Evaluation of Electronic Cigarette Use (Vaping) Topography and Estimation of Liquid Consumption: Implications for Research Protocol Standards Definition and for Public Health Authorities’ Regulation
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Sample
2.2. Evaluation of Use Pattern
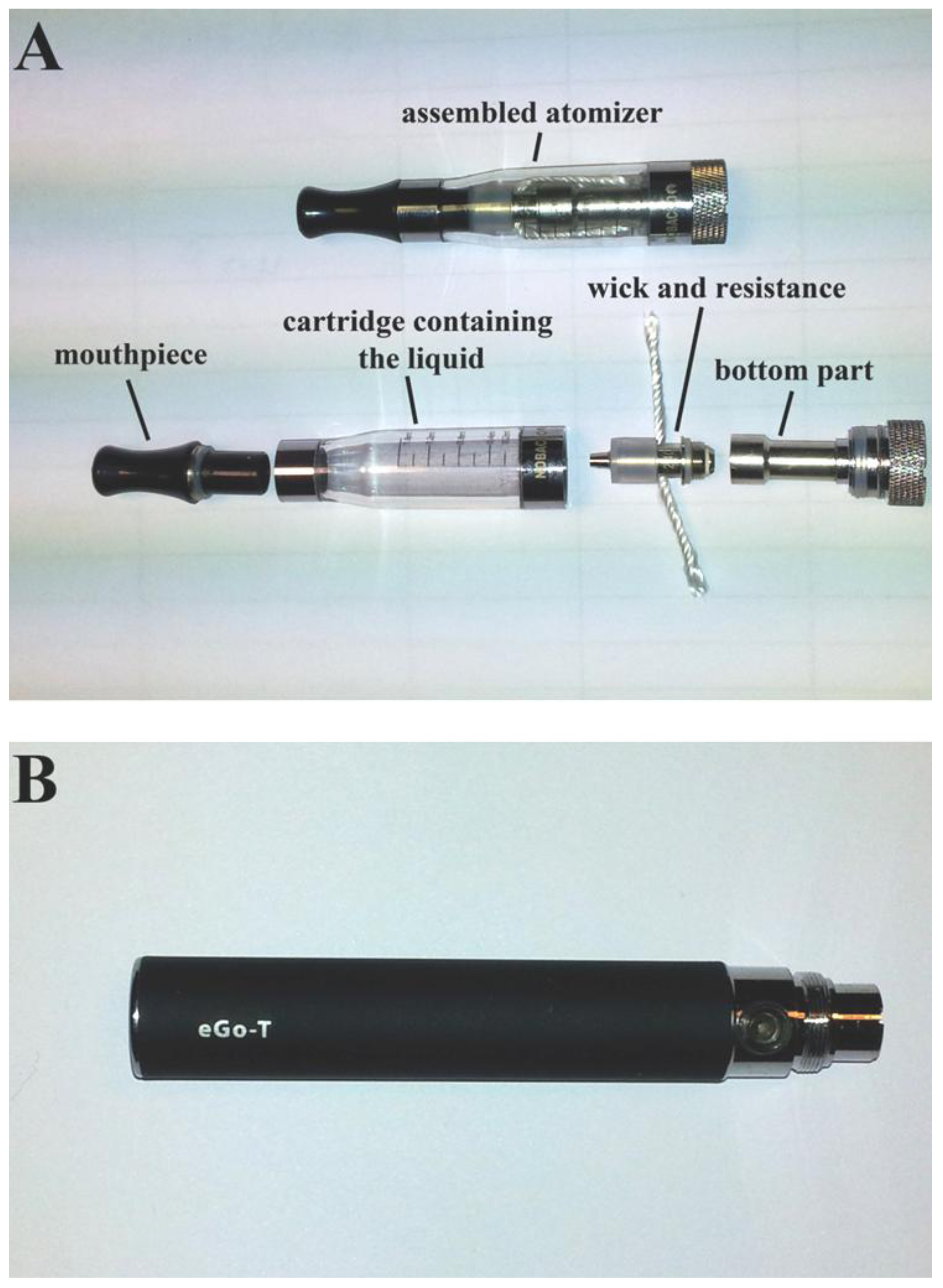
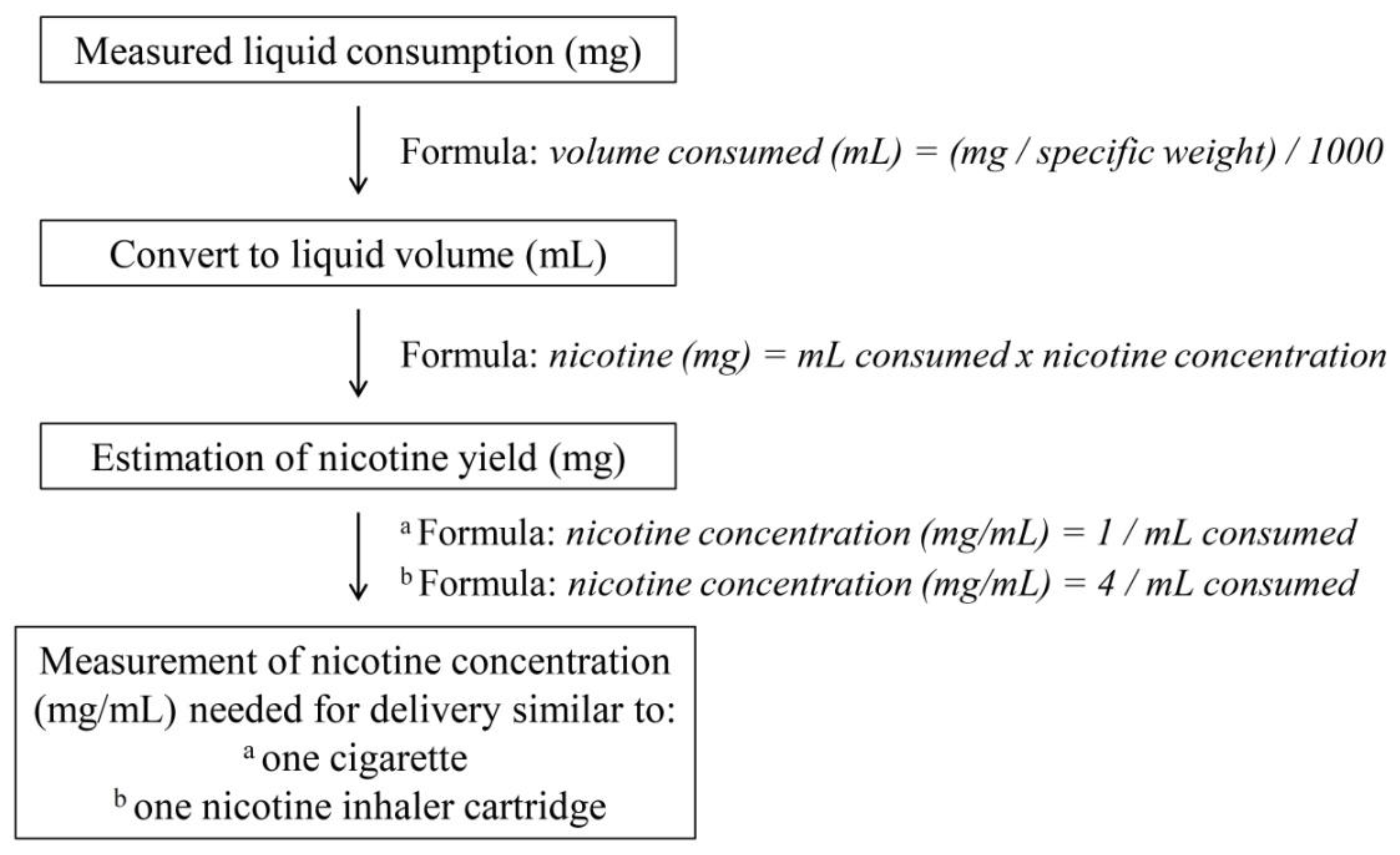
2.3. Evaluation of EC Liquid Consumption
2.4. Statistical Analysis
3. Results and Discussion
3.1. Characteristics of Study Groups
| Characteristic | Electronic cigarette users (n = 45) | Smokers (n = 35) | p-value |
|---|---|---|---|
| Age (years) | 38 ± 5 | 37 ± 5 | 0.219 |
| Gender (male) | 35 (77.8%) | 26 (74.3%) | 0.716 |
| Cigarette consumption (number per day) | 26 ± 8 | 24 ± 5 | 0.125 |
