Potential Anti-HIV Agents from Marine Resources: An Overview
Abstract
:1. Introduction
2. Potential Marine-Derived Anti-HIV Agents and Their Anti-HIV Activity
2.1. Phlorotannins
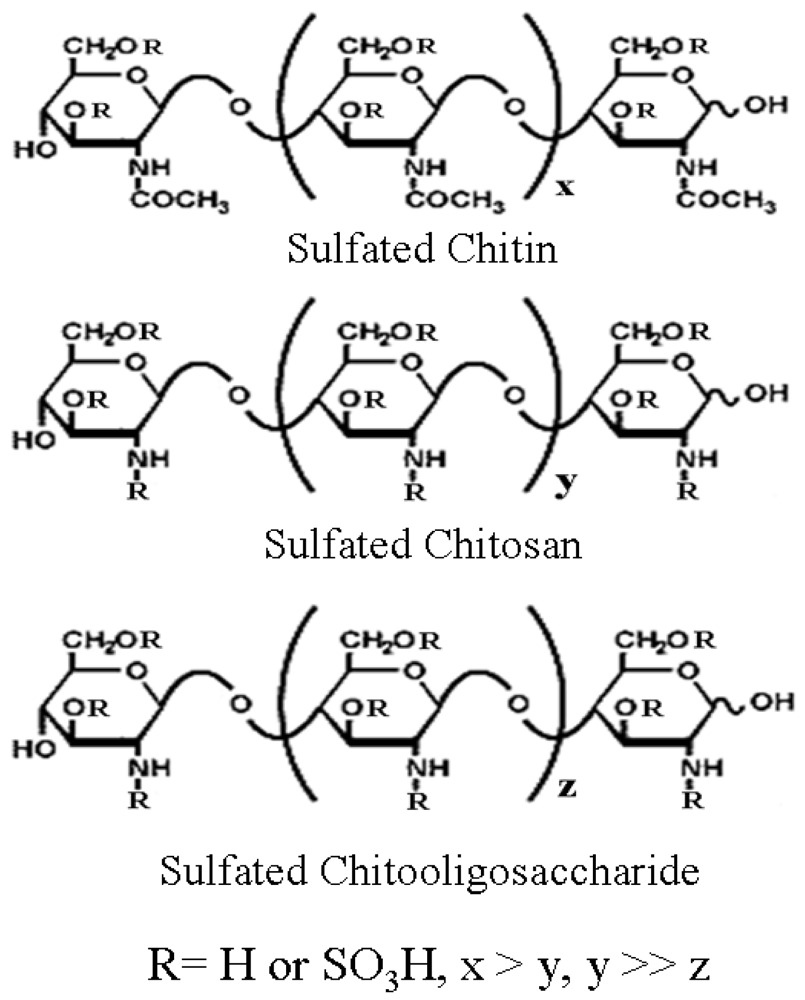
2.2. Chitin, Chitosan and Chitooligosaccharide Derivatives
2.3. Sulfated Polysaccharides
2.4. Lectins
2.5. Bioactive Peptides
3. Prospects of Marine Anti-HIV Drugs
4. Conclusion
Acknowledgements
References
- Ojewole, E; Mackraj, I; Naidoo, P; Govender, T. Exploring the use of novel drug delivery systems for antiretroviral drugs. Eur J Pharm Biopharm 2008, 70, 697–710. [Google Scholar]
- Govender, T; Ojewole, E; Naidoo, P. Polymeric nanoparticles for enhancing antiretroviral drug therapy. Drug Deliv 2008, 15, 493–501. [Google Scholar]
- Clavel, F; Hance, AJ. HIV drug resistance. N Engl J Med 2004, 350, 1023–1035. [Google Scholar]
- Lee, SA; Hong, SK; Suh, CI; Oh, MH; Park, JH; Choi, BW; Park, SW; Paik, SY. Anti-HIV-1 efficacy of extracts from medicinal plants. J Microbiol 2010, 48, 249–252. [Google Scholar]
- Tantillo, C; Ding, J; Jacobo-Molina, A; Nanni, RG; Boyer, PL; Hughes, SH; Pauwels, R; Andries, K; Janssen, PAJ; Arnold, E. Locations of anti-AIDS drug binding sites and resistance mutations in the three-dimensional structure of HIV-1 reverse transcriptase. Implications for mechanisms of drug inhibition and resistance. J Mol Biol 1994, 243, 369–387. [Google Scholar]
- Lipsky, JJ. Antiretroviral drugs for AIDS. Lancet 1996, 348, 800–803. [Google Scholar]
- Tzileleka, LA; Vagias, C; Roussis, V. Natural products with anti-HIV activity from marine organisms. Curr Top Med Chem 2003, 3, 1512–1535. [Google Scholar]
- Singh, IP; Bharate, SB; Bhutani, KK. Anti-HIV natural products. Curr Sci 2005, 89, 269–290. [Google Scholar]
- Schaeffer, DJ; Krylov, VS. Anti-HIV activity of extracts and compounds from algae and cyanobacteria. Ecotox Environ Safe 2000, 45, 208–227. [Google Scholar]
- Vlietinck, AJ; De Bruyne, T; Apers, S; Pieters, LA. Plantderived leading compounds for chemotherapy of human immunodeficiency virus (HIV) infection. Planta Med 1998, 64, 97–109. [Google Scholar]
- De Clercq, E. Current lead natural products for the chemotherapy of human immuno deficiency virus infection. Med Res Rev 2000, 20, 323–349. [Google Scholar]
- Kong, JM; Goh, NK; Chia, LS; Chia, TF. Recent advances in traditional plant drugs and orchids. Acta Pharmacol Sin 2003, 24, 7–21. [Google Scholar]
- Wang, RR; Gu, Q; Wang, YH; Zhang, XM; Yang, LM; Zhou, J; Chen, JJ; Zheng, YT. Anti-HIV-1 activities of compounds isolated from the medicinal plant Rhus chinensis. J Ethnopharmacol 2008, 17, 249–256. [Google Scholar]
- Hupfeld, J; Efferth, T. Drug resistance of human immunodeficiency virus and overcoming it by natural products. In Vivo 2009, 23, 1–6. [Google Scholar]
