Inventory of Engineered Nanoparticle-Containing Consumer Products Available in the Singapore Retail Market and Likelihood of Release into the Aquatic Environment
Abstract
:1. Introduction
2. Methods
2.1. Nano-Inventory
2.2. Determination of Emission of ENP into the Aquatic Environment
- (a)
- The article’s lifetime;
- (b)
- The ways that ENP is incorporated in the products (i.e., suspended in liquid/solid, surface bound, or in the bulk);
- (c)
- Release pattern (i.e., down the drain, runoff, and incineration and landfill).
2.2.1. Daily Release Quantity
3. Results and Discussion
3.1. Types of ENP and Their Application in Consumer Products in the Singapore Retail Market
| ENMs | Function | Application Areas | Reference |
|---|---|---|---|
| Ag | Antimicrobial protection Conductivity or electrical properties | Textiles, food packaging, medical devices, water treatment process, surface coating, electronics such as transparent conducting films, transparent electrodes for flexible devices | [3,8,9,20,28,29,30] |
| TiO2 | Absorption of ultraviolet radiation Catalyst | Electronic devices, sunscreen, cleaning, water treatment, solar cell, health and fitness | [8,9,20,28,29] |
| Al2O3 | Extremely fine powder with great capability of polishing Absorption of light Antimicrobial protection Adsorbent | Cleaning, cosmetics, water treatment, coatings, food additives, catalysts, ceramics, electrical insulators | [8,9,20,28] |
| Al | Dispersion strength catalyst | Cosmetics, catalysts, coating, optical biomaterials, drug delivery | [8,20,28,31] |
| ZnO | Sun protection Antimicrobial protection | Sunscreen, cleaning, paints, cosmetics, food packaging, personal care products/sprays | [8,9,20,28] |
| SiO2 | Extend life of paints and coatings | Paints, coatings, food packaging | [8,9,20,28] |
| Zr/ZrO2 | Good absorbability and hydrophilicity Bio-corrosion resistant and bio-compatible | Drug delivery, water treatment (membrane, ion exchanger), coatings, fuel, battery, pigment, abrasive material, medical implant | [8,28,32,33] |
| Hydroxyapatite | Biocompatibility | Therapeutic and/or diagnostic agents coatings, drug delivery, sensor, biomaterials | [8,20,28,34,35,36] |
| Ceramics | Filtration with great rejection efficiency Hardness and strength | Home and health/filter, paint, personal care products, cosmetics, food and beverage, coatings | [3,8,20,28] |
| CeO2 | Catalyst Sun protection Optical property | Coatings, paints, automotive/fuel catalysts, biomedicine | [8,28,37,38,39] |
| Fe/FexOy | Hardness and strength magnetic and catalytic properties | Food and beverage, home and health/sporting goods, environmental remediation, water treatment, catalysts | [8,9,28,40] |
| Carbon nanomaterials (C60, carbon black, carbon nanotubes (CNT) | Hardness and strength Light with high density Conductivity Metallic property Mechanical and Magnetic property | Probing electronic devices, catalysts, adsorbent in water treatment, detection devices, molecular filtration membrane, detection sensor or probe, automotive, sporting goods, clothing, food packaging | [3,8,9,20,22] |
| Nanoclays | Hardness and strength Improvements in mechanical, thermal, flame resistance, and barrier properties | Food packaging, automotive, cosmetics and toiletries, environment and water treatment, flame retardants, medical devices, packaging, paints, pigments and coatings | [3,8,20,28] |
| Organic nanomaterials | Organic nanomaterials are widely used in a variety of field, including vitamins, anti-oxidants, color agents, flavors, preservatives, drug delivery, cosmetics, nutrients and supplements. | [8,20,28] | |
| Other nanomaterials | Many other nanomaterials are being increasingly used in the commercial field, including Cu and CuxOy, Ti, metal nitrides, and alkaline earth metals. Quantum dots composed of metal (oxide), or semiconductor materials with novel electronic, optical, magnetic and catalytic properties are used in medical imaging, diagnostics and security printing at increasing rates. | [3,8,20,28,41] | |
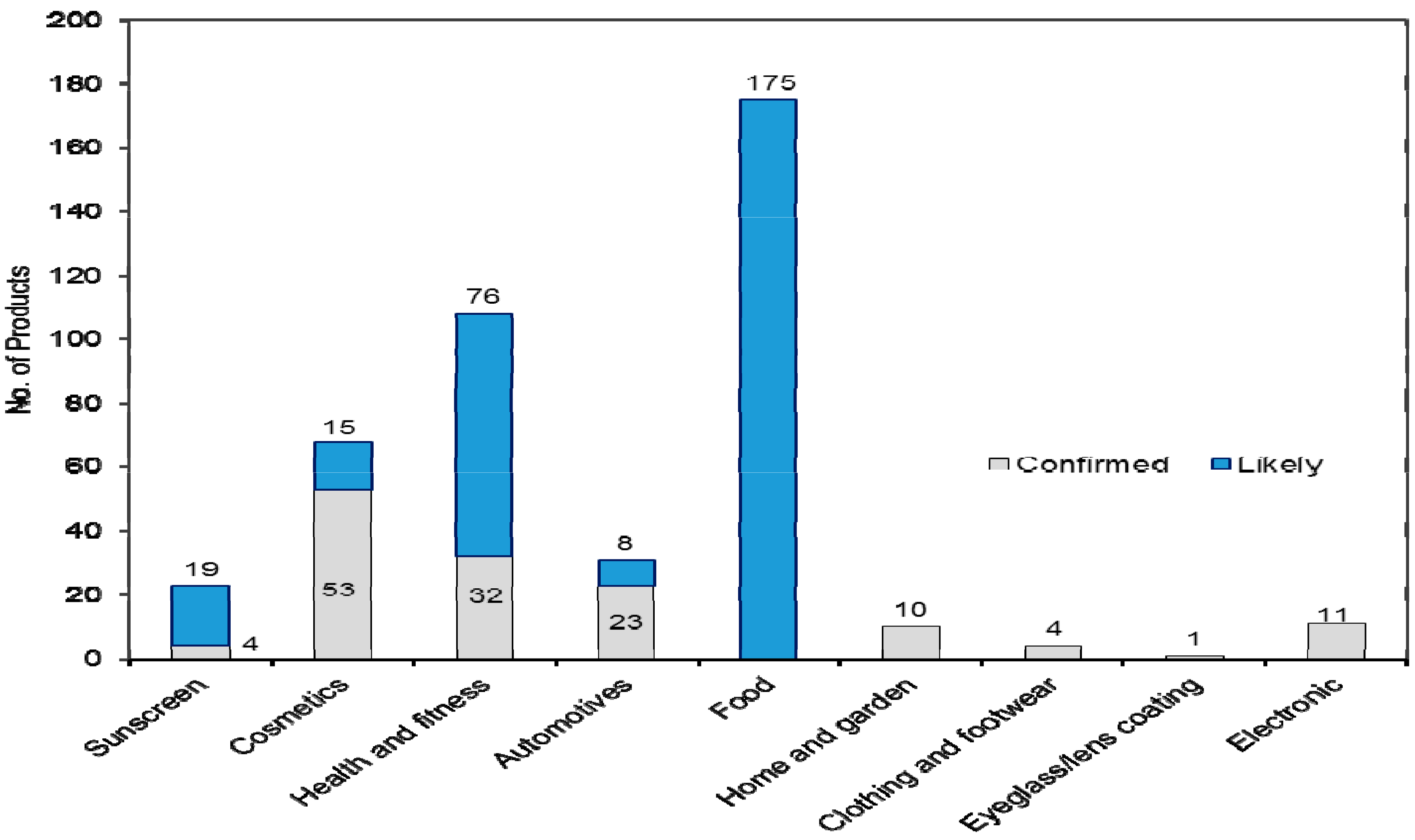
3.2. Release of ENP during Use and Disposal of Consumer Products
- Health and fitness (personal care): TiO2, SiO2, metal oxides, and nano carbon are widely used in this category. Products include: toothpaste, shampoo, shower gel/milk, facial cleaning gel, and body lotion with UV protection. The use of products in this category will lead to direct release of ENP into the wastewater. For example, TiO2 can be easily released from toothpaste during tooth brushing. In addition, SiO2-containing facial cleaning gel and shampoo have great potential to release particulate SiO2 into wastewater.