| Smoking duration (years) | 21 ± 6 | 19 ± 5 | 0.151 |
| Brinkman index | 571 ± 258 | 451 ± 178 | 0.021 |
| Electronic cigarette use (months) | 7 ± 3 | ||
| Electronic cigarette consumption (mL/day) | 5 ± 2 |
3.2. Topography in EC Users Compared with Smokers
| Measurements | ECIG | SM-S | SM-E | p-value (ECIG vs. SM-S) | p-value (ECIG vs. SM-E) | p-value (SM-S vs. SM-E) |
|---|---|---|---|---|---|---|
| Puff duration (s) | 4.2 ± 0.7 | 2.1 ± 0.4 | 2.4 ± 0.5 | <0.001 | <0.001 | 0.017 |
| Inhalation duration (s) | 1.3 ± 0.4 | 2.2 ± 0.4 | 2.0 ± 0.4 | <0.001 | <0.001 | <0.001 |
| Exhalation duration (s) | 1.7 ± 0.5 | 1.8 ± 0.4 | 1.7 ± 0.3 | 0.379 | 0.614 | 0.426 |
| Intraobserver error puff duration (%) | 3.6 ± 0.6 | 4.2 ± 1.2 | 3.3 ± 1.2 | |||
| Interobserver error puff duration (%) | 3.9 ± 1.4 | 5.2 ± 2.2 | 4.0 ± 1.6 | |||
| Intraobserver error inhalation duration (%) | 5.8 ± 2.5 | 4.6 ± 1.2 | 4.9 ± 1.8 | |||
| Interobserver error inhalation duration (%) | 8.3 ± 2.1 | 5.7 ± 1.5 | 7.0 ± 1.9 | |||
| Intraobserver error exhalation duration (%) | 5.8 ± 2.6 | 5.0 ± 1.7 | 6.0 ± 2.0 | |||
| Interobserver error exhalation duration (%) | 7.4 ± 1.5 | 6.6 ± 1.5 | 6.4 ± 2.1 |
3.3. Evaluation of EC Liquid Consumption
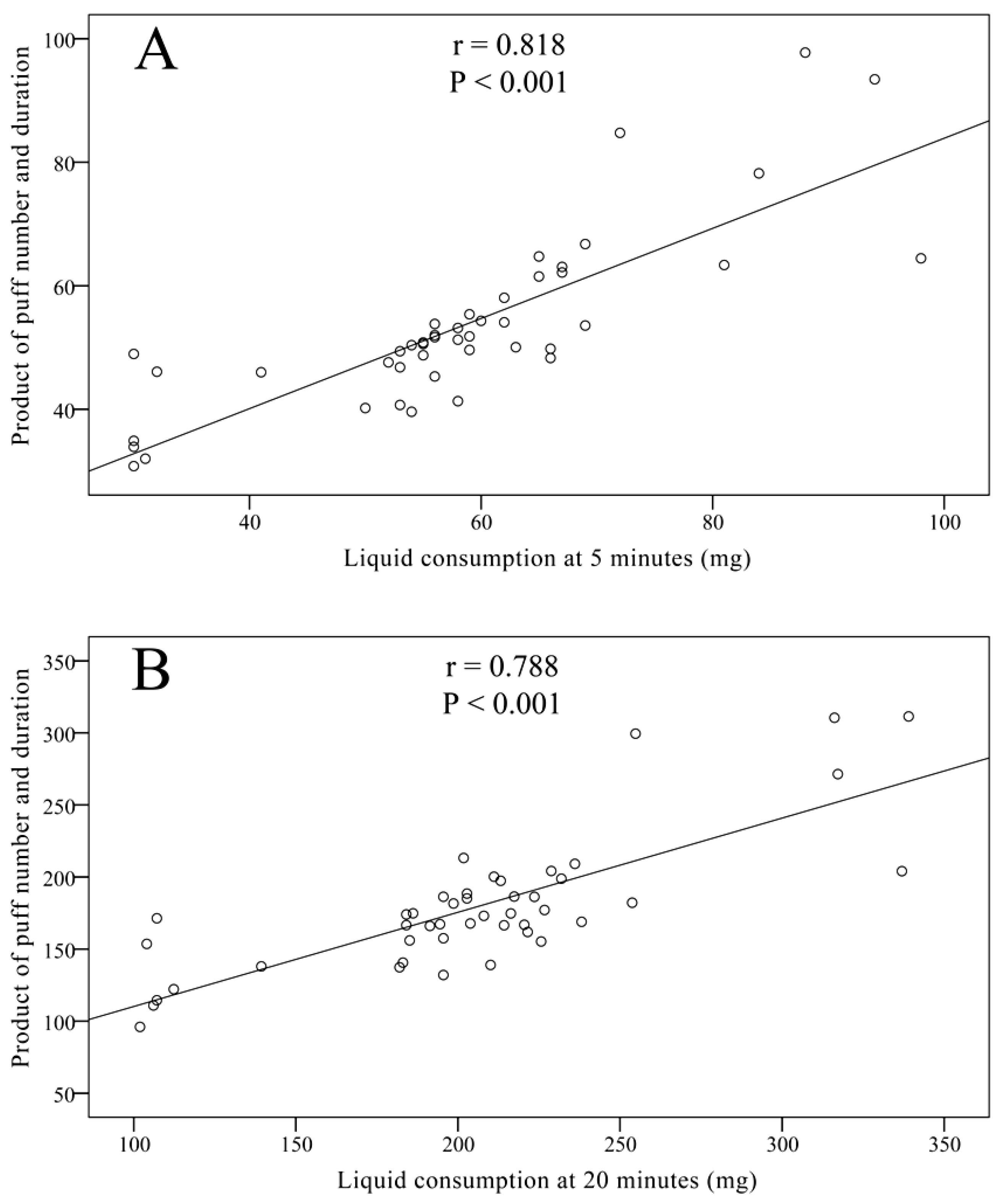
3.4. Discussion
3.4.1. Vaping vs. Smoking Topography
3.4.2. Implications from Evaluating Vaping Topography
3.4.3. Nicotine Delivery
3.4.4. Limitations
4. Conclusions
Acknowledgements
Conflict of Interest
References
- Rigotti, N.A.; Pipe, A.L.; Benowitz, N.L.; Arteaga, C.; Garza, D.; Tonstad, S. Efficacy and safety of varenicline for smoking cessation in patients with cardiovascular disease: A randomized trial. Circulation 2010, 121, 221–229. [Google Scholar] [CrossRef]
- Rodu, B.; Godshall, W.T. Tobacco harm reduction: An alternative cessation strategy for inveterate smokers. Harm Reduct. J. 2006, 3, 37. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C.; et al. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob. Control 2013. [Google Scholar] [CrossRef]
- ISO 3308: Routine Analytical Cigarette-Smoking Machine—Definitions and Standard Conditions; International Organization for Standardization (ISO): Geneva, Switzerland, 2000.
