Lobocrassins A–E: New Cembrane-Type Diterpenoids from the Soft Coral Lobophytum crassum
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
3.4. Acetylation of Lobocrassin C (3)
3.5. Molecular Mechanics Calculations
3.6. Cytotoxicity Testing
3.7. Superoxide Anion Generation and Elastase Release by Human Neutrophils
4. Conclusions
Acknowledgments
- Samples Availability: Not Available.
References
- Blunt, JW; Copp, BR; Munro, MHG; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep. 2011, 28, 196–268. [Google Scholar]
- Bowden, BF; Brittle, JA; Coll, JC; Liyanage, N; Mitchell, SJ; Stokie, GJ. Studies of Australian soft corals. VI-A new cembranolide diterpene from the soft coral Lobophytum crassum (Coelenterata, Anthozoa, Octocorallia, Alcyonacea). Tetrahedron Lett. 1977, 18, 3661–3662. [Google Scholar]
- Kinamoni, Z; Groweiss, A; Carmely, S; Kashman, Y; Loya, Y. Several new cembranoid diterpenes from three soft corals of the Red Sea. Tetrahedron 1983, 39, 1643–1648. [Google Scholar]
- Kashman, Y; Carmely, S; Groweiss, A. Further cembranoid derivatives from the Red Sea soft corals Alcyonium flaccidum and Lobophytum crassum. J. Org. Chem. 1981, 46, 3592–3596. [Google Scholar]
- Matthée, GF; König, GM; Wright, AD. Three new diterpenes from the marine soft coral Lobophytum crassum. J. Nat. Prod. 1998, 61, 237–240. [Google Scholar]
- Duh, C-Y; Wang, S-K; Huang, B-T; Dai, C-F. Cytotoxic cembrenolide diterpenes from the Formosan soft coral Lobophytum crassum. J. Nat. Prod. 2000, 63, 884–885. [Google Scholar]
- Yin, SW; Shi, YP; Li, XM; Wang, BG. A novel hydroperoxyl substituted cembranolide diterpene from marine soft coral Lobophytum crassum. Chin. Chem. Lett. 2005, 16, 1489–1491. [Google Scholar]
- Yin, S-W; Shi, Y-P; Li, X-M; Wang, B-G. A new cembranoid diterpene and other related metabolites from the South-China-Sea soft coral Lobophytum crassum. Helv. Chim. Acta 2006, 89, 567–572. [Google Scholar]
