Toxicity of Gutkha, a Smokeless Tobacco Product Gone Global: Is There More to the Toxicity than Nicotine?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Preparation and Exposure of Smokeless Tobacco Extract
2.3. Experimental Design
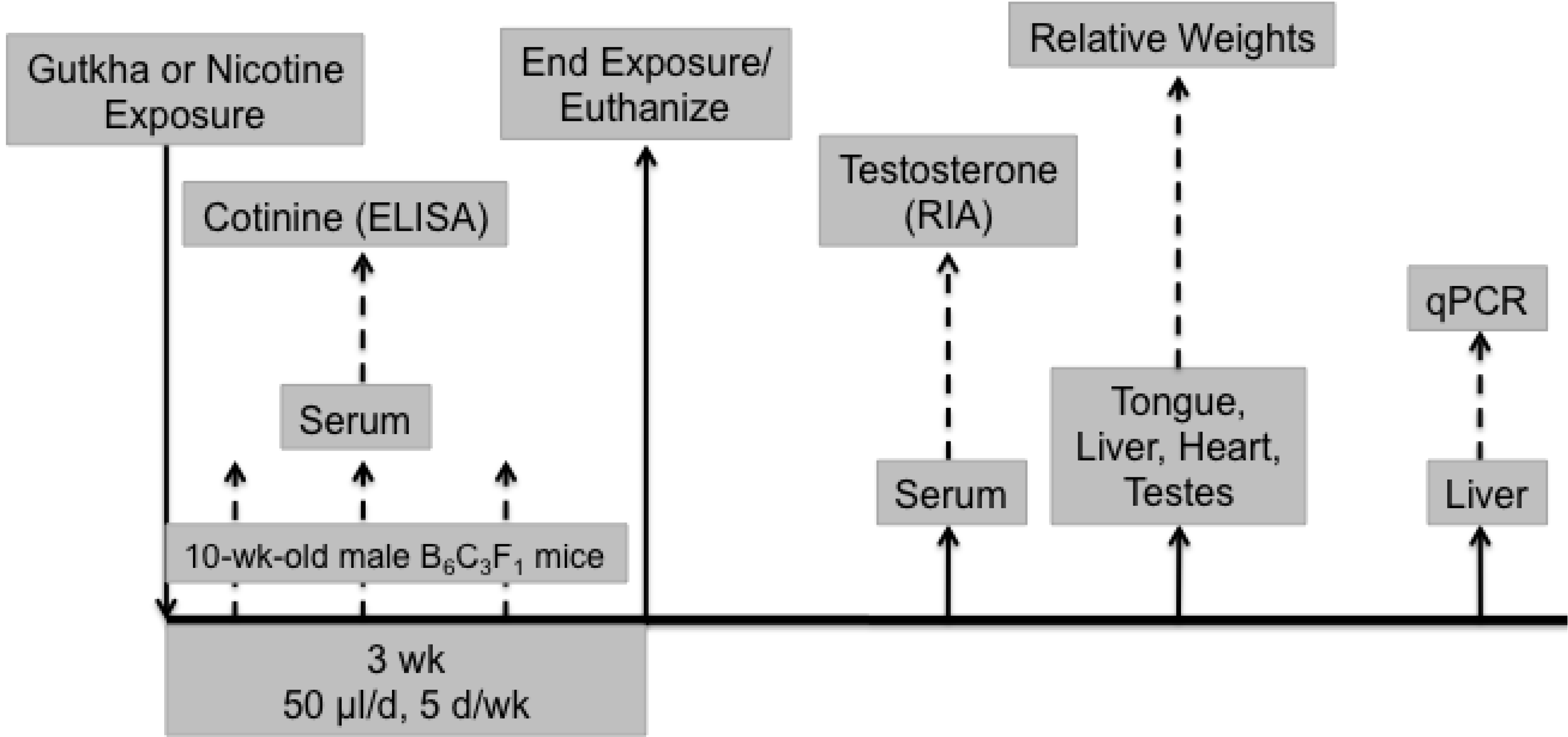
2.4. RIA of Serum Testosterone
2.5. RNA Extraction and Real-Time PCR Analyses
2.6. Statistics
3. Results
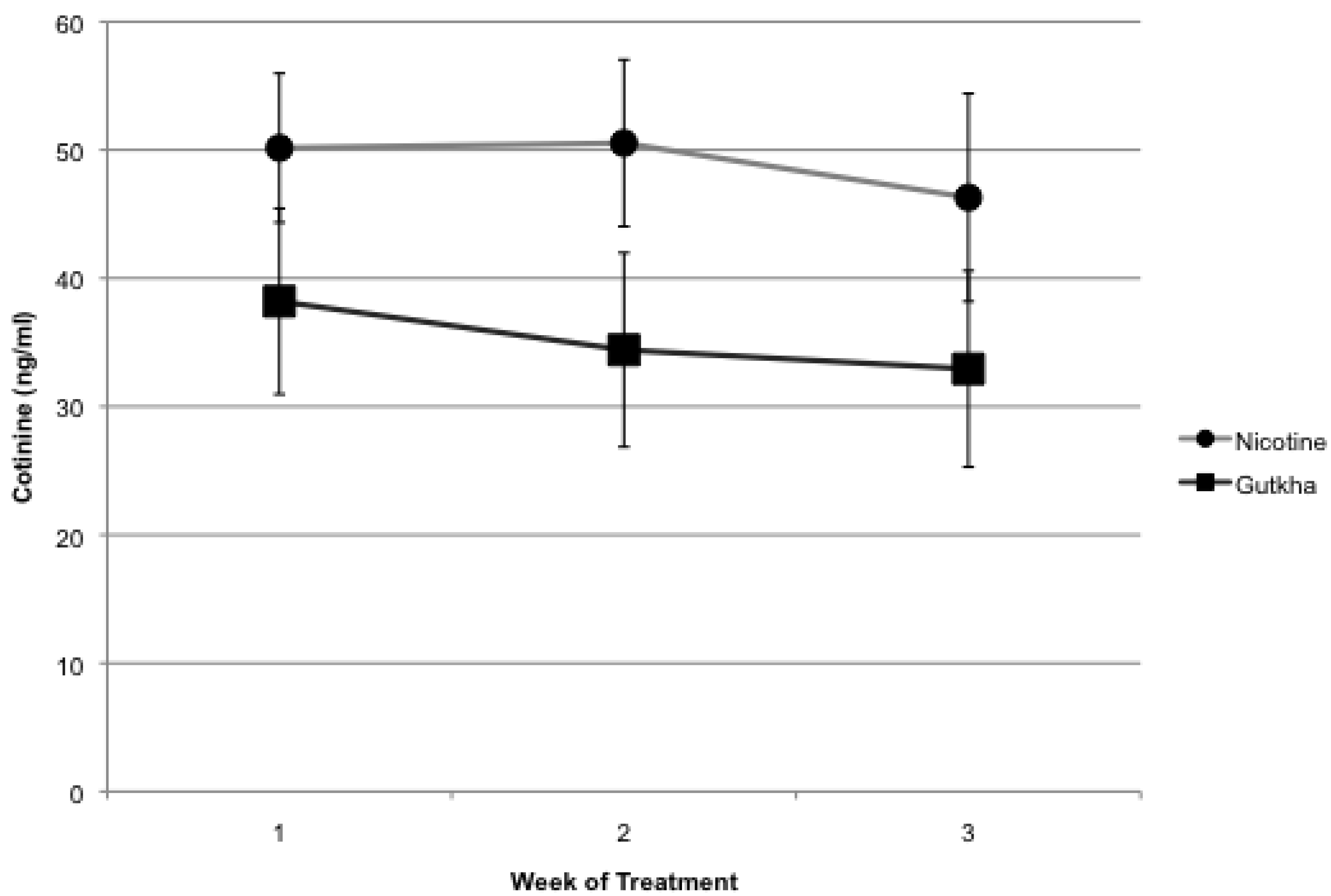
3.1. Serum Cotinine
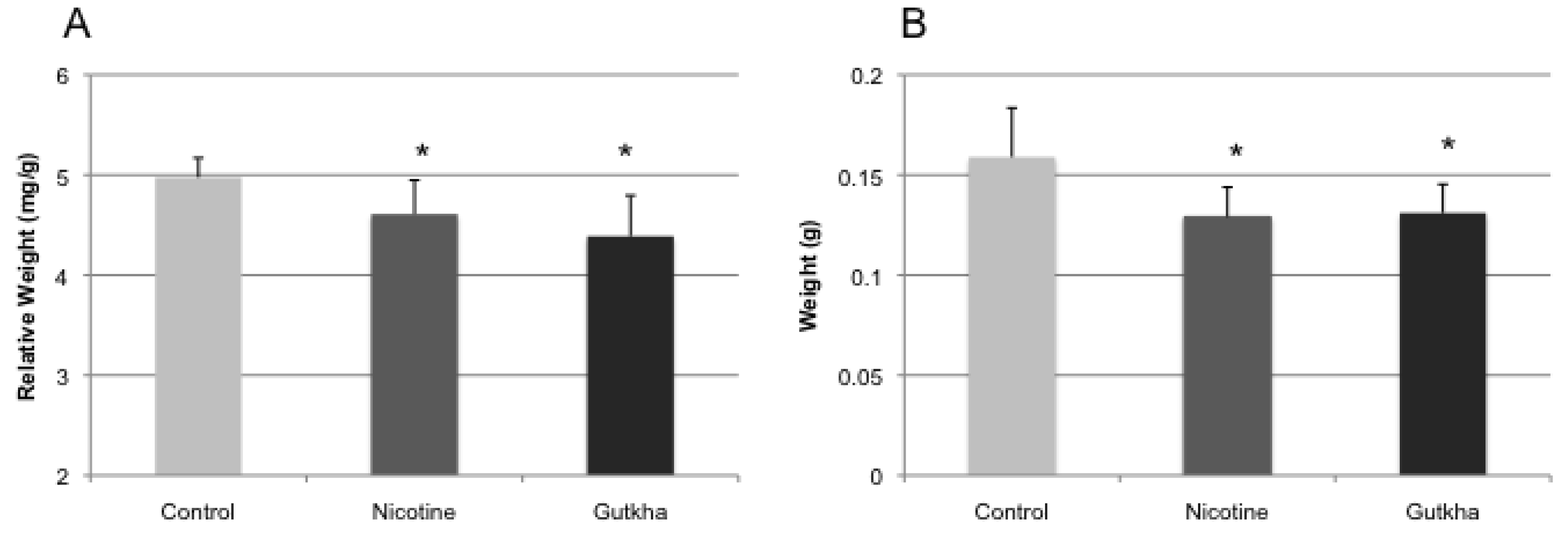
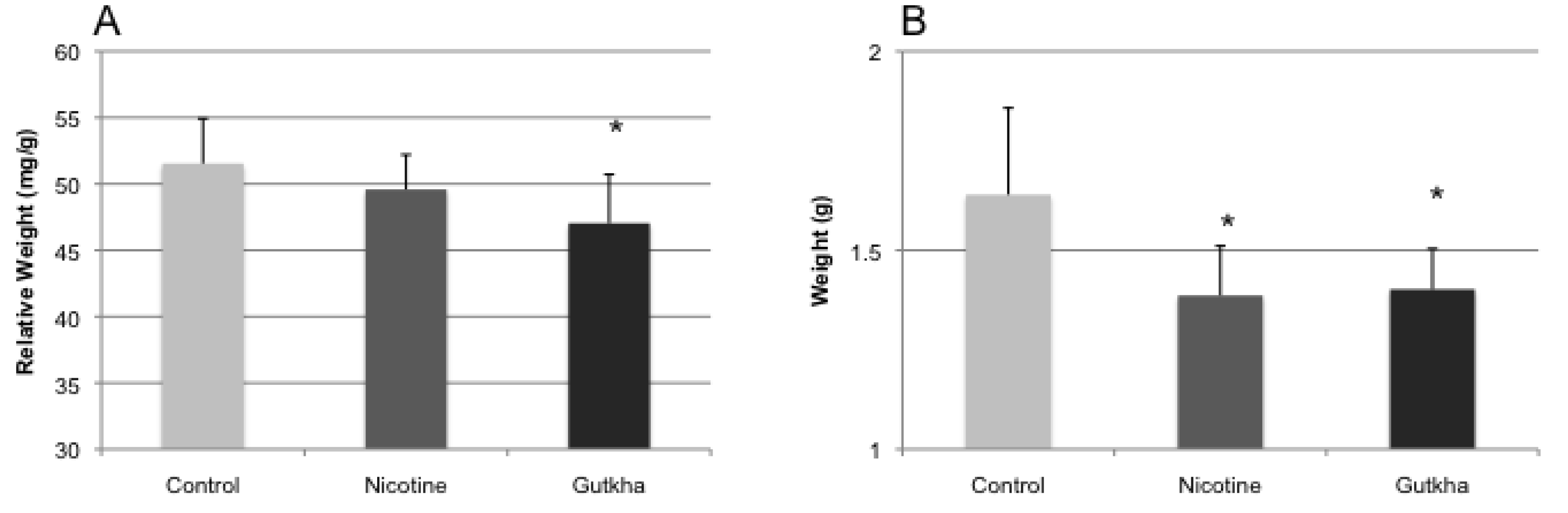
3.2. Body and Organ Weights

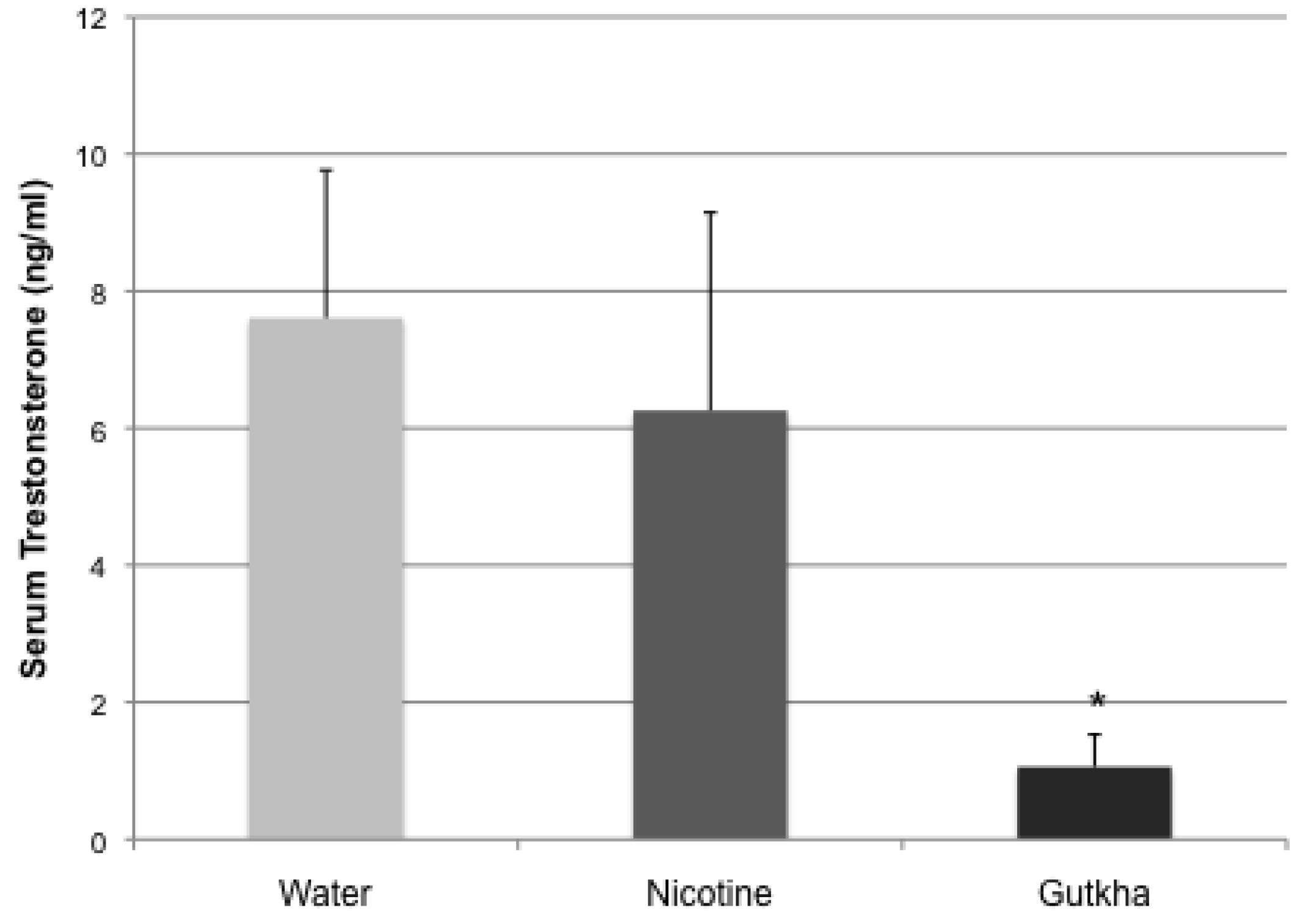
3.3. Serum Testosterone