- Automotive (i.e., tire, polish and wax, coating, fuel additives): This category was found to be important primarily because of consumables such as tires, which are worn down during their lifetime. This can lead to direct release of ENP into the air and onto road surfaces, from where ENP can go directly into the aquatic environment via road runoff into surface water and in small quantities by air transport and dry deposition. ENP from maintenance products and accessories, such as car coatings or wax products, can be released into road runoff but also into wastewater during their application at private homes. The predominant ENP in this category include carbon black, ceramics, and SiO2.
- Home and garden (e.g., coatings, cleaning products): TiO2, Ti, and Ag are widely used in this category. Products in this category are relevant in terms of potential for direct release of ENP into surface water by outdoor use, and into wastewater during indoor use and disposal of products.
- Clothing and footwear: Ag is used in nano form in textiles as an antimicrobial agent. Wearing clothing with Ag is expected to lead to only minimal direct release of Ag into the aquatic environment. The main release will occur during washing of clothing containing nanoparticles of Ag into the wastewater systems [44]. The amount of ionic and particulate Ag released from the fabrics depends on the type of Ag incorporation into the textile.
- Eyeglass/lens coating: This is a small-scale use category. ENP may be released into wastewater when washing glasses but only if the ENP were non-permanently coated on the surface of the lenses. The main ENP involved include TiO2, SiO2, and polymer thin films, which are all permanent coatings.
- Construction materials, paint, and coating: Most nanomaterials being used in this category are fixed in the products. Some ENP such as Ag will be released in ionic form. Environmental degradation over time and rainfall flushing during heavy storms may cause small quantities of ENP to be released into the aquatic environment. Most of the released nanosized particles are expected to go into surface runoff.
| Category | # | TiO2 | ZnO | Pt | Nano–hyaluronic acid | Si | Cu | Nano capsule | Nano vitamin E | Nano–lipobelle E Q10 | Nano Liposome | Au | Ag | SiO2 | MnO2 | Ti | Ceramic | Iron oxides | Al2O3 | Alumino–silicate oxides | Copper oxides | Nano Collagen | C60 | Carbon nanotube | Carbon nanofiber | Nano Fiber | Carbon black | Nano glass | Unclassified |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sunscreen | 23 | 19 | 12 | ||||||||||||||||||||||||||
| Cosmetics | 68 | 22 | 2 | 5 | 4 | 1 | 1 | 2 | 1 | 2 | 1 | 12 | 1 | 1 | 4 | 4 | 1 | 1 | 2 | 1 | 19 | ||||||||
| Health and fitness | 108 | 69 | 1 | 1 | 1 | 18 | 2 | 1 | 1 | 1 | 1 | 2 | 22 | ||||||||||||||||
| Automotive | 31 | 1 | 1 | 5 | 1 | 1 | 22 | ||||||||||||||||||||||
| Food | 175 | 11 | 5 | 159 | |||||||||||||||||||||||||
| Home and garden | 10 | 6 | 4 | 1 | |||||||||||||||||||||||||
| Clothing and footwear | 4 | 1 | 3 | ||||||||||||||||||||||||||
| Eyeglass/lens coatings | 1 | 1 | |||||||||||||||||||||||||||
| Electronics | 11 | 4 | 4 | 1 | 2 | ||||||||||||||||||||||||
| Total | 431 | 128 | 20 | 6 | 4 | 1 | 1 | 2 | 1 | 2 | 1 | 13 | 10 | 179 | 1 | 7 | 6 | 5 | 4 | 1 | 1 | 2 | 1 | 1 | 1 | 2 | 1 | 1 | 69 |
| Product | Category | ENP Type | Type of Matrix Containing ENP | Release Pattern | Release Factor (%) | Reference |
|---|---|---|---|---|---|---|
| Sunscreen | Sunscreen | TiO2, ZnO | Suspended in liquid | Down the drain | 90 | Only 0.03% ENP can penetrate into skin after 24 h exposure [45] and 10% loss in the sea |
| Day cream | Skincare/Cosmetics | Pt | Suspended in liquid | Down the drain | 95 | Assumption: approximately 5% losses to solid waste and other solid phases. |
| Facial cleaning gel | Skincare/Cosmetics | Nano-hyaluronic acid | Suspended in liquid | – | 0 | Based on the assumption: present in form of small vesicles that get approximately 100% adsorbed on the skin and loose nano form. |
| Facial cleaning gel | Skincare/Cosmetics | MnO2 | Suspended in liquid | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Day cream | Skincare/Cosmetics | Au, TiO2 | Suspended in liquid | Down the drain | 95 | Assumption: approximately 5% loss to solid waste and other solid phases. |
| Mask cream | Skincare/Cosmetics | Pt | Suspended in liquid | Down the drain | 95 | Based on the assumption: approximately 5% loss to solid waste and other solid phases. |
| Day cream | Skincare/Cosmetics | Nano-capsules | Suspended in liquid | – | 0 | Nano-capsules were described to be adsorbed completely by Lademann et al., 2013 [47]. |
