Aberrant Production of Th1/Th2/Th17-Related Cytokines in Serum of C57BL/6 Mice after Short-Term Formaldehyde Exposure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Formaldehyde Exposure
2.3. Collection of Samples
2.4. CBA
2.5. Statistical Analysis
3. Results
3.1. Effects of Exposure to FA on Th1-Related Cytokines
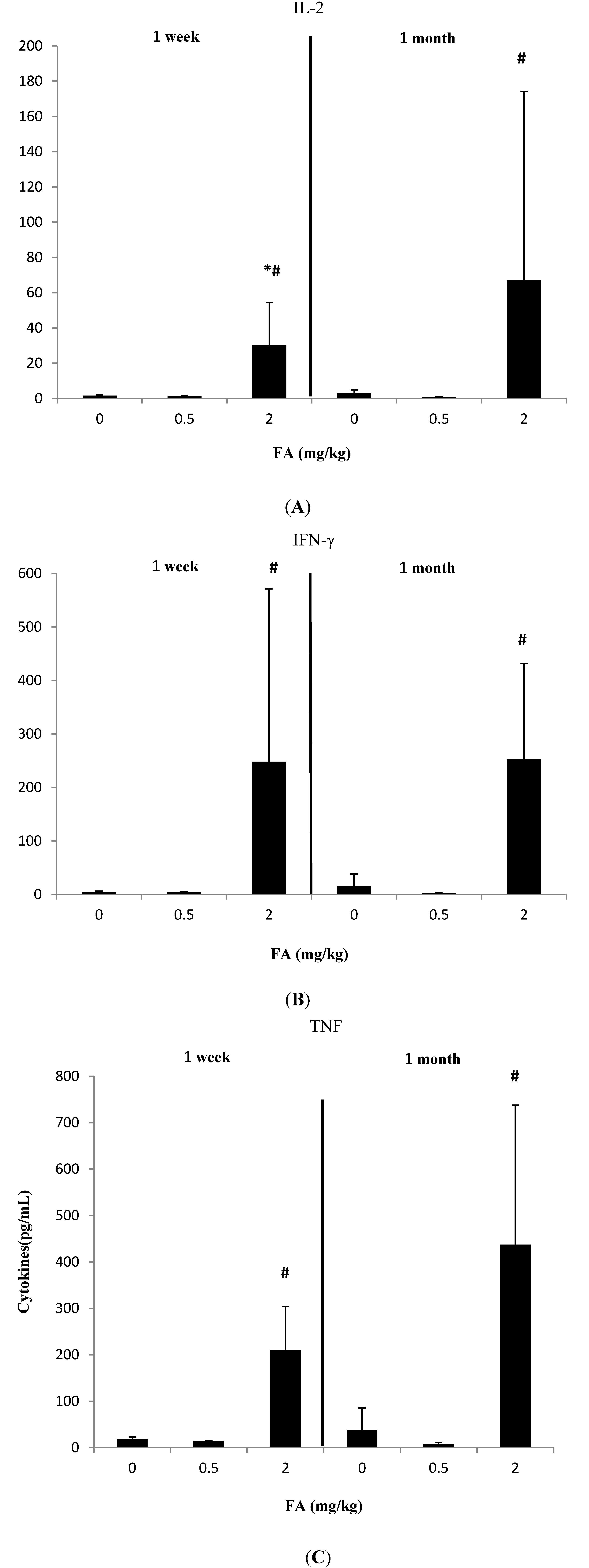
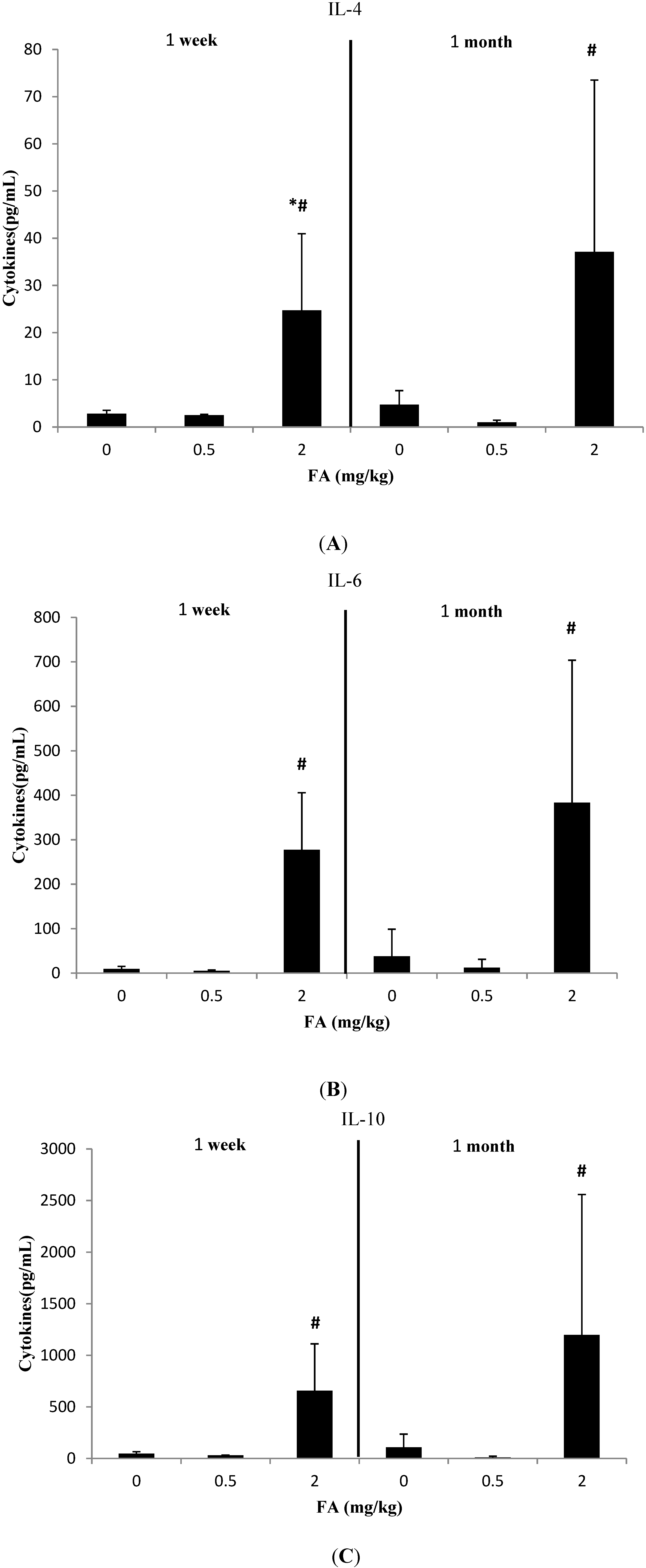
3.2. Effects of Exposure to FA on Th2-Related Cytokines
3.3. Effects of Exposure to FA on Th17-Related Cytokines
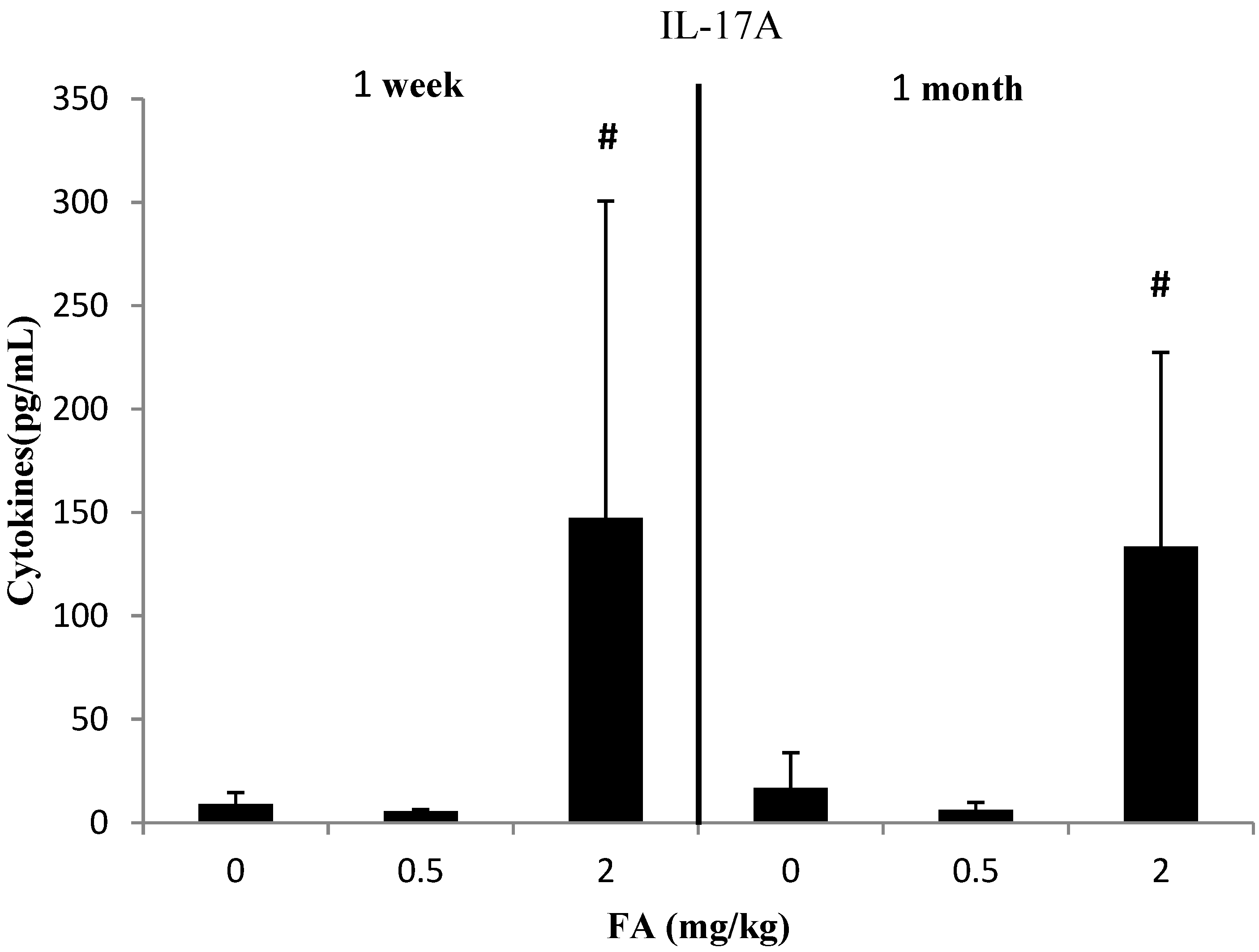
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Syamlal, G.; Mazurek, J.M.; Bang, K.M. Prevalence of lifetime asthma and current asthma attacks in U.S. Working adults: An analysis of the 1997–2004 national health interview survey data. J. Occup. Environ. Med. 2009, 51, 1066–1074. [Google Scholar]