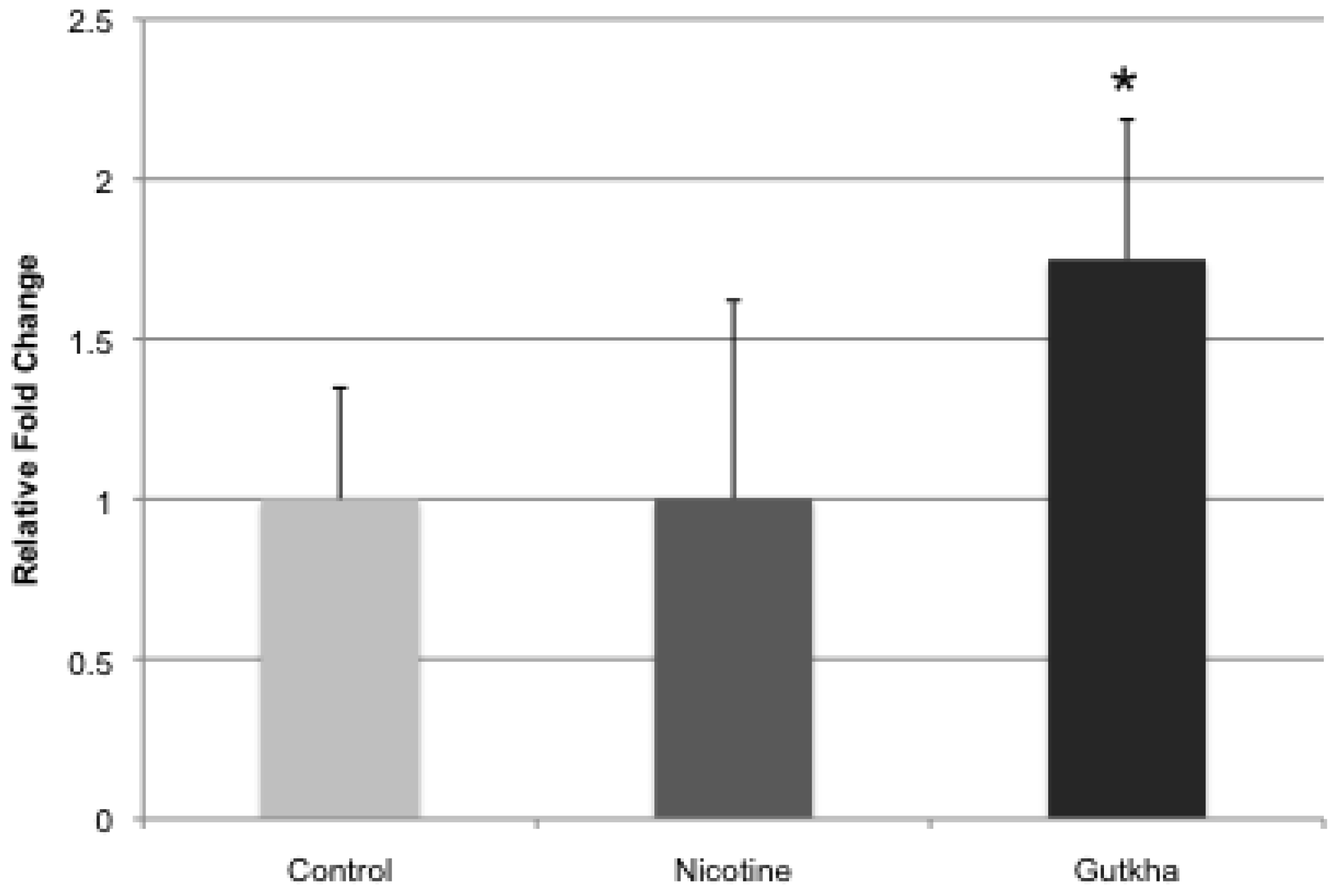
3.4. Liver CYP2A5 Expression
4. Discussion and Conclusions
Acknowledgements
Conflicts of Interest
References
- Tobacco Free Initiative. Tobacco: Deadly in Any Form or Disguise; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Mejia, A.B.; Ling, P.M. Tobacco industry consumer research on smokeless tobacco users and product development. Amer. J. Public Health 2010, 100, 78–87. [Google Scholar] [CrossRef]
- Bahreinifar, S.; Sheon, N.M.; Ling, P.M. Is snus the same as dip? Smokers’ perceptions of new smokeless tobacco advertising. Tob. Control 2013, 22, 84–90. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). State-specific prevelance of cigarette smoking and smokeless tobacco use among adults—United States, 2009. MMWR Morb. Mortal. Wkly. Rep. 2010, 59, 1400–1406. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Current tobacco use among middle and high school students—United States, 2011. MMWR Morb. Mortal. Wkly. Rep. 2012, 161, 581–585. [Google Scholar]
- Changrani, J.; Gany, F. Paan and Gutka in the United States: An emerging threat. J. Immigr. Health 2005, 7, 103–108. [Google Scholar] [CrossRef]
- CDC. Fact Sheet: Betel Quid with Tobacco (Gutka) 2007. Available online: http://www.cdc.gov/tobacco/data_statistics/fact_sheets/smokeless/betel_quid/ (accessed on 8 January 2014).
- Strickland, S. Anthropological perspectives on the use of areca nut. Add. Biol. 2002, 7, 85–97. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer (IARC). Smokeless Tobacco and Some Tobacco-Specific N-Nitrosamines. International Agency for Research on Cancer Monographs on the Evaluation of Carcinogenic Risks to Humans; World Health Organization: Geneva, Switzerland, 2007; Volume 89. [Google Scholar]
- Reddy, K.S.; Perry, C.L.; Stigler, M.H.; Arora, M. Differences in tobacco use among young people in urban India by sex, socioeconomic status, age, and school grade: Assessment of baseline survey data. Lancet 2006, 367, 589–594. [Google Scholar] [CrossRef]
- World Health Organization, Regional Office for South-East Asia. Global Adult Tobacco Survey (GATS): India Country Report. New Delhi: WHO-SEARO. 2009. Available online: http://www.searo.who.int/LinkFiles/Regional_Tobacco_Surveillance_System_GATS_India.pdf (accessed on 8 January 2014).
- Population Reference Bureau. 2009. Available online: http://www.prb.org/Publications/Datasheets/2009/2009wpds.aspx (accessed on 8 January 2014).
