Scoping the Impact of Changes in Population Age-Structure on the Future Burden of Foodborne Disease in The Netherlands, 2020–2060
Abstract
:1. Introduction
2. Data and Methods
2.1. Population Data
2.2. Incidence Estimation
2.3. Excess Mortality Estimation
3. Results and Discussion
3.1. Demographic Changes
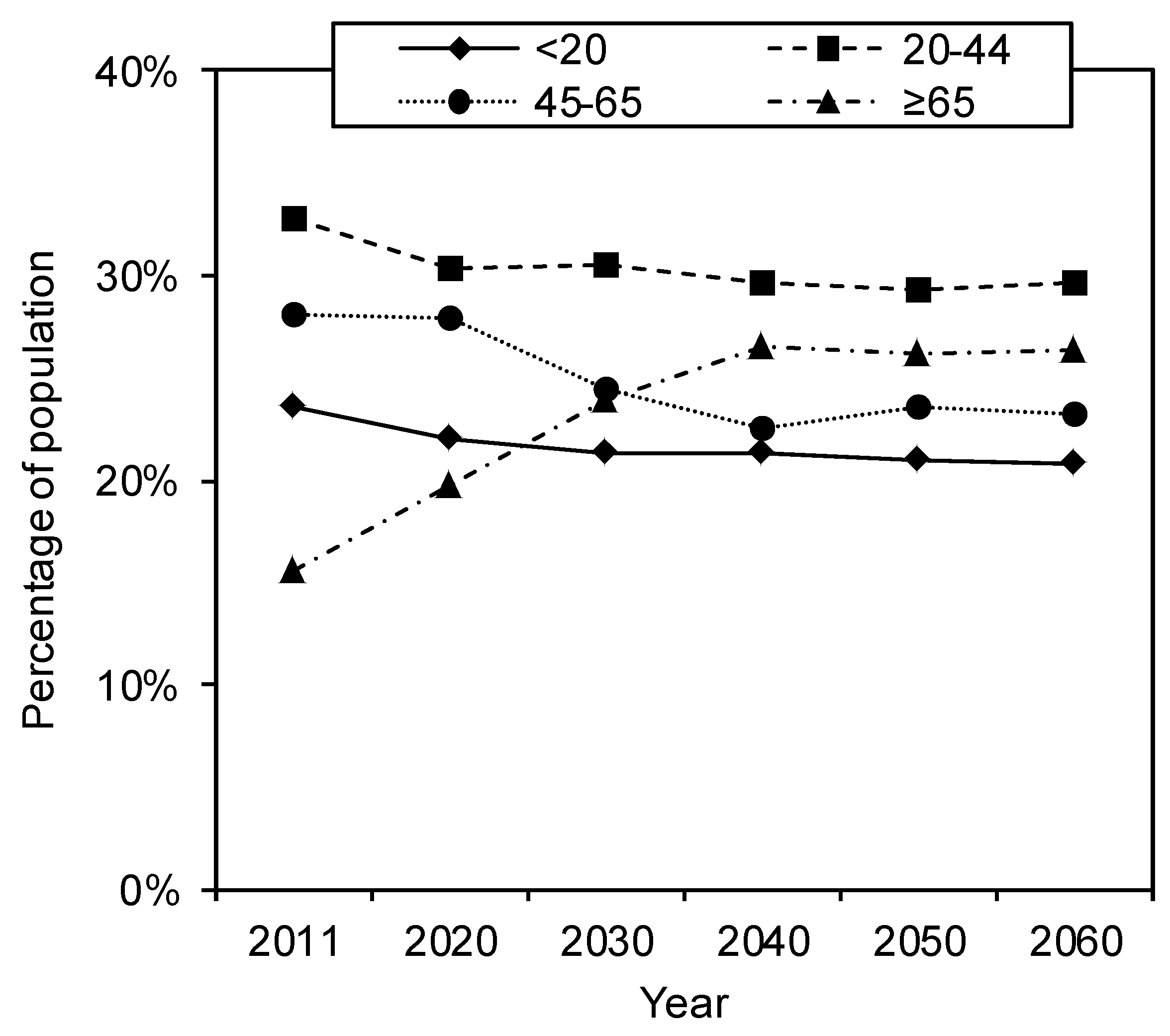
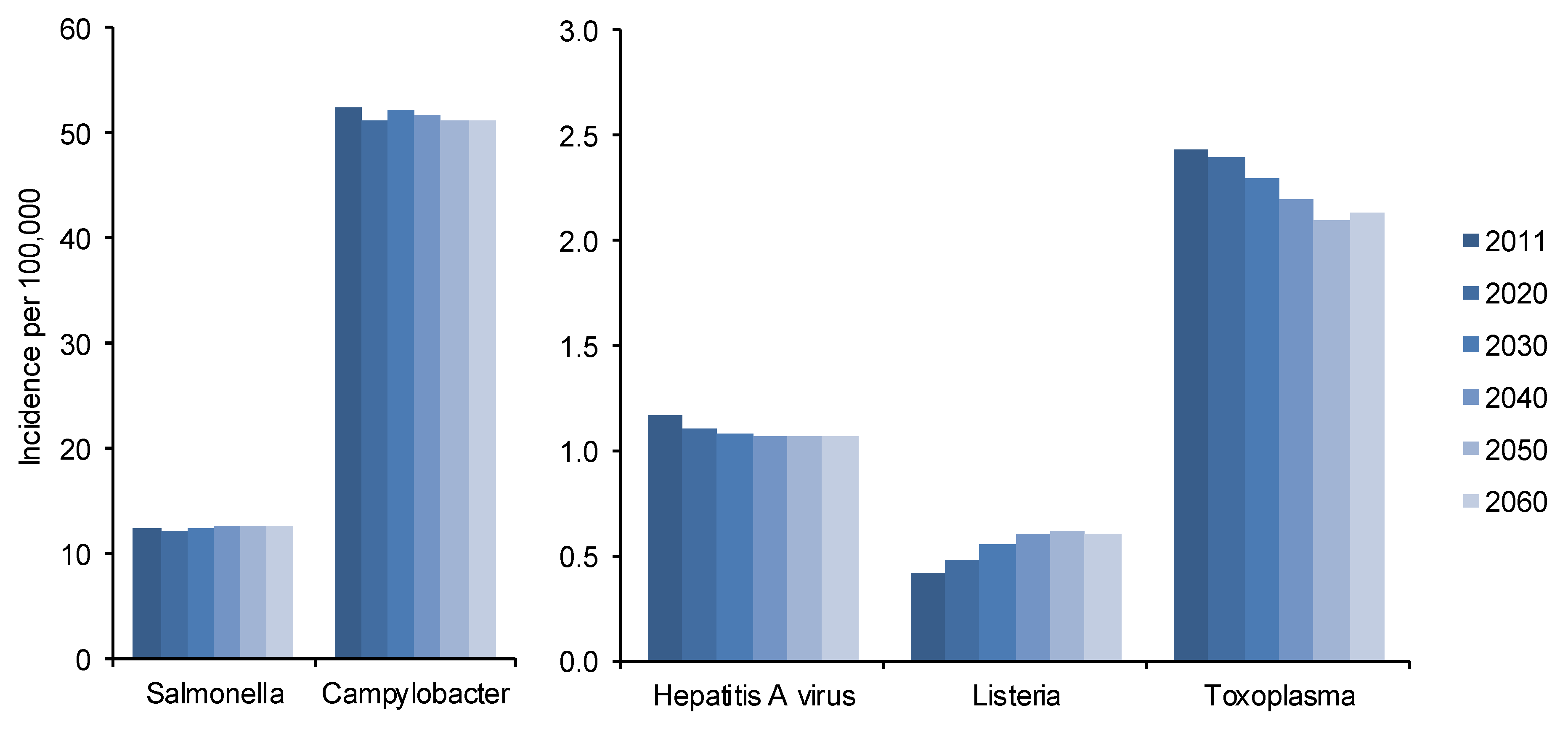
3.2. Incidence Estimation
| Age | Salmonella 1 | Campylobacter 1 | HAV 2 | Listeria 3 | Fatal List 3 | Toxoplasma 4 |
|---|---|---|---|---|---|---|
| 0 | 0.03 | 0.06 | 1.2 × 10−4 | 7.3 × 10−5 | 0 | 0 |
| 1–4 | 0.03 | 0.05 | 1.5 × 10−3 | 1.8 × 10−5 | 0 | 0 |
| 5–9 | 0.02 | 0.03 | 2.9 × 10−3 | 1.4 × 10−5 | 0 | 0 |
| 10–14 | 0.01 | 0.03 | 2.2 × 10−3 | 0 | 0 | 0 |
| 15–19 | 0.02 | 0.07 | 1.6 × 10−3 | 6.7 × 10−5 | 0 | 0 |
| 20–24 | 0.02 | 0.10 | 1.8 × 10−3 | 9.1 × 10−5 | 0 | 0.85 |
| 25–29 | 0.01 | 0.06 | 1.3 × 10−3 | 1.8 × 10−4 | 0 | 0.85 |
| 30–34 | 0.01 | 0.04 | 1.0 × 10−3 | 2.3 × 10−4 | 0 | 0.85 |
| 35–39 | 0.01 | 0.04 | 1.6 × 10−3 | 2.5 × 10−4 | 0 | 0.85 |
| 40–44 | 0.01 | 0.04 | 1.2 × 10−3 | 5.2 × 10−5 | 1.1 × 10−5 | 0.85 |
| 45–49 | 0.01 | 0.04 | 9.4 × 10−4 | 1.5 × 10−4 | 1.1 × 10−5 | 0.85 |
| 50–54 | 0.01 | 0.06 | 7.8 × 10−4 | 2.3 × 10−4 | 8.4 × 10−5 | 0.85 |
| 55–59 | 0.01 | 0.06 | 7.0 × 10−4 | 4.5 × 10−4 | 1.2 × 10−4 | 0.85 |