- Liu, D.; Zheng, Y.; Li, B.; Yao, H.; Li, R.; Zhang, Y.; Yang, X. Adjuvant effects of gaseous formaldehyde on the hyper-responsiveness and inflammation in a mouse asthma model immunized by ovalbumin. J. Immunotoxicol. 2011, 8, 305–314. [Google Scholar]
- Hosgood, H.D.; Zhang, L.; Tang, X.; Vermeulen, R.; Hao, Z.; Shen, M.; Qiu, C.; Ge, Y.; Hua, M.; Ji, Z.; et al. Occupational exposure to formaldehyde and alterations in lymphocyte subsets. Amer. J. Ind. Med. 2013, 56, 252–257. [Google Scholar]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Formaldehyde, 2-butoxyethanol and 1-tert-butoxypropan-2-ol. IARC Monogr. Eval. Carcinog. Risks Hum. 2006, 88, 1–287. [Google Scholar]
- Coggon, D.; Harris, E.C.; Poole, J.; Palmer, K.T. Extended follow-up of a cohort of British chemical workers exposed to formaldehyde. J. Natl. Cancer Instit. 2003, 95, 1608–1615. [Google Scholar]
- Hauptmann, M.; Lubin, J.H.; Stewart, P.A.; Hayes, R.B.; Blair, A. Mortality from lymphohematopoietic malignancies among workers in formaldehyde industries. J. Natl. Cancer Instit. 2003, 95, 1615–1623. [Google Scholar]
- Hauptmann, M.; Lubin, J.H.; Stewart, P.A.; Hayes, R.B.; Blair, A. Mortality from solid cancers among workers in formaldehyde industries. Amer. J. Epidemiol. 2004, 159, 1117–1130. [Google Scholar]
- IARC monographs on the evaluation of carcinogenic risks to humans. Chemical agents and related occupations. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100, 9–562.