| Eyeliner | Makeup/Cosmetics | Iron oxides | Suspended in solid | Down the drain | 10 | 90% of the product retention on the facial cotton |
| Concealer | Makeup/Cosmetics | Al2O3 | Suspended in liquid | Down the drain | 30 | 70% retention on the facial cotton |
| Foundation powder | Makeup/Cosmetics | ZnO | Suspended in solid | Down the drain | 30 | 70% retention on the facial cotton |
| Toothpaste | Health and fitness | All | Suspended in liquid | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Toothbrush | Health and fitness | Ag, Au | Surface bound | Solid waste | 0 | Permanent coating |
| Body lotion | Health and fitness | TiO2 | Suspended in liquid | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Shower spray | Health and fitness | SiO2 | Suspended in liquid | Down the drain | 80 | 20% loss during the spray |
| Shower gel | Health and fitness | ZnO, TiO2 | Suspended in liquid | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Shampoo | Shampoo/Health and fitness | TiO2 | Suspended in liquid | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Badminton Rackets | Sporting goods | All | Surface bound | Unclassified | 0 | Disposal of the products as solid wastes |
| Tire | Automotive | SiO2, Carbon black | Suspended in solid | Runoff | 70 | Based on the assumption: 70% of the particles deposited on roadsides are washed off of roadsides and reach urban runoff during rainfall. |
| Car coating (& polish wax) | Automotive | Ceramic, TiO2 | Suspended in liquid | Down the drain | 100 | Kojima et al., 2011 [48] |
| Beverages, instant noodles, seasoning, snacks, sweets | Food | TiO2, SiO2 | Suspended in solid | Down the drain | 60 | Oral exposure is assumed to be the main route in terms of Food. SiO2 and TiO2 are assumed to not dissolve during passage through the gastrointestinal tract. However, according to UNEP 2011, 30% of food gets thrown away [49]. Long-term retention in the body is not clear, but assumed to be 10%. |
| Beverage | Food | ZnO | Suspended in solid | –(as ions) | 0 | Oral exposure is assumed to be the main route in terms of Food. ZnO is assumed to be transferred into ionic form during passage through the gastrointestinal tract. |
| Cleaning detergent | Home and garden | TiO2, Ag, Ti | Suspended in liquid | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Filter | Home and garden | Ag | Surface bound | Unclassified | 0 | Permanent coating |
| Socks | Clothing and footwear | Ag | Surface bound | Down the drain | 100 | Technical guide document on risk assessment, 2003 [46] |
| Hair curling machine | Household appliances/Electronics | Ag, Ti, and ceramic | Surface bound | – | 0 | Permanent coating |
| Washing machine | Household appliances/Electronics | Ag | Surface bound | Down the drain | 100 | Tiede et al., 2011 [50] |
3.3. Estimation of ENP Reaching the Aquatic Environment
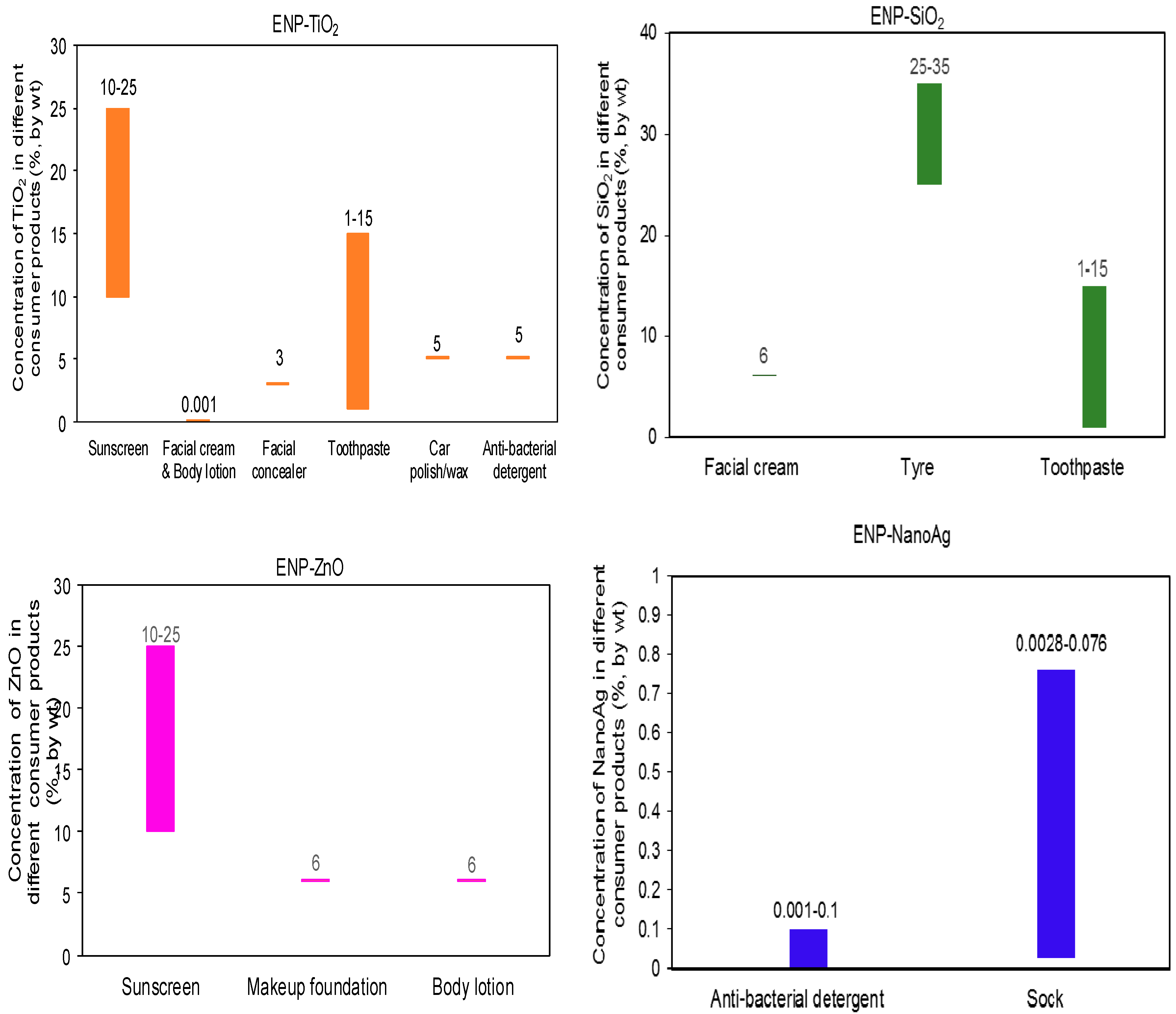
| Category | ENP Type | ENP Concentration (% by Weight) | References | |
|---|---|---|---|---|
| Low | High | |||
| Sunscreen | TiO2 | 10 | 25 | Weir et al., 2012 [42], EC regulation on Sunscreen [24] |
| Sunscreen | ZnO | 10 | 25 | Weir et al., 2012 [42], EC regulation on Sunscreen [24] |
| Day cream/Cosmetics | Pt | 10 | 20 | Purest colloids: http://www.purestcolloids.com/colloidal-skin-conditioners.php [51] |
| Day cream/Cosmetics | Nano-lipobelle EQ10 | 0.5 | 4 | Müller et al., 2007 [52] |
| Lotion/Cosmetics | Nano hyaluronic acid | 60 | 100 | Based on the similar product information: http://www.ebay.com.au/bhp/hyaluronic-acid-serum [53] |
| Cream/Cosmetics | Nano-capsules | 0.5 | 4 | Müller et al., 2007 [52] |
| Cream/Cosmetics | Au | 20 | 20 | Taufikurohmah et al., 2011 [54] |
| Cream/Cosmetics | SiO2 | 6 | 15 | Tiede et al., 2011 [55] |
| Facial cleaning gel/Cosmetics | MnO2 | 0.001 | 0.001 | Weir et al., 2012 [42] |
| Facial cleaning gel/Cosmetics | TiO2 | 0.001 | 0.001 | Weir et al., 2012 [42] |
| Foundation/Cosmetic | TiO2 | 0.5 | 4 | Müller et al., 2007 [52] |
| Eyeliner/Cosmetic | Iron oxides | 3 | 3 | Tiede et al., 2011 [55] |
| Eyeliner/Cosmetic | Nano Collagen | 0.5 | 4 | Müller et al., 2007 [52] |
| Concealer/Cosmetic | Al2O3 | 3 | 3 | Tiede et al., 2011 [55] |
| Foundation/Cosmetic | Alumino-silicate oxides | 3 | 3 | Tiede et al., 2011 [55] |
| Eye shadow/Cosmetic | Au | 3 | 3 | Tiede et al., 2011 [55] |
| Foundation/Cosmetic | ZnO | 3 | 3 | Tiede et al., 2011 [55] |
| Toothpaste/Health and fitness | TiO2 | 1 | 15 | Weir et al., 2012 [42], Tiede et al., 2011 [55] |
| Toothpaste/Health and fitness | SiO2 | 1 | 15 | Weir et al., 2012 [42], Tiede et al., 2011 [55] |
| Toothbrush/Health and fitness | Ag | 0.001 | 0.1 | Weir et al., 2012 [42], Tiede et al., 2011 [55] |
