Chitosan Based Polyelectrolyte Complexes as Potential Carrier Materials in Drug Delivery Systems
Abstract
:1. Introduction
2. Polyelectrolyte Complexes between Chitosan and Natural Polymers
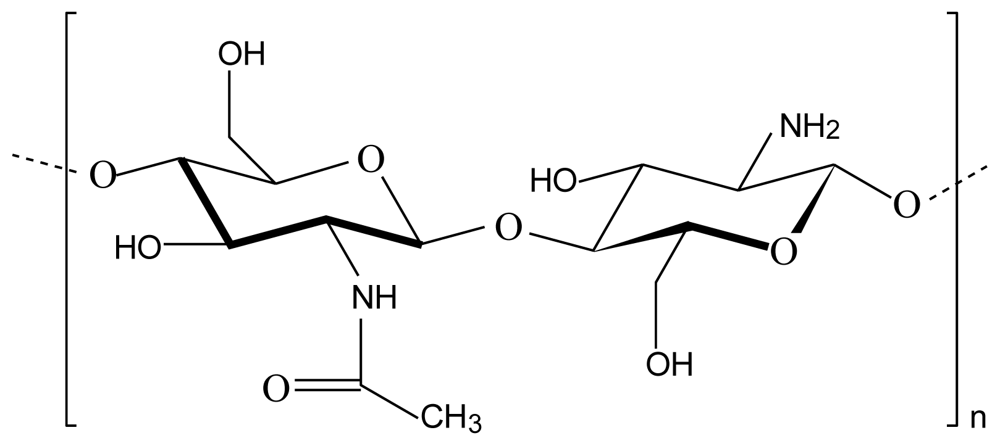
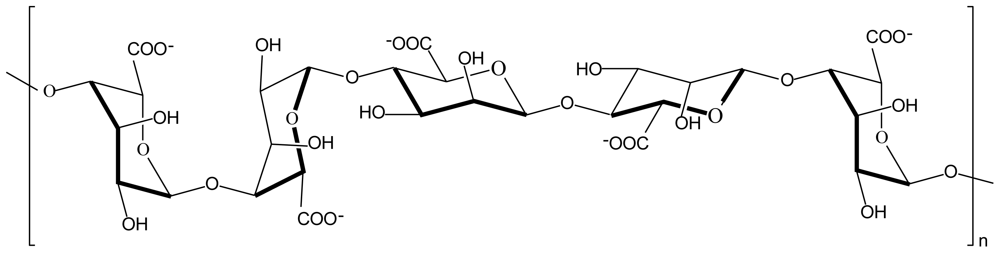
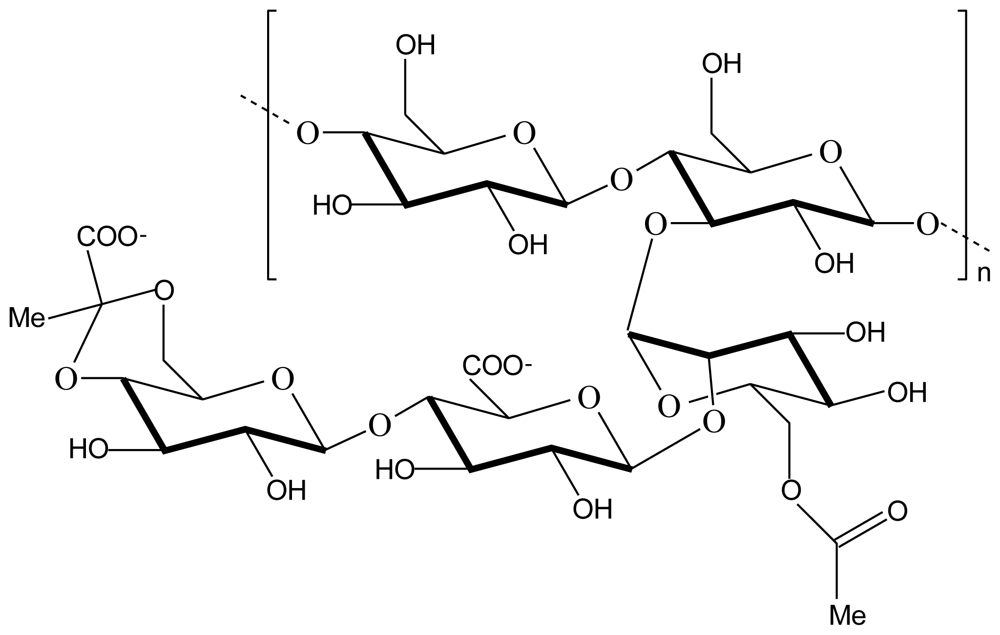
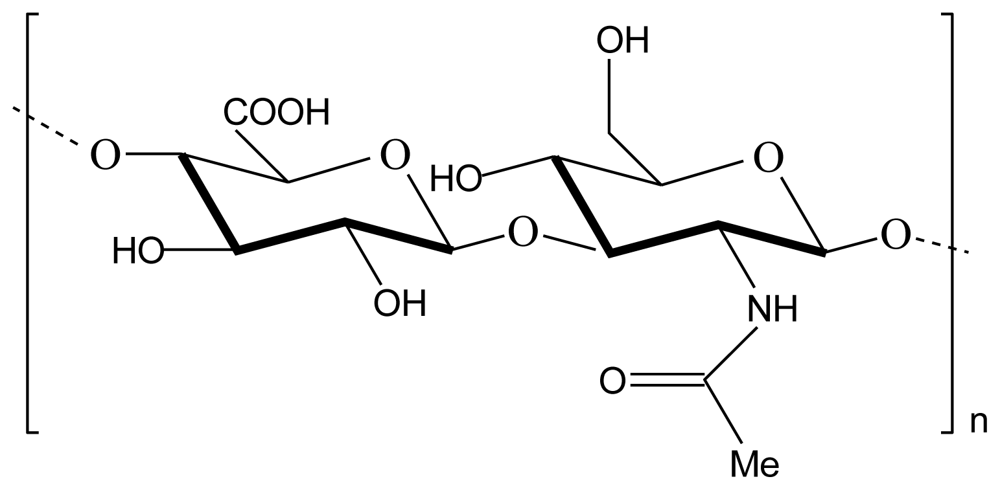
2.1. Chitosan-alginate polyelectrolyte complex
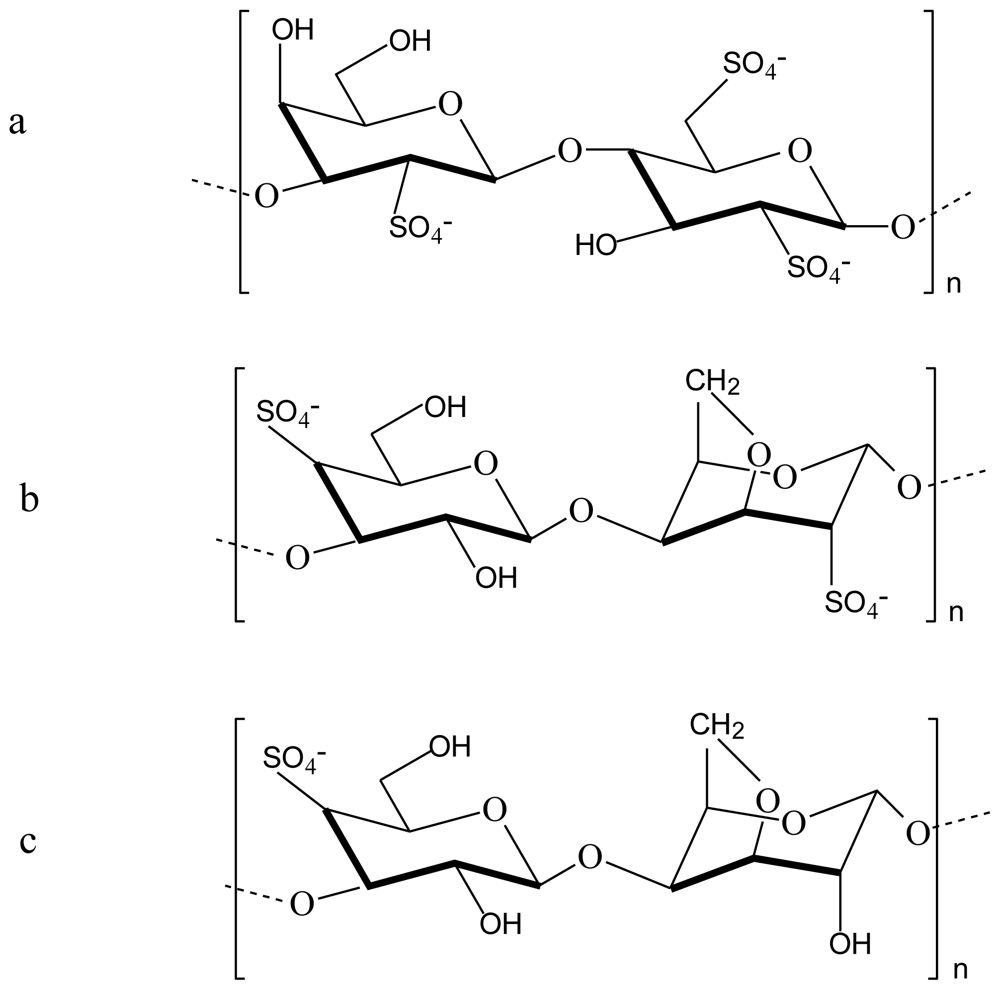
2.2. Chitosan-carrageenan polyelectrolyte complex
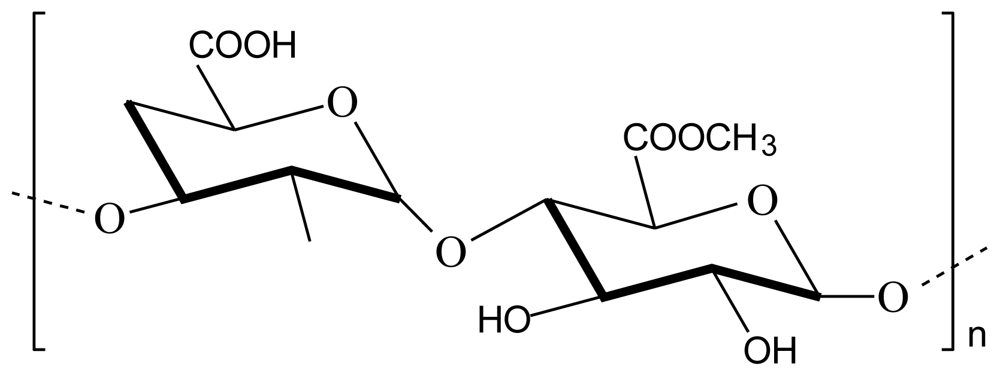
2.3. Chitosan-pectin polyelectrolyte complex
2.4. Chitosan-xanthan gum polyelectrolyte complex
2.5. Chitosan-hyaluronic acid polyelectrolyte complex
2.6. Chitosan-gum kondagogu polyelectrolyte complex
2.7. Chitosan-gelatine polyelectrolyte complex
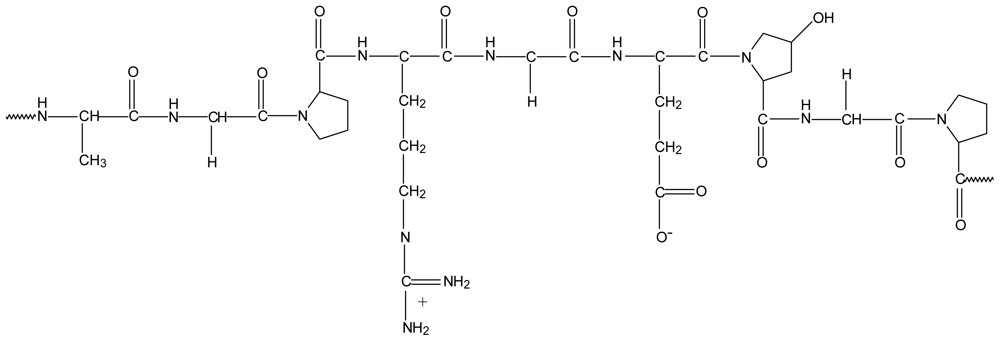
2.8. Chitosan-γ-poly(glutamic acid) polyelectrolyte complex
2.9. Chitosan-maleic starch half-ester acid polyelectrolyte complex
3. Polyelectrolyte Complexes between Chitosan and Synthetic Polymers
3.1. Chitosan-cross-linked-poly(acrylic acid) polyelectrolyte complexes
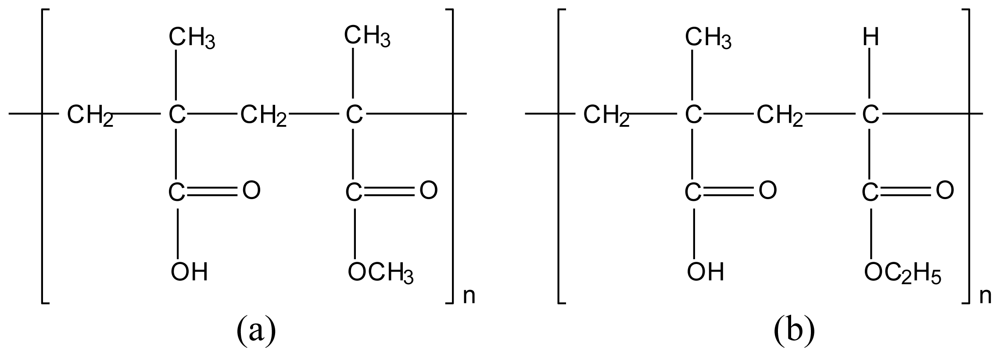
3.2. Chitosan-polymethacrylate copolymer (Eudragit) polyelectrolyte complexes
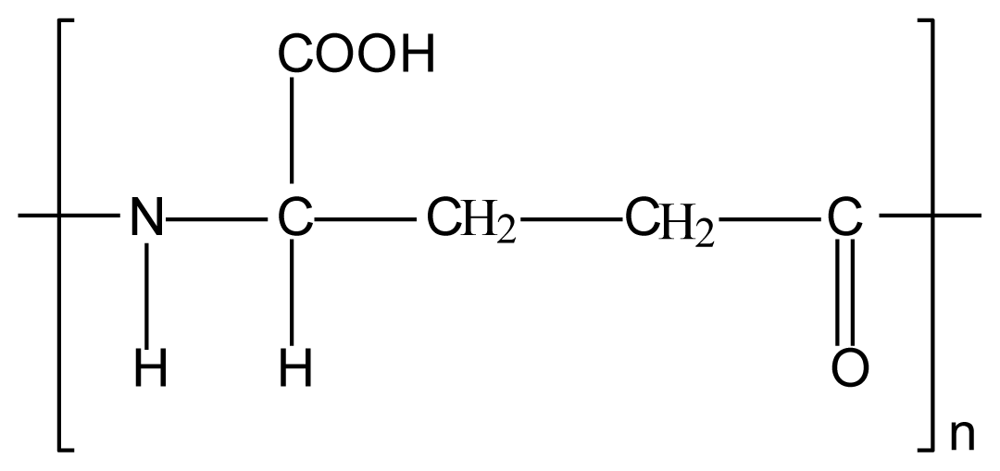
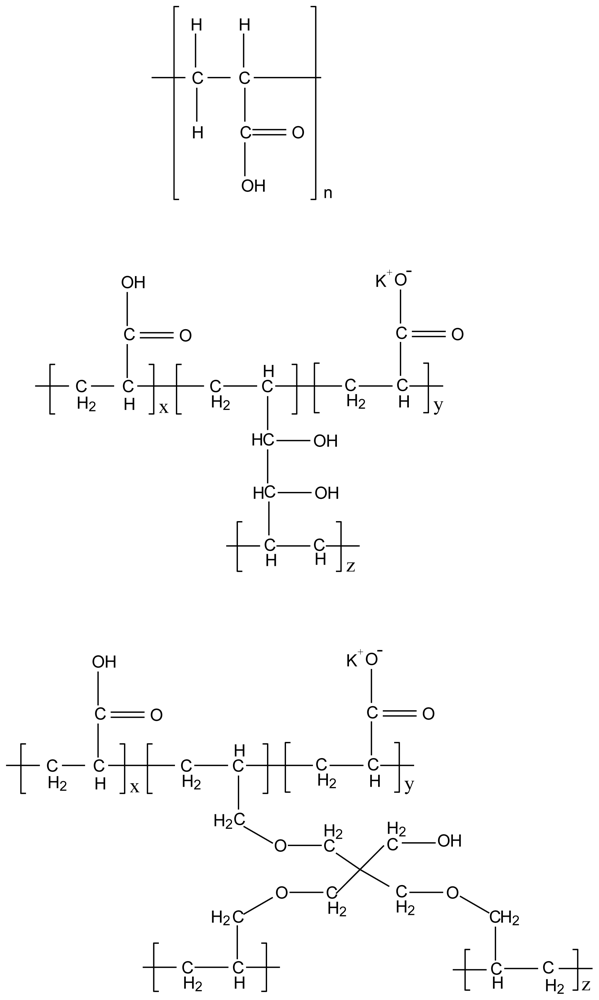
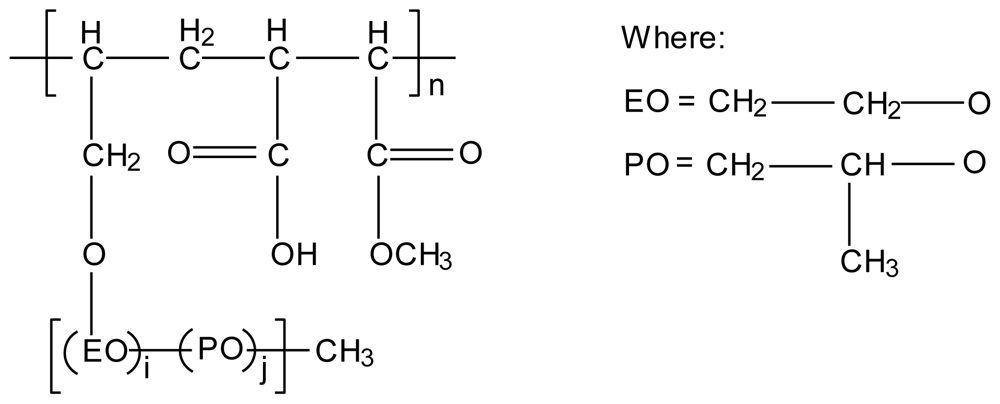
3.3. Chitosan-polyalkylenoxide-maleic acid polyelectrolyte complex
4. Conclusions
References
- Berger, J; Reist, M; Mayer, JM; Felt, O; Peppas, NA; Gurny, R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur J Pharm Biopharm 2004, 57, 19–34. [Google Scholar]
- Berger, J; Reist, M; Mayer, JM; Felt, O; Gurny, R. Structure and interactions in chitosan hydrogels formed by complexation or aggregation for biomedical applications. Eur J Pharm Biopharm 2004, 57, 35–52. [Google Scholar]
- Il’ina, AV; Varlamov, VP. Chitosan-based polyelectrolyte complexes: A review. Appl Biochem Microbiol 2005, 41, 5–11. [Google Scholar]