- Barnes, P.M.; Adams, P.F.; Powell-Griner, E. Health characteristics of the Asian adult population: United States, 2004–2006. Adv. Data 2008, 394, 1–22. [Google Scholar]
- National Cancer Institute, Centers for Disease Control and Prevention & Stockholm Centre of Public Health. Smokeless Tobacco Fact Sheets. In Proceedings of the Paper Presented at the Third International Conference on Smokeless Tobacco, Stockholm, Sweden, 22–25 September 2002.
- Borgerding, M.F.; Bodnar, J.A.; Curtin, G.M.; Swauger, J.E. The chemical composition of smokeless tobacco: A survey of products sold in the United States in 2006 and 2007. Regul. Toxicol. Pharmacol. 2012, 64, 367–387. [Google Scholar] [CrossRef]
- Willis, D.; Popovech, M.; Gany, F.; Zelikoff, J. Toxicology of smokeless tobacco: Implications for immune, reproductive, and cardiovascular systems. J. Toxicol. Environ. Health B Crit. Rev. 2012, 15, 317–331. [Google Scholar] [CrossRef]
- Johnson, G.K.; Slach, N.A. Impact of tobacco use on periodontal status. J. Dent. Educ. 2001, 65, 313–321. [Google Scholar]
- Aro, P.; Ronkainen, J.; Storskrubb, T.; Vieth, M.; Engstrand, L.; Johansson, S.E.; Bolling-Sternevald, E.; Bolinder, G.; Alving, K.; Talley, N.J.; et al. Use of tobacco products and gastrointestinal morbidity: An endoscopic population-based study (the Kalixanda study). Eur. J. Epidemiol. 2010, 25, 741–750. [Google Scholar] [CrossRef]
- Shankaran, K.; Kandarkar, S.V.; Contractor, Q.Q.; Kalro, R.H.; Desai, H.G. Electron microscopic observations in gastric mucosa of habitual tobacco chewers. Indian J. Med. Res. 1994, 99, 267–271. [Google Scholar]
- Boffetta, P.; Aagnes, B.; Weiderpass, E.; Andersen, A. Smokeless tobacco use and risk of cancer of the pancreas and other organs. Int. J. Cancer 2005, 114, 992–995. [Google Scholar] [CrossRef]
- Johnson, G.K.; Poore, T.K.; Squier, C.A.; Wertz, P.W.; Reinhardt, R.A.; Vincent, R.A. Prostaglandin E2 and interleukin-1 levels in smokeless tobacco-induced oral mucosal lesions. J. Period. Res. 1994, 29, 430–438. [Google Scholar] [CrossRef]
- Trivedy, C.R.; Craig, G.; Warnakulasuriya, S. The oral health consequences of chewing areca nut. Add. Biol. 2002, 7, 115–125. [Google Scholar] [CrossRef]
- Chao, A.; Thun, M.J.; Henley, S.J.; Jacobs, E.J.; McCullough, M.L.; Calle, E.E. Cigarette smoking, use of other tobacco products and stomach cancer mortality in US adults: The cancer prevention study II. Int. J. Cancer 2002, 101, 380–389. [Google Scholar] [CrossRef]
- Rodu, B.; Cole, P. Smokeless tobacco use among men in the United States, 2000 and 2005. J. Oral Path. Med. 2009, 38, 545–550. [Google Scholar] [CrossRef]
- Timberlake, D.S.; Huh, J. Demographic pro- files of smokeless tobacco users in the U.S. S. Amer. J. Prev. Med. 2009, 37, 29–34. [Google Scholar] [CrossRef]
- Foulds, J.; Ramstrom, L.; Burke, M.; Fagerstrom, K. Effect of smokeless tobacco (snus) on smoking and public health in Sweden. Tob. Control. 2003, 12, 349–359. [Google Scholar] [CrossRef]
- Hatsukami, D.K.; Lemmonds, C.; Tomar, S.L. Smokeless tobacco use: Harm reduction or induction approach? Prev. Med. 2004, 38, 309–317. [Google Scholar] [CrossRef]
- Levy, D.T.; Mumford, E.A.; Cummings, K.M.; Gilpin, E.A.; Giovino, G.; Hyland, A.; Warner, K.E. The relative risks of a low-nitrosamine smokeless tobacco product compared with smoking cigarettes: Estimates of a panel of experts. Cancer Epidem. Biomarker. Prev. 2004, 13, 2035–2042. [Google Scholar]
- O’Connor, R.J.; Hyland, A.; Giovino, G.A.; Fong, G.T.; Cummings, K.M. Smoker awareness of and beliefs about supposedly less-harmful tobacco products. Amer. J. Prev. Med. 2005, 29, 85–90. [Google Scholar] [CrossRef]
