Tobacco-Specific Nitrosamines in Electronic Cigarettes: Comparison between Liquid and Aerosol Levels
Abstract
:1. Introduction
2. Methods
2.1. Sample Selection
2.2. Protocol Design and Methods of Analysis
2.3. Statistical Analysis
3. Results
| Liquid Samples | Company | Nicotine Content (mg/mL) | NNN (ng/g) | NAT (ng/g) | NAB (ng/g) | NNK (ng/g) |
|---|---|---|---|---|---|---|
| Mystique Tobacco Echo | Nobacco, Athens, Greece | 18 | 7.7 | ND | 1.5 | ND |
| Maxx-Blend | Flavourart, Oleggio, Italy | 18 | ND | ND | 2.3 | ND |
| Numbers Three | Nobacco, Athens, Greece | 18 | ND | ND | 1.2 | ND |
| Spiked Sample a | 18 | 46.9 | 53.9 | 45.6 | 42.0 |
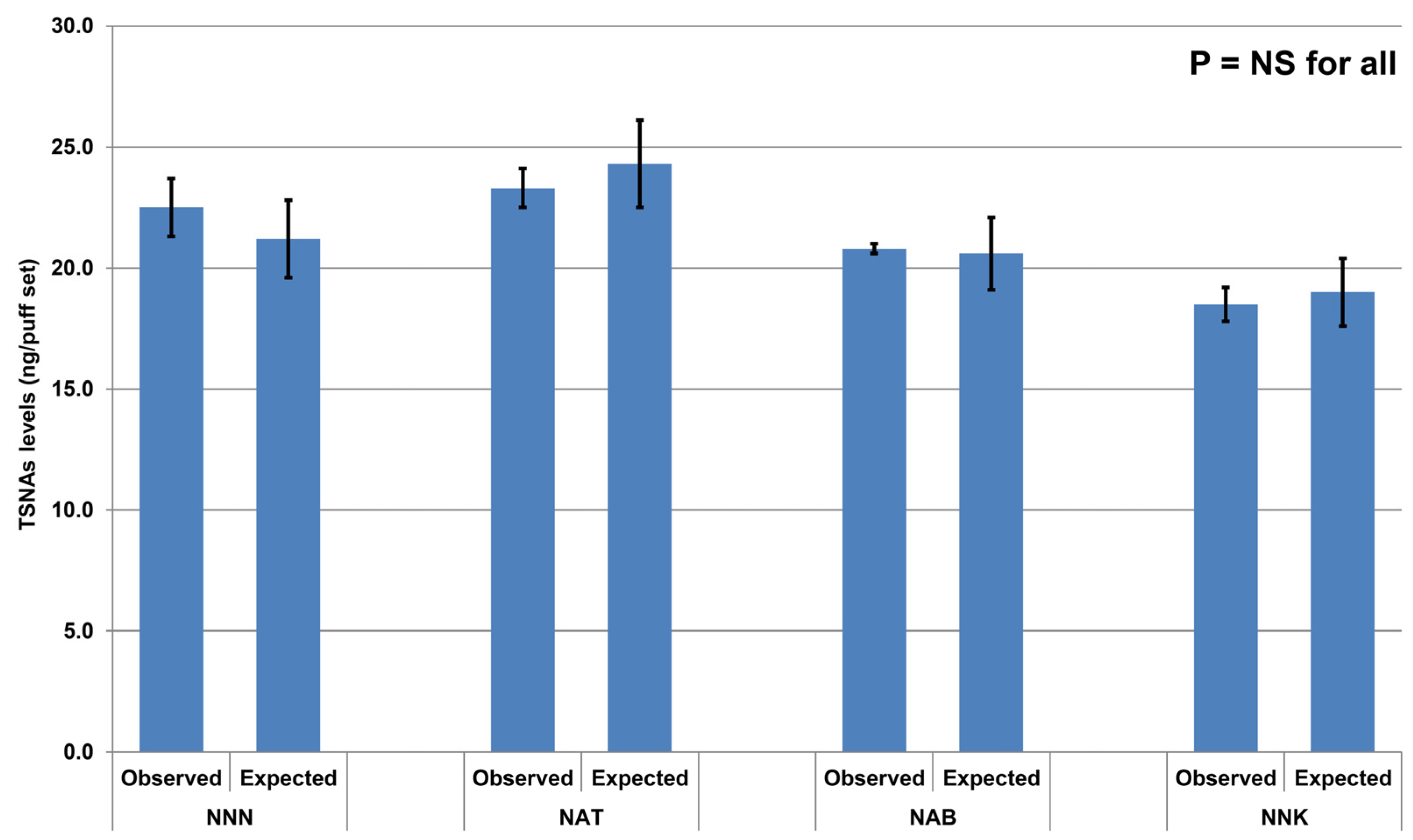
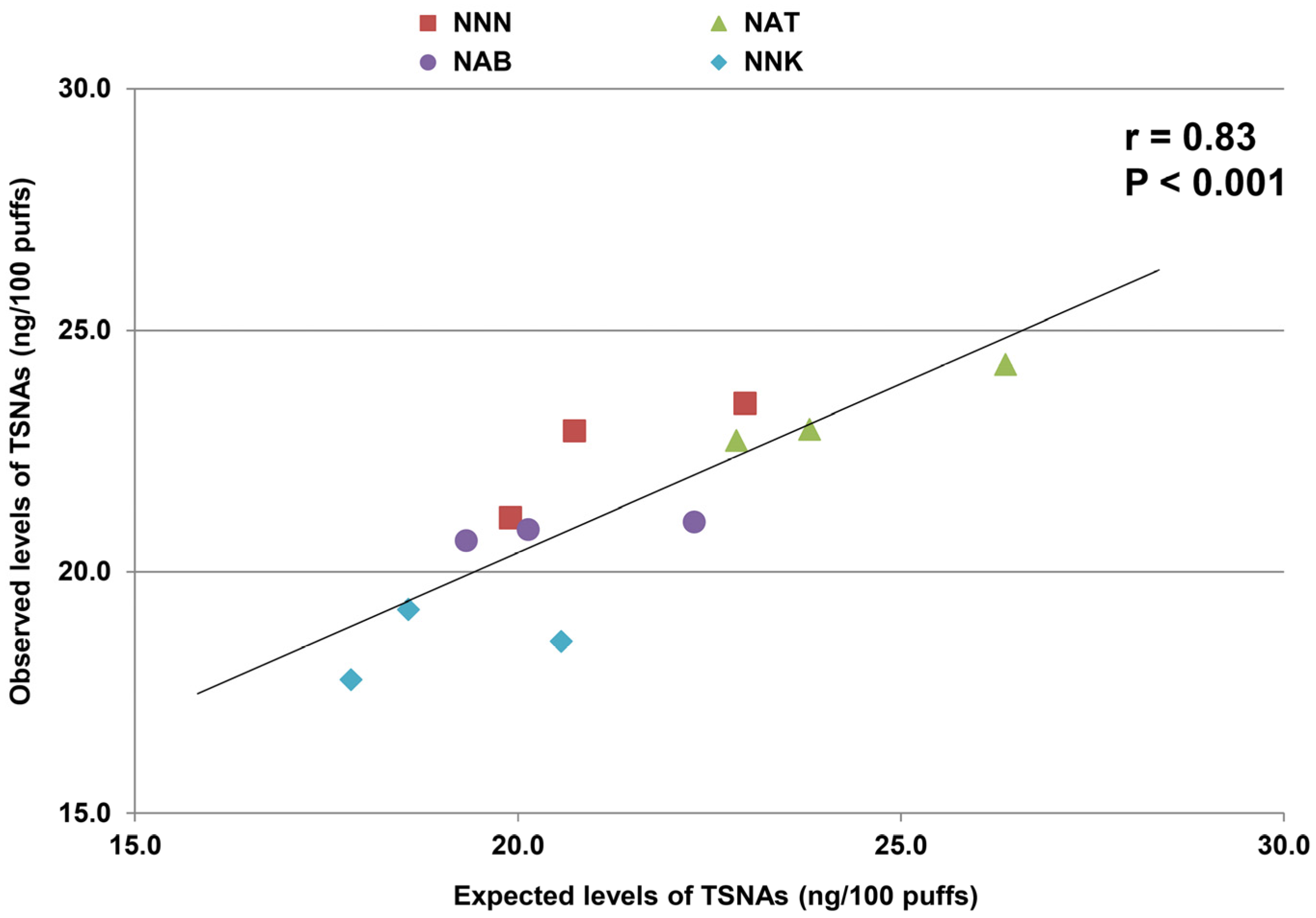
4. Discussion
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Farsalinos, K.E.; Polosa, R. Safety evaluation and risk assessment of electronic cigarettes as tobacco cigarette substitutes: A systematic review. Ther. Adv. Drug Saf. 2014, 5, 67–86. [Google Scholar] [CrossRef] [PubMed]
- Hajek, P.; Etter, J.F.; Benowitz, N.; Eissenberg, T.; McRobbie, H. Electronic cigarettes: Review of use, content, safety, effects on smokers and potential for harm and benefit. Addiction 2014, 109, 1801–1810. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S.; Hoffmann, D. Tobacco-specific nitrosamines, an important group of carcinogens in tobacco and tobacco smoke. Carcinogenesis 1988, 9, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S. Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem. Res. Toxicol. 1998, 11, 559–603. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Shin, H.S. Determination of tobacco-specific nitrosamines in replacement liquids of electronic cigarettes by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2013, 1291, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Romagna, G.; Voudris, V. Authors miss the opportunity to discuss important public health implications. J. Chromatogr. A 2013, 1312, 155–156. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Gillman, I.G.; Melvin, M.S.; Paolantonio, A.R.; Gardow, W.J.; Humphries, K.E.; Brown, S.E.; Poulas, K.; Voudris, V. Nicotine levels and presence of selected tobacco-derived toxins in tobacco flavoured electronic cigarette refill liquids. Int. J. Environ. Res. Public Health 2015, 12, 3439–3452. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, D.; Dong, M.; Hecht, S.S. Origin in tobacco smoke of N-nitrosonornicotine, a tobacco-specific carcinogen. J. Natl. Cancer Inst. 1977, 58, 1841–1844. [Google Scholar] [PubMed]
- Adams, J.D.; Lee, S.J.; Vinchkoski, N.; Castonguay, A.; Hoffmann, D. On the formation of the tobacco-specific carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone during smoking. Cancer Lett. 1983, 17, 336–346. [Google Scholar] [CrossRef]
- Fischer, S.; Spiegelhalder, B.; Eisenbarth, J.; Preussmann, R. Investigations on the origin of tobacco-specific nitrosamines in mainstream smoke of cigarettes. Carcinogenesis 1990, 11, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Zuber, J.; D’Andres, S.; Boudoux, R.; Renaud, J.M. TSNA Levels in the mainstream smoke of simplified blend prototypes. Beitr. Tabakforsch. Int. 2014, 20, 331–340. [Google Scholar]
- McAuley, T.R.; Hopke, P.K.; Zhao, J.; Babaian, S. Comparison of the effects of e-cigarette vapor and cigarette smoke on indoor air quality. Inhal. Toxicol. 2012, 24, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C.; Jacob, P., 3rd; Benowitz, N. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob. Control 2014, 23, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Schober, W.; Jörres, R.A.; Fromme, H. Response to letter regarding article, “use of electronic cigarettes (e-cigarettes) impairs indoor air quality and increases FeNO levels of e-cigarette consumers”. Int. J. Hyg. Environ. Health 2014, 217, 707–708. [Google Scholar] [CrossRef] [PubMed]
- Stepanov, I.; Jensen, J.; Hatsukami, D.; Hecht, S.S. Tobacco-specific nitrosamines in new tobacco products. Nicotine Tob. Res. 2006, 8, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Counts, M.E.; Hsu, F.S.; Laffoon, S.W.; Dwyer, R.W.; Cox, R.H. Mainstream smoke constituent yields and predicting relationships from a worldwide market sample of cigarette brands: ISO smoking conditions. Regul. Toxicol. Pharmacol. 2004, 39, 111–134. [Google Scholar] [CrossRef] [PubMed]
