Resistance to Extended-Spectrum β-Lactamases in Salmonella from a Broiler Supply Chain
Abstract
:1. Introduction
2. Materials and Methods
2.1. Salmonella Isolates
2.2. Antimicrobial Susceptibility Testing and Phenotypic Screening for ESBL Production

2.3. Detection of Inducible Chromosomal β-Lactamases
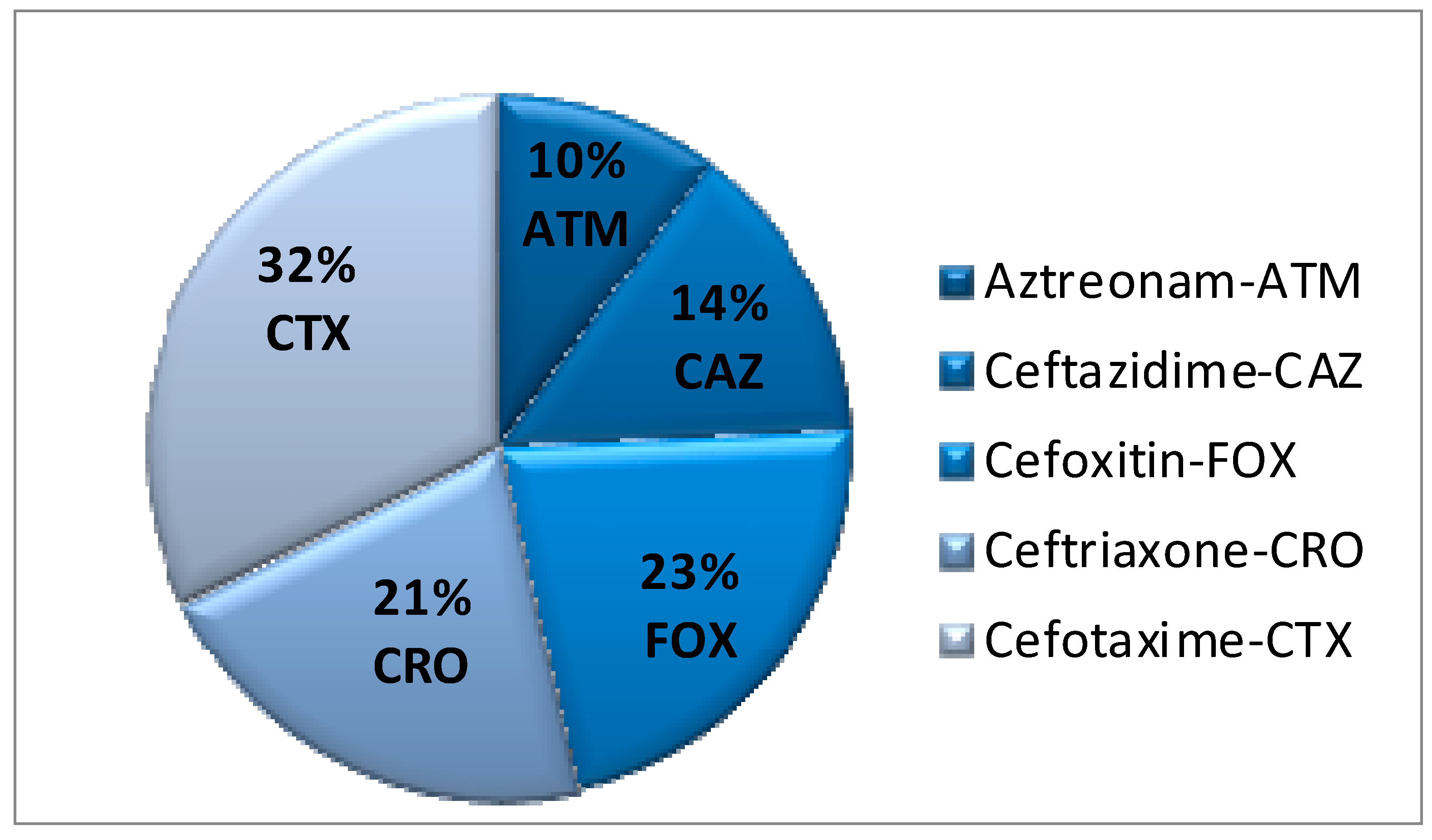
3. Results and Discussion
| Salmonella enterica subsp. enterica (Serotypes) | ATM | CAZ | FOX | CRO | CTX |
|---|---|---|---|---|---|
| S. Linvingstone | 31 | 25 | 25 | 29 | 31 |
| S. Virchow | 30 | 26 | 25 | 30 | 30 |
| S. Mbandaka | 30 | 22 | 24 | 30 | 28 |
| S. Albany | 33 | 27 | 27 | 33 | 32 |
| S. Carrau | 30 | 23 | 26 | 27 | 30 |
| S. Shwarzenground | 28 | 26 | 26 | 27 | 27 |
| S. Minnesota | 30 | 25 | 25 | 24 | 26 |
| S. Oranienburg | 32 | 25 | 25 | 30 | 32 |
| S. Meleagridis | 30 | 26 | 25 | 28 | 30 |
| S. Worthington | 28 | 26 | 25 | 29 | 27 |
| S. Muenchen | 28 | 24 | 24 | 27 | 24 |
| S. Tennessee | 30 | 26 | 26 | 27 | 28 |
| S. Enteritidis | 0 | 7 | 27 | 24 | 19 |
| S. Mandelia | 10 | 11 | 25 | 21 | 18 |
| S. Hadar | 0 | 10 | 19 | 16 | 17 |
| S. Anatum | 33 | 26 | 27 | 27 | 29 |
| S. Agona | 31 | 25 | 25 | 27 | 28 |
| S. Cubana | 21 | 21 | 24 | 29 | 28 |
| S. Rissen | 29 | 26 | 22 | 28 | 28 |
| Serotypes | ESBL | |||
|---|---|---|---|---|
| 1° test | 2° test | 3° test | AmpC | |
| S. Linvingstone | − | − | + | − |
| S. Virchow | + | + | − | + |
| S. Mbandaka | + | + | + | + |
| S. Albany * | − | − | − | − |
| S. Carrau | + | + | − | + |
| S. Schwarzenground | + | + | + | + |
| S. Minnesota | + | + | + | + |
| S. Oranienburg | − | + | − | + |
| S. Meleagridis * | − | − | − | − |