- Zhang, W; Krohn, K; Ding, J; Miao, Z-H; Zhou, X-H; Chen, S-H; Pescitelli, G; Salvadori, P; Kurtan, T; Guo, Y-W. Structural and stereochemical studies of α-methylene-γ-lactone-bearing cembrane diterpenoids from a South China Sea soft coral Lobophytum crassum. J. Nat. Prod. 2008, 71, 961–966. [Google Scholar]
- Chao, C-H; Wen, Z-H; Wu, Y-C; Yeh, H-C; Sheu, J-H. Cytotoxic and anti-inflammatory cembranoids from the soft coral Lobophytum crassum. J. Nat. Prod. 2008, 71, 1819–1824. [Google Scholar]
- Lin, S-T; Wang, S-K; Cheng, S-Y; Duh, C-Y. Lobocrasol, a new diterpenoid from the soft coral Lobophytum crassum. Org. Lett. 2009, 11, 3012–3014. [Google Scholar]
- Wanzola, M; Furuta, T; Kohno, Y; Fukumitsu, S; Yasukochi, S; Watari, K; Tanaka, C; Higuchi, R; Miyamoto, T. Four new cembrane diterpenes isolated from an Okinawan soft coral Lobophytum crassum with inhibitory effects on nitric oxide production. Chem. Pharm. Bull. 2010, 58, 1203–1209. [Google Scholar]
- Vanisree, M; Subbaraju, GV. Alcyonacean metabolites VIII—Antibacterial metabolites from Labophytum crassum of the Indian Ocean. Asian J. Chem. 2002, 14, 957–960. [Google Scholar]
- Rodríguez, AD; Li, Y; Dhasmana, H; Barnes, CL. New marine cembrane diterpenoids isolated from the Caribbean gorgonian Eunicea mammosa. J. Nat. Prod. 1993, 56, 1101–1113. [Google Scholar]
- Wei, X; Rodríguez, AD; Baran, P; Raptis, RG; Sánchez, JA; Ortega-Barria, E; González, J. Antiplasmodial cembradiene diterpenoids from a Southwestern Caribbean gorgonian octocoral of the genus Eunicea. Tetrahedron 2004, 60, 11813–11819. [Google Scholar]
- Rudi, A; Shmul, G; Benayahu, Y; Kashman, Y. Sinularectin, a new diterpenoid from the soft coral Sinularia erecta. Tetrahedron Lett. 2006, 47, 2937–2939. [Google Scholar]
- Huang, H-C; Chao, C-H; Kuo, Y-H; Sheu, J-H. Crassocolides G–M, cembranoids from the Formosan soft coral Sarcophyton crassocaule. Chem. Biodivers. 2009, 6, 1232–1242. [Google Scholar]
- Rodríguez, AD; Martínez, N. Marine antitumor agents: 14-deoxycrassin and pseudoplexaurol, new cembranoid diterpenes from the Caribbean gorgonian Pseudoplexaura porosa. Experientia 1993, 49, 179–181. [Google Scholar]