- Etrych, T; Leclercq, L; Boustta, M; Vert, M. Polyelectrolyte complex formation and stability when mixing polyanions and polycations in salted media: A model study related to the case of body fluids. Eur J Pharm Sci 2005, 2, 281–288. [Google Scholar]
- Bhattarai, N; Gunn, JJ; Zhang, M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv Drug Deliv Rev 2010, 62, 83–99. [Google Scholar]
- Park, JH; Saravanakumar, G; Kim, K; Kwon, IC. Targeted delivery of low molecular drugs using chitosan and its derivatives. Adv Drug Deliv Rev 2010, 62, 28–41. [Google Scholar]
- Kramarenko, EY; Khokhlov, AR; Reineker, P. Stoichiometric polyelectrolyte complexes of ionic block copolymers and oppositely charged polyions. J Chem Phys 2006, 125, 1–8. [Google Scholar]
- Malafaya, PB; Silva, GA; Reis, RL. Natural-origin polymers as carriers and scaffolds for biomolecules and cell delivery in tissue engineering applications. Adv Drug Deliv Rev 2007, 59, 207–233. [Google Scholar]
- Esmaeili, F; Heuking, S; Junginger, HE; Borchard, G. Progress in chitosan-based vaccine delivery systems. J Drug Del Sci Tech 2010, 20, 53–61. [Google Scholar]
- Caramella, C; Ferrari, F; Bonferoni, MC; Rossi, S; Sandri, G. Chitosan and its derivatives as drug penetration enhancers. J Drug Del Sci Tech 2010, 20, 5–13. [Google Scholar]
- Naidu, VGM; Madhusudhana, K; Sashidhar, RB; Ramakrishna, S; Khar, RK; Ahmed, FJ; Diwan, PV. Polyelectrolyte complexes of gum kondagogu and chitosan, as diclofenac carriers. Carbohydr Polym 2009, 76, 464–471. [Google Scholar]
- Moustafine, R; Margulis, EB; Sibgatullina, LF; Kemenova, VA; Van den Mooter, G. Comparative evaluation of interpolyelectrolyte complexes of chitosan with Eudragit L100 and Eudragit L100-55 as potential carriers for oral controlled drug delivery. Eur J Pharm Biopharm 2008, 70, 215–225. [Google Scholar]
- Danielsen, S; Strand, S; de Lange Davies, C; Stokke, BT. Glycosaminoglycan destabilization of DNA-chitosan polyplexes for gene delivery depends on chitosan chain length and GAG properties. Biochim Biophys Acta 2005, 1721, 44–54. [Google Scholar]
