The Effect of Improved Water Supply on Diarrhea Prevalence of Children under Five in the Volta Region of Ghana: A Cluster-Randomized Controlled Trial
Abstract
:1. Introduction
2. Methods
2.1. Study Setting
2.2. Implementation of Intervention
2.3. Study Design
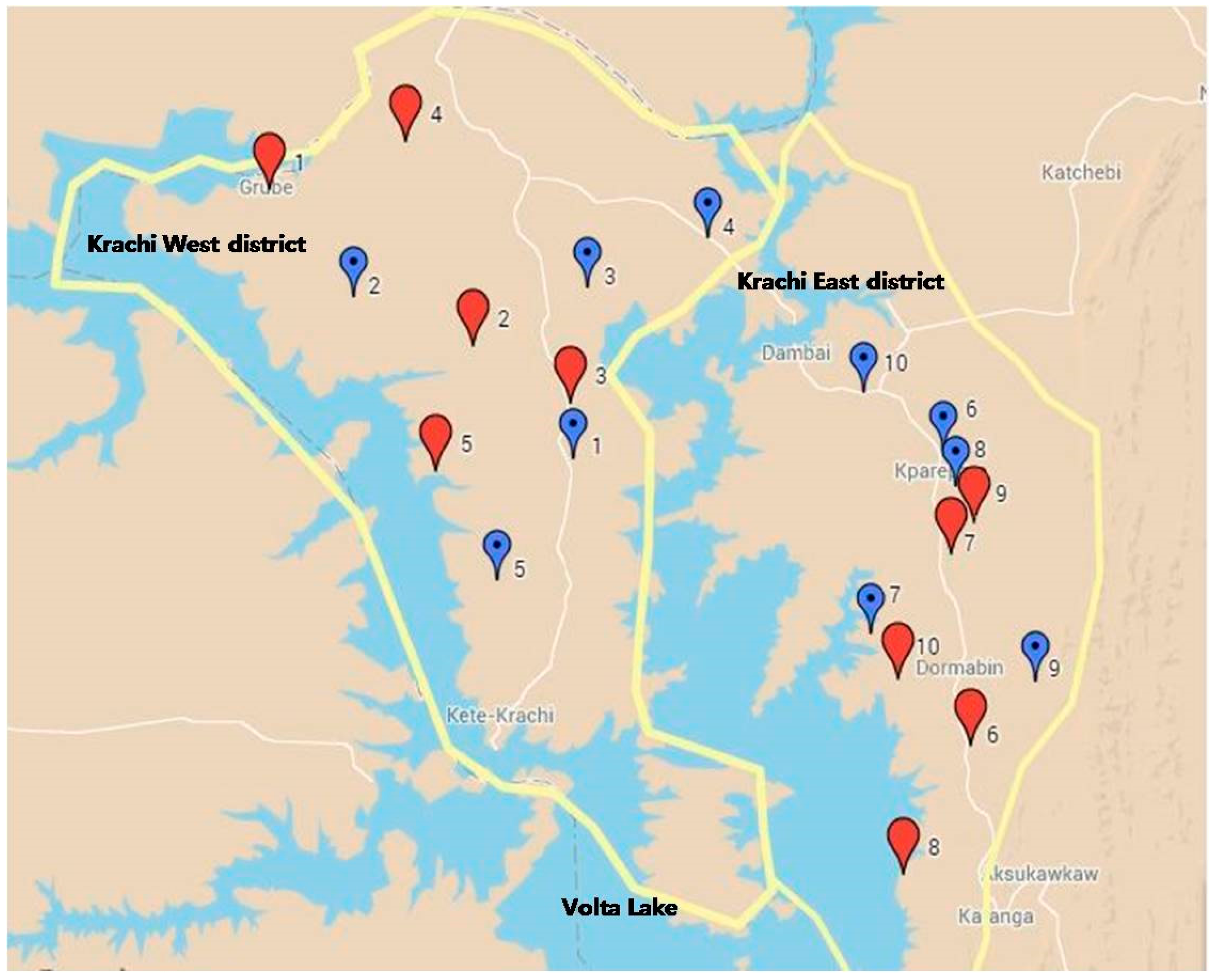
2.4. Randomization and Eligibility Criteria
2.5. Primary End Point
2.6. Sample Size Calculation
2.7. Data Collection
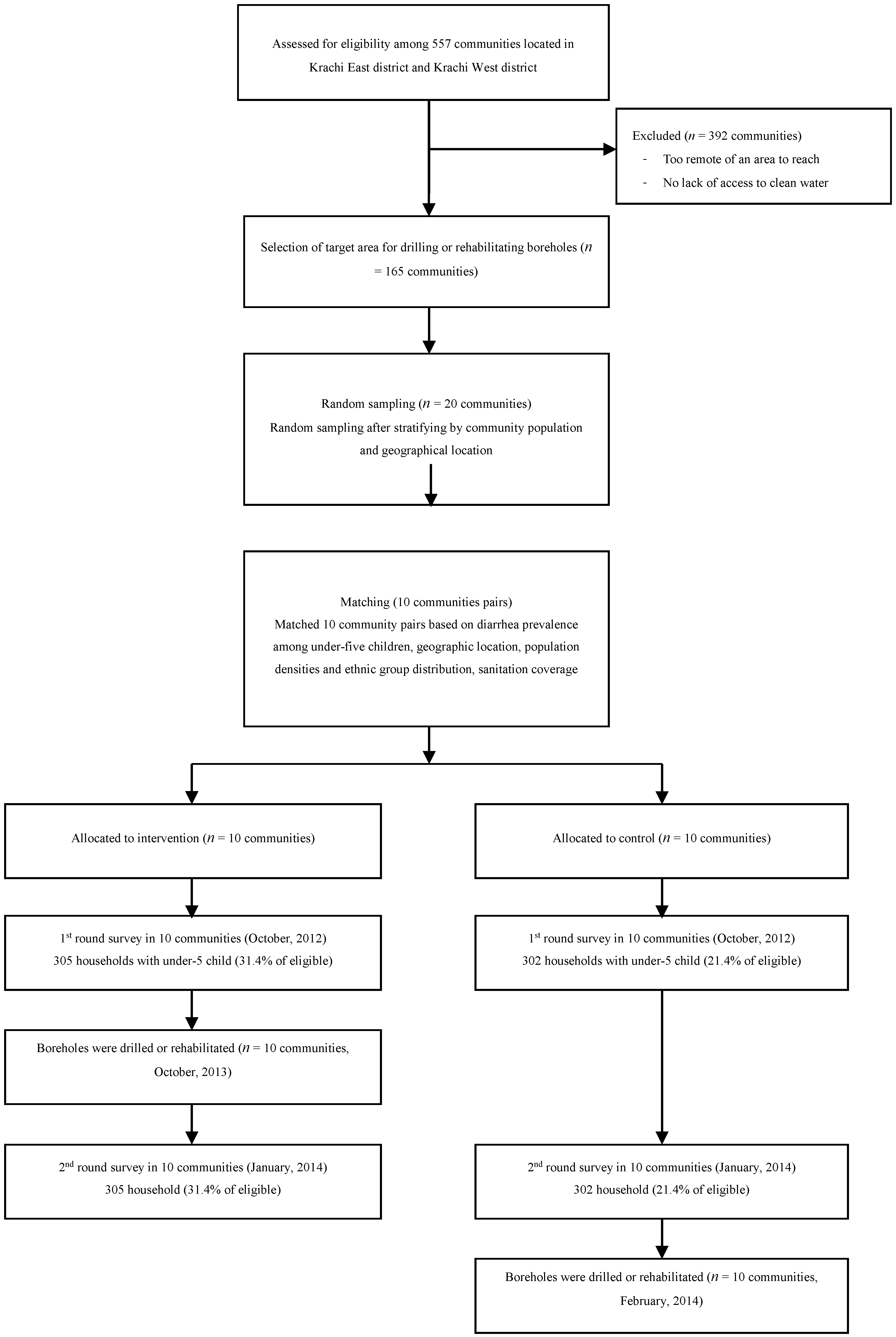
3. Results
3.1. Baseline Characteristics
3.2. Hand-Washing Practices after the Intervention
3.3. Diarrhea Prevalence in Intervention and Control Communities
| Variables | % Or Unit (Standard Deviation) | |
|---|---|---|
| Control | Intervention | |
| Main Indicator | ||
| Prevalence of diarrhea | 31.30% | 27.90% |
| Age of head of household (years) | 44.65 (14.97) | 44.14 (14.61) |
| Level of Education | ||
| Not educated or primary level not completed | 60.10% | 62.80% |
| Completed more than primary school | 39.90% | 37.20% |
| No. of children under five | 1.64 (1.33) | 1.48 (1.32) |
| Household expenditures per month (US $) | 38.26 (36.82) | 39.89 (43.95) |
| Household income per month (US $) | 50.10 (48.49) | 60.70 (94.49) |
| Sanitation | ||
| Household toilet | 16.90% | 9.60% |
| Open defecation | 56.70% | 53.70% |
| Other (neighbor’s latrine, communal latrine) | 26.4% | 36.7% |
| Water Storage | ||
| Storage tank | 7.50% | 4.70% |
| Barrel | 67.80% | 70.80% |
| Basin | 7.50% | 10.10% |
| Bucket | 2.00% | 1.00% |
| Gallon | 8.20% | 5.70% |
| Container Type | ||
| Containers with lid, cover | 68.20% | 71.30% |
| Containers with tap | 0.30% | 0.70% |
| Narrow mouth (<10 cm), uncovered | 4.80% | 4.00% |
| Narrow mouth (>10 cm), uncovered | 29.30% | 30.60% |
| Average storage days | 2.28 (2.10) | 2.02 (1.46) |
| Total quantity of water per day per person (L) | 22.61 (17.12) | 19.52 (12.85) |
| Water treatment (no treatment) | 88.00% | 90.40% |
| Hand Washing Practice | ||
| Before eating | 94.80% | 94.20% |
| After defecation | 90.90% | 87.80% |
| Before food preparation | 34.80% | 32.50% |
| After cleaning a child’s buttocks | 7.00% | 7.50% |
| Before feeding a child | 2.80% | 2.70% |