- Liu, Z; Peng, L; Li, WZ; Li, Y. Asymmetric total synthesis of pseudoplexaurol and 14-deoxycrassin, two antitumor marine cembrane diterpenoids. Synlett 2003, 13, 1977–1980. [Google Scholar]
- Ravi, BN; Faulkner, DJ. Cembranoid diterpenes from a South Pacific soft coral. J. Org. Chem. 1978, 43, 2127–2131. [Google Scholar]
- Allinger, NL. Conformational analysis. 130. MM2. A hydrocarbon force field utilizing V1 and V2 torsional terms. J. Am. Chem. Soc. 1977, 99, 8127–8134. [Google Scholar]
- Alley, MC; Scudiero, DA; Monks, A; Hursey, ML; Czerwinski, MJ; Fine, DL; Abbott, BJ; Mayo, JG; Shoemaker, RH; Boyd, MR. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar]
- Scudiero, DA; Shoemaker, RH; Paull, KD; Monks, A; Tierney, S; Nofziger, TH; Currens, MJ; Seniff, D; Boyd, MR. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar]
- Hwang, T-L; Su, Y-C; Chang, H-L; Leu, Y-L; Chung, P-J; Kuo, L-M; Chang, Y-J. Suppression of superoxide anion and elastase release by C18 unsaturated fatty acids in human neutrophils. J. Lipid Res. 2009, 50, 1395–1408. [Google Scholar]
- Hwang, T-L; Li, G-L; Lan, Y-H; Chia, Y-C; Hsieh, P-W; Wu, Y-H; Wu, Y-C. Potent inhibition of superoxide anion production in activated human neutrophils by isopedicin, a bioactive component of the Chinese medicinal herb Fissistigma oldhamii. Free Radical Biol. Med. 2009, 46, 520–528. [Google Scholar]
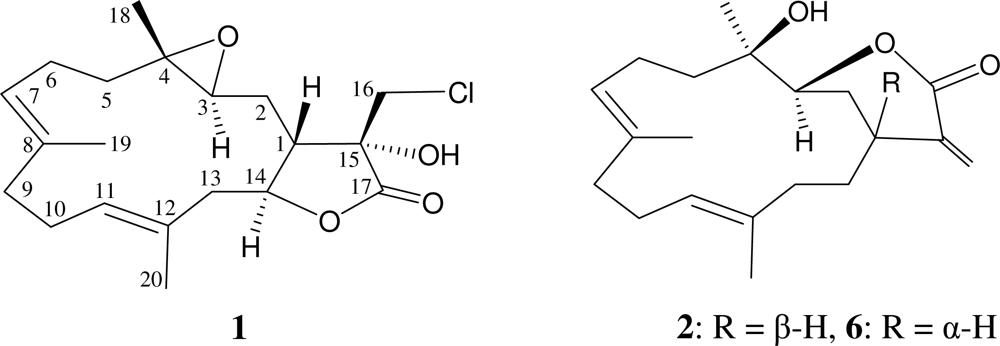
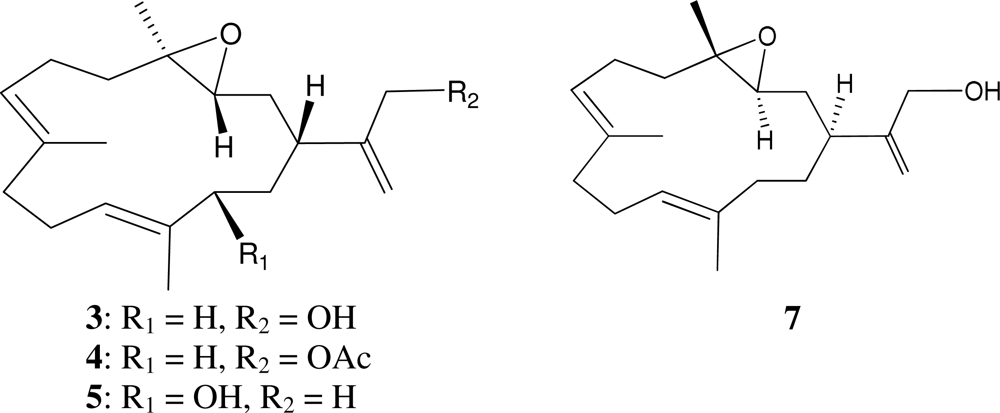
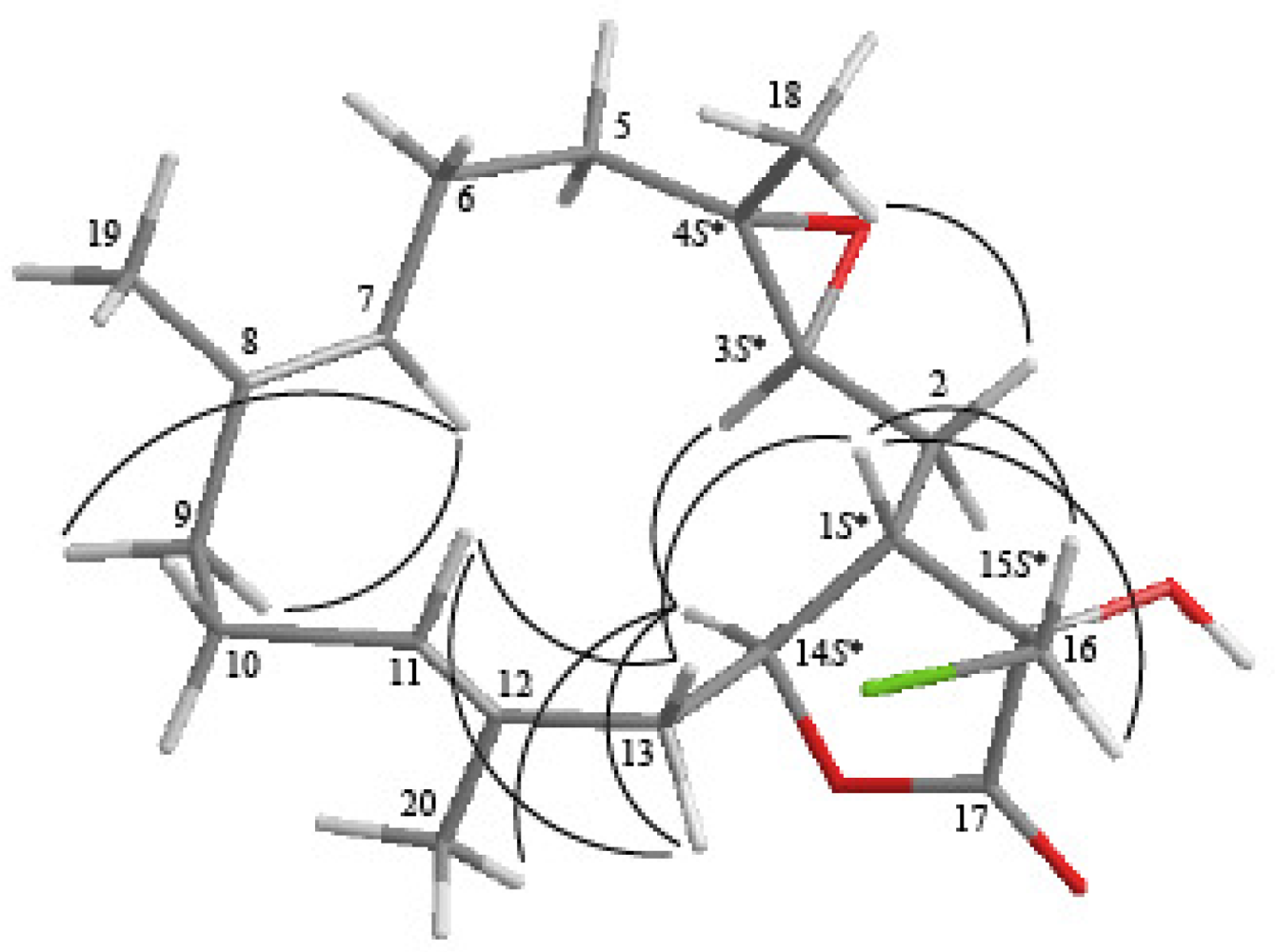
| C/H | 1H a | 13C b | 1H–1H COSY | HMBC (H→C) | |
|---|---|---|---|---|---|
| 1 | 2.76 ddd (10.0, 5.2, 4.0) c | 44.4 | (CH) d | H2-2, H-14 | C-2, C-13, C-14, C-16 |
| 2a | 2.14 ddd (15.6, 5.2, 4.4) | 23.5 | (CH2) | H-1, H-2b, H-3 | C-1, C-3, C-4, C-14, C-15 |
| b | 1.68 ddd (15.6, 8.4, 4.0) | H-1, H-2a, H-3 | C-1, C-3, C-4, C-14 | ||
| 3 | 2.86 dd (8.4, 4.4) | 60.3 | (CH) | H2-2 | C-2 |
| 4 | 64.0 | (C) | |||
| 5a | 2.07 m | 38.0 | (CH2) | H-5b, H2-6 | C-3, C-4, C-7 |
| b | 1.29 m | H-5a, H2-6 | C-3 | ||