- Bernabe, P; Peniche, C; Argüelles-Monal, W. Swelling behavior of chitosan/pectin polyelectrolyte complex membranes. Effect of thermal cross-linking. Polym Bull 2005, 55, 367–375. [Google Scholar]
- Krayukhina, MA; Samoilova, NA; Yamskov, IA. Polyelectrolyte complexes of chitosan, formation, properties and applications. Russ Chem Rev 2008, 77, 799–813. [Google Scholar]
- Argüelles-Monal, W; Cabrera, G; Peniche, C; Rinaudo, M. Conductimetric study of the interpolyelectrolyte reaction between chitosan and polygalacturonic acid. Polymer 2000, 41, 2373–2378. [Google Scholar]
- Gamzazade, AI; Nasibov, SM. Formation and properties of polyelectrolyte complexes of chitosan hydrochloride and sodium dextransulfate. Carbohydr Polym 2002, 50, 339–343. [Google Scholar]
- Maciel, JS; Silva, DA; Paula, HCB; de Paula, RCM. Chitosan/carboxymethyl cashew gum polyelectrolyte complex: synthesis and thermal stability. Eur Polym J 2005, 41, 2726–2733. [Google Scholar]
- Sashina, ES; Novoselov, NP. Polyelectrolyte complexes of fibroin with chitosan. Macromol Chem Polym Mater 2005, 78, 493–497. [Google Scholar]
- Zhao, Q; Qian, J; An, Q; Gao, C; Gui, Z; Jin, H. Synthesis and characterization of soluble chitosan/sodium carboxymethyl cellulose polyelectrolyte complexes and the pervaporation dehydration of their homogenous membranes. J Membr Sci 2009, 333, 68–78. [Google Scholar]
- Oliveira, MA; Ciarlini, PC; Feitosa, JPA; de Paula, RCM; Paula, HCB. Chitosan/”angico” gum nanoparticles: Synthesis and characterization. Mater Sci Eng C 2009, 29, 448–451. [Google Scholar]
- Sankalia, MG; Mashru, RC; Sankalia, JM; Sutariya, VB. Reversed chitosan-alginate polyelectrolyte complex for stability improvement of alpha-amylase: Optimization and physicochemical characterization. Eur J Pharm Biopharm 2007, 65, 215–232. [Google Scholar]
- Beneke, CE; Viljoen, AM; Hamman, JH. Polymeric plant-derived excipients in drug delivery. Molecules 2009, 14, 2602–2620. [Google Scholar]