- Aneiros, A; Garateix, A. Bioactive peptides from marine sources: pharmacological properties and isolation procedures. J Chromatogr B Analyt Technol Biomed Life Sci 2004, 803, 41–53. [Google Scholar]
- Wijesekara, I; Kim, SK. Angiotensin-I-converting enzyme (ACE) inhibitors from marine resources: prospects in the pharmaceutical industry. Mar Drugs 2010, 8, 1080–1093. [Google Scholar]
- Pomponi, SA. The oceans and human health: the discovery and development of marine-derived drugs. Oceonogrophy 2001, 14, 78–87. [Google Scholar]
- Targett, NM; Arnold, TM. Predicting the effects of brown algal phlorotannins on marine herbivores in tropical and temperate oceans. J Phycol 1998, 34, 195–205. [Google Scholar]
- Shibata, T; Kawaguchi, S; Hama, Y; Inagaki, M; Yamaguchi, K; Nakamura, T. Local and chemical distribution of phlorotannins in brown algae. J Appl Phycol 2004, 16, 291–296. [Google Scholar]
- Ahn, GN; Kim, KN; Cha, SH; Song, CB; Lee, J; Heo, MS; Yeo, IK; Lee, NH; Jee, YH; Kim, JS; Heu, MS; Jeon, YJ. Antioxidant activities of phlorotannins purified from Ecklonia cava on free radical scavenging using ESR and H2O2-mediated DNA damage. Eur Food Res Technol 2007, 226, 71–79. [Google Scholar]
- Singh, IP; Bharate, SB. Phloroglucinol compounds of natural origin. Nat Prod Rep 2006, 23, 558–591. [Google Scholar]
- Glombitza, KW; Li, SM. Hydroxyphlorethols from the brown alga Carpophyllum maschalocarpum. Phytochemistry 1991, 30, 2741–2745. [Google Scholar]
- La Barre, S; Potin, P; Leblanc, C; Delage, L. The halogenated metabolism of brown algae (Phaeophyta), its biological importance and its environmental significance. Mar Drugs 2010, 8, 988–1010. [Google Scholar]
- Ahn, MJ; Yoon, KD; Min, SY; Lee, JS; Kim, JH; Kim, TG; Kim, SH; Kim, NG; Huh, H; Kim, J. Inhibition of HIV-1 reverse transcriptase and protease by phlorotannins from the brown alga Ecklonia cava. Biol Pharm Bull 2004, 27, 544–547. [Google Scholar]
- Ahn, MJ; Yoon, KD; Kim, CY; Kim, JH; Shin, CG; Kim, J. Inhibitory activity on HIV-1 reverse transcriptase and integrase of a carmalol derivative from a brown alga Ishige okamurae. Phytother Res 2006, 20, 711–713. [Google Scholar]
- Artan, M; Li, Y; Karadeniz, F; Lee, SH; Kim, MM; Kim, SK. Anti-HIV-1 activity of phloroglucinol derivative, 6,6-bieckol, from. Eckloniacava Bioorg Med Chem 2008, 16, 7921–7926. [Google Scholar]
- Carroll, SS; Olsen, DB; Bennett, CD; Gotlib, L; Graham, DJ; Condraq, JH; Stern, AM; Shafer, JA; Kuo, LC. Inhibition of HIV-I reverse transcriptase by pyridinone derivatives. J Biol Chem 1993, 268, 276–281. [Google Scholar]
- Zhang, H; Vrang, L; Backbro, K; Lind, P; Sahlberg, C; Unge, T; Oberg, B. Inhibition of human immunodeficiency virus type 1 wild-type and mutant reverse transcriptases by the phenyl ethyl thiazolyl thiourea derivatives trovirdine and MSC-127. Antivir Res 1995, 28, 331–342. [Google Scholar]