- McQueen, A.; Tower, S.; Sumner, W. Interviews with “vapers”: Implications for future research with electronic cigarettes. Nicotine Tob. Res. 2011, 13, 860–867. [Google Scholar] [CrossRef]
- Bullen, C.; Mc Robbie, H.; Thornley, S.; Glover, M.; Lin, R.; Laugesen, M. Effects of an electronic nicotine delivery device (ecigarette) on desire to smoke and withdrawal, user preferences and nicotine delivery: Randomised cross-over trial. Tob. Control 2010, 19, 98–103. [Google Scholar] [CrossRef]
- Vansickel, A.R.; Cobb, C.C.; Weaver, M.F.; Eissenberg, T.E. A clinical laboratory model for evaluating the acute effects of electronic “cigarettes”: Nicotine delivery profile and cardiovascular subjective effects. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1945–1953. [Google Scholar] [CrossRef]
- Vansickel, A.R.; Eissenberg, T. Electronic cigarettes: Effective nicotine delivery after acute administration. Nicotine Tob. Res. 2013, 15, 267–270. [Google Scholar] [CrossRef]
- European Commission. Proposal for a Directive of the European Parliament and of the Council on the Approximation of Laws, Regulations and Administrative Provisions of the Member States Concerning the Manufacture, Presentation and Sale of Tobacco and Related Products. Available online: http://ec.europa.eu/health/tobacco/docs/com_2012_ 788_en.pdf (accessed on 15 March 2013).
- Benowitz, N.L.; Jacob, P.; Savanapridi, C. Determinants of nicotine intake while chewing nicotine polacrilex gum. Clin. Pharmacol. Ther. 1987, 41, 467–473. [Google Scholar] [CrossRef]
- Schneider, N.G.; Olmstead, R.E.; Franzon, M.A.; Lunell, E. The nicotine inhaler: Clinical pharmacokinetics and comparison with other nicotine treatments. Clin. Pharmacokinet. 2001, 40, 661–684. [Google Scholar] [CrossRef]
- Dawkins, L.; Turner, J.; Roberts, A.; Soar, K. “Vaping” profiles and preferences: An online survey of electronic cigarette users. Addiction 2013, 108, 1115–1125. [Google Scholar] [CrossRef]
- Blank, M.D.; Dispharoon, S.; Eissenberg, T. Comparison of methods for measurement of smoking behaviour: Mouthpiece-based computerized devices versus direct observation. Nicotine Tob. Res. 2009, 11, 896–903. [Google Scholar]
- Breland, A.B.; Kleykamp, B.A.; Eissenberg, T. Clinical laboratory evaluation of potential reduced exposure products for smokers. Nicotine Tob. Res. 2006, 8, 727–738. [Google Scholar] [CrossRef]
- Pfizer. Nicotrol Inhaler Prescribing Information. Available online: http://www.pfizer.com/files/ products/uspi_nicotrol_inhaler.pdf (accessed on 24 May 2013).
- European Commision. Directive 2001/37/EC of the European Parliament and of the Council of 5 June 2001 on the Approximation of the Laws, Regulations and Administrative Provisions of the Member States Concerning the Manufacture, Presentation and Sale of Tobacco Products. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CONSLEG:2001 L0037: 20090807:EN:PDF (accessed on 25 May 2013).
- Purkis, S.W.; Troude, V.; Hill, C.A. Effect of puffing intensity on cigarette smoke yields. Regul. Toxicol. Pharmacol. 2013, 66, 72–82. [Google Scholar] [CrossRef]
- Djordjevic, M.V.; Fan, J.; Ferguson, S.; Hoffmann, D. Self-regulation of smoking intensity. Smoke yields of the low-nicotine, low-“tar” cigarettes. Carcinogenesis 1995, 16, 2015–2021. [Google Scholar]
- Eberhardt, H.J.; Scherer, G. Human smoking behavior in comparison with machine smoking methods: A summary of the five papers presented at the 1995 meeting of the CORESTA Smoke and Technology Groups in Vienna. Beitr. Tab. Intl. 1995, 16, 131–140. [Google Scholar]
- Rodu, B. The scientific foundation for tobacco harm reduction, 2006–2011. Harm Reduct. J. 2011, 8, 19. [Google Scholar] [CrossRef]