- Hein, S; Wang, K; Stevens, WF; Kjems, J. Chitosan composites for biomedical applications: status, challenges and perspectives. Mater Sci Technol 2008, 24, 1053–1061. [Google Scholar]
- Li, X; Xie, H; Lin, J; Xie, W; Ma, X. Characterization and biodegradation of chitosan-alginate polyelectrolyte complexes. Polym Degrad Stab 2009, 94, 1–6. [Google Scholar]
- Sæther, HV; Holme, HK; Maurstad, G; Smidsrød, O; Stokke, BT. Polyelectrolyte complex formation using alginate and chitosan. Carbohydr Polym 2008, 74, 813–821. [Google Scholar]
- Liao, IC; Wan, ACA; Yim, EKF; Leong, KW. Controlled release from fibers of polyelectrolyte complexes. J Control Release 2005, 104, 347–358. [Google Scholar]
- Tapia, C; Escobar, Z; Costa, E; Sapag-Hagar, J; Valenzuela, F; Basualto, C; Gai, MN; Yazdani-Pedram, M. Comparative studies on polyelectrolyte complexes and mixtures of chitosan-alginate and chitosan-carrageenan as prolonged diltiazem clorhydrate release systems. Eur J Pharm Biopharm 2004, 57, 65–75. [Google Scholar]
- Coviello, T; Alhaique, F; Dorigo, A; Matricardi, P; Grassi, M. Two galactomannans and scleroglucan as matrices for drug delivery: Preparation and release studies. Eur J Pharm Biopharm 2007, 66, 200–209. [Google Scholar]
- Shumilina, EV; Shchipunov, YA. Chitosan-carrageenan gels. Colloid J 2002, 64, 372–378. [Google Scholar]
- Briones, AV; Sato, T. Encapsulation of glucose oxidase (GOD) in polyelectrolyte complexes of chitosan-carrageenan. React Funct Polym 2010, 70, 19–27. [Google Scholar]
- Fry, SC. Primary cell wall metabolism, tracking the careers of wall polymers in living plant cells. New Phytol 2004, 161, 641–675. [Google Scholar]
- Sriamornsak, P; Thirawong, N; Weerapol, Y; Nunthanid, J; Sungthongjeen, S. Swelling and erosion of pectin matrix tablets and their impact on drug release behavior. Eur J Pharm Biopharm 2007, 67, 211–219. [Google Scholar]
- Cárdenas, A; Goycoolea, FM; Rinaudo, M. On the gelling behaviour of nopal (Opuntia ficus indica) low metholoxyl pectin. Carbohydr Polym 2008, 73, 212–222. [Google Scholar]