| 6a | 2.24 m | 23.4 | (CH2) | H2-5, H-6b, H-7 | C-4, C-7, C-8 |
| b | 2.09 m | H2-5, H-6a, H-7 | C-7, C-8 | ||
| 7 | 5.07 dd (6.4, 6.4) | 122.5 | (CH) | H2-6, H3-19 | C-6, C-9, C-19 |
| 8 | 135.2 | (C) | |||
| 9a | 2.26 m | 38.8 | (CH2) | H-9b, H2-10 | C-7, C-8, C-10, C-11, C-19 |
| b | 2.04 m | H-9a, H2-10 | C-7, C-8, C-10, C-11, C-19 | ||
| 10a | 2.32 m | 24.8 | (CH2) | H2-9, H-10b, H-11 | C-8, C-9, C-11, C-12 |
| b | 2.21 m | H2-9, H-10a, H-11 | C-8, C-9, C-11, C-12 | ||
| 11 | 5.23 dd (6.4, 6.4) | 130.2 | (CH) | H2-10, H3-20 | C-9, C-10, C-20 |
| 12 | 130.1 | (C) | |||
| 13a | 2.67 br d (14.4) | 43.0 | (CH2) | H-13b, H-14 | C-1, C-11, C-12, C-14 |
| b | 2.52 dd (14.4, 7.2) | H-13a, H-14 | C-1, C-11, C-12, C-14 | ||
| 14 | 4.66 ddd (10.0, 7.2, 2.8) | 80.0 | (CH) | H-1, H2-13 | C-12 |
| 15 | 77.2 | (C) | |||
| 16a | 3.79 d (11.6) | 44.5 | (CH2) | H-16b | C-1, C-15, C-17 |
| b | 3.53 d (11.6) | H-16a | C-1, C-15, C-17 | ||
| 17 | 173.4 | (C) | |||
| 18 | 1.34 s | 17.0 | (CH3) | C-3, C-4, C-5 | |
| 19 | 1.60 s | 15.7 | (CH3) | H-7 | C-7, C-8, C-9 |
| 20 | 1.74 s | 17.4 | (CH3) | H-11 | C-11, C-12, C-13 |
| OH-15 | 4.03 br s | C-1, C-15, C-16, C-17 | |||
| C/H | 1 H a | 13C b | 1H–1H COSY | HMBC (H→C) | |
|---|---|---|---|---|---|
| 1 | 2.70 m | 35.5 | (CH) d | H2-2, H-14 | C-15, C-16, C-17 |
| 2 | 1.98 m | 25.2 | (CH2) | H-1, H-3 | C-1, C-3, C-4, C-14, C-15 |
| 3 | 4.29 dd (8.0, 5.5) c | 79.9 | (CH) | H2-2 | C-1, C-4, C-5, C-18 |
| 4 | 74.6 | (C) | |||
| 5a | 1.87 m | 37.4 | (CH2) | H-5b, H2-6 | C-3, C-4, C-6, C-7, C-18 |
| b | 1.68 ddd (14.5, 9.5, 4.5) | H-5a, H2-6 | C-3, C-4, C-6, C-7, C-18 | ||
| 6a | 2.22 m | 22.4 | (CH2) | H2-5, H-6b, H-7 | C-4, C-5, C-7, C-8 |
| b | 2.16 m | H2-5, H-6a, H-7 | C-4, C-5, C-7, C-8 | ||
| 7 | 5.21 dd (7.0, 7.0) | 125.6 | (CH) | H2-6, H3-19 | C-5, C-6, C-9, C-19 |
| 8 | 135.7 | (C) | |||
| 9 | 2.14 m | 38.8 | (CH2) | H2-10 | C-7, C-8, C-10 |
| 10a | 2.22 m | 24.2 | (CH2) | H2-9, H-10b, H-11 | C-8, C-11, C-12 |
| b | 2.15 m | H2-9, H-10a, H-11 | C-8, C-9, C-11, C-12 | ||