| After handling a sick person | 3.10% | 5.40% |
| After returning from a social gathering | 8.70% | 7.90% |
| Washing with soap | 96.60% | 97.00% |
| Knowledge of diarrhea | 65.60% | 61.00% |
| Hand Washing Practices | Control Group | Intervention Group | p Value | ||
|---|---|---|---|---|---|
| Baseline | After Intervention | Baseline | After Intervention | ||
| Before eating | 94.80% | 74.40% | 94.20% | 74.50% | 0.971 |
| After defecation | 90.90% | 89.00% | 87.80% | 85.60% | 0.225 |
| Before food preparation | 34.80% | 40.90% | 32.50% | 44.10% | 0.435 |
| After cleaning a child’s buttocks | 7.00% | 23.10% | 7.50% | 21.90% | 0.720 |
| Before feeding a child | 2.80% | 19.90% | 2.70% | 13.70% | 0.044 |
| After handling a sick person | 3.10% | 4.00% | 5.40% | 4.70% | 0.675 |
| After returning from a social gathering | 8.70% | 8.90% | 7.90% | 8.50% | 0.853 |
| Washing with soap | 96.60% | 96.20% | 97.00% | 97.10% | 0.530 |
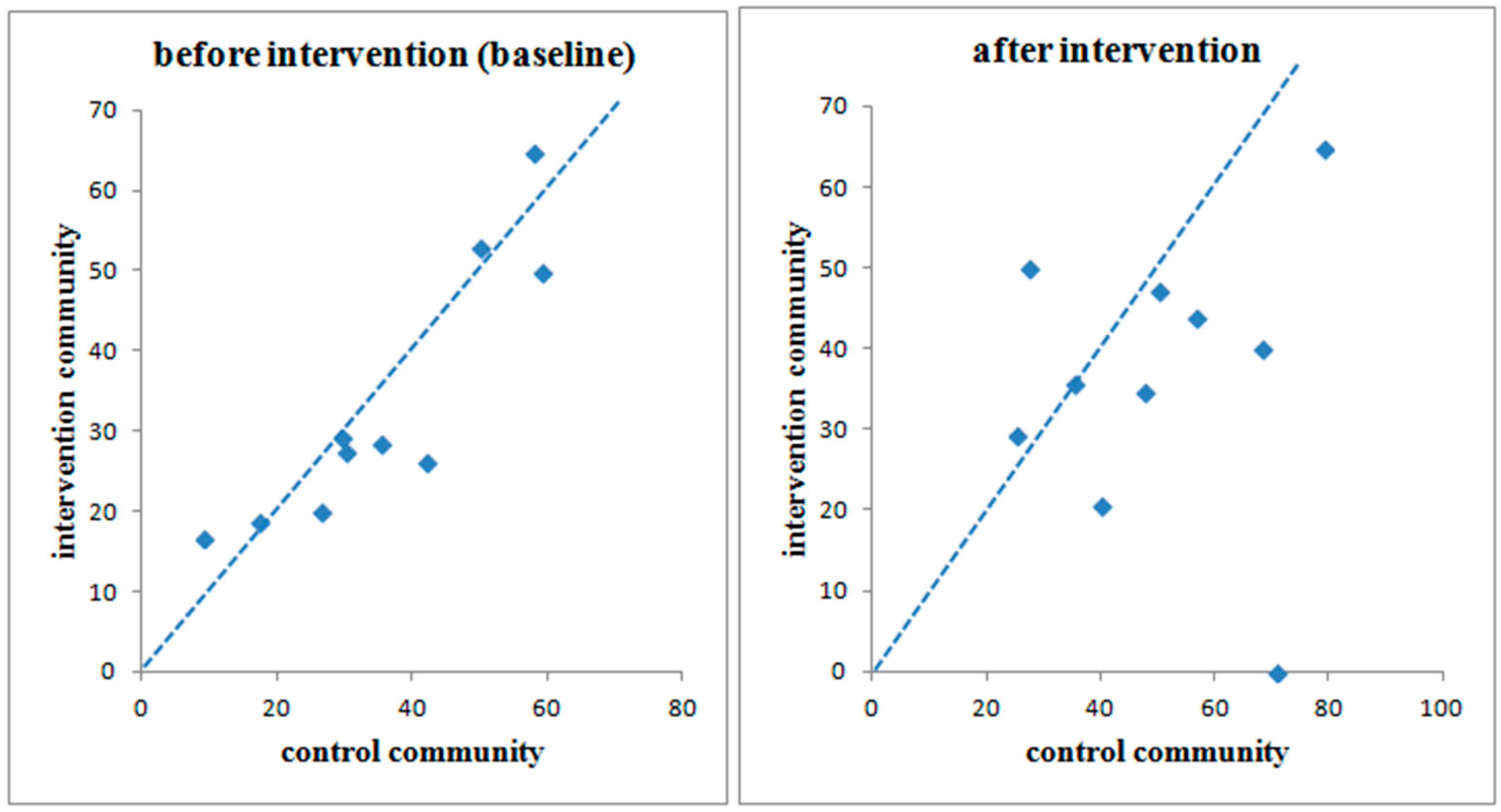
3.4. Impact on Diarrhea Prevalence Following the Intention-to-Treat Analysis Method
| District | Paired Communities | Baseline | After Intervention | Result (Crude) | Result (Adjusted 3) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pair | Control | Intervention | Control | Intervention | Control | Intervention | PR 1 | CI 2 | PR | CI | |
| Krachi West | pair 1 | Grubi | Ntewusae | 20/76 (26.3%) | 15/75 (20.0%) | 43/76 (56.6%) | 33/75 (44.0%) | 0.88 | 0.76–1.03 | 0.81 | 0.68–0.96 |
| pair 2 | Kpollo | Kaliako | 11/19 (57.9%) | 11/17 (64.7%) | 15/19 (78.9%) | 11/17 (64.7%) | 0.92 | 0.48–1.76 | 0.90 | 0.44–1.84 | |
| pair 3 | Ankaase | Bakam | 8/16 (50.0%) | 9/17 (52.9%) | 8/16 (50.0%) | 8/17 (47.1%) | 1.00 | 0.62–1.62 | 1.00 | 0.62–1.62 | |
| pair 4 | Majimaji | Papaye | 13/22 (59.1%) | 10/20 (50.0%) | 15/22 (68.2%) | 8/20 (40.0%) | 0.66 | 0.41–1.07 | 0.64 | 0.40–1.04 | |
| pair 5 | Shitor Kope | Gyeasayor | 8/19 (42.1%) | 6/23 (26.1%) | 9/19 (47.4%) | 8/23 (34.8%) | 0.79 | 0.56–1.12 | 0.70 | 0.45–1.09 | |
| Subtotal | 60/152 (39.5%) | 51/152 (33.6%) | 90/152 (59.2%) | 68/152 (44.7%) | 0.85 | 0.74–0.97 | 0.82 | 0.71–0.96 | |||