- Bigucci, F; Luppi, B; Cerchiara, T; Sorrenti, M; Bettinetti, G; Rodriquez, L; Zecchi, V. Chitosan/pectin polyelectrolyte complexes: Selection of suitable preparative conditions for colon-specific delivery of vancomycin. Eur J Pharm Sci 2008, 35, 435–441. [Google Scholar]
- Mundargi, R; Patil, SA; Aminabhavi, TM. Evaluation of acrylamide-grafted-xanthan gum copolymer matrix tablets for oral controlled delivery of antihypertensive drugs. Carbohydr Polym 2007, 69, 130–141. [Google Scholar]
- Argin-Soysal, S; Kofinas, P; Lo, YM. Effect of complexation conditions on xanthan-chitosan polyelectrolyte complex gels. Food Hydrocol 2009, 23, 202–209. [Google Scholar]
- Magnin, D; Lefebvre, J; Chornet, E; Dumitriu, S. Physicochemical and structural characterisation of a polyionic matrix of interest in biotechnology, in the pharmaceutical and biomedical fields. Carbohydr Polym 2004, 55, 437–453. [Google Scholar]
- Dumitriu, S; Magny, P; Montane, D; Vidal, PF; Chornet, E. Polyionic hydrogels obtained by complexation: Their properties as support for enzyme immobilization. J Bioact Compat Polym 1994, 9, 184–209. [Google Scholar]
- Dumitriu, S; Chornet, E; Vidal, PF; Moresoli, C. Polyionic hydrogels as support for immobilization of lipase. Biotechnol Tech 1995, 9, 833–836. [Google Scholar]
- Kim, SJ; Shin, SR; Lee, SM; Kim, IY; Kim, SI. Thermal characteristics of polyelectrolyte complexes composed of chitosan and hyaluronic acid. J Macromol Sci 2003, A40, 807–815. [Google Scholar]
- Rinaudo, M. Properties and degradation of selected polysaccharides: hyaluronan and chitosan. Corros Eng Sci Technol 2007, 42, 324–334. [Google Scholar]
- Denuziere, A; Ferrier, D; Damour, O; Domard, A. Chitosan-chondroitin sulphate and chitosan-hyaluronate polyelectrolyte complexes: biological properties. Biomaterials 1998, 19, 1275–1285. [Google Scholar]
- Vasiliu, S; Popa, M; Rinaudo, M. Polyelectrolyte capsules made of two biocompatible natural polymers. Eur Polym J 2005, 41, 923–932. [Google Scholar]