| S. Worthington | + | + | + | + |
| S. Muenchen | + | + | + | + |
| S. Tennessee | − | + | − | − |
| S. Enteritidis | + | + | + | + |
| S. Mandelia | + | + | + | + |
| S. Hadar | + | + | + | + |
| S. Anatum | − | − | + | + |
| S. Agona | − | + | − | − |
| S. Cubana | + | + | + | + |
| S. Rissen | + | + | + | + |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- The Poultry Site. Global Poultry Trends. Available online: www.thepoultrysite.com/articles/2960/global-poultry-trendsamericas-to-produce-42-million-tonnes-of-chicken-in-2014 (accessed on 24 July 2014).
- Mondelli, A.C.; Sadatsune, T. Antibioticoterapia para o clínico. Rev. Soc. Bras. Microbiologia. 2001, 47, 7–53. [Google Scholar]
- Naiemi, N.A.; Zwart, B.; Rijnsburger, M.C.; Roosendaal, R.; Debets-Ossenkopp, Y.J.; Mulder, J.A.; Fijen, C.A.; Maten, W.; Vandenbroucke-Grauls, C.M.; Savelkoul, P.H. Extended-spectrum-beta-lactamase production in a Salmonella enterica serotype Typhi strain from the Philippines. J. Clin. Microbiol. 2008, 46, 2794–2795. [Google Scholar] [CrossRef]
- Rupp, M.E.; Fey, P.D. Enterobactérias produtoras de beta-lactamasas de espectro extendido (BLEE): Diagnóstico, prevencion y tratamiento farmacológico. Drugs 2003, 63, 353–365. [Google Scholar] [CrossRef]
- Bradford, P.A. Extended-spectrum beta-lactamases in the 21st century: Characterization, epidemiology, and detection of this important resistance threat. Clin. Microbiol. Rev. 2001, 14, 933–951. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; Fifteenth Informational Supplement. CLSI document 2007, M100-S17 (Replaces M100-S16). Available online: www.microbiolab-bg.com/CLSI.pdf (accessed on 24 June 2014).