| Krachi East | pair 6 | Tokurano Attafie | Kparekpare | 13/75 (17.3%) | 14/75 (18.7%) | 19/75 (25.3) | 22/75 (29.3) | 1.03 | 0.92–1.16 | 0.99 | 0.89–1.09 |
| pair 7 | Nwane Akura | Tsikatakope | 9/30 (30.0%) | 8/29 (27.6%) | 12/30 (40.0) | 6/29 (20.7) | 0.86 | 0.68–1.08 | 0.83 | 0.65–1.07 | |
| pair 8 | Adokwata Tornu | Okuma Akura | 6/17 (35.3%) | 4/14 (28.6%) | 6/17 (35.3) | 5/14 (35.7) | 0.95 | 0.67–1.36 | 1.00 | 0.69–1.46 | |
| pair 9 | Abongo Akura | Katafua Junction | 5/17 (29.4%) | 5/17 (29.4%) | 12/17 (70.6) | 0/17 (0.00) | 0.53 | 0.36–0.77 | 0.53 | 0.36–0.77 | |
| pair 10 | Atsigode Kope | Kwame Akura | 1/11 (9.1%) | 3/18 (16.7%) | 3/11 (27.3) | 9/18 (50.0) | 1.15 | 0.89–1.48 | 1.15 | 0.89–1.48 | |
| Subtotal | 34/150 (22.7%) | 34/153 (22.2%) | 52/150 (34.7%) | 42/153 (27.5%) | 0.96 | 0.87–1.05 | 0.95 | 0.86–1.04 | |||
| Total | 94/302 (31.1%) | 85/305 (27.9%) | 142/302 (47.0%) | 110/305 (36.1%) | 0.91 | 0.83–0.98 | 0.89 | 0.82–0.97 | |||
3.5. Compliance Rate
3.6. Per-Protocol and As-Treated Analyses
| District | Pair | Compliance | Diarrhea Prevalence | Per-Protocol | As Treated | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Intervention | Control | Intervention | ||||||||||
| Name | NO 1 | Name | YES 2 | Access | No | Access | No | PR 3 | CI 4 | PR | CI | ||
| Krachi West | pair 1 | Grubi | 44/76 (57.9%) | Ntewusae | 70/75 (93.3%) | 13/32 (40.6%) | 30/44 (68.2%) | 32/70 (45.7%) | 1/5 (20.0%) | 0.86 | 0.73–1.02 | 0.86 | 0.74–1.01 |
| pair 2 | Kpollo | 19/19 (100%) | Kaliako | 13/17 (76.5%) | - | 15/19 (78.9%) | 8/13 (61.5%) | 3/4 (75.0%) | 0.97 | 0.49–1.93 | 0.90 | 0.46–1.74 | |
| pair 3 | Ankaase | 1/16 (6.3%) | Bakam | 17/17 (100%) | 8/15 (53.3%) | 0/1 (0.0%) | 8/17 (47.1%) | - | 1.44 | 0.80–2.60 | 1.21 | 0.65–2.27 | |
| pair 4 | Majimaji | 16/22 (72.7%) | Papaye | 20/20 (100%) | 4/6 (66.7%) | 11/16 (68.8%) | 8/20 (40.0%) | - | 0.67 | 0.41–1.11 | 0.70 | 0.43–1.16 | |
| pair 5 | Shitor Kope | 19/19 (100%) | Gyeasayor | 23/23 (100%) | - | 9/19 (47.4%) | 8/23 (34.8%) | - | 0.79 | 0.56–1.12 | 0.79 | 0.56–1.12 | |
| Subtotal | 99/152 (65.1%) | 143/152 (94.1%) | 25/53 (47.17%) | 65/99 (65.7%) | 64/143 (44.8%) | 4/9 (44.44%) | 0.82 | 0.71–0.96 | 0.83 | 0.72–0.96 | |||