- Fang, J-Y; Chen, J-P; Leu, Y-L; Hu, J-W. Temperature-sensitive hydrogels composed of chitosan and hyaluronic acid as injectable carriers for drug delivery. Eur J Pharm Biopharm 2008, 68, 626–636. [Google Scholar]
- Oyarzun-Ampuero, FA; Brea, J; Loza, MI; Torres, D; Alonso, MJ. Chitosan-hyaluronic acid nanoparticles loaded with heparin for the treatment of asthma. Int J Pharm 2009, 381, 122–129. [Google Scholar]
- Vinod, VTP; Sashidhar, RB; Suresh, KI; Rama Rao, B; Vijaya Saradhi, UVR; Prabhakar Rao, T. Morphological, physic-chemical and structural characterisation of gum kondagogu (Cochlospermum gossypium): A tree gum from India. Food Hydrocol 2008, 22, 899–915. [Google Scholar]
- Vinod, VTP; Sashidhar, RB. Solution and conformational properties of gum kondagogu (Cochlospermum gossypium) A natural product with immense potential as a food additive. Food Chem 2009, 116, 686–692. [Google Scholar]
- Attama, AA. Polyelectrolyte complexes of EudragitL30D-55 and gelatine: Antinociceptive activity of entrapped piroxicam. Drug Deliv 2007, 14, 155–162. [Google Scholar]
- Yin, Y; Li, Z; Sun, Y; Yao, K. A preliminary study on chitosan/gelatine polyelectrolyte complex formation. J Mater Sci (Letters) 2005, 40, 4649–4652. [Google Scholar]
- Foda, NH; El-laithy, HM; Tadros, MI. Implantable biodegradable sponges: Effect of interpolymer complex formation of chitosan with gelatine on the release behaviour of Tramadol hydrochloride. Drug Dev Ind Pharm 2007, 33, 7–17. [Google Scholar]
- Hsieh, C-Y; Tsai, S-P; Wang, D-M; Chang, Y-N; Hsieh, H-J. Preparation of γ-PGA/chitosan composite tissue engineering matrices. Biomaterials 2005, 26, 5617–5623. [Google Scholar]
- Hajdu, I; Bodnár, M; Filipscei, G; Hartmann, JF; Daróczi, L; Zrínyi, M; Borbély, J. Nanoparticles prepared by self-assembly of chitosan and poly-γ-glutamic acid. Colloid Polym Sci 2008, 286, 343–350. [Google Scholar]
- Keresztessy, Z; Bodnár, M; Ber, E; Hajdu, I; Zhang, M; Hartmann, JF. Self-assembling chitosan/poly-γ- glutamic acid nanoparticles for targeted drug delivery. Colloid Polym Sci 2009, 287, 759–765. [Google Scholar]