- Ngo, DN; Kim, MM; Kim, SK. Chitin oligosaccharides inhibit oxidative stress in live cells. Carbohydr Polym 2008, 74, 228–234. [Google Scholar]
- Shahidi, F; Arachchi, JKV; Jeon, YJ. Food applications of chitin and chitosans. Trends Food Sci Technol 1999, 10, 37–51. [Google Scholar]
- Kim, SK; Nghiep, ND; Rajapakse, N. Therapeutic prospectives of chitin, chitosan and their derivatives. J Chitin Chitosan 2006, 11, 1–10. [Google Scholar]
- Jayakumar, R; New, N; Nagagama, H; Furuike, T; Tamura, H. Synthesis, characterization and biospecific degradation behavior of sulfated chitin. Macromol Symp 2008, 264, 163–167. [Google Scholar]
- Suwan, J; Zhang, Z; Li, B; Vongchan, P; Meepowpan, P; Zhang, F; Mousa, SA; Mousa, S; Premanode, B; Kongtawelert, P; Linhardt, RJ. Sulfonation of papain-treated chitosan and its mechanism for anticoagulant activity. Carbohydr Res 2009, 344, 1190–1196. [Google Scholar]
- He, Q; Ao, Q; Wang, A; Gong, Y; Zhao, N; Zhang, X. In vitro cytotoxicity and protein drug release properties of chitosan/heparin microspheres. Tsinghua Sci Technol 2007, 12, 361–365. [Google Scholar]
- Thierry, B; Merhi, Y; Silver, J; Tabrizian, M. Biodegradable membrane-covered stent from chitosan-based polymers. J Biomed Mater Res A 2005, 75, 556–566. [Google Scholar]
- Prabaharan, M; Reis, RL; Mano, JF. Carboxymethyl chitosan-graft-phosphatidylethanolamine: Amphiphilic matrices for controlled drug delivery. React Funct Polym 2007, 67, 43–52. [Google Scholar] [Green Version]
- Jayakumar, R; Nwe, N; Tokura, S; Tamura, H. Sulfated chitin and chitosan as novel biomaterials. Int J Biol Macromol 2007, 40, 175–181. [Google Scholar]
- Saiki, I; Murata, J; Nakajima, M; Tokura, S; Azuma, I. Inhibition by sulfated chitin derivatives of invasion through extracellular matrix and enzymatic degradation by metastatic melanoma cells. Cancer Res 1990, 50, 3631–3637. [Google Scholar]
- Vasyukova, NI; Chalenko, GI; Gerasimova, NG; Perekhod, EA; Ozeretskovskaya, OL; Irina, AV; Varlamov, VP; Albulov, AI. Chitin and chitosan derivatives as elicitors of potato resistance to late blight. Appl Biochem Micro 2000, 36, 372–376. [Google Scholar]
- Xing, R; Liu, S; Yu, H; Zhang, Q; Li, Z; Li, P. Preparation of low-molecular-weight and highsulfate-content chitosans under microwave radiation and their potential antioxidant activity in vitro. Carbohyd Res 2004, 339, 2515–2519. [Google Scholar]
- Sosa, MA; Fazely, F; Koch, JA; Vercellotti, SV; Ruprecht, RM. N-carboxymethylchitosan-N,O-sulfate as an anti-HIV-1 agent. Biochem Biophys Res Commun 1991, 174, 489–496. [Google Scholar]
- Nishimura, SI; Kai, H; Shinada, K; Yoshida, T; Tokura, S; Kurita, K; Nakashima, H; Yamamoto, N; Uryu, T. Regioselective syntheses of sulfated polysaccharides: specific anti-HIV-1 activity of novel chitin sulfates. Carbohydr Res 1998, 306, 427–433. [Google Scholar]
