The Association between Season of Pregnancy and Birth-Sex among Chinese
Abstract
:1. Introduction
2. Methods
2.1. Participants
2.2. Statistic
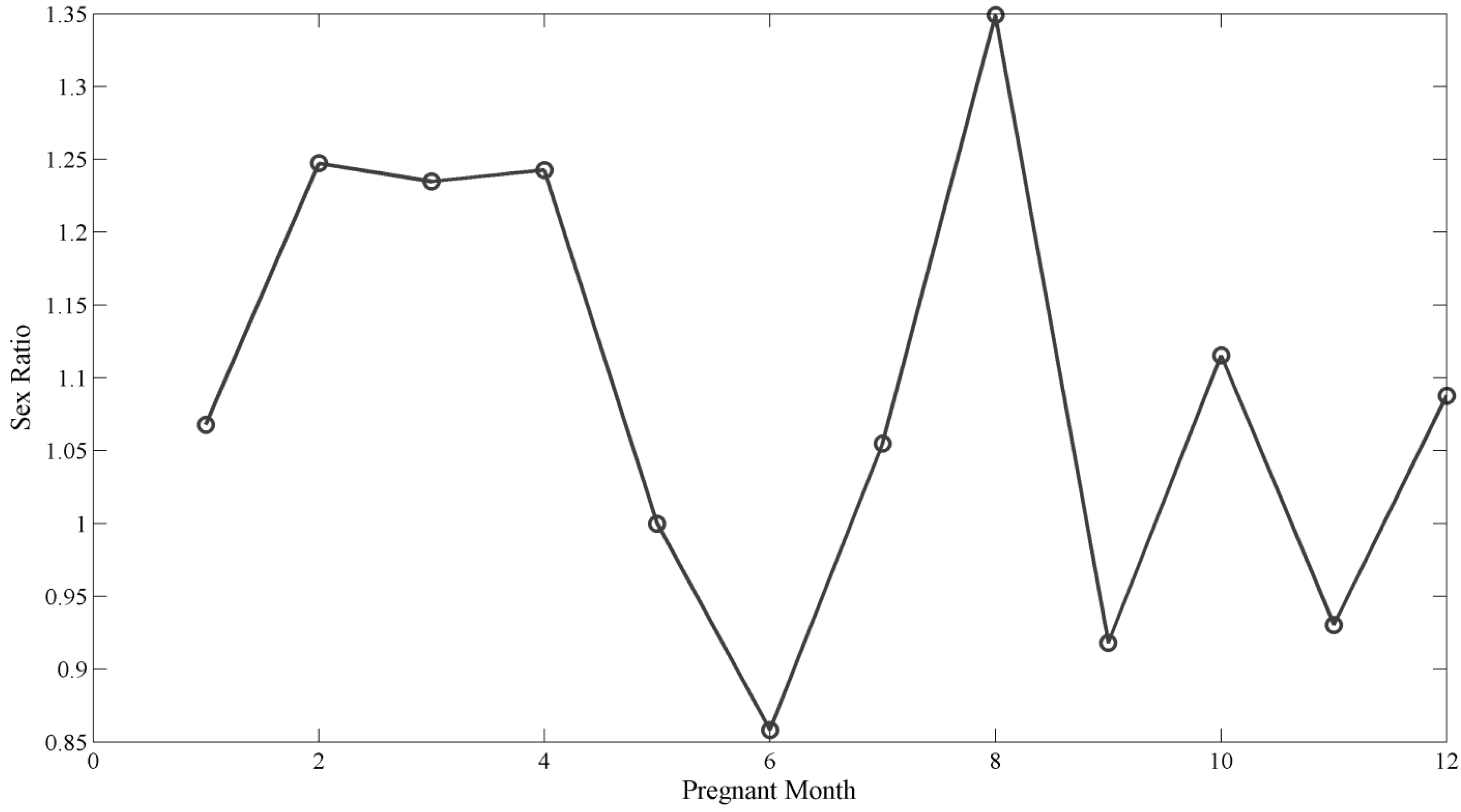
3. Results
| Characteristic | Male | Female | Sex Ratio (M/F) | p-value |
|---|---|---|---|---|
| Father Age (Year) | ||||
| ≥30 | 425 | 435 | 0.977 | 0.0999 |
| <30 | 1157 | 1034 | 1.119 | |
| Mother Age | ||||
| ≥30 | 305 | 290 | 1.052 | 0.7825 |
| <30 | 1277 | 1179 | 1.083 | |
| Region | ||||
| City | 1057 | 974 | 1.085 | 0.7946 |
| Rural | 525 | 495 | 1.061 | |
| Pregnant Season | ||||
| Spring | 386 | 311 | 1.241 | 0.0843 |
| Summer | 379 | 392 | 0.967 | |
| Autumn | 399 | 356 | 1.121 | |
| Winter | 418 | 410 | 1.020 | |
| Nation | ||||
| Non-Mongol | 828 | 754 | 1.098 | 0.6014 |
| Mongol | 754 | 715 | 1.055 | |
| Mother education | ||||
| Junior high school | 981 | 866 | 1.133 | 0.0910 |
| High school | 601 | 603 | 0.997 | |
| Mother profession | ||||
| Famer | 684 | 589 | 1.161 | 0.1263 |
| Workers | 250 | 278 | 0.899 | |
| Civil servants | 179 | 166 | 1.078 | |
| Merchant | 239 | 207 | 1.155 | |
| Others | 230 | 229 | 1.004 | |
| Miscarriage history | ||||
| No | 1522 | 1408 | 1.081 | 0.6774 |
| Yes | 60 | 61 | 0.984 |
| Characteristic | Odd Ratio | 95% CI | p-value | |
|---|---|---|---|---|
| Father Age (Year) | ||||
| ≥30 | Reference | |||
| <30 | 0.815 | 0.65 | 1.02 | 0.075 |
| Mother Age | ||||
| ≥30 | Reference | |||
| <30 | 1.158 | 0.902 | 1.489 | 0.2516 |
| Region | ||||
| City | Reference | |||
| Rural | 0.859 | 0.684 | 1.078 | 0.1912 |
| Pregnant Season | ||||
| Spring | Reference | |||
| Summer | 0.782 | 0.636 | 0.961 | 0.0195 * |
| Autumn | 0.912 | 0.74 | 1.123 | 0.3852 |
| Winter | 0.809 | 0.66 | 0.991 | 0.0412 * |
| Nation | ||||
| Non-Mongol | Reference | |||
| Mongol | 0.949 | 0.789 | 1.142 | 0.5796 |
| Mother education | ||||
| Junior high school | Reference | |||
| High school | 0.859 | 0.71 | 1.039 | 0.1178 |
| Mother profession | ||||