| Krachi East | pair 6 | Tokurano Attafie | 56/75 (74.7%) | Kparekpare | 61/75 (81.3%) | 3/19 (15.8%) | 16/56 (28.6%) | 17/61 (27.9%) | 5/14 (35.7%) | 1.01 | 0.89–1.14 | 0.99 | 0.87–1.11 |
| pair 7 | Nwane Akura | 2/30 (6.7%) | Tsikatakope | 23/29 (79.3%) | 10/28 (35.7%) | 2/2 (100%) | 5/23 (21.7%) | 1/6 (16.7%) | 0.87 | 0.63–1.21 | 0.94 | 0.71–1.25 | |
| pair 8 | Adokwata Tornu | 14/17 (82.4%) | Okuma Akura | 7/14 (50.0%) | 1/3 (33.3%) | 5/14 (35.7%) | 2/7 (28.6%) | 3/7 (42.9%) | 0.90 | 0.62–1.32 | 0.90 | 0.63–1.28 | |
| pair 9 | Abongo Akura | 17/17 (100%) | Katafua Junction | 11/17 (64.7%) | - | 12/17 (70.6%) | 0/11 (0.0%) | 0/6 (0.0%) | 0.53 | 0.36–0.77 | 0.60 | 0.45–0.81 | |
| pair10 | Atsigode Kope | 9/11 (81.8%) | Kwame Akura | 12/18 (66.7%) | 0/5 (0.0%) | 3/6 (50.0%) | 6/12 (50.0%) | 3/6 (50.0%) | 1.08 | 0.83–1.42 | 1.03 | 0.80–1.33 | |
| Subtotal | 98/150 (65.3%) | 114/153 (74.5%) | 14/55 (25.45%) | 38/95 (40.0%) | 30/114 (26.3%) | 12/39 (30.77%) | 0.94 | 0.85–1.04 | 0.93 | 0.85–1.03 | |||
| Total | 197/302 (65.2%) | 257/305 (84.3%) | 39/108 (36.11%) | 103/194 (53.1%) | 94/257 (36.6%) | 16/48 (33.33%) | 0.89 | 0.81–0.97 | 0.90 | 0.83–0.98 | |||
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO/UNICEF. Progress on Sanitation and Drinking Water: 2015 Update and MDG Assessment; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Liu, L.; Oza, S.; Hogan, D.; Perin, J.; Rudan, I.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of child mortality in 2000–2013, with projections to inform post-2015 priorities: An updated systematic analysis. Lancet 2015, 385, 430–440. [Google Scholar] [CrossRef]
- WHO. Ghana Statistics Profile; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- GHS. Ghana Demographic and Health Survey 2008; Ghana Health Service: Accra, Republic of Ghana, 2009. [Google Scholar]
- Kirchhoff, L.V.; McClelland, K.E.; do Carmo Pinho, M.; Araujo, J.G.; de Sousa, M.A.; Guerrant, R.L. Feasibility and efficacy of in-home water chlorination in rural North eastern Brazil. J. Hyg. 1985, 94, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Mahfouz, A.A.; Abdel-Moneim, M.; al-Erian, R.A.; al-Amari, O.M. Impact of chlorination of water in domestic storage tanks on childhood diarrhoea: A community trial in the rural areas of Saudi Arabia. J. Trop. Med. Hyg. 1995, 98, 126–130. [Google Scholar] [PubMed]