- Lee, DS; Kim, YM; Lee, MS; Ahn, CB; Jung, WK; Je, JY. Synergistic effects between aminoethyl-chitosans and β-lactams against methicillin-resistant Staphylococcus aureus (MRSA). Bioorg Med Chem Lett 2010, 20, 975–978. [Google Scholar]
- Artan, M; Karadeniz, F; Kim, MM; Kim, SK. Chitosan derivatives as HIV-1 inhibitors. J Biotechnol 2008, 136S, S527–540. [Google Scholar]
- Dou, JL; Tan, CY; Du, YG; Bai, XF; Wang, KY; Ma, XJ. Effects of chitooligosaccharides on rabbit neutrophils in vitro. Carbohydr Polym 2007, 69, 209–213. [Google Scholar]
- Jeon, YJ; Kim, SK. Continuous production of chitooligosaccharides using a dual reactor system. Process Biochem 2000, 35, 623–632. [Google Scholar]
- Jeon, YJ; Kim, SK. Production of chitooligosaccharides using ultrafiltration membrane reactor and their antibacterial activity. Carbohydr Polym 2000, 41, 133–141. [Google Scholar]
- Yang, EJ; Kim, JG; Kim, JY; Kim, SC; Lee, NH; Hyun, CG. Anti-inflammatory effect of chitosan oligosaccharides in RAW 264.7 cells. Cent Eur J Biol 2010, 5, 95–102. [Google Scholar]
- Chae, SY; Jang, MK; Nah, JW. Influence of molecular weight on oral absorption of water soluble chitosans. J Control Release 2005, 102, 383–394. [Google Scholar]
- Hong, SP; Kim, MH; Oh, SW; Han, CH; Kim, YH. ACE inhibitory and antihypertensive effect of chitosan oligosaccharides in SHR. Korean J Food Sci Technol 1998, 30, 1476–1479. [Google Scholar]
- Park, PJ; Je, JY; Kim, SK. Free radical scavenging activity of chitooligosaccharides by electron spin resonance spectrometry. J Agric Food Chem 2003, 51, 4624–4627. [Google Scholar]
- Park, PJ; Lee, HK; Kim, SK. Preparation of hetero-chitooligosaccharides and their antimicrobial activity on Vibrio parahaemolyticus. J Microbiol Biotechnol 2004, 14, 41–47. [Google Scholar]
- Shen, KT; Chen, MH; Chan, HY; Jeng, JH; Wang, YJ. Inhibitory effects of chitooligosaccharides on tumor growth and metastasis. Food Chem Toxicol 2009, 47, 1864–1871. [Google Scholar]
- Jeon, YJ; Kim, SK. Antitumor activity of chitosan oligosaccharides produced in ultrafiltration membrane reactor system. J Microbiol Biotechnol 2002, 12, 503–507. [Google Scholar]
- Jeon, YJ; Kim, SK. Potential immuno-stimulating effect of antitumoral fraction of chitosan oligosaccharides. J Chitin Chitosan 2001, 6, 163–167. [Google Scholar]
- Liu, B; Liu, WS; Han, BQ; Sun, YY. Antidibetic effects of chito-oligosaccharides on pancreatic islet cells in streptozotocin-induced diabetic rats. World J Gastroenterol 2007, 13, 725–731. [Google Scholar]
- Kim, KN; Joo, ES; Kim, KI; Kim, SK; Yang, HP; Jeon, YJ. Effect of chitosan oligosaccharides on cholesterol level and antioxidant enzyme activities in hypercholesterolemic rat. J Korean Soc Food Sci Nutr 2005, 34, 36–41. [Google Scholar]
