Effects of Oral Glucosamine Hydrochloride Administration on Plasma Free Amino Acid Concentrations in Dogs
Abstract
:1. Introduction
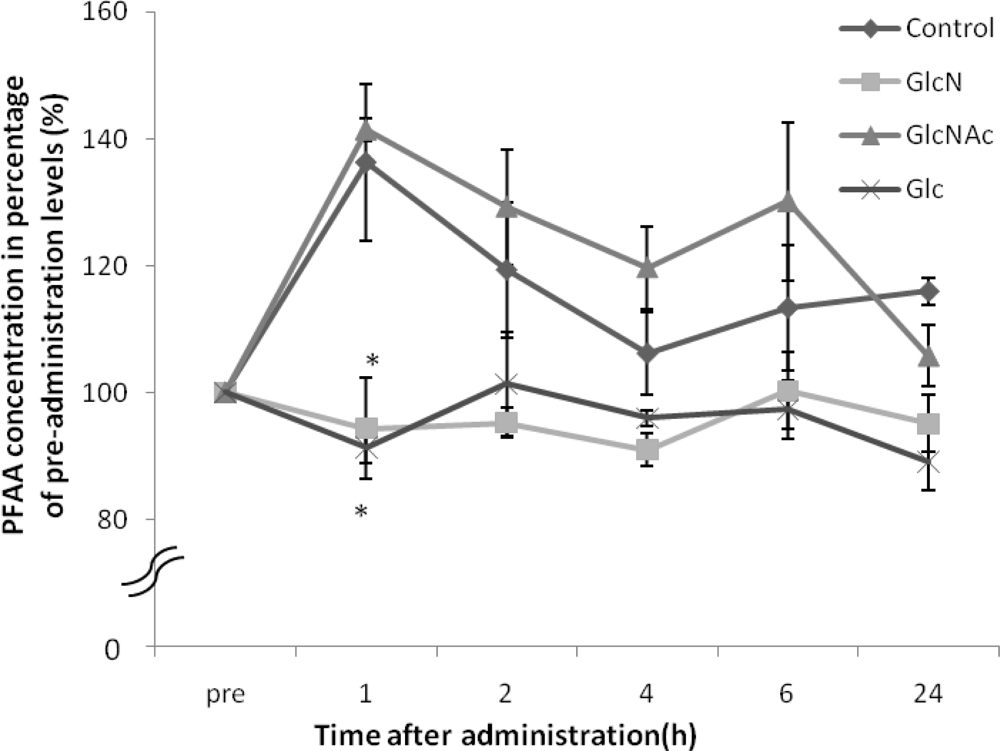
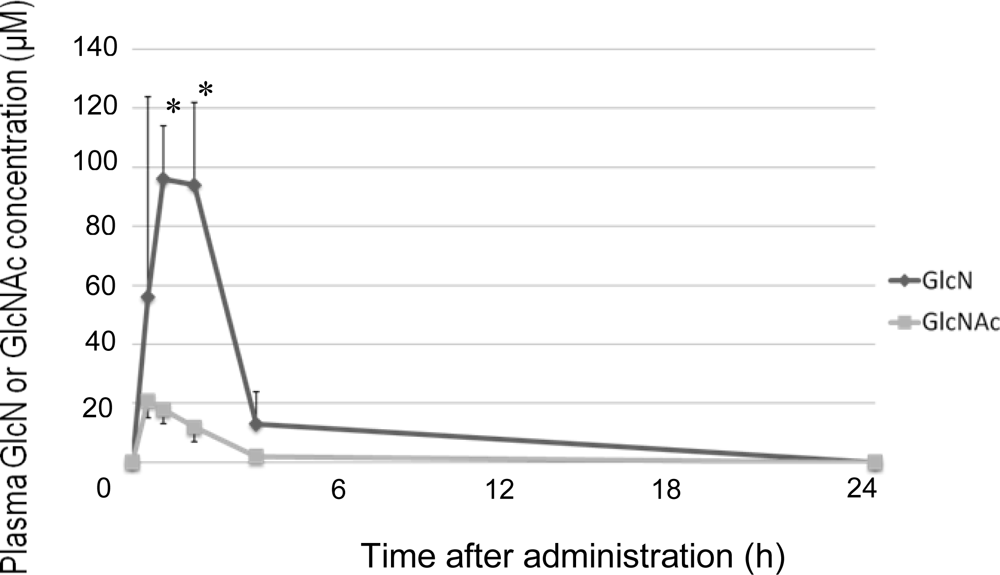
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Animals
3.3. Administration and Blood Sampling
3.4. Measurement of PFAA Concentrations
3.5. Measurement of Plasma GlcN and GlcNAc Concentrations
3.6. Statistical Analysis
4. Conclusions
- Sample Availability: Available from the authors.