- Piadé, J.J.; Wajrock, S.; Jaccard, G.; Janeke, G. Formation of mainstream cigarette smoke constituents prioritized by the World Health Organization—Yield patterns observed in market surveys, clustering and inverse correlations. Food Chem. Toxicol. 2013, 55, 329–347. [Google Scholar] [CrossRef] [PubMed]
- Cahn, Z.; Siegel, M. Electronic cigarettes as a harm reduction strategy for tobacco control: A step forward or a repeat of past mistakes? J. Public Health Policy 2011, 32, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Alderman, S.L.; Song, C.; Moldoveanu, S.C.; Cole, S.K. Particle size distribution of e-cigarette aerosols and the relationship to Cambridge filter pad collection efficiency. Beitr. Tabakforsch. 2015, 26, 183–190. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farsalinos, K.E.; Gillman, G.; Poulas, K.; Voudris, V. Tobacco-Specific Nitrosamines in Electronic Cigarettes: Comparison between Liquid and Aerosol Levels. Int. J. Environ. Res. Public Health 2015, 12, 9046-9053. https://doi.org/10.3390/ijerph120809046
Farsalinos KE, Gillman G, Poulas K, Voudris V. Tobacco-Specific Nitrosamines in Electronic Cigarettes: Comparison between Liquid and Aerosol Levels. International Journal of Environmental Research and Public Health. 2015; 12(8):9046-9053. https://doi.org/10.3390/ijerph120809046
Chicago/Turabian StyleFarsalinos, Konstantinos E., Gene Gillman, Konstantinos Poulas, and Vassilis Voudris. 2015. "Tobacco-Specific Nitrosamines in Electronic Cigarettes: Comparison between Liquid and Aerosol Levels" International Journal of Environmental Research and Public Health 12, no. 8: 9046-9053. https://doi.org/10.3390/ijerph120809046
APA StyleFarsalinos, K. E., Gillman, G., Poulas, K., & Voudris, V. (2015). Tobacco-Specific Nitrosamines in Electronic Cigarettes: Comparison between Liquid and Aerosol Levels. International Journal of Environmental Research and Public Health, 12(8), 9046-9053. https://doi.org/10.3390/ijerph120809046