- Miura, T; Usami, M; Tsuura, Y; Ishida, H; Seino, Y. Hypoglycemic and hypolipidemic effect of chitosan in normal and neonatal streptozotocin-induced diabetic mice. Biol Pharm Bull 1995, 18, 1623–1625. [Google Scholar]
- Yoon, NY; Ngo, DN; Kim, SK. Acetylcholinesterase inhibitory activity of novel chitooligosaccharide derivatives. Carbohydr Polym 2009, 78, 869–872. [Google Scholar]
- Park, PJ; Je, JY; Jung, WK; Kim, SK. Anticoagulant activity of heterochitosans and their oligosaccharide sulfates. Eur Food Res Technol 2004, 219, 529–533. [Google Scholar]
- Cho, EJ; Rahman, A; Kim, SW; Baek, YM; Hwang, HJ; Oh, JY; Hwang, HS; Lee, SK; Yun, JW. Chitosan oligosaccharides inhibit adipogenesis in 3T3-L1 adipocytes. J Microbiol Biotechnol 2008, 18, 80–87. [Google Scholar]
- Artan, M; Karadeniz, F; Karagozlu, MZ; Kim, MM; Kim, SK. Anti-HIV-1 activity of low molecular weight sulfated chitooligosaccharides. Carbohydr Res 2010, 345, 656–662. [Google Scholar]
- Vijayavel, K; Anbuselvam, C; Balasubramanian, MP. Free radical scavenging activity of the marine mangrove Rhizophora apiculata bark extract with reference to naphthalene induced mitochondrial dysfunction. Chem Biol Interact 2006, 163, 170–175. [Google Scholar]
- Alban, S; Franz, G. Partial synthetic glucan sulfates as potential new antithrombotics: a review. Biomacromolecules 2001, 2, 354–361. [Google Scholar]
- Costa, LS; Fidelis, GP; Cordeiro, SL; Oliveira, RM; Sabry, DA; Camara, RBG; Nobre, LTDB; Costa, MSSP; Almeida-Lima, J; Farias, EHC; Leite, EL; Rocha, HAO. Biological activities of sulfated polysaccharides from tropical seaweeds. Biomed Pharmacother 2010, 64, 21–28. [Google Scholar]
- Ley, K; Cerrito, M; Arfors, KE. Sulfated polysaccharides inhibit leukocyte rolling in rabbit mesentery venules. Am J Physiol Heart Circ Physiol 1991, 260, H1667–1673. [Google Scholar]
- Majdoub, H; Ben Mansour, M; Chaubet, F; Roudesli, MS; Maaroufi, RM. Anticoagulant activity of a sulfated polysaccharide from the green alga Arthrospira platensis. Biochim Biophys Acta 2009, 1790, 1377–1381. [Google Scholar]
- Synytsya, A; Kim, WJ; Kim, SM; Pohl, R; Synytsya, A; Kvasnicka, F; Copikova, J; Park, YI. Structure and antitumor activity of fucoidan isolated from sporophyll of Korean seaweed Undaria pinnatifida. Carbohydr Polym 2010, 81, 41–48. [Google Scholar]
- Na, YS; Kim, WJ; Kim, SM; Park, JW; Lee, SM; Kim, SO; Synytsya, A; Park, YI. Purification, characterization and immunostimulating activity of water-soluble polysaccharide isolated from Capsosiphon fulvescens. Int Immunopharmacol 2010, 10, 364–370. [Google Scholar]
- Witvrouw, M; De Clercq, E. Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen Pharmacol 1997, 29, 497–511. [Google Scholar]