- Sandikci, M.; Eren, U.; Kum, S. Effects of formaldehyde and xylene on cd4- and cd8-positive T cells in bronchus-associated lymphoid tissue in rats. Toxicol. Ind. Health 2007, 23, 471–477. [Google Scholar]
- Aydin, S.; Canpinar, H.; Undeger, U.; Guc, D.; Colakoglu, M.; Kars, A.; Basaran, N. Assessment of immunotoxicity and genotoxicity in workers exposed to low concentrations of formaldehyde. Arch. Toxicol. 2013, 87, 145–153. [Google Scholar]
- Zhang, L.; Tang, X.; Rothman, N.; Vermeulen, R.; Ji, Z.; Shen, M.; Qiu, C.; Guo, W.; Liu, S.; Reiss, B.; et al. Occupational exposure to formaldehyde, hematotoxicity, and leukemia-specific chromosome changes in cultured myeloid progenitor cells. Cancer Epidemiol. Biomark. Prev. 2010, 19, 80–88. [Google Scholar]
- Lee, C.G.; Sahoo, A.; Im, S.H. Epigenetic regulation of cytokine gene expression in T lymphocytes. Yonsei Med. J. 2009, 50, 322–330. [Google Scholar]
- Ohtsuka, R.; Shutoh, Y.; Fujie, H.; Yamaguchi, S.; Takeda, M.; Harada, T.; Doi, K. Rat strain difference in histology and expression of Th1- and Th2-related cytokines in nasal mucosa after short-term formaldehyde inhalation. Exp. Toxicol. Pathol. 2003, 54, 287–291. [Google Scholar]
- Jimenez, R.; Ramirez, R.; Carracedo, J.; Aguera, M.; Navarro, D.; Santamaria, R.; Perez, R.; del Castillo, D.; Aljama, P. Cytometric bead array (CBA) for the measurement of cytokines in urine and plasma of patients undergoing renal rejection. Cytokine 2005, 32, 45–50. [Google Scholar]
- Wyns, H.; Croubels, S.; Demeyere, K.; Watteyn, A.; de Backer, P.; Meyer, E. Development of a cytometric bead array screening tool for the simultaneous detection of pro-inflammatory cytokines in porcine plasma. Vet. Immunol. Immunopathol. 2013, 151, 28–36. [Google Scholar]
- Ulrich, P.; Grenet, O.; Bluemel, J.; Vohr, H.W.; Wiemann, C.; Grundler, O.; Suter, W. Cytokine expression profiles during murine contact allergy: T helper 2 cytokines are expressed irrespective of the type of contact allergen. Arch. Toxicol. 2001, 75, 470–479. [Google Scholar]
- Fujimaki, H.; Kurokawa, Y.; Kunugita, N.; Kikuchi, M.; Sato, F.; Arashidani, K. Differential immunogenic and neurogenic inflammatory responses in an allergic mouse model exposed to low levels of formaldehyde. Toxicology 2004, 197, 1–13. [Google Scholar]
- Wang, F.; Li, C.; Liu, W.; Jin, Y. Effect of exposure to volatile organic compounds (VOCs) on airway inflammatory response in mice. J. Toxicol. Sci. 2012, 37, 739–748. [Google Scholar]
- Kim, J.Y.; Jeong, M.S.; Park, K.Y.; Seo, S.J. Aggravation of atopic dermatitis-like symptoms by consecutive low concentration of formaldehyde exposure in NC/NGA mice. Exp. Dermatol. 2013, 22, 219–221. [Google Scholar]
- Thrasher, J.D.; Broughton, A.; Madison, R. Immune activation and autoantibodies in humans with long-term inhalation exposure to formaldehyde. Arch. Environ. Health 1990, 45, 217–223. [Google Scholar]
- Persoz, C.; Achard, S.; Momas, I.; Seta, N. Inflammatory response modulation of airway epithelial cells exposed to formaldehyde. Toxicol Lett. 2012, 211, 159–163. [Google Scholar]