| 60–64 | 0.01 | 0.05 | 4.1 × 10−4 | 5.8 × 10−4 | 1.8 × 10−4 | 0.85 |
| 65–69 | 0.01 | 0.06 | 2.8 × 10−4 | 1.0 × 10−3 | 1.6 × 10−4 | 0 |
| 70–74 | 0.01 | 0.06 | 1.4 × 10−4 | 1.6 × 10−3 | 3.1 × 10−4 | 0 |
| 75–79 | 0.02 | 0.06 | 2.6 × 10−4 | 1.9 × 10−3 | 3.1 × 10−4 | 0 |
| 80–84 | 0.02 | 0.04 | 3.0 × 10−4 | 2.7 × 10−3 | 1.6 × 10−4 | 0 |
| 85–89 | 0.01 | 0.05 | 1.0 × 10−4 | 2.0 × 10−3 | 0 | 0 |
| ≥90 | 0.01 | 0.02 | 6.9 × 10−4 | 1.9 × 10−3 | 1.5 × 10−4 | 0 |
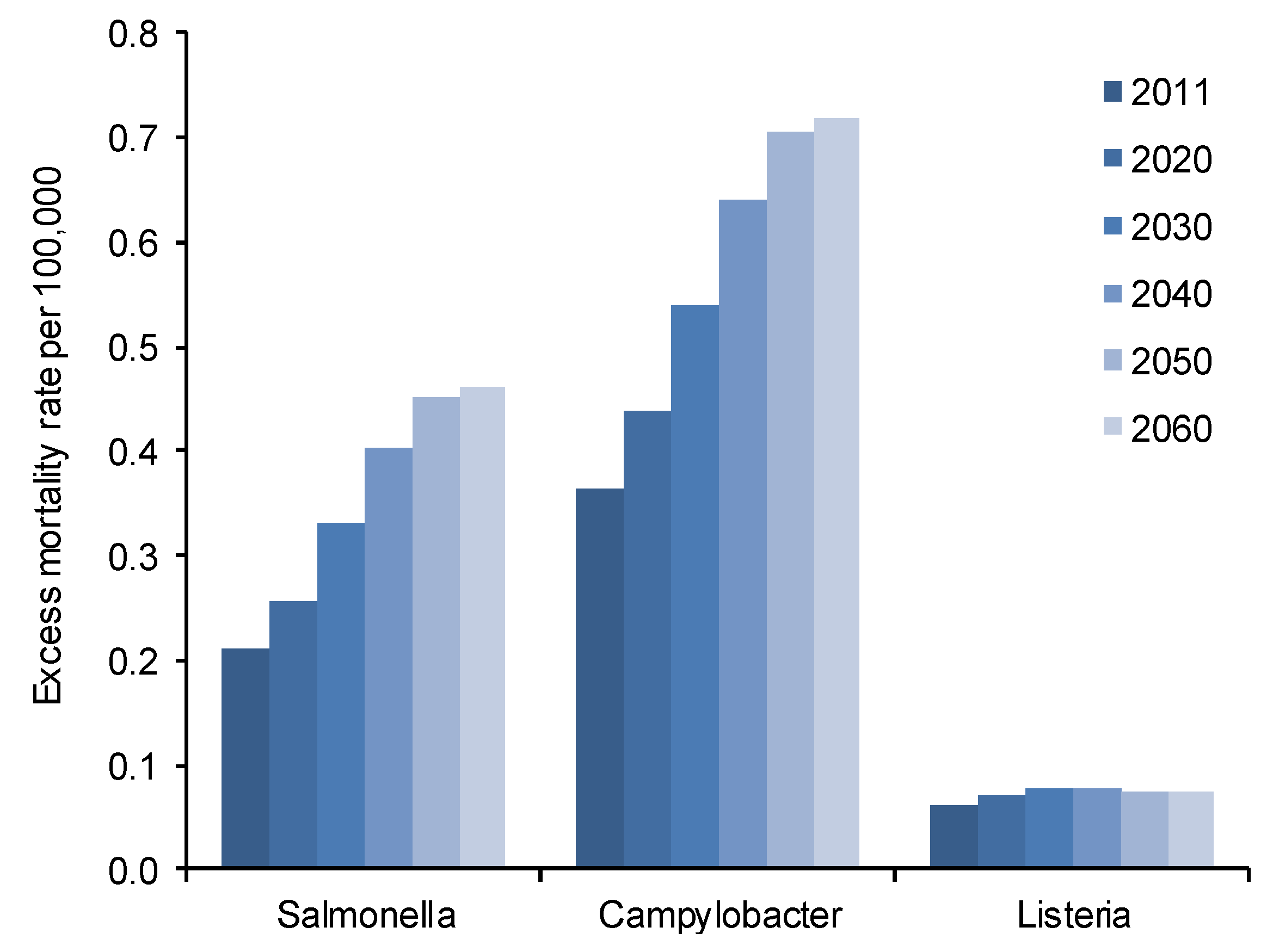
3.3. Excess Mortality Estimation
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Statistics Netherlands (CBS) Statline Database 2012. Available online: www.cbs.nl (accessed on 20 December 2012).
- Miller, R.A. The aging immune system: Primer and prospectus. Science 1996, 273, 70–74. [Google Scholar]
- Gavazzi, G.; Krause, K.H. Ageing and infection. Lancet Infect. Dis. 2002, 2, 659–666. [Google Scholar] [CrossRef]
- Russell, T.L.; Berardi, R.R.; Barnett, J.L.; Dermentzoglou, L.C.; Jarvenpaa, K.M.; Schmaltz, S.P.; Dressman, J.B. Upper gastrointestinal pH in seventy-nine healthy, elderly, North American men and women. Pharm. Res. 1993, 10, 187–196. [Google Scholar] [CrossRef]
- Bavishi, C.; Dupont, H.L. Systematic review: The use of proton pump inhibitors and increased susceptibility to enteric infection. Aliment. Pharmacol. Therapeut. 2011, 34, 1269–1281. [Google Scholar]