- Conroy, R.M.; Elmore-Meegan, M.; Joyce, T.; McGuigan, K.G.; Barnes, J. Solar disinfection of drinking water and diarrhoea in Maasai children: A controlled field trial. Lancet 1996, 348, 1695–1697. [Google Scholar] [CrossRef]
- Sathe, A.A.; Hinge, D.V.; Watve, M.G. Water treatment and diarrhoea. Lancet 1996, 348, 335–336. [Google Scholar] [CrossRef]
- Xiao, S.; Lin, C.; Chen, K. Evaluation of effectiveness of comprehensive control for diarrhea diseases in rural areas of East Fujian and analysis of its cost-benefit. Zhonghua Yu Fang Yi Xue Za Zhi 1997, 31, 40–41. (In Chinese) [Google Scholar] [PubMed]
- Semenza, J.C.; Roberts, L.; Henderson, A.; Bogan, J.; Rubin, C.H. Water distribution system and diarrheal disease transmission: A case study in Uzbekistan. Am. J. Trop. Med. Hyg. 1998, 59, 941–946. [Google Scholar] [PubMed]
- Quick, R.E.; Venczel, L.; Mintz, E.; Soleto, L.; Aparicio, J.; Gironaz, M.; Hutwagner, L.; Greene, K.; Bopp, C.; Maloney, K. Diarrhoea prevention in bolivia through point-of-use water treatment and safe storage: A promising new strategy. Epidemiol. Infect. 1999, 122, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Sobsey, M.D.; Handzel, T.; Venczel, L. Chlorination and safe storage of household drinking water in developing countries to reduce waterborne disease. Water Sci. Technol. 2003, 47, 221–228. [Google Scholar] [PubMed]
- Iijima, Y.; Karama, M.; Oundo, J.O.; Honda, T. Prevention of bacterial diarrhea by pasteurization of drinking water in Kenya. Microbiol. Immunol. 2001, 45, 413–416. [Google Scholar] [CrossRef] [PubMed]
- Quick, R.E.; Kimura, A.; Thevos, A.; Tembo, M.; Shamputa, I.; Hutwagner, L.; Mintz, E. Diarrhea prevention through household level water disinfection and safe storage in Zambia. Am. J. Trop. Med. Hyg. 2002, 66, 584–589. [Google Scholar] [PubMed]