- Kastner, P.E.; le Calve, S.; Zheng, W.; Casset, A.; Pons, F. A dynamic system for single and repeated exposure of airway epithelial cells to gaseous pollutants. Toxicol. In Vitro 2013, 27, 632–640. [Google Scholar]
- Bardet, G.; Achard, S.; Loret, T.; Desauziers, V.; Momas, I.; Seta, N. A model of human nasal epithelial cells adapted for direct and repeated exposure to airborne pollutants. Toxicol. Lett. 2014, 229, 144–149. [Google Scholar]
- Rager, J.E.; Smeester, L.; Jaspers, I.; Sexton, K.G.; Fry, R.C. Epigenetic changes induced by air toxics: Formaldehyde exposure alters mirna expression profiles in human lung cells. Environ. Health Perspect. 2011, 119, 494–500. [Google Scholar]
- Zhang, Y.; Liu, X.; McHale, C.; Li, R.; Zhang, L.; Wu, Y.; Ye, X.; Yang, X.; Ding, S. Bone marrow injury induced via oxidative stress in mice by inhalation exposure to formaldehyde. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Larsen, S.T.; Wolkoff, P.; Hammer, M.; Kofoed-Sorensen, V.; Clausen, P.A.; Nielsen, G.D. Acute airway effects of airborne formaldehyde in sensitized and non-sensitized mice housed in a dry or humid environment. Toxicol. Appl. Pharmacol. 2013, 268, 294–299. [Google Scholar]
- Lino dos Santos Franco, A.; Domingos, H.V.; Damazo, A.S.; Breithaupt-Faloppa, A.C.; de Oliveira, A.P.; Costa, S.K.; Oliani, S.M.; Oliveira-Filho, R.M.; Vargaftig, B.B.; Tavares-de-Lima, W. Reduced allergic lung inflammation in rats following formaldehyde exposure: Long-term effects on multiple effector systems. Toxicology 2009, 256, 157–163. [Google Scholar]
- Casset, A.; Marchand, C.; Purohit, A.; le Calve, S.; Uring-Lambert, B.; Donnay, C.; Meyer, P.; de Blay, F. Inhaled formaldehyde exposure: Effect on bronchial response to mite allergen in sensitized asthma patients. Allergy 2006, 61, 1344–1350. [Google Scholar]
- Frank, J.W.; Carroll, J.A.; Allee, G.L.; Zannelli, M.E. The effects of thermal environment and spray-dried plasma on the acute-phase response of pigs challenged with lipopolysaccharide. J. Anim. Sci. 2003, 81, 1166–1176. [Google Scholar]
- Elshal, M.F.; McCoy, J.P. Multiplex bead array assays: Performance evaluation and comparison of sensitivity to ELISA. Methods 2006, 38, 317–323. [Google Scholar]
- Williams, P.N.; Collier, C.T.; Carroll, J.A.; Welsh, T.H., Jr.; Laurenz, J.C. Temporal pattern and effect of sex on lipopolysaccharide-induced stress hormone and cytokine response in pigs. Domest. Anim. Endocrinol. 2009, 37, 139–147. [Google Scholar]
- Williams, A.; Steffens, F.; Reinecke, C.; Meyer, D. The Th1/Th2/Th17 cytokine profile of HIV-infected individuals: A multivariate cytokinomics approach. Cytokine 2013, 61, 521–526. [Google Scholar]
- Yujie, W.; Yu, Z.; Sixuan, Q.; Li, W.; Peng, L.; Zeng, G.; Sujiang, Z.; Jianyong, L. Detection of bcr-abl fusion proteins in patients with leukemia using a cytometric bead array. Leuk. Lymphoma 2012, 53, 451–455. [Google Scholar]
- Talat, N.; Shahid, F.; Perry, S.; Dawood, G.; Hussain, R. Th1/th2 cytometric bead array can discriminate cytokine secretion from endogenously activated cells in pulmonary disease, recent and remote infection in tuberculosis. Cytokine 2011, 54, 136–143. [Google Scholar]