- Sonaje, K; Lin, YH; Juang, JH; Wey, SP; Chen, CT; Sung, HW. In vivo evaluation of safety and efficacy of self-assembled nanoparticles for oral insulin delivery. Biomaterials 2009, 30, 2329–2339. [Google Scholar]
- Sonaje, K; Chen, Y-J; Chen, H-L; Wey, SP; Juang, JH; Nguyen, HN; Hsu, CW; Lin, KJ; Sung, HW. Enteric-coated capsules filled with freeze-dried chitosan/poly (γ-glutamic acid) nanoparticles for oral insulin delivery. Biomaterials 2010, 31, 3384–3394. [Google Scholar]
- Xiao, C; Fang, F. Ionic self-assembly and characterization of a polysaccharide-based polyelectrolyte complex of maleic starch half-ester acid with chitosan. J Appl Polym Sci 2008, 112, 2255–2260. [Google Scholar]
- Kriwet, B; Kissel, T. Interactions between bioadhesive poly (acrylic acid) and calcium ions. Int J Pharm 1996, 127, 135–145. [Google Scholar]
- Lee, M-H; Chun, M-K; Choi, H-K. Preparation of Carbopol/Chitosan interpolymer complex as a controlled release tablet matrix; effects of complex formation medium on drug release characteristics. Arch Pharm Res 2008, 31, 932–937. [Google Scholar]
- Park, S-H; Chun, M-K; Choi, H-K. Preparation of an extended release matrix tablet using chitosan/Carbopol interpolymer complex. Int J Pharm 2008, 347, 39–44. [Google Scholar]
- Silva, CL; Pereira, JC; Ramalho, A; Pais, AACC; Sousa, JSJ. Films based on chitosan polyelectrolyte complexes for skin drug delivery: Development and characterisation. J Memb Sci 2008, 320, 268–279. [Google Scholar]
- Lu, Z; Chen, W; Hamman, JH. Chitosan-polycarbophil complexes in swellable matrix systems for controlled drug release. Curr Drug Deliv 2007, 4, 257–263. [Google Scholar]
- Lu, Z; Chen, W; Olivier, EI; Hamman, JH. Matrix polymeric excipients: Comparing a novel polyelectrolyte complex with hydroxypropylmethylcellulose. Drug Deliv 2008, 15, 87–96. [Google Scholar]
- Lu, Z; Chen, W; Hamman, JH; Ni, J; Zhai, X. Chitosan-polycarbophil interpolyelectrolyte complex as an excipient for bioadhesive matrix systems to control macromolecular drug delivery. Pharm Dev Technol 2008, 13, 37–47. [Google Scholar]