| 11 | 5.01 dd (6.5, 6.5) | 124.7 | (CH) | H2-10 | C-10, C-13, C-20 |
| 12 | 135.3 | (C) | |||
| 13a | 2.21 m | 36.1 | (CH2) | H-13b, H2-14 | C-14 |
| b | 2.02 m | H-13a, H2-14 | C-1, C-11, C-12, C-14, C-20 | ||
| 14a | 1.38 m | 31.5 | (CH2) | H-1, H2-13, H-14b | C-1, C-2, C-12 |
| b | 1.90 m | H-1, H2-13, H-14a | C-12 | ||
| 15 | 140.2 | (C) | |||
| 16 | 166.5 | (C) | |||
| 17a | 6.34 s | 125.7 | (CH2) | H-17b | C-1, C-15, C-16 |
| b | 5.55 s | H-17a | C-1, C-16 | ||
| 18 | 1.27 s | 24.2 | (CH3) | C-3, C-4, C-5 | |
| 19 | 1.56 s | 15.3 | (CH3) | H-7 | C-7, C-8, C-9 |
| 20 | 1.61 s | 15.6 | (CH3) | C-11, C-12, C-13 | |
| OH-4 | 1.89 s | C-3, C-4, C-5, C-18 | |||
| C/H | 1 H a | 13C b | 1H–1H COSY | HMBC (H→C) | |
|---|---|---|---|---|---|
| 1 | 2.05 m | 39.3 | (CH) d | H2-2, H-14 | n.o. e |
| 2a | 1.89 ddd (14.5, 5.0, 4.0) c | 33.9 | (CH2) | H-1, H-2b, H-3 | C-1, C-3, C-4, C-14, C-15 |
| b | 1.46 ddd (14.5, 10.5, 3.5) | H-1, H-2a, H-3 | C-1, C-3, C-4, C-14, C-15 | ||
| 3 | 2.85 dd (10.5, 4.0) | 62.8 | (CH) | H2-2 | C-2, C-5 |
| 4 | 61.0 | (C) | |||
| 5a | 2.03 m | 38.0 | (CH2) | H-5b, H2-6 | C-3, C-4, C-6, C-7 |
| b | 1.35 m | H-5a, H2-6 | C-6, C-7 | ||
| 6a | 1.99 m | 23.1 | (CH2) | H2-5, H-6b, H-7 | C-4, C-7 |
| b | 2.17 m | H2-5, H-6a, H-7 | C-7 | ||
| 7 | 5.11 dd (6.5, 6.5) | 125.2 | (CH) | H2-6, H3-19 | C-6, C-9, C-19 |
| 8 | 134.6 | (C) | |||
| 9 | 2.25 m | 39.6 | (CH2) | H2-10 | C-8, C-11 |
| 10a | 2.23 m | 24.4 | (CH2) | H2-9, H-10b, H-11 | C-9, C-12 |
| b | 2.21 m | H2-9, H-10a, H-11 | C-9, C-12 | ||
| 11 | 5.39 dd (7.0, 7.0) | 128.7 | (CH) | H2-10, H3-20 | C-10, C-13, C-20 |
| 12 | 136.1 | (C) | |||
| 13 | 4.19 m | 76.6 | (CH) | H2-14 | n.o. |
| 14 | 1.72 m | 40.3 | (CH2) | H-1, H-13 | C-1, C-2, C-12, C-13, C-15 |
| 15 | 150.1 | (C) | |||
| 16 | 1.71 s | 18.8 | (CH3) | H2-17 | C-1, C-15, C-17 |
| 17a | 4.68 s | 109.8 | (CH2) | H3-16, H-17b | C-1, C-16 |
| b | 4.65 s | H3-16, H-17a | C-1, C-16 | ||
| 18 | 1.20 s | 17.6 | (CH3) | C-3, C-4, C-5 | |
| 19 | 1.62 s | 15.3 | (CH3) | H-7 | C-7, C-8, C-9 |