- Choi, C. Bacterial meningitis in aging adults. Clin. Infect. Dis. 2001, 33, 1380–1385. [Google Scholar] [CrossRef]
- Van Pelt, W.; de Wit, M.A.; Wannet, W.J.; Ligtvoet, E.J.; Widdowson, M.A.; van Duynhoven, Y.T. Laboratory surveillance of bacterial gastroenteric pathogens in The Netherlands, 1991–2001. Epidemiol. Infect. 2003, 130, 431–441. [Google Scholar]
- Friesema, I.H.M.; De Jager, C.M.; Van der Zwaluw, W.K.; Notermans, D.W.; Van Heerwaarden, C.A.M.; Heuvelink, A.E.; Van der Ende, A.; Spanjaard, L.; Van Pelt, W. Surveillance of Listeria monocytogenes in The Netherlands. Infectieziekten Bulletin 2012, 23, 15–18. [Google Scholar]
- Havelaar, A.H.; Haagsma, J.A.; Mangen, M.J.; Kemmeren, J.M.; Verhoef, L.P.; Vijgen, S.M.; Wilson, M.; Friesema, I.H.; Kortbeek, L.M.; van Duynhoven, Y.T.; van Pelt, W. Disease burden of foodborne pathogens in the Netherlands, 2009. Int. J. Food Microbiol. 2012, 156, 231–238. [Google Scholar] [CrossRef]
- Hofhuis, A.; van Pelt, W.; van Duynhoven, Y.T.; Nijhuis, C.D.; Mollema, L.; van der Klis, F.R.; Havelaar, A.H.; Kortbeek, L.M. Decreased prevalence and age-specific risk factors for Toxoplasma gondii IgG antibodies in The Netherlands between 1995/1996 and 2006/2007. Epidemiol. Infect. 2011, 139, 530–538. [Google Scholar] [CrossRef]