| Toothbrush/Health and fitness | Au | 0.001 | 0.1 | Weir et al., 2012 [42], Tiede et al., 2011 [55] |
| Body lotion/Health and fitness | TiO2 | 0.001 | 0.001 | Weir et al., 2012 [42] |
| Shower spray/Health and fitness | SiO2 | 0.001 | 0.001 | Weir et al., 2012 [42] |
| Shampoo/Health and fitness | TiO2 | 0.001 | 0.001 | Weir et al., 2012 [42] |
| Tire/Automotive | SiO2 | 25 | 35 | Wik et al., 2005 [56] |
| Tire/Automotive | Carbon black | 25 | 35 | Wik et al., 2005 [56] |
| Coating/Automotive | Ceramic | 10 | 10 | Boxall et al., 2007 [11] |
| Coating/Automotive | Nano glass | 10 | 10 | Boxall et al., 2007 [11] |
| Coating/Automotive | TiO2 | 5 | 5 | Boxall et al., 2007 [11] |
| Beverage/Food | SiO2 | 0 | <1.5 | Health Canada: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/1-anti-eng.php [57] |
| Beverage/Food | ZnO | <1.5 | Health Canada: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/1-anti-eng.php [57] | |
| Beverage/Food | TiO2 | <1.5 | Health Canada: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/1-anti-eng.php [57] | |
| Instant noodle/Food | SiO2 | 1 | 1 | Health Canada: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/1-anti-eng.php [57] |
| Sweets/Food | TiO2 | 0.34 | 0.34 | European food safety authority, 2004 [58]. |
| Snack/Food | SiO2 | 1 | 1 | Health Canada: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/1-anti-eng.php [57] |
| Seasoning/Food | SiO2 | 1 | 1 | Health Canada: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/1-anti-eng.php [57] |
| Cleaning/Home and garden | TiO2 | 5 | 5 | Boxall et al., 2007 [11] |
| Cleaning/Home and garden | Ag | 0.001 | 0.1 | Boxall et al., 2007 [11] |
| Cleaning/Home and garden | Ti | 5 | 5 | Boxall et al., 2007 [11] |
| Antimicrobial coating/Home and garden | TiO2 | 5 | 5 | Boxall et al., 2007 [11] |
| Clothing and footwear | Ag | 0.0028 | 0.076 | Benn and Westerhoff, 2008 [44] |
| Household electrical appliances/Electronics | Ag | 100 | 100 | Tiede et al., 2011 [55] |
| Product Name | Category | Usage Quantity (g/pc/d) | ||
|---|---|---|---|---|
| Low | High | References | ||
| Sunscreen | Sunscreen | 0.04 | 1.90 | Biesterbos et al., 2013 [59] |
| Day cream | Skincare/Cosmetics | 0.1 | 1.1 | Biesterbos et al., 2013 [59] |
| Eye cream | Skincare/Cosmetics | 0.005 | 0.055 | Assumption: 1/20 of the day cream. |
| Toner | Skincare/Cosmetics | 0.5 | 4.3 | Biesterbos et al., 2013 [59] |
| Night cream | Skincare/Cosmetics | 0.09 | 0.9 | Biesterbos et al., 2013 [59] |
| Facial cleaning gel | Skincare/Cosmetics | 0.5 | 4.3 | Biesterbos et al., 2013 [59] |
| Cream mask | Skincare/Cosmetics | 0.3 | 3.3 | Assumption: 3 times more than day cream |
| Foundation | Makeup/Cosmetics | 0.002 | 0.132 | Loretz et al., 2006 [50] |
| Foundation powder | Makeup/Cosmetics | 0.002 | 0.132 | Loretz et al., 2006 [50] |
| Eyeliner | Makeup/Cosmetics | 0.0002 | 0.0003 | Biesterbos et al., 2013 [59] |
| Concealer | Makeup/Cosmetics | 0.006 | 0.06 | Assumption: 10% of the usage quantity of the foundation. |
| Eye shadow | Makeup/Cosmetics | 0.006 | 0.06 | Assumption: 10% of the usage quantity of the foundation. |
| Toothpaste | Toothpaste/Health and fitness | 1.515 | 2.669 | Hall et al., 2007 [60] |
| Shampoo | Health and fitness | 3.79 | 21.91 | Loretz et al., 2006 [50] |
| Body lotion | Health and fitness | 1.836 | 7.25 | Hall et al., 2007 [60] |
| Shower gel | Health and fitness | 6 | 23 | Loretz et al., 2006 [50] |
| Tire | Automotive | 0.120 (unit: g/km) | 0.120 | Gehrig et al., 2005 [64] |
| Coating | Automotive | 0.3 | 0.3 | Tiede et al., 2011 [55] |
| Instant coffee | Beverage/Food | 1.0 | 1.0 | Derived from annual instant coffee consumption of 2000 tons in Singapore [65] |
| Instant noodles | Food | 3.6 | 3.6 | Seasoning powder (15 g) is 14% of an instant noodle package weight (110 g). Daily intake of grain products is 3.7 g/kg-day, cited from 2011 Exposure Factors Handbook of the USEPA [63]. The range of BMI for Asians is 17–35 kg/m2 (WHO expert consultation report, 2004) [66]. The average height for male and female in Singapore is 1.7 and 1.6 meters, respectively [67]. Therefore, the estimated average body weight for Asians is assumed to be 70 kg. The percentage of instant noodles in total grain products is assumed to be 10%. Therefore, daily intake of instant noodles is 14% × 259 × 10% = 3.6 g. |
| Seasoning powder | Food | 0.032 | 0.032 | Carlsen et al., 2011 [68] |
| Snacks (Chips) | Food | 0.42 | 0.98 | Daily intake of snacks for Asians is 0.1 (± 0.04) g/kg-day (USEPA, 2011, Exposure factors handbook) [63]. The estimated average body weight for Asians is assumed to be 70 kg. The percentage of chips in total snacks is assumed to be 10%. Daily intake of chips is = 7(± 2.8) × 10% = 0.7(± 0.28) g. |