References
- Gregorym, PJ; Sperry, M; Wilson, AF. Dietary supplements for osteoarthritis. Am Fam Physician 2008, 77, 177–184. [Google Scholar]
- Minami, S; Hata, M; Tamai, Y; Hashida, M; Takayama, T; Yamamoto, S; Okada, M; Funatsu, T; Tsuka, T; Imagawa, T; Okamoto, Y. Clinical application of d-glucosamine and scale collagen peptide on canine and feline orthopedic diseases and spondylitis deformans. Carbohydr Polym 2010, 84, 831–834. [Google Scholar]
- Goodrich, LR; Nixon, AJ. Medical treatment of osteoarthritis in the horse-a review. Vet J 2006, 171, 51–69. [Google Scholar]
- Barclay, TS; Tsourounis, C; McCart, GM. glucosamine. Ann Pharmacother 1998, 32, 574–579. [Google Scholar]
- Aghazadeh-Habashi, A; Sattari, S; Pasutto, F; Jamali, F. Single dose pharmacokinetics and bioavailability of glucosamine in the rat. J Pharm Pharm Sci 2002, 5, 181–184. [Google Scholar]
- Adebowale, A; Du, J; Liang, Z; Leslie, JL; Eddington, ND. The bioavailability and pharmacokinetics of glucosamine hydrochloride and low molecular weight chondroitin sulfate after single and multiple doses to beagle dogs. Biopharm Drug Dispos 2002, 23, 217–225. [Google Scholar]
- Laverty, S; Sandy, JD; Celeste, C; Vachon, P; Marier, JF; Plaas, AH. Synovial fluid levels and serum pharmacokinetics in a large animal model following treatment with oral glucosamine at clinically relevant doses. Arthritis Rheum 2005, 52, 181–191. [Google Scholar]
- Du, J; White, N; Eddington, ND. The bioavailability and pharmacokinetics of glucosamine hydrochloride and chondroitin sulfate after oral and intravenous single dose administration in the horse. Biopharm Drug Dispos 2004, 25, 109–116. [Google Scholar]
- Meulyzer, M; Vachon, P; Beaudry, F; Vinardell, T; Richard, H; Beauchamp, G; Laverty, S. Comparison of pharmacokinetics of glucosamine and synovial fluid levels following administration of glucosamine sulphate or glucosamine hydrochloride. Osteoarthritis Cartilage 2008, 16, 973–979. [Google Scholar]
- Shikhman, AR; Brinson, DC; Valbracht, J; Lotz, MK. Differential metabolic effects of glucosamine and N-acetylglucosamine in human articular chondrocytes. Osteoarthritis Cartilage 2009, 17, 1022–1028. [Google Scholar]
- Tamai, Y; Miyatake, K; Okamoto, Y; Takamori, Y; Sakamoto, H; Minami, S. Enhanced healing of cartilaginous injuries by glucosamine hydrochloride. Carbohydr Polym 2002, 48, 369–378. [Google Scholar]
- Tamai, Y; Miyatake, K; Okamoto, Y; Takamori, Y; Sakamoto, K; Minami, S. Enhanced healing of cartilaginous injuries by N-acetyl-d-glucosamine and glucuronic acid. Carbohydr Polym 2002, 54, 251–262. [Google Scholar]
- Naito, K; Watari, T; Furuhata, A; Yomogida, S; Sakamoto, K; Kurosawa, H; Kaneko, K; Nagaoka, I. Evaluation of the effect of glucosamine on an experimental rat osteoarthritis model. Life Sci 2010, 86, 538–543. [Google Scholar]