- Mesa, R.J.; Blanc, V.; Blanch, A.R.; Cortes, P.; Gonzalez, J.J.; Lavilla, S.; Miro, E.; Muniesa, M.; Saco, M.; Tortola, M.T.; et al. Extended-spectrum β-lactamase-producing Enterobacteriaceae in different environments (humans, food, animal farms and sewage). J. Antimicrob. Chemother. 2006, 58, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Mo, S.S.; Norström, M.; Slettemeås, J.S.; Løvland, A.; Urdahl, A.M.; Sunde, M. Emergence of AmpC-producing Escherichia coli in the broiler production chain in a country with a low antimicrobial usage profile. Vet. Microbiol. 2014, 171, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Hrabák, J. Clinically importante beta-lactamases of gram-negative bacteria: Extended-spectrum β-lactamase (ESBL). Epidemiol. Mikrobiol. Imunol. 2007, 56, 103–111. [Google Scholar] [PubMed]
- Sanders, C.C.; Sanders, W.E., Jr. Emergence of resistance to cefamandole: Possible role of cefoxitin-inducible beta-lactamases. Antimicrob. Agents Chemother. 2007, 15, 792–797. [Google Scholar] [CrossRef]
- Tenover, F.C. Development and spread of bacterial resistance to antimicrobial agents: An overview. Clin. Infect. Dis. 2011, 33, S108–S115. [Google Scholar] [CrossRef]
- Gupta, V. An update on newer β-lactamases. Indian J. Med. Res. 2007, 126, 417–427. [Google Scholar] [PubMed]
- Giamarellou, H. Multidrug resistance in Gram-negative bacteria that produce extended-spectrum β-lactamases (ESBLs). Clin. Microbiol. Infect. 2005, 11, 1–16. [Google Scholar] [CrossRef] [PubMed]
- U.S. FDA. Bacteriological Analytical Manual Chapter 5: Salmonella. Available online: www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm070149.htm (accessed on 20 December 2013).
- Paterson, D.L.; Bonomo, R.A. Extended-spectrum-lactamases: A clinical update. Clin. Microbiol. Rev. 2005, 18, 657–686. [Google Scholar] [CrossRef] [PubMed]
- Lago, A.; Fuentefria, S.R.; Fuentefria, D.B. Enterobactérias produtoras de ESBL em Passo Fundo, Estado do Rio Grande do Sul, Brasil. Rev. Soc. Bras. Med. Trop. 2010, 43, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.D. β-lactamases and β-lactamase inhibitors. Inter. J. Antimicrob. Agents 1999, 12, S3–S7. [Google Scholar] [CrossRef]
- Shah, A.A.; Hasan, F.; Ahmed, S.; Hameed, A. Characteristics, epidemiology and clinical importance of emerging strains of Gram-negative bacilli producing extended-spectrum beta-lactamases. Res. Microbiol. 2004, 155, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.R. Fagotipagem e análise por RAPD/PCR (DNA Polimórfico Amplificado ao Acaso) de amostras de Salmonella Enteritidis isoladas de materiais de origem avícola e de alimentos e humanos envolvidos em casos de toxinfecções alimentares. M.D. Thesis, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, 2001. [Google Scholar]
- Borsoi, A.; de Souza Moraes, H.L.; Salle, C.T.P.; do Nascimento, V.P. Número mais provável de Salmonella isoladas de carcaças de frango resfriadas. Ciência. Rural 2010, 40, 2338–2342. [Google Scholar] [CrossRef]
- Cortez, A.L.L.; Carvalho, A.C. de F.B. de.; Ikuno, A.A.; Bürger, K.P.; Vidal-Martins, A.M.C. Antibiotic resistance of Salmonella strains isolated from a chicken abattoir. Revista Arquivos do Instituto. Biológico. 2006, 73, 157–163. [Google Scholar]
- Van den Bogaard, A.E.; London, N.; Driessen, C.; Stobberingh, E.E. Antibiotic resistance of faecal Escherichia coli in poultry, poultry farmers, and poultry slaughterers. J. Antimicrob. Chemother. 2001, 47, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Hasman, H.; Mevius, D.; Veldman, K.; Olesen, I.; Aarestrup, F.M. β-Lactamases amonng extended-spectrum β-lactamase (ESBL)-resistant Salmonella from poultry, poultry products, and human patients in The Netherlands. J. Antimicrob. Chemother. 2005, 56, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.N.; Duarte, A. Salmonella Enteritidis em aves: Retrospectiva no Brasil. Rev. Bras. Cienc. Avic. 2002, 4, 85–100. [Google Scholar] [CrossRef]
- Portaria Ministerial No.193. No. 89, de 13 de maio de 1998. (Revoked). Available online: http://pesquisa.in.gov.br/imprensa/jsp/visualiza/index.jsp?data=13/05/1998&jornal=1&pagina=163&totalArquivos=224 (accessed on 24 July 2014).