| Sweets (Chocolate candy) | Food | 0.14 | 0.14 | Daily per capita intake of sweets is 0.4 g/kg-day in Exposure Factors Handbook [63]. The estimated average body weight for Asians is assumed to be 70 kg. The percentage of chocolate candy investigated in our inventory in total sweets is assumed to be 5%. Individual daily intake of chocolate candy is 0.4 × 70 × 5% = 0.14 g. |
| Cleaning | Home and garden | 110 | 110 | Technical guide document on risk assessment, 2003 [46] |
| Clothing | Clothing and footwear | 4.45 | 4.45 | Assumption: 1/20 of emission from clothing. Tiede et al., 2011 [55] |
| Washing machine | Household electrical appliances | 1.375 | 1.375 | Tiede et al., 2011 [55] |
| Category | Market Share (%) | Reference |
|---|---|---|
| Sunscreen | 85 | Market survey: out of 27 products, 23 are likely to contain ENP. |
| Skincare/Cosmetics | 1 | Tiede et al., 2011 [55] |
| Makeup/Cosmetics | 2 | Tiede et al., 2011 [55] |
| Toothpaste/Health and fitness | 78 | Market survey: Out of 79 toothpaste, 62 are likely to use ENP. |
| Hair treatment/Health and fitness | 3 | Market survey: Out of 309 products 9 likely to contain ENP |
| Body lotion/Health and fitness | 0.5 | Tiede et al., 2011 [55] |
| Shower spray/Health and fitness | 0.1 | Tiede et al., 2011 [55] |
| Shower gel/Health and fitness | 2.5 | Market survey: Out of 162 products, 4 are likely to contain ENP. |
| Tire/Automotive | 10 | Tiede et al., 2011 [55] |
| Coating/Automotive | <1 | Tiede et al., 2011 [55] |
| Beverages/Food | 31 | Market survey: Out of 148 products, 46 are likely to use ENP. |
| Instant noodles/Food | 26 | Market survey: Out of 232 products, 60 are likely to use ENP. |
| Seasoning/Food | 0.1 | Based on the assumption: market share is very low, 0.1%. |
| Snack/Food | 21 | Market survey: Out of 247 products, 53 are likely to use ENP. |
| Sweets/Food | 13 | Market survey: Out of 79 products, 10 are likely to use ENP. |
| Cleaning/Home and garden | <1 | Tiede et al., 2011 [55] |
| Clothing and footwear | <1 | Tiede et al., 2011 [55] |
| Household electrical appliances | <1 | Tiede et al., 2011 [55] |
| No. | ENP Type | Product/Category | Release into Water | Emission Quantity (g/pc/d) | Release Elsewhere | Emission Quantity (g/pc/d) | ||
|---|---|---|---|---|---|---|---|---|
| Low | High | Low | High | |||||
| 1 | TiO2 | Sunscreen, automotive, cleaning, skincare, makeup, toothpaste, shampoo, body lotion, shower gel, beverages | Down the drain | 3.80E–02 | 5.01E–01 | Losses in the sea, solid waste | 2.30E–04 | 2.50E–02 |
| 2 | ZnO | Sunscreen, makeup, skincare | Down the drain | 5.93E–04 | 1.41E–01 | Solid waste | 6.59E–05 | 1.56E–02 |
| 3 | Pt | Skincare | Down the drain | 7.20E–07 | 3.17E–04 | Solid waste | 3.79E–08 | 1.67E–05 |
| 4 | Au | Skincare, makeup | Down the drain | 1.21E–06 | 1.36E–03 | Solid waste | 2.25E–07 | 7.33E–05 |
| 5 | MnO2 | Skincare | Down the drain | 1.52E–09 | 1.30E–08 | N/A | N/A | N/A |
| 6 | SiO2 | Skincare, toothpaste, shower spray, beverages, instant noodles, seasoning, snacks | Down the drain | 1.02E–02 | 8.59E–02 | Solid waste and aerosol in the air | 4.12E–03 | 9.14E–03 |
| 7 | Alumino–silicate oxides | Makeup | Down the drain | 1.80E–08 | 1.19E–06 | Solid waste | 4.20E–08 | 2.77E–06 |
| 8 | Iron oxides | Makeup | Down the drain | 3.00E–08 | 9.00E–06 | Solid waste | 2.70E–07 | 8.10E–05 |
| 9 | Al2O3 | Makeup | Down the drain | 2.40E–08 | 7.20E–06 | Solid waste | 5.60E–08 | 1.68E–05 |
| 10 | Cu | Makeup | Down the drain | 1.08E–08 | 1.08E–07 | Solid waste | 2.52E–08 | 2.52E–07 |
| 11 | Si | Makeup | Down the drain | 1.08E–08 | 1.08E–07 | Solid waste | 2.52E–08 | 2.52E–07 |
| 12 | Collagen | Makeup | Down the drain | 2.00E–10 | 2.40E–09 | solid waste | 1.80E–09 | 2.16E–08 |
| 13 | Carbon black * | Tire | Runoff | 5.98E–02 | 8.38E–02 | Other environmental compartments: air and soil. | 2.56E–02 | 3.59E–02 |
| 14 | SiO2 * | Tire | Runoff | 5.98E–02 | 8.38E–02 | Other environmental compartments: air (airborne particles) and soil. | 2.56E–02 | 3.59E–02 |
| 15 | Ceramic | Car coating | Down the drain | 1.20E–04 | 1.20E–04 | N/A | N/A | N/A |
| 16 | Nano glass | Car coating | Down the drain | 3.00E–05 | 3.00E–05 | N/A | N/A | N/A |
| 17 | Ti | Home cleaning | Down the drain | 5.50E–03 | 5.50E–03 | N/A | N/A | N/A |
| 18 | Ag | Cleaning, clothing and footwear, household electrical appliances | Down the drain | 1.38E–02 | 1.42E–02 | N/A | N/A | N/A |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nanotechnology and Human Health: Scientific Evidence and Risk Governance. Available online: http://apps.who.int/iris/handle/10665/108626 (assessed on 1 July 2015).