- Colwell, R.R.; Huq, A.; Islam, M.S.; Aziz, K.; Yunus, M.; Khan, N.H.; Mahmud, A.; Sack, R.B.; Nair, G.; Chakraborty, J. Reduction of cholera in Bangladeshi villages by simple filtration. Proc. Natl. Acad. Sci. USA. 2003, 100, 1051–1055. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, L.; Kaufmann, R.B.; Kay, D.; Enanoria, W.; Haller, L.; Colford, J.M. Water, sanitation, and hygiene interventions to reduce diarrhoea in less developed countries: A systematic review and meta-analysis. Lancet. Infect. Dis. 2005, 5, 42–52. [Google Scholar] [CrossRef]
- Clasen, T.; Schmidt, W.P.; Rabie, T.; Roberts, I.; Cairncross, S. Interventions to improve water quality for preventing diarrhea: Systematic review and meta-analysis. BMJ 2007, 334. [Google Scholar] [CrossRef] [PubMed]
- Cairncross, S.; Hunt, C.; Boisson, S.; Bostoen, K.; Curtis, V.; Fung, I.C.; Schmidt, W.-P. Water, sanitation and hygiene for the prevention of diarrhoea. Int. J. Epidemiol. 2010, 39, i193–i205. [Google Scholar] [CrossRef] [PubMed]
- Wolf, J.; Prüss-Ustün, A.; Cumming, O.; Bartram, J.; Bonjour, S.; Cairncross, S.; Clasen, T.; Colford, J.M.; Curtis, V.; France, J. Assessing the impact of drinking water and sanitation on diarrhoeal disease in low-and middle-income settings: Systematic review and meta-regression. Trop. Med. Int. Heahlth 2014, 19, 928–942. [Google Scholar] [CrossRef] [PubMed]
- Bahl, M.R. Impact of piped water supply on the incidence of typhoid fever and diarrheal diseases in Lusaka. Med. J. Zambia 1976, 10, 98–99. [Google Scholar] [PubMed]
- Esrey, S.A.; Habicht, J.P.; Latham, M.C.; Sisler, D.G.; Casella, G. Drinking water source, diarrheal morbidity, and child growth in villages with both traditional and improved water supplies in rural Lesotho, Southern Africa. Am. J. Public Health 1988, 78, 1451–1455. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shepard, D.; Zhu, Y.; Cash, R.A.; Zhao, R.; Zhu, Z.; Shen, F. Reduction of enteric infectious disease in rural china by providing deep-well tap water. Bull. WHO 1989, 67, 171–180. [Google Scholar] [PubMed]
- Tonglet, R.; Isu, K.; Mpese, M.; Dramix, M.; Hennart, P. Can improvements in water supply reduce childhood diarrhea? Health Policy Pan. 1992, 7, 260–68. [Google Scholar] [CrossRef]