- Helms, M.; Vastrup, P.; Gerner-Smidt, P.; Molbak, K. Short and long term mortality associated with foodborne bacterial gastrointestinal infections: Registry based study. BMJ 2003, 326, 357. [Google Scholar] [CrossRef]
- Bouwknegt, M.; Van Pelt, W.; Kubbinga, M.; Weda, M.; Havelaar, A.H. Recent increase in campylobacteriosis incidence in The Netherlands potentially related to proton-pump inhibitor use. Lancet 2013, 381, S22. [Google Scholar] [CrossRef]
- Schenzle, D.; Dietz, K.; Frosner, G.G. Antibody against hepatitis A in seven European countries. II. Statistical analysis of cross-sectional surveys. Amer. J. Epidemiol. 1979, 110, 70–76. [Google Scholar]
- Verhoef, L.; Boot, H.J.; Koopmans, M.; Mollema, L.; Van Der Klis, F.; Reimerink, J.; Van Pelt, W. Changing risk profile of hepatitis A in The Netherlands: A comparison of seroprevalence in 1995–1996 and 2006–2007. Epidemiol. Infect. 2011, 139, 1172–1180. [Google Scholar]
- McDonald, S.A.; Mangen, M.J.; Suijkerbuijk, A.; Colzani, E.; Kretzschmar, M.E.E. Effects of an ageing population and the replacement of immune birth cohorts on the burden of hepatitis A in the Netherlands. BMC Infect. Dis. 2013, 13, 120. [Google Scholar] [CrossRef]