- O’Connor, R.J.; McNeill, A.; Borland, R.; Hammond, D.; King, B.; Boudreau, C.; Cummings, K.M. Smokers’ beliefs about the relative safety of other tobacco products: Findings from the ITC collaboration. Nicotine Tob. Res. 2007, 9, 1033–1042. [Google Scholar] [CrossRef]
- Kumari, A.; Mojidra, B.; Gautam, A.; Verma, Y.; Kumar, S. Reproductive toxic potential of panmasala in male Swiss albino mice. Toxicol. Ind. Health. 2011, 27, 683–690. [Google Scholar] [CrossRef]
- Avti, P.K.; Kumar, S.; Pathak, C.M.; Vaiphei, K.; Khanduja, K.L. Smokeless tobacco impairs the antioxidant defense in liver, lung, and kidney of rats. Toxicol. Sci. 2006, 89, 547–553. [Google Scholar] [CrossRef]
- Blum, J.L.; Nyagode, B.A.; James, M.O.; Denslow, N.D. Effects of the pesticide methoxychlor on gene expression in the liver and testes of the male largemouth bass (Micropterus salmoides). Aquat. Toxicol. 2008, 86, 459–469. [Google Scholar] [CrossRef]
- Coggins, C.R.; Ballantyne, M.; Curvall, M; Rutqvist, L.E. The in vitro toxicology of Swedish snus. Crit. Rev. Toxicol. 2012, 42, 304–313. [Google Scholar] [CrossRef]
- Costea, D.E.; Lukandu, O.; Bui, L.; Ibrahim, M.J.; Lygre, R.; Neppelberg, E.; Ibrahim, S.O.; Vintermyr, O.K.; Johannessen, A.C. Adverse effects of Sudanese toombak vs. Swedish snuff on human oral cells. J. Oral Pathol. Med. 2010, 39, 128–140. [Google Scholar] [CrossRef]
- Joyce, A.R.; Hawkins, W.; Fariss, M.W.; Sengupta, T.K. Role of plasma membrane disruption in reference moist smokeless tobacco-induced cell death. Toxicol. Lett. 2010, 198, 191–199. [Google Scholar] [CrossRef]
- Archana, K.; Gautam, A.K.; Lakkad, B.C.; Kumar, S. In utero and lactation exposure of mice to pan masala: Effect on dams and pregnancy outcome. J. Environ. Pathol. Toxicol. Oncol. 2011, 30, 71–81. [Google Scholar] [CrossRef]
- Mojidra, B.N.; Archana, K.; Gautam, A.K.; Verma, Y.; Lakkad, B.C.; Kumar, S. Evaluation of genotoxicity of pan masala employing chromosomal aberration and micronucleus assay in bone marrow cells of the mice. Toxicol. Ind. Health 2009, 25, 467–471. [Google Scholar] [CrossRef]
- Armitage, A.K.; Turner, D.M. Absorption of nicotine in cigarette and cigar smoke through the oral mucosa. Nature 1970, 226, 1231–1232. [Google Scholar] [CrossRef]
- Jana, K.; Jana, S.; Samanta, P.K. Effects of chronic exposure to sodium arsenite on hypothalamo-pituitary- testicular activities in adult rats: Possible an estrogenic mode of action. Reprod. Biol. Endocrinol. 2006, 4, 9–22. [Google Scholar] [CrossRef]
- Sarma, A.B.; Charkravarti, J.; Chakraborti, A.; Banerjee, T.S.; Roy, D.; Mukherjee, D.; Mukherjee, A. Evaluation of panmasala for toxic effects on liver and other organs. Food Chem. Toxicol. 1992, 30, 161–163. [Google Scholar] [CrossRef]
- Said, T.M.; Ranga, G.; Agarwal, A. Relationship between semen quality and tobacco chewing in men undergoing infertility evaluation. Fert. Steril. 2005, 84, 649–653. [Google Scholar] [CrossRef]
- Dikshit, R.K.; Buch, J.G.; Mansuri, S.M. Effect of tobacco consumption on semen quality of a population of hypofertile males. Fert. Steril. 1987, 48, 334–336. [Google Scholar]
- Patterson, T.R.; Stringham, J.D.; Meikle, A.W. Nicotine and cotinine inhibit steroidogenesis in mouse Leydig cells. Life Sci. 1990, 46, 265–272. [Google Scholar] [CrossRef]
- Audrain-McGovern, J.; Benowitz, N.L. Cigarette smoking, nicotine, and body weight. Clin. Pharmacol. Ther. 2011, 90, 164–168. [Google Scholar] [CrossRef]
- Chiolero, A.; Faeh, D.; Paccaud, F.; Cornuz, J. Consequences of smoking for body weight, body fat distribution, and insulin resistance. Amer. J. Clin. Nut. 2008, 87, 801–809. [Google Scholar]
- Nicklas, B.J.; Tomoyasu, N.; Muir, J.; Goldberg, A.P. Effects of cigarette smoking and its cessation on body weight and plasma leptin levels. Metabolism 1999, 48, 804–808. [Google Scholar] [CrossRef]