- Sadakane, K.; Takano, H.; Ichinose, T.; Yanagisawa, R.; Shibamoto, T. Formaldehyde enhances mite allergen-induced eosinophilic inflammation in the murine airway. J. Environ. Pathol. Toxicol. Oncol. 2002, 21, 267–276. [Google Scholar]
- Nandate, K.; Ishidao, T.; Hori, H.; Morimoto, Y.; Ogata, M. The effect of formaldehyde exposure on cytokine production in murine alveolar macrophages. J. UOEH 2003, 25, 197–205. [Google Scholar]
- Thrasher, J.D.; Wojdani, A.; Cheung, G.; Heuser, G. Evidence for formaldehyde antibodies and altered cellular immunity in subjects exposed to formaldehyde in mobile homes. Arch. Environ. Health 1987, 42, 347–350. [Google Scholar]
- Tarkowski, M.; Kur, B.; Polakowska, E.; Jablonska, E. Comparative studies of lymph node cell subpopulations and cytokine expression in murine model for testing the potentials of chemicals to induce respiratory sensitization. Int. J. Occupational Med. Environ. Health 2008, 21, 253–262. [Google Scholar]
- Sasaki, Y.; Ohtani, T.; Ito, Y.; Mizuashi, M.; Nakagawa, S.; Furukawa, T.; Horii, A.; Aiba, S. Molecular events in human T cells treated with diesel exhaust particles or formaldehyde that underlie their diminished interferon-gamma and interleukin-10 production. Int. Arch. Allergy Immunol. 2009, 148, 239–250. [Google Scholar]
- Hoyer, K.K.; Dooms, H.; Barron, L.; Abbas, A.K. Interleukin-2 in the development and control of inflammatory disease. Immunol. Rev. 2008, 226, 19–28. [Google Scholar]
- Hofmann, S.R.; Ettinger, R.; Zhou, Y.J.; Gadina, M.; Lipsky, P.; Siegel, R.; Candotti, F.; O’Shea, J.J. Cytokines and their role in lymphoid development, differentiation and homeostasis. Curr. Opin. Allergy Clin. Immunol. 2002, 2, 495–506. [Google Scholar]
- Jones, S.A. Directing transition from innate to acquired immunity: Defining a role for IL-6. J. Immunol. 2005, 175, 3463–3468. [Google Scholar]
- Huang, M.; Wang, J.; Lee, P.; Sharma, S.; Mao, J.T.; Meissner, H.; Uyemura, K.; Modlin, R.; Wollman, J.; Dubinett, S.M. Human non-small cell lung cancer cells express a type 2 cytokine pattern. Cancer Res. 1995, 55, 3847–3853. [Google Scholar]
- Mills, K.H. Induction, function and regulation of IL-17-producing T cells. Eur. J. Immunol. 2008, 38, 2636–2649. [Google Scholar]
- Weaver, C.T.; Hatton, R.D.; Mangan, P.R.; Harrington, L.E. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu. Rev. Immunol. 2007, 25, 821–852. [Google Scholar]
- Cousens, L.P.; Orange, J.S.; Biron, C.A. Endogenous IL-2 contributes to t cell expansion and ifn-gamma production during lymphocytic choriomeningitis virus infection. J. Immunol. 1995, 155, 5690–5699. [Google Scholar]
- Dallman, M.J.; Wood, K.J.; Hamano, K.; Bushell, A.R.; Morris, P.J.; Wood, M.J.; Charlton, H.M. Cytokines and peripheral tolerance to alloantigen. Immunol. Rev. 1993, 133, 5–18. [Google Scholar]
- Dallman, M.J. Cytokines as mediators of organ graft rejection and tolerance. Curr. Opin. Immunol. 1993, 5, 788–793. [Google Scholar]
- Ezratty, V.; Bonay, M.; Neukirch, C.; Orset-Guillossou, G.; Dehoux, M.; Koscielny, S.; Cabanes, P.A.; Lambrozo, J.; Aubier, M. Effect of formaldehyde on asthmatic response to inhaled allergen challenge. Environ. Health Perspect. 2007, 115, 210–214. [Google Scholar]
