from a national study. Am. J. Public Health 2004, 94, 1580–1586. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.F.; Kuo, F.E.; Sullivan, W.C. Views of nature and self-discipline: Evidence from inner city children. J. Environ. Psychol. 2002, 22, 49–63. [Google Scholar] [CrossRef]
- Putnam, R. Bowling alone: America’s declining social capital. J. Democr. 1995, 6, 65–78. [Google Scholar] [CrossRef]
- Kim, D.; Kawachi, I. US state-level social capital and health-related quality of life: Multilevel evidence of main, mediating, and modifying effects. Ann. Epidemiol. 2007, 17, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Helliwell, J. How’s life? Combining individual and national variables to explain subjective well-being. Econ. Model. 2003, 20, 331–360. [Google Scholar] [CrossRef]
- Jackson, L. The relationship of urban design to human health and condition. Landsc. Urban Plan. 2003, 64, 191–200. [Google Scholar] [CrossRef]
- Holt-Lunstad, J.; Smith, T.B.; Layton, J.B. Social relationships and mortality risk: A meta-analytic review. PLoS Med. 2010, 7, e1000316. [Google Scholar] [CrossRef] [PubMed]
- Cooper, H.; Arber, S.; Fee, L.; Ginn, J. The Influence of Social Support and Social Capital on Health; Health Education Authority: London, UK, 1999. [Google Scholar]
- Kuo, F.E. The role of arboriculture in a healthy social ecology. J. Arboric. 2003, 29, 148–155. [Google Scholar]
- Kuo, F.E.; Bacaicoa, M.; Sullivan, W.C. Transforming inner-city landscapes: Trees, sense of safety, and preference. Environ. Behav. 1998, 30, 28–59. [Google Scholar] [CrossRef]
- Wilson, E.O. Biophilia; Harvard University Press: Cambridge, MA, USA, 1984. [Google Scholar]
- Coley, R.L.; Kuo, F.E. Where does community grow? The social context created by nature in urban public housing. Environ. Behav. 1997, 29, 468. [Google Scholar] [CrossRef]
- Sullivan, W.C.; Kuo, F.; Depooter, S. The fruit of urban nature vital neighborhood spaces. Environ. Behav. 2004, 36, 678–700. [Google Scholar] [CrossRef]
- Kuo, F.E.; Sullivan, W.C.; Levine-Coley, R.; Brunson, L. Fertile ground for community: Inner-city neighborhood common spaces. Am. J. Community Psychol. 1998, 26, 823–851. [Google Scholar] [CrossRef]
- Kweon, B.-S.; Sullivan, W.C.; Wiley, A.R. Green Common Spaces and the Social Integration of Inner-City Older Adults. Environ. Behav. 1998, 30, 832–858. [Google Scholar] [CrossRef]
- Maas, J.; van Dillen, S.M.E.; Verheij, R.A.; Groenewegen, P.P. Social contacts as a possible mechanism behind the relation between green space and health. Health Place 2009, 15, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Routledge. In Innovative Approaches to Researching Landscape and Health: Open Space: People Space 2; Ward Thompson, C.; Aspinall, P.; Bell, S. (Eds.) Routledge: Abingdon, UK, 2010.
- Sullivan, W.C.; Frumkin, H.; Jackson, R.J.; Chang, C.Y. Gaia meets Asclepius: Creating healthy places. Landsc. Urban Plan. 2014, 127, 182–184. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coutts, C.; Hahn, M. Green Infrastructure, Ecosystem Services, and Human Health. Int. J. Environ. Res. Public Health 2015, 12, 9768-9798. https://doi.org/10.3390/ijerph120809768
Coutts C, Hahn M. Green Infrastructure, Ecosystem Services, and Human Health. International Journal of Environmental Research and Public Health. 2015; 12(8):9768-9798. https://doi.org/10.3390/ijerph120809768
Chicago/Turabian StyleCoutts, Christopher, and Micah Hahn. 2015. "Green Infrastructure, Ecosystem Services, and Human Health" International Journal of Environmental Research and Public Health 12, no. 8: 9768-9798. https://doi.org/10.3390/ijerph120809768
APA StyleCoutts, C., & Hahn, M. (2015). Green Infrastructure, Ecosystem Services, and Human Health. International Journal of Environmental Research and Public Health, 12(8), 9768-9798. https://doi.org/10.3390/ijerph120809768