- Chaudhry, Q.; Blackburn, J.; Floyd, P. A scoping study to identify gaps in environmental regulation for the products and applications of nanotechnologies. Available online: http://randd.defra.gov.uk/Default.aspx?Menu=Menu&Module=More&Location=None&Completed=0&ProjectID=13855 (assessed on 23 July 2015).
- Aitken, R.J.; Chaudhry, M.Q.; Boxall, A.B.A.; Hull, M. In-depth reivew: Manufacture and use of nanomaterials: current status in the UK and global trends. Occup. Med. (Lond). 2006, 56, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Nanomaterials in Consumer Products—Update of Products on the European Market in 2010. Available online: http://rivm.openrepository.com/rivm/handle/10029/259837 (assessed on 1 July 2015).
- Zhang, W. Nanoscale iron particles for environmental remediation: An overview. J. Nanopart. Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- USEPA. Nanomaterial Case Studies: Nanoscale Titanium Dioxide in Water Treatment and in Topical Sunscreen. Available online: http://cfpub.epa.gov/ncea/cfm/recordisplay.cfm?deid=230972 (assessed on 1 July 2015).
- Nowack, B.; Krug, H.F.; Height, M. 120 Years of Nanosilver History: Implications for Policy Makers. Environ. Sci. Technol. 2011, 45, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Nanoscience and Nanotechnologies: Opportunities and Uncertainties. Available online: https://royalsociety.org/~/media/Royal_Society_Content/policy/publications/2004/9693.pdf (assessed on 1 July 2015).
- Schmid, K.; Riediker, M. Use of nanoparticles in swiss industry: A targeted survey. Environ. Sci. Technol. 2008, 42, 2253–2260. [Google Scholar] [CrossRef] [PubMed]
- Maynard, A.D.; Aitken, R.J.; Butz, T.; Colvin, V.; Donaldson, K.; Oberdörster, G.; Philbert, M.A.; Ryan, J.; Seaton, A.; Stone, V.; et al. Safe handling of nanotechnology. Nature 2006, 444, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Boxall, A.; Chaudhry, Q.; Sinclair, C.; Jones, A.; Aitken, R.; Jefferson, B.; Watts, C. Current and Future Predicted Environmental Exposure to Engineered Nanoparticles; Central Science Laboratory: Yorkshire, UK, 2007. [Google Scholar]
- Savolainen, K.; Pylkkänen, L.; Norppa, H.; Falck, G.; Lindberg, H.; Tuomi, T.; Vippola, M.; Alenius, H.; Hämeri, K.; Koivisto, J.; et al. Nanotechnologies, engineered nanomaterials and occupational health and safety—A review. Safety Sci. 2010, 48, 957–963. [Google Scholar] [CrossRef]
- Breggin, L.K.; Falkner, R.; Pendergrass, J.; Porter, R.; Jaspers, N. Addressing the risks of nanomaterials under united states and european union regulatory frameworks for chemicals. In Assessing Nanoparticle Risks to Human Health; Ramachandran, G., Ed.; William Andrew: Waltham, MA, USA, 2011; pp. 195–272. [Google Scholar]
- Lee, J.H.; Lee, S.-B.; Bae, G.N.; Jeon, K.S.; Yoon, J.U.; Ji, J.H.; Sung, J.H.; Lee, B.G.; Lee, J.H.; Yang, J.S.; et al. Exposure assessment of carbon nanotube manufacturing workplaces. Inhal. Toxicol. 2010, 22, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, C.; Von Goetz, N.; Scheringer, M.; Wormuth, M.; Hungerbühler, K. Potential exposure of German consumers to engineered nanoparticles in cosmetics and personal care products. Nanotoxicology 2011, 5, 12–29. [Google Scholar] [CrossRef] [PubMed]
- Perrenoud, A.; Gasser, M.; Rutishauser, B.R.; Gehr, P.; Riediker, M. Characterisation of nanoparticles resulting from different braking behaviours. Int. J. Biomed. Nanosci. Nanotechnol. 2010, 1. [Google Scholar] [CrossRef]
- Fröhlich, E.; Roblegg, E. Models for oral uptake of nanoparticles in consumer products. Toxicology 2012, 291, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Handy, R.D.; Van Den Brink, N.; Chappell, M.; Mühling, M.; Behra, R.; Dušinská, M.; Simpson, P.; Ahtiainen, J.; Jha, A.N.; Seiter, J.; et al. Practical considerations for conducting ecotoxicity test methods with manufactured nanomaterials: What have we learnt so far? Ecotoxicology 2012, 21, 933–972. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Definition of a Nanomaterial. Available online: http://ec.europa.eu/environment/chemicals/nanotech/faq/definition_en.htm (accessed on 1 July 2015.).
- Woodrow Wilson Institute. Nanotechnology Consumer Product Inventory. Available online: http://www.nanotechproject.org/cpi/ (accessed on 1 July 2015).
- Hansen, S.F.; Michelson, E.S.; Kamper, A.; Borling, P.; Stuer-Lauridsen, F.; Baun, A. Categorization framework to aid exposure assessment of nanomaterials in consumer products. Ecotoxicology 2008, 17, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Köhler, A.R.; Som, C.; Helland, A.; Gottschalk, F. Studying the potential release of carbon nanotubes throughout the application life cycle. J. Clean. Prod. 2008, 16, 927–937. [Google Scholar] [CrossRef]
- Mueller, N.C.; Nowack, B. Exposure modeling of engineered nanoparticles in the environment. Environ. Sci. Technol. 2008, 42, 4447–4453. [Google Scholar] [CrossRef] [PubMed]
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex:32009R1223 (accessed on 1 July 2015).
- Vogel, U.; Savolainen, K.; Wu, Q.; Tongeren, M.V.; Brouwer, D.; Berges, M. Handbook of Nanosafety: Measurement, Exposure and Toxicology; Academic Press: Londn, UK, 2014. [Google Scholar]
- Brake and Tire Wear Emissions From On-Road Vehicles in MOVES2014. Available online: http://www.epa.gov/otaq/models/moves/documents/420r14013.pdf (accessed on 1 July 2015).
- Statistics In Brief 2013—Land Transport Authority. Available online: https://www.lta.gov.sg/content/dam/ltaweb/corp/PublicationsResearch/files/FactsandFigures/Statistics%20in%20Brief%202014.pdf (accessed on 1 July 2015).
- Wijnhoven, S.W.P.; Dekkers, S.; Hagens, W.I.; de Jong, W.H. Exposure to Nanomaterials in Consumer Products. Available online: http://www.rivm.nl/milieuportaal/images/RIVMrapport_%27Exposure_to_nanomaterials_in_consumer_products%27%5B1%5D%5B1%5D.pdf (accessed on 1 July 2015).