| Famer | Reference | |||
| Workers | 0.726 | 0.564 | 0.933 | 0.0124 * |
| Civil servants | 0.981 | 0.716 | 1.346 | 0.9074 |
| Merchant | 0.983 | 0.759 | 1.273 | 0.8968 |
| Others | 0.852 | 0.664 | 1.092 | 0.206 |
| Miscarriage history | ||||
| No | Reference | |||
| Yes | 0.920 | 0.633 | 1.336 | 0.6602 |
4. Discussion
5. Limitations
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gazala, E.; Ron-Feldman, V.; Alterman, M.; Kama, S.; Novack, L. The association between birth season and future development of childhood asthma. Pediatr. Pulmonol. 2006, 41, 1125–1128. [Google Scholar] [CrossRef]
- Crump, C. Season of birth and risk of Hodgkin and non-Hodgkin lymphoma. Int. J. Cancer 2014. [Google Scholar] [CrossRef]
- Natale, V.; Adan, A.; Fabbri, M. Season of birth, gender, and social-cultural effects on sleep timing preferences in humans. Sleep 2009, 32, 423–426. [Google Scholar]
- Ma, Q.; Xu, W.; Zhou, X.; Cui, C.; Pan, C.W. The relationship of season of birth with refractive error in very young children in eastern China. PLoS One 2014. [Google Scholar] [CrossRef]
- Trivers, R.L.; Willard, D.E. Natural selection of parental ability to vary the sex ratio of offspring. Science 1973, 179, 90–92. [Google Scholar]
- McLachlan, J.C.; Storey, H. Hot male: Can sex in humans be modified by temperature? J. Theor. Biol. 2003, 222, 71–72. [Google Scholar] [CrossRef]
- Kumari, J.R.; Rao, T.V. Seasonal variation of human sex ratio at birth and the modifying factors. Indian J. Pediatr. 1982, 49, 541–546. [Google Scholar] [CrossRef]
- Melnikov, V.N.; Grech, V. Seasonality of live birth sex ratio in south western Siberia, Russia, 1959–2001. J. Epidemiol. Community Health 2003, 57, 471–472. [Google Scholar] [CrossRef]
- Jongbloet, P.H. Siberian live birth sex ratios and the SPrOO hypothesis. J. Epidemiol. Community Health. 2004, 58, p. 528. Available online: www.ncbi.nlm.nih.gov/pmc/articles/PMC1732792/pdf/v058p00528.pdf (accessed on 1 August 2014).