| 20 | 1.62 s | 10.5 | (CH3) | H-11 | C-11, C-12, C-13 |
| Compounds | Cell lines IC50 (μg/mL) | ||||
|---|---|---|---|---|---|
| K562 | CCRF-CEM | Molt4 | HepG2 | Huh 7 | |
| 1 | 15.39 | 5.33 | 11.86 | 32.16 | 26.13 |
| 2 | 2.97 | 0.48 | 0.34 | 3.44 | 8.17 |
| 3 | >40 | 11.55 | 9.51 | >40 | 39.77 |
| 4 | 24.00 | 10.53 | 10.99 | 34.91 | >40 |
| Doxorubicin a | 0.24 | 0.05 | 0.07 | 0.71 | 0.46 |
| Compounds | Superoxide anion | Elastase release |
|---|---|---|
| IC50 (μg/mL) or (Inh %) a | IC50 (μg/mL) or (Inh %) a | |
| 1 | (2.8 ± 1.9) | (0.9 ± 2.5) |
| 2 | 4.8 ± 0.7 | 4.9 ± 0.4 |
| 3 | (1.4 ± 2.4) | (9.6 ± 9.4) |
| 4 | (−1.9 ± 7.3) | (11.0 ± 3.9) |
| 5 | (−1.2 ± 1.5) | (−4.4 ± 9.5) |
| DPI b | 0.8 ± 0.2 | |
| Elastatinal b | 30.8 ± 5.7 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kao, C.-Y.; Su, J.-H.; Lu, M.-C.; Hwang, T.-L.; Wang, W.-H.; Chen, J.-J.; Sheu, J.-H.; Kuo, Y.-H.; Weng, C.-F.; Fang, L.-S.; et al. Lobocrassins A–E: New Cembrane-Type Diterpenoids from the Soft Coral Lobophytum crassum. Mar. Drugs 2011, 9, 1319-1331. https://doi.org/10.3390/md9081319
Kao C-Y, Su J-H, Lu M-C, Hwang T-L, Wang W-H, Chen J-J, Sheu J-H, Kuo Y-H, Weng C-F, Fang L-S, et al. Lobocrassins A–E: New Cembrane-Type Diterpenoids from the Soft Coral Lobophytum crassum. Marine Drugs. 2011; 9(8):1319-1331. https://doi.org/10.3390/md9081319
Chicago/Turabian StyleKao, Chia-Ying, Jui-Hsin Su, Mei-Chin Lu, Tsong-Long Hwang, Wei-Hsien Wang, Jih-Jung Chen, Jyh-Horng Sheu, Yueh-Hsiung Kuo, Ching-Feng Weng, Lee-Shing Fang, and et al. 2011. "Lobocrassins A–E: New Cembrane-Type Diterpenoids from the Soft Coral Lobophytum crassum" Marine Drugs 9, no. 8: 1319-1331. https://doi.org/10.3390/md9081319
APA StyleKao, C.-Y., Su, J.-H., Lu, M.-C., Hwang, T.-L., Wang, W.-H., Chen, J.-J., Sheu, J.-H., Kuo, Y.-H., Weng, C.-F., Fang, L.-S., Wen, Z.-H., & Sung, P.-J. (2011). Lobocrassins A–E: New Cembrane-Type Diterpenoids from the Soft Coral Lobophytum crassum. Marine Drugs, 9(8), 1319-1331. https://doi.org/10.3390/md9081319