- Overview on Promising Nanomaterials for Industrial Applications. Available online: http://www.tut.ee/public/m/Mehaanikateaduskond/Instituudid/Materjalitehnika_instituut/MTX9100/Additional_reading/NanoMat2015.pdf (accessed on 1 July 2015).
- AmericanElements: Silver Nanoparticles. Available online: http://www.americanelements.com/agnp.html (accessed on 1 July 2015).
- AzoNano: Aluminium/Aluminum Nanoparticles—Properties, Application. Available online: http://www.azonano.com/article.aspx?ArticleID=3257 (accessed on 1 July 2015).
- AmericanElements: Zirconium Oxide Nanopowder. Available online: http://www.americanelements.com/zroxnp.html (accessed on 1 July 2015).
- DaNa: Zirconium Dioxide. Available online: http://nanopartikel.info/en/nanoinfo/materials/zirconium-dioxide (accessed on 1 July 2015).
- Uskoković, V.; Uskoković, D.P. Nanosized hydroxyapatite and other calcium phosphates: Chemistry of formation and application as drug and gene delivery agents. J. Biomed. Mater. Res. B. 2011, 96, 152–191. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.; Ghosh, S.; Agarwal, A. Carbon nanotube reinforced hydroxyapatite composite for orthopedic application: A review. Mater. Sci. Eng. C 2012, 32, 1727–1758. [Google Scholar] [CrossRef]
- Du, M.; Song, W.; Cui, Y.; Yang, Y.; Li, J. Fabrication and biological application of nano–hydroxyapatite (nHA)/alginate (ALG) hydrogel as scaffolds. J. Mater. Chem. 2011, 21, 2228–2236. [Google Scholar] [CrossRef]
- Fauchadour, D.; Jeanson, T.; Bousseau, J.-N.; Echalier, B. Nanoparticles of Cerium Oxide—Application to Coatings Technologies. Available online: http://www.pcimag.com/articles/83469-nanoparticles-of-cerium-oxide-application-to-coatings-technologies (accessed on 1 July 2015).
- Goharshadi, E.K.; Samiee, S.; Nancarrow, P. Fabrication of cerium oxide nanoparticles: Characterization and optical properties. J. Colloid Interf. Sci. 2011, 356, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Patent Application Title: Cerium Oxide Nanoparticles. Available online: http://www.faqs.org/patents/app/20090233098 (accessed on 1 July 2015).
- Xu, P.; Zeng, G.M.; Huang, D.L.; Feng, C.L.; Hu, S.; Zhao, M.H.; Lai, C.; Wei, Z.; Huang, C.; Xie, G.X.; et al. Use of iron oxide nanomaterials in wastewater treatment: A review. Sci. Total Environ. 2012, 424, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Esteve-turrillas, F.A.; Abad-fuentes, A. Applications of quantum dots as probes in immunosensing of small-sized analytes. Biosens. Bioelectron. 2013, 41, 12–29. [Google Scholar] [CrossRef] [PubMed]
- Weir, A.; Westerhoff, P.; Fabricius, L.; Hristovski, K.; von Goetz, N. Titanium dioxide nanoparticles in food and personal care products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.; Kramer, E.; Oomen, A.G.; Rivera, Z.E.H.; Oegema, G.; Tromp, P.C.; Fokkink, R.; Rietveld, A.; Marvin, H.J.P.; Weigel, S.; et al. Presence of nano-sized silica during in vitro digestion of foods containing silica as a food additive. ACS Nano 2012, 6, 2441–2451. [Google Scholar] [CrossRef] [PubMed]
- Benn, T.M.; Westerhoff, P. Nanoparticle silver released into water from commercially available sock fabrics. Environ. Sci. Technol. 2008, 42, 4133–4139. [Google Scholar] [CrossRef] [PubMed]
- Cross, S.E.; Innes, B.; Roberts, M.S.; Tsuzuki, T.; Robertson, T.A.; McCormick, P. Human skin penetration of sunscreen nanoparticles: In vitro assessment of a novel micronized zinc oxide formulation. Skin Pharmacol. Phys. 2007, 20, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Technical Guidance Document on Risk Assessment. Available online: http://enfo.agt.bme.hu/drupal/en/etanfolyam/11424 (accessed on 1 July 2015).
- Lademann, J.; Patzelt, A.; Richter, H.; Lademann, O.; Baier, G.; Breucker, L.; Landfester, K. Nanocapsules for drug delivery through the skin barrier by tissue-tolerable plasma. Laser Phys. Lett. 2013, 10. [Google Scholar] [CrossRef]
- Kojima, K.; Murakami, M.; Yoshimizu, C.; Tayasu, I.; Nagata, T.; Furumai, H. Evaluation of surface runoff and road dust as sources of nitrogen using nitrate isotopic composition. Chemosphere 2011, 84, 1716–1722. [Google Scholar] [CrossRef] [PubMed]
- World Environment Day—Food Waste Facts. Available online: http://www.unep.org/wed/quickfacts/ (accessed on 1 July 2015).
- Loretz, L.; Api, A.M.; Barraj, L.; Burdick, J.; Davis, D.A.; Dressler, W.; Gilberti, E.; Jarrett, G.; Mann, S.; Laurie Pan, Y.H.; et al. Exposure data for personal care products: hairspray, spray perfume, liquid foundation, shampoo, body wash, and solid antiperspirant. Food Chem. Toxicol. 2006, 44, 2008–2018. [Google Scholar] [CrossRef] [PubMed]
- Purest colloids: Colloidal Skin Conditioners by Purest Colloids. Available online: https://www.purestcolloids.com/colloidal-skin-conditioners.php (accessed on 1 July 2015).
- Müller, R.H.; Petersen, R.D.; Hommoss, A.; Pardeike, J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv. Drug Deliver. Rev. 2007, 59, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Hyaluronic Acid Serum. Available online: http://www.ebay.com.au/bhp/hyaluronic-acid-serum (accessed on 1 July 2015).
- Taufikurohmah, T.; Sanjaya, I.G.M.; Syahrani, A. Nanogold synthesis using matrix mono glyceryl stearate as antiaging compounds in modern cosmetics. J. Mater. Sci. Eng. A 2011, 1, 857–864. [Google Scholar]
- Tiede, K.; Westerhoff, P.; Hansen, S.F.; Fern, G.J.; Hankin, S.M.; Aitken, R.J.; Chaudhry, Q.; Boxall, A.B.A. Review of the Risks Posed to Drinking Water by Man-Made Nanoparticles. Available online: http://forskningsbasen.deff.dk/Share.external?sp=S9b311d78-5049-46a3-9a87-a6a35a4c76f6&sp=Sdtu (accessed on 1 July 2015).
- Wik, A.; Dave, G. Environmental labeling of car tires––Toxicity to Daphnia magna can be used as a screening method. Chemosphere 2005, 58, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Permitted Colouring Agents—Lists of Permitted Food Additives. Available online: http://www.hc-sc.gc.ca/fn-an/securit/addit/list/3-colour-color-eng.php (accessed on 1 July 2015).