- Harada, T.; Kobayashi, R.; Wada, K.; Nishihara, R.; Kondo, A.; Akimitsu, O.; Noji, T.; Taniwaki, N.; Nakade, M.; Krejci, M.; Takeuchi, H. Effect of birth season on circadian typology appearing in Japanese young children aged 2 to 12 years disappears in older students aged 18 to 25 years. Chronobiol. Int. 2011, 28, 638–642. [Google Scholar] [CrossRef]
- McMahon, G.; Zayats, T.; Chen, Y.P.; Prashar, A.; Williams, C.; Guggenheim, J.A. Season of birth, daylight hours at birth, and high myopia. Ophthalmology 2009, 116, 468–473. [Google Scholar] [CrossRef]
- James, W.H. Interpregnancy intervals, high maternal age and seasonal effects on the human sex ratio. Hum. Reprod. 1996, 11, 7–8. [Google Scholar] [CrossRef]
- Helle, S.; Helama, S.; Jokela, J. Temperature-related birth sex ratio bias in historical Sami: Warm years bring more sons. Biol. Lett. 2008, 4, 60–62. [Google Scholar] [CrossRef]
- Neuwald, J.L.; Valenzuela, N. The lesser known challenge of climate change: Thermal variance and sex-reversal in vertebrates with temperature-dependent sex determination. PLoS One 2011. [Google Scholar] [CrossRef]
- Rhen, T.; Schroeder, A.; Sakata, J.T.; Huang, V.; Crews, D. Segregating variation for temperature-dependent sex determination in a lizard. Heredity 2011, 106, 649–660. [Google Scholar] [CrossRef]
- Mork, L.; Czerwinski, M.; Capel, B. Predetermination of sexual fate in a turtle with temperature-dependent sex determination. Dev. Biol. 2014, 386, 264–271. [Google Scholar] [CrossRef]
- Shen, Z.G.; Wang, H.P. Molecular players involved in temperature-dependent sex determination and sex differentiation in Teleost fish. Genet. Sel. Evol. 2014, 46. [Google Scholar] [CrossRef]
- Wedekind, C.; Evanno, G.; Szekely, T.; Pompini, M.; Darbellay, O.; Guthruf, J. Persistent unequal sex ratio in a population of grayling (Salmonidae) and possible role of temperature increase. Conserv. Biol. 2013, 27, 229–234. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Kitano, T. High temperature induces cyp26b1 mRNA expression and delays meiotic initiation of germ cells by increasing cortisol levels during gonadal sex differentiation in Japanese flounder. Biochem. Biophys. Res. Commun. 2012, 419, 287–292. [Google Scholar] [CrossRef]
- Navarro-Martin, L.; Vinas, J.; Ribas, L.; Diaz, N.; Gutierrez, A.; di Croce, L. Piferrer FDNA methylation of the gonadal aromatase (cyp19a) promoter is involved in temperature-dependent sex ratio shifts in the European sea bass. PLoS Genet. 2011. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Buemio, A.; Chu, R.; Vafaee, M.; Crews, D. Epigenetic control of gonadal aromatase (cyp19a1) in temperature-dependent sex determination of red-eared slider turtles. PLoS One 2013. [Google Scholar] [CrossRef]
- Sakata, N.; Miyazaki, K.; Wakahara, M. Up-regulation of P450arom and down-regulation of Dmrt-1 genes in the temperature-dependent sex reversal from genetic males to phenotypic females in a salamander. Dev. Genes. Evol. 2006, 216, 224–228. [Google Scholar] [CrossRef]
- Shoemaker, C.M.; Queen, J.; Crews, D. Response of candidate sex-determining genes to changes in temperature reveals their involvement in the molecular network underlying temperature-dependent sex determination. Mol. Endocrinol. 2007, 21, 2750–2763. [Google Scholar] [CrossRef]
- Roche, J.R.; Lee, J.M.; Berry, D.P. Climatic factors and secondary sex ratio in dairy cows. J. Dairy Sci. 2006, 89, 3221–3227. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, T.; Lin, D.; Liang, H.; Chen, M.; Tong, W.; Mu, Y.; Feng, C.X.; Gao, Y.; Zheng, Y.; Sun, W. The Association between Season of Pregnancy and Birth-Sex among Chinese. Int. J. Environ. Res. Public Health 2014, 11, 8166-8174. https://doi.org/10.3390/ijerph110808166
Xu T, Lin D, Liang H, Chen M, Tong W, Mu Y, Feng CX, Gao Y, Zheng Y, Sun W. The Association between Season of Pregnancy and Birth-Sex among Chinese. International Journal of Environmental Research and Public Health. 2014; 11(8):8166-8174. https://doi.org/10.3390/ijerph110808166
Chicago/Turabian StyleXu, Tan, Dongdong Lin, Hui Liang, Mei Chen, Weijun Tong, Yongping Mu, Cindy Xin Feng, Yongqing Gao, Yumei Zheng, and Wenjie Sun. 2014. "The Association between Season of Pregnancy and Birth-Sex among Chinese" International Journal of Environmental Research and Public Health 11, no. 8: 8166-8174. https://doi.org/10.3390/ijerph110808166
APA StyleXu, T., Lin, D., Liang, H., Chen, M., Tong, W., Mu, Y., Feng, C. X., Gao, Y., Zheng, Y., & Sun, W. (2014). The Association between Season of Pregnancy and Birth-Sex among Chinese. International Journal of Environmental Research and Public Health, 11(8), 8166-8174. https://doi.org/10.3390/ijerph110808166