- Shrestha, S.; Aihara, Y.; Yoden, K.; Yamagata, Z.; Nishida, K.; Kondo, N. Access to improved water and its relationship with diarrhoea in Kathmandu Valley, Nepal: A cross-sectional study. BMJ Open 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Teklemariam, S.; Getaneh, T.; Bekele, F. Environmental determinants of diarrheal morbidity in under-five children, Keffa-Sheka zone, South West Ethiopia. Ethiop. Med. J. 2000, 38, 27–34. [Google Scholar] [PubMed]
- Tumwine, J.K.; Thompson, J.; Katua-Katua, M.; Mujwajuzi, M.; Johnstone, N.; Wood, E.; Porras, I. Diarrhoea and effects of different water sources, sanitation and hygiene behaviour in East Africa. Trop. Med. Int. Health 2002, 7, 750–756. [Google Scholar] [CrossRef] [PubMed]
- Escamilla, V.; Wagner, B.; Yunus, M.; Streatfield, P.K.; Geen, A.; Emch, M. Effect of deep tube well use on childhood diarrhoea in Bangladesh. Bull. WHO 2011, 89, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Plate, D.K.; Strassmann, B.I.; Wilson, M.L. Water sources are associated with childhood diarrhea prevalence in rural East-Central Mali. Trop. Med. Int. Health 2004, 9, 416–425. [Google Scholar] [CrossRef] [PubMed]
- World Vision Ghana. Krachi East Baseline Survey Result Report; Krachi East Area Development Progarmme Office of World Vision: Krachi East District, Ghana, 2008. [Google Scholar]
- World Vision Ghana. Krachi West Baseline Survey Result Report; Krachi West Area Development Progarmme Office of World Vision Ghana: Krachi West District, Ghana, 2008. [Google Scholar]
- Hayes, R.J.; Bennett, S. Simple sample size calculation for cluster-randomized trials. Int. J. Epidemiol. 1999, 28, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Zou, G. A modified poisson regression approach to prospective studies with binary data. Am. J. Epidemiol. 2004, 159, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Sussman, J.B.; Hayward, R.A. An IV for the RCT: Using instrumental variables to adjust for treatment contamination in randomized controlled trials. BMJ 2010, 340. [Google Scholar] [CrossRef] [PubMed]