- Kijlstra, A.; Jongert, E. Control of the risk of human toxoplasmosis transmitted by meat. Int. J. Parasitol. 2008, 38, 1359–1370. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bouwknegt, M.; Van Pelt, W.; Havelaar, A.H. Scoping the Impact of Changes in Population Age-Structure on the Future Burden of Foodborne Disease in The Netherlands, 2020–2060. Int. J. Environ. Res. Public Health 2013, 10, 2888-2896. https://doi.org/10.3390/ijerph10072888
Bouwknegt M, Van Pelt W, Havelaar AH. Scoping the Impact of Changes in Population Age-Structure on the Future Burden of Foodborne Disease in The Netherlands, 2020–2060. International Journal of Environmental Research and Public Health. 2013; 10(7):2888-2896. https://doi.org/10.3390/ijerph10072888
Chicago/Turabian StyleBouwknegt, Martijn, Wilfrid Van Pelt, and Arie H. Havelaar. 2013. "Scoping the Impact of Changes in Population Age-Structure on the Future Burden of Foodborne Disease in The Netherlands, 2020–2060" International Journal of Environmental Research and Public Health 10, no. 7: 2888-2896. https://doi.org/10.3390/ijerph10072888
APA StyleBouwknegt, M., Van Pelt, W., & Havelaar, A. H. (2013). Scoping the Impact of Changes in Population Age-Structure on the Future Burden of Foodborne Disease in The Netherlands, 2020–2060. International Journal of Environmental Research and Public Health, 10(7), 2888-2896. https://doi.org/10.3390/ijerph10072888