- Antal, M.J.; Mok, W.S.L.; Roy, J.C.; T-Raissi, A. Pyrolytic sources of hydrocarbons from biomass. J. Anal. Appl. Pyrolysis 1985, 8, 291–303. [Google Scholar]
- Stein, Y.S.; Antal, M.J.; Jones, M.J. A study of the gas-phase pyrolysis of glycerol. J. Anal. Appl. Pyrolysis 1983, 4, 283–296. [Google Scholar] [CrossRef]
- Etter, J.F.; Bullen, C. Electronic cigarette: Users profile, utilization, satisfaction and perceived efficacy. Addiction 2011, 106, 2017–2028. [Google Scholar] [CrossRef]
- Cahn, Z.; Siegel, M. Electronic cigarettes as a harm reduction strategy for tobacco control: A step forward or a repeat of past mistakes? J. Public Health Policy 2011, 32, 16–31. [Google Scholar]
- McNeill, A.; Munafò, M.R. Reducing harm from tobacco use. J. Psychopharmacol. 2013, 27, 13–18. [Google Scholar]
- Farsalinos, K.; Tsiapras, D.; Kyrzopoulos, S.; Voudris, V. Acute and chronic effects of smoking on myocardial function in healthy heavy smokers: A study of Doppler flow, Doppler tissue velocity and two-dimensional speckle tracking echocardiography. Echocardiography 2013, 30, 285–292. [Google Scholar] [CrossRef]
- Bolliger, C.T.; Zellweger, J.P.; Danielsson, T.; van Biljon, X.; Robidou, A.; Westin, A.; Perruchoud, A.P.; Sawe, U. Influence of long-term smoking reduction on health risk markers and quality of life. Nicotine Tob. Res. 2002, 4, 433–439. [Google Scholar] [CrossRef]
- Hatsukami, D.K.; Kotlyar, M.; Allen, S.; Jensen, J.; Li, S.; Le, C.; Murphy, S. Effects of cigarette reduction on cardiovascular risk factors and subjective measures. Chest 2005, 128, 2528–2537. [Google Scholar] [CrossRef]
- Godtfredsen, N.S.; Prescott, E.; Osler, M. Effect of smoking reduction on lung cancer risk. JAMA 2005, 294, 1505–1510. [Google Scholar] [CrossRef]
- Djordjevic, M; Stellman, S.D.; Zang, E. Doses of nicotine and lung carcinogens delivered to cigarette smokers. J. Natl. Cancer Inst. 2000, 92, 106–111. [Google Scholar]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb. Exp. Pharmacol. 2009, 192, 29–60. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Farsalinos, K.E.; Romagna, G.; Tsiapras, D.; Kyrzopoulos, S.; Voudris, V. Evaluation of Electronic Cigarette Use (Vaping) Topography and Estimation of Liquid Consumption: Implications for Research Protocol Standards Definition and for Public Health Authorities’ Regulation. Int. J. Environ. Res. Public Health 2013, 10, 2500-2514. https://doi.org/10.3390/ijerph10062500
Farsalinos KE, Romagna G, Tsiapras D, Kyrzopoulos S, Voudris V. Evaluation of Electronic Cigarette Use (Vaping) Topography and Estimation of Liquid Consumption: Implications for Research Protocol Standards Definition and for Public Health Authorities’ Regulation. International Journal of Environmental Research and Public Health. 2013; 10(6):2500-2514. https://doi.org/10.3390/ijerph10062500
Chicago/Turabian StyleFarsalinos, Konstantinos E., Giorgio Romagna, Dimitris Tsiapras, Stamatis Kyrzopoulos, and Vassilis Voudris. 2013. "Evaluation of Electronic Cigarette Use (Vaping) Topography and Estimation of Liquid Consumption: Implications for Research Protocol Standards Definition and for Public Health Authorities’ Regulation" International Journal of Environmental Research and Public Health 10, no. 6: 2500-2514. https://doi.org/10.3390/ijerph10062500
APA StyleFarsalinos, K. E., Romagna, G., Tsiapras, D., Kyrzopoulos, S., & Voudris, V. (2013). Evaluation of Electronic Cigarette Use (Vaping) Topography and Estimation of Liquid Consumption: Implications for Research Protocol Standards Definition and for Public Health Authorities’ Regulation. International Journal of Environmental Research and Public Health, 10(6), 2500-2514. https://doi.org/10.3390/ijerph10062500