- Byass, P.; Hanlon, P.W. Daily morbidity records: Recall and reliability. Int. J. Epidemiol. 1994, 23, 757–63. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W.-P.; Sophie, B.; Bernd, G.; Stephen, P.L.; Mauricio, L.B. Epidemiological methods in diarrhoea studies—An update. Int. J. Epidemiol. 2011, 40, 1678–1692. [Google Scholar] [CrossRef] [PubMed]
- Luby, S.P.; Agboatwalla, M.; Hoekstra, R.M. The variability of childhood diarrhea in Karachi, Pakistan, 2002–2006. Am. J. Trop. Med. Hyg. 2011, 84, 870–77. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W.-P.; Genser, B.; Barreto, M.L.; Clasen, T.; Luby, S.P.; Cairncross, S. Sampling strategies to measure the prevalence of common recurrent infections in longitudinal studies. Emerg. Themes. Epidemiol. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cha, S.; Kang, D.; Tuffuor, B.; Lee, G.; Cho, J.; Chung, J.; Kim, M.; Lee, H.; Lee, J.; Oh, C. The Effect of Improved Water Supply on Diarrhea Prevalence of Children under Five in the Volta Region of Ghana: A Cluster-Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2015, 12, 12127-12143. https://doi.org/10.3390/ijerph121012127
Cha S, Kang D, Tuffuor B, Lee G, Cho J, Chung J, Kim M, Lee H, Lee J, Oh C. The Effect of Improved Water Supply on Diarrhea Prevalence of Children under Five in the Volta Region of Ghana: A Cluster-Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2015; 12(10):12127-12143. https://doi.org/10.3390/ijerph121012127
Chicago/Turabian StyleCha, Seungman, Douk Kang, Benedict Tuffuor, Gyuhong Lee, Jungmyung Cho, Jihye Chung, Myongjin Kim, Hoonsang Lee, Jaeeun Lee, and Chunghyeon Oh. 2015. "The Effect of Improved Water Supply on Diarrhea Prevalence of Children under Five in the Volta Region of Ghana: A Cluster-Randomized Controlled Trial" International Journal of Environmental Research and Public Health 12, no. 10: 12127-12143. https://doi.org/10.3390/ijerph121012127
APA StyleCha, S., Kang, D., Tuffuor, B., Lee, G., Cho, J., Chung, J., Kim, M., Lee, H., Lee, J., & Oh, C. (2015). The Effect of Improved Water Supply on Diarrhea Prevalence of Children under Five in the Volta Region of Ghana: A Cluster-Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 12(10), 12127-12143. https://doi.org/10.3390/ijerph121012127