- Kibbe, AH. Handbook of Pharmaceutical Excipients; Pharmaceutical Press: Washington, DC, USA, 2000; pp. 401–406. [Google Scholar]
- Moustafine, RI; Kabanova, TV; Kemenova, VA; Van den Mooter, G. Characteristics of interpolyelectrolyte complexes of Eudragit E100 with Eudragit L100. J Control Release 2005, 103, 191–198. [Google Scholar]
- Lorenzo-Lamosa, ML; Remuñán-Lopez, C; Vila-Jato, JL; Alonso, MJ. Design of microencapsulated chitosan microspheres for colonic drug delivery. J Control Release 1998, 52, 109–118. [Google Scholar]
- Yoshizawa, T; Shin-ya, Y; Hong, K-J; Kajiuchi, T. PH and temperature-sensitive release behaviours from polyelectrolyte complex films composed of chitosan and PAOMA copolymer. Eur J Pharm Biopharm 2005, 59, 307–313. [Google Scholar]
- Lankalapalli, S; Kolapalli, VRM. Polyelectrolyte complexes: A review of their applicability in drug delivery technology. Ind J Pharm Sci 2009, 71, 481–487. [Google Scholar]
- Shiraishi, S; Imai, T; Otagiri, M. Controlled release of indomethacin by chitosan-polyelectrolyte complex: Optimization and in vivo/in vitro evaluation. J Control Release 1993, 25, 217–225. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hamman, J.H. Chitosan Based Polyelectrolyte Complexes as Potential Carrier Materials in Drug Delivery Systems. Mar. Drugs 2010, 8, 1305-1322. https://doi.org/10.3390/md8041305
Hamman JH. Chitosan Based Polyelectrolyte Complexes as Potential Carrier Materials in Drug Delivery Systems. Marine Drugs. 2010; 8(4):1305-1322. https://doi.org/10.3390/md8041305
Chicago/Turabian StyleHamman, Josias H. 2010. "Chitosan Based Polyelectrolyte Complexes as Potential Carrier Materials in Drug Delivery Systems" Marine Drugs 8, no. 4: 1305-1322. https://doi.org/10.3390/md8041305
APA StyleHamman, J. H. (2010). Chitosan Based Polyelectrolyte Complexes as Potential Carrier Materials in Drug Delivery Systems. Marine Drugs, 8(4), 1305-1322. https://doi.org/10.3390/md8041305