- Tochikubo, O; Ando, T. Amino Acids and Life-style Related Diseases–Application of New Aminograms in Life Science; Kagawa Nutrition University Publishing Division: Tokyo, Japan, 2010; pp. 52–68. [Google Scholar]
- Derfoul, A; Miyoshi, AD; Freeman, DE; Tuan, RS. Glucosamine promotes chondrogenic phenotype in both chondrocytes and mesenchymal stem cells and inhibits MMP-13 expression and matrix degradation. Osteoarthritis Cartilage 2007, 15, 646–655. [Google Scholar]
- Reginster, JY. Chondromodulation in 2003: dream or reality? Rev Med Suisse Romande 2004, 124, 85–87. [Google Scholar]
- Deshmukh, K; Nimni, ME. Isolation and characterization of cyanogen bromide peptides from the collagen of bovine articular cartilage. Biochem J 1973, 133, 615–622. [Google Scholar]
| Essential amino acids (EAA) | Nonessential amino acids (NEAA) |
|---|---|
| Threonine (Thr) | Asparate (Asp) |
| Valine (Val) | Serine (Ser) |
| Methionine (Met) | Asparagine (Asn) |
| Isoleucine (Ile) | Glutamate (Glu) |
| Leucine (Leu) | Glutamine (Gln) |
| Phenylalanine (Phe) | Glycine (Gly) |
| Lysine (Lys) | Alanine (Ala) |
| Histidine (His) | Tyrosine (Tyr) |
| Tryptophan (Trp) | Proline (Pro) |
| Arginine (Arg) | Citrulline (Cit) |
| Ornithine (Orn) | |
| Hydroxyproline (Hypro) |
| Control | GlcN | GlcNAc | Glc | |
|---|---|---|---|---|
| Glu | 128.3 ± 6.4 | 98.6 ± 9.0* | 184.0 ± 20.7 | 103.6 ± 10.1* |
| Gly | 126.8 ± 12.3 | 95.3 ± 5.8* | 145.0 ± 6.1 | 100.9 ± 2.9* |
| Ala | 135.3 ± 21.9 | 82.8 ± 4.1* | 148.3 ± 4.1 | 85.4 ± 5.6* |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Azuma, K.; Osaki, T.; Tsuka, T.; Imagawa, T.; Okamoto, Y.; Takamori, Y.; Minami, S. Effects of Oral Glucosamine Hydrochloride Administration on Plasma Free Amino Acid Concentrations in Dogs. Mar. Drugs 2011, 9, 712-718. https://doi.org/10.3390/md9050712
Azuma K, Osaki T, Tsuka T, Imagawa T, Okamoto Y, Takamori Y, Minami S. Effects of Oral Glucosamine Hydrochloride Administration on Plasma Free Amino Acid Concentrations in Dogs. Marine Drugs. 2011; 9(5):712-718. https://doi.org/10.3390/md9050712
Chicago/Turabian StyleAzuma, Kazuo, Tomohiro Osaki, Takeshi Tsuka, Tomohiro Imagawa, Yoshiharu Okamoto, Yoshimori Takamori, and Saburo Minami. 2011. "Effects of Oral Glucosamine Hydrochloride Administration on Plasma Free Amino Acid Concentrations in Dogs" Marine Drugs 9, no. 5: 712-718. https://doi.org/10.3390/md9050712
APA StyleAzuma, K., Osaki, T., Tsuka, T., Imagawa, T., Okamoto, Y., Takamori, Y., & Minami, S. (2011). Effects of Oral Glucosamine Hydrochloride Administration on Plasma Free Amino Acid Concentrations in Dogs. Marine Drugs, 9(5), 712-718. https://doi.org/10.3390/md9050712