- Adhikari, U; Mateu, CG; Chattopadhyay, K; Pujol, CA; Damonte, EB; Ray, B. Structure and antiviral activity of sulfated fucans from Stoechospermum marginatum. Phytochemistry 2006, 67, 2474–2482. [Google Scholar]
- Damonte, EB; Matulewicz, MC; Cerezo, AS. Sulfated seaweed polysaccharides as antiviral agents. Curr Med Chem 2004, 11, 2399–2419. [Google Scholar]
- Luscher-Mattil, M. Polyanions–a lost chance in the fight against HIV and other virus diseases. Antivir Chem Chemoth 2000, 11, 249–259. [Google Scholar]
- Chattopadhyay, N; Ghosh, T; Sinha, S; Chattopadhyay, K; Karmakar, P; Ray, B. Polysaccharides from Turbinaria conoides: Structural features and antioxidant capacity. Food Chem 2010, 118, 823–829. [Google Scholar]
- Mandal, P; Mateu, CG; Chattopadhyay, K; Pujol, CA; Damonte, EB; Ray, B. Structural features and antiviral activity of sulfated fucans from the brown seaweed Cystoseira indica. Antivir Chem Chemother 2007, 18, 153–162. [Google Scholar]
- Mandal, P; Pujol, CA; Carlucci, MJ; Chattopadhyay, K; Damonte, EB; Ray, B. Sulfated xylomannan of Scinaia hetai: Isolation, structural features and antiviral activity. Phytochemistry 2008, 69, 2193–2199. [Google Scholar]
- Bourgougnon, N; Lahaye, M; Quemener, B; Chermann, JC; Rimbert, M; Cormaci, M; Furnari, G; Komprobst, JM. Annual variation in composition and in vitro anti-HIV-1 activity of the sulfated glucuronogalactan from Schizymenia dubyi (Rhodophyta, Gigartinales). J App Phycol 1996, 8, 155–161. [Google Scholar]
- Riccio, R; Kinnel, RB; Bifulco, G; Scheuer, PJ. Kakelokelose, a sulfated mannose polysaccharide with anti-HIV activity from the pacific tunicate Didemnum molle. Tetrahedron Lett 1996, 37, 1979–1982. [Google Scholar]
- Amornrut, C; Toida, T; Imanari, T; Woo, ER; Park, H; Linhardt, R; Wu, SJ; Kim, YS. A new sulfated β-galactan from clams with anti-HIV activity. Carbohyd Res 1999, 321, 121–127. [Google Scholar]
- Meiyu, G; Fuchuan, L; Xianliang, X; Jing, L; Zuowei, Y; Huashi, G. The potential molecular targets of marine sulfated polymannuroguluronate interfering with HIV-1 entry. Interaction between SPMG and HIV-1 rgp120 and CD4 molecule. Antivir Res 2003, 59, 127–135. [Google Scholar]
- Liu, H; Geng, M; Xin, X; Li, F; Zhang, Z; Li, J; Ding, J. Multiple and multivalent interactions of novel anti-AIDS drug candidates, sulfated polymannuronate (SPMG)-derived oligosaccharides, with gp120 and their anti-HIV activities. Glycobiology 2005, 15, 501–510. [Google Scholar]
- Lee, JB; Hayashi, K; Hirata, M; Kuroda, E; Suzuki, E; Kubo, Y; Hayashi, T. Antiviral sulfated polysaccharide from Navicula directa, a diatom collected from deep-sea water in Toyama bay. Biol Pharm Bull 2006, 29, 2135–2139. [Google Scholar]
- Wang, SC; Bligh, SWA; Shi, SS; Wang, ZT; Hu, ZB; Crowder, J; Branford-White, C; Vella, C. Structural features and anti-HIV