- Moser, B.; Bodrogi, F.; Eibl, G.; Lechner, M.; Rieder, J.; Lirk, P. Mass spectrometric profile of exhaled breath—Field study by PTR-MS. Respir. Physiol. Neurobiol. 2005, 145, 295–300. [Google Scholar]
- Sibirny, V.; Demkiv, O.; Klepach, H.; Honchar, T.; Gonchar, M. Alcohol oxidase- and formaldehyde dehydrogenase-based enzymatic methods for formaldehyde assay in fish food products. Food Chem. 2011, 127, 774–779. [Google Scholar]
- Kim, E.M.; Lee, H.Y.; Lee, E.H.; Lee, K.M.; Park, M.; Ji, K.Y.; Jang, J.H.; Jeong, Y.H.; Lee, K.H.; Yoon, I.J.; et al. Formaldehyde exposure impairs the function and differentiation of NK cells. Toxicol. Lett. 2013, 223, 154–161. [Google Scholar]
- Intlekofer, A.M.; Takemoto, N.; Wherry, E.J.; Longworth, S.A.; Northrup, J.T.; Palanivel, V.R.; Mullen, A.C.; Gasink, C.R.; Kaech, S.M.; Miller, J.D.; et al. Effector and memory cd8+ T cell fate coupled by T-bet and eomesodermin. Nat. Immunol. 2005, 6, 1236–1244. [Google Scholar]
- Witowski, J.; Wisniewska, J.; Korybalska, K.; Bender, T.O.; Breborowicz, A.; Gahl, G.M.; Frei, U.; Passlick-Deetjen, J.; Jorres, A. Prolonged exposure to glucose degradation products impairs viability and function of human peritoneal mesothelial cells. J. Amer. Soc. Nephrol. 2001, 12, 2434–2441. [Google Scholar]
- Kitagaki, H.; Ono, N.; Hayakawa, K.; Kitazawa, T.; Watanabe, K.; Shiohara, T. Repeated elicitation of contact hypersensitivity induces a shift in cutaneous cytokine milieu from a T helper cell type 1 to a T helper cell type 2 profile. J. Immunol. 1997, 159, 2484–2491. [Google Scholar]
- Kitagaki, H.; Kimishima, M.; Teraki, Y.; Hayakawa, J.; Hayakawa, K.; Fujisawa, S.; Shiohara, T. Distinct in vivo and in vitro cytokine profiles of draining lymph node cells in acute and chronic phases of contact hypersensitivity: Importance of a type 2 cytokine-rich cutaneous milieu for the development of an early-type response in the chronic phase. J. Immunol. 1999, 163, 1265–1273. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, H.; Tan, K.; Sun, R.; Yin, L.; Zhang, J.; Pu, Y. Aberrant Production of Th1/Th2/Th17-Related Cytokines in Serum of C57BL/6 Mice after Short-Term Formaldehyde Exposure. Int. J. Environ. Res. Public Health 2014, 11, 10036-10050. https://doi.org/10.3390/ijerph111010036
Wei H, Tan K, Sun R, Yin L, Zhang J, Pu Y. Aberrant Production of Th1/Th2/Th17-Related Cytokines in Serum of C57BL/6 Mice after Short-Term Formaldehyde Exposure. International Journal of Environmental Research and Public Health. 2014; 11(10):10036-10050. https://doi.org/10.3390/ijerph111010036
Chicago/Turabian StyleWei, Haiyan, Kehong Tan, Rongli Sun, Lihong Yin, Juan Zhang, and Yuepu Pu. 2014. "Aberrant Production of Th1/Th2/Th17-Related Cytokines in Serum of C57BL/6 Mice after Short-Term Formaldehyde Exposure" International Journal of Environmental Research and Public Health 11, no. 10: 10036-10050. https://doi.org/10.3390/ijerph111010036
APA StyleWei, H., Tan, K., Sun, R., Yin, L., Zhang, J., & Pu, Y. (2014). Aberrant Production of Th1/Th2/Th17-Related Cytokines in Serum of C57BL/6 Mice after Short-Term Formaldehyde Exposure. International Journal of Environmental Research and Public Health, 11(10), 10036-10050. https://doi.org/10.3390/ijerph111010036