- Ministério da Agricultura, Pecuária e Abastecimento—Instrução Normativa No. 26 de 09 de julho de 2009. Available online: http://pesquisa.in.gov.br/imprensa/jsp/visualiza/index.jsp?data=10/07/2009&jornal=1&pagina=14&totalArquivos=152 (accessed on 24 July 2014).
- Névoa, M.L.; Caramori, J.G., Jr.; Vieites, F M.; Nunes, R.V.; de Vargas Junior, J.G.; Kamimura, R. Antimicrobianos e prebióticos nas dietas de animais não ruminantes. Scientia Agraria Paranaensis 2013, 12, 85–95. [Google Scholar]
- Sader, H.S.; Mendes, R.E.; Gales, A.C.; Jones, R.N.; Pfaller, M.A.; Zoccoli, C.; Sampaio, J.; Latin America Study Group. Perfil de Sensibilidade a antimicrobianos de bactérias isoladas do trato respiratório baixo de pacientes com pneumonia internados em hospitais brasileiros: Resultados do Programa SENTRY, 1997 e 1998. J. Pneumol. 2001, 27, 59–67. [Google Scholar] [CrossRef]
- Sader, H.S.; Jones, R.N.; Gales, A.C.; Winokur, P.; Kugler, K.C.; Pfaller, M.A.; Doern, G.V.; The SENTRY Latin America Study Group. Antimicrobial susceptibility of patterns for pathogens isolated from patients in Latin American medical centers with a diagnosis of pneumonia: Results from the SENTRY Antimicrobial Surveillance Program 1997. Diagn. Microbiol. Infect. Dis. 1998, 32, 289–301. [Google Scholar] [CrossRef]
- Aarestrup, F.M.; Hendriksen, R.S.; Lockett, J.; Gay, K.; Teates, K.; McDermott, P.F.; White, D.G.; Hasman, H.; Sørensen, G.; Bangtrakulnonth, A.; et al. International spread of multidrug-resistant Salmonella schwarzengrund in food products. Emerg. Infect. Dis. 2007, 13, 726–731. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gelinski, J.M.L.N.; Bombassaro, A.; Baratto, C.M.; Vicente, V.A. Resistance to Extended-Spectrum β-Lactamases in Salmonella from a Broiler Supply Chain. Int. J. Environ. Res. Public Health 2014, 11, 11718-11726. https://doi.org/10.3390/ijerph111111718
Gelinski JMLN, Bombassaro A, Baratto CM, Vicente VA. Resistance to Extended-Spectrum β-Lactamases in Salmonella from a Broiler Supply Chain. International Journal of Environmental Research and Public Health. 2014; 11(11):11718-11726. https://doi.org/10.3390/ijerph111111718
Chicago/Turabian StyleGelinski, Jane Mary Lafayette Neves, Amanda Bombassaro, César Milton Baratto, and Vânia Aparecida Vicente. 2014. "Resistance to Extended-Spectrum β-Lactamases in Salmonella from a Broiler Supply Chain" International Journal of Environmental Research and Public Health 11, no. 11: 11718-11726. https://doi.org/10.3390/ijerph111111718
APA StyleGelinski, J. M. L. N., Bombassaro, A., Baratto, C. M., & Vicente, V. A. (2014). Resistance to Extended-Spectrum β-Lactamases in Salmonella from a Broiler Supply Chain. International Journal of Environmental Research and Public Health, 11(11), 11718-11726. https://doi.org/10.3390/ijerph111111718