- Moffatt, R.J.; Owens, S.G. Cessation from cigarette smoking: Changes in body weight, body composition, resting metabolism, and energy consumption. Metabolism. 1991, 40, 465–470. [Google Scholar] [CrossRef]
- Stamford, B.A.; Matter, S.; Fell, R.D.; Papanek, P. Effects of smoking cessation on weight gain, metabolism rate, caloric consumption, and blood lipids. Amer. J. Clin. Nutr. 1986, 43, 486–494. [Google Scholar]
- Filozof, C.; Fernandez-Pinilla, M.C.; Fernandez-Cruz, A. Smoking cessation and weight gain. Obes. Rev. 2004, 5, 95–103. [Google Scholar] [CrossRef]
- Bellinger, L.; Cepeda-Benito, A.; Wellman, P.J. Meal patterns in male rats during and after intermittent nicotine administration. Pharmacol. Biochem. Behav. 2003, 74, 495–504. [Google Scholar] [CrossRef]
- Zhou, X.; Zhuo, X.; Xie, F.; Kluetzman, K.; Shu, Y.Z.; Humphreys, W.G.; Ding, X. Role of CYP2A5 in the clearance of nicotine and cotinine: Insights from studies on a Cyp2a5-nullmouse model. J. Pharmacol. Exp. Ther. 2010, 332, 578–587. [Google Scholar] [CrossRef]
- Wong, H.L.; Murphy, S.E.; Hecht, S.S. Cytochrome P450 2A-catalyzed metabolic activation of structurally similar carcinogenic nitrosamines: N-nitrosonornicotine enantiomers, N-nitrosopiperidine, and N-nitrosopyrrolidine. Chem. Res. Toxicol. 2005, 18, 61–69. [Google Scholar] [CrossRef]
- Richter, P.; Hodge, K.; Stanfill, S.; Zhang, L.; Watson, C. Surveillance of moist snuff: Total nicotine, moisture, pH, un-ionized nicotine, and tobacco-specific nitrosamines. Nicotine Tob. Res. 2008, 10, 1645–1652. [Google Scholar] [CrossRef]
- Stanfill, S.B.; Connolly, G.N.; Zhang, L.; Jia, L.T.; Henningfield, J.E.; Richter, P.; Lawler, T.S.; Ayo-Yusuf, O.A.; Ashley, D.L.; Watson, C.H. Global surveillance of oral tobacco products: Total nicotine, unionised nicotine and tobacco-specific N-nitrosamines. Tob. Control. 2011, 20. [Google Scholar] [CrossRef]
- Gu, J.; Dudley, C.; Su, T.; Spink, D.C.; Zhang, Q.Y.; Moss, R.L.; Ding, X. Cytochrome P450 and steroid hydroxylase activity in mouse olfactory and vomeronasal mucosa. Biochem. Biophys. Res. Commun. 1999, 266, 262–267. [Google Scholar] [CrossRef]
- Raunio, H.; Hakkola, J.; Pelkonen, O. The CYP2A Subfamily, in Cytochrome P450 Role in Metabolism and Toxicity of Drugs and Other Xenobiotics; Ioannides, C., Ed.; Advancing the Chemical Sciences Publishing: Cambridge, UK, 2008; pp. 151–171. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Willis, D.N.; Popovech, M.A.; Gany, F.; Hoffman, C.; Blum, J.L.; Zelikoff, J.T. Toxicity of Gutkha, a Smokeless Tobacco Product Gone Global: Is There More to the Toxicity than Nicotine? Int. J. Environ. Res. Public Health 2014, 11, 919-933. https://doi.org/10.3390/ijerph110100919
Willis DN, Popovech MA, Gany F, Hoffman C, Blum JL, Zelikoff JT. Toxicity of Gutkha, a Smokeless Tobacco Product Gone Global: Is There More to the Toxicity than Nicotine? International Journal of Environmental Research and Public Health. 2014; 11(1):919-933. https://doi.org/10.3390/ijerph110100919
Chicago/Turabian StyleWillis, Daniel N., Mary A. Popovech, Francesca Gany, Carol Hoffman, Jason L. Blum, and Judith T. Zelikoff. 2014. "Toxicity of Gutkha, a Smokeless Tobacco Product Gone Global: Is There More to the Toxicity than Nicotine?" International Journal of Environmental Research and Public Health 11, no. 1: 919-933. https://doi.org/10.3390/ijerph110100919
APA StyleWillis, D. N., Popovech, M. A., Gany, F., Hoffman, C., Blum, J. L., & Zelikoff, J. T. (2014). Toxicity of Gutkha, a Smokeless Tobacco Product Gone Global: Is There More to the Toxicity than Nicotine? International Journal of Environmental Research and Public Health, 11(1), 919-933. https://doi.org/10.3390/ijerph110100919