- Opinion of the Scientific Panel on Food Additives, Flavorings, Processing Aids and Materials in Contact With Food on A Request From the Commission Related to the Safety in Use of Rutile Titanium Dioxide As An Alternative to the Presently Permitted Anat. Available online: http://www.w3.org/1999/xlink" xlink:href="forskningsbasen.deff.dk/Share.external?sp=Saa52aaa0-91b2-4b1d-98c9-85e56238e6a1&sp=Sdtu (accessed on 1 July 2015).
- Biesterbos, J.W.H.; Dudzina, T.; Delmaar, C.J.E.; Bakker, M.I.; Russel, F.G.M.; von Goetz, N.; Scheepers, P.T.J.; Roeleveld, N. Usage patterns of personal care products: important factors for exposure assessment. Food Chem. Toxicol. 2013, 55, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Hall, B.; Tozer, S.; Safford, B.; Coroama, M.; Steiling, W.; Leneveu-Duchemin, M.C.; McNamara, C.; Gibney, M. European consumer exposure to cosmetic products, a framework for conducting population exposure assessments. Food Chem. Toxicol. 2007, 45, 2097–2108. [Google Scholar] [CrossRef] [PubMed]
- Loretz, L.J.; Api, A.M.; Barraj, L.M.; Burdick, J.; Dressler, W.E.; Gettings, S.D.; Han Hsu, H.; Pan, Y.H.L.; Re, T.A.; Renskers, K.J.; et al. Exposure data for cosmetic products: lipstick, body lotion, and face cream. Food Chem. Toxicol. 2005, 43, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Loretz, L.J.; Api, A.M.; Babcock, L.; Barraj, L.M.; Burdick, J.; Cater, K.C.; Jarrett, G.; Mann, S.; Pan, Y.H.L.; Re, T.A.; et al. Exposure data for cosmetic products: facial cleanser, hair conditioner, and eye shadow. Food Chem. Toxicol. 2008, 46, 1516–1524. [Google Scholar] [CrossRef] [PubMed]
- Exposure Factors Handbook. Available online: http://www.osti.gov/scitech/biblio/5192412 (accessed on 1 July 2015).
- Gehrig, R.; Hill, M.; Buchmann, B.; Imhof, D.; Weingartner, E.; Baltensperger, U. Separate determination of PM10 emission factors of road traffic for tailpipe emissions and emissions from abrasion and resuspension processes. Int. J. Environ. Pollut. 2005, 22. [Google Scholar] [CrossRef]
- S’poreans Drinking More Coffee: The Malaysian Times, 2013. Available online: http://www.themalaysiantimes.com.my/sporeans-drinking-more-coffee/ (accessed on 1 July 2015).
- WHO expert consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004, 363, 157–163. [Google Scholar]
- Deurenberg, P.; Bhaskaran, K.; Lim, P.; Lian, K. Singaporean Chinese adolescents have more subcutaneous adipose tissue than Dutch Caucasians of the same age and body mass index. ASA Pac. J. Clin. Nutr. 2003, 12, 261–265. [Google Scholar]
- Carlsen, M.H.; Blomhoff, R.; Andersen, L.F. Intakes of culinary herbs and spices from a food frequency questionnaire evaluated against 28-days estimated records. Nutr. J. 2011, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Walser, T.; Limbach, L.K.; Brogioli, R.; Erismann, E.; Flamigni, L.; Hattendorf, B.; Juchli, M.; Krumeich, F.; Ludwig, C.; Prikopsky, K.; et al. Persistence of engineered nanoparticles in a municipal solid-waste incineration plant. Nat. Nanotechnol. 2012, 7, 520–524. [Google Scholar] [CrossRef] [PubMed]
- National Population and Talent Division: Singapore Department of Statistics. Available online: http://www.nptd.gov.sg/portals/0/news/population-in-brief-2014.pdf (accessed on 1 July 2015).
- Local Catchment Water Singapore. Available online: http://www.pub.gov.sg/water/Pages/LocalCatchment.aspx (accessed on 1 July 2015).
- ABC Waters Programme Singapore. Available online: http://www.pub.gov.sg/abcwaters/Pages/default.aspx (accessed on 1 July 2015).
- Kaegi, R.; Voegelin, A.; Sinnet, B.; Zuleeg, S.; Hagendorfer, H.; Burkhardt, M.; Siegrist, H. Behavior of metallic silver nanoparticles in a pilot wastewater treatment plant. Environ. Sci. Technol. 2011, 45, 3902–3908. [Google Scholar] [CrossRef] [PubMed]
- Benn, T.; Wang, Y.; Hristovski, K. Titanium nanomaterial removal and release from wastewater treatment plants. Environ. Sci. Technol. 2009, 43, 6757–6763. [Google Scholar]
- Hou, L.; Xia, J.; Li, K.; Chen, J.; Wu, X.; Li, X. Removal of ZnO nanoparticles in simulated wastewater treatment processes and its effects on COD and NH(4)(+)–N reduction. Water Sci. Technol. 2013, 67, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, R.; Smeraldi, J.; Hosseini, T.; Khatib, L.; Olson, B.H.; Rosso, D. Evaluation of nanocopper removal and toxicity in municipal wastewaters. Environ. Sci. Technol. 2010, 44, 7808–7813. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Leu, Y.-R.; Aitken, R.J.; Riediker, M. Inventory of Engineered Nanoparticle-Containing Consumer Products Available in the Singapore Retail Market and Likelihood of Release into the Aquatic Environment. Int. J. Environ. Res. Public Health 2015, 12, 8717-8743. https://doi.org/10.3390/ijerph120808717
Zhang Y, Leu Y-R, Aitken RJ, Riediker M. Inventory of Engineered Nanoparticle-Containing Consumer Products Available in the Singapore Retail Market and Likelihood of Release into the Aquatic Environment. International Journal of Environmental Research and Public Health. 2015; 12(8):8717-8743. https://doi.org/10.3390/ijerph120808717
Chicago/Turabian StyleZhang, Yuanyuan, Yu-Rui Leu, Robert J. Aitken, and Michael Riediker. 2015. "Inventory of Engineered Nanoparticle-Containing Consumer Products Available in the Singapore Retail Market and Likelihood of Release into the Aquatic Environment" International Journal of Environmental Research and Public Health 12, no. 8: 8717-8743. https://doi.org/10.3390/ijerph120808717
APA StyleZhang, Y., Leu, Y.-R., Aitken, R. J., & Riediker, M. (2015). Inventory of Engineered Nanoparticle-Containing Consumer Products Available in the Singapore Retail Market and Likelihood of Release into the Aquatic Environment. International Journal of Environmental Research and Public Health, 12(8), 8717-8743. https://doi.org/10.3390/ijerph120808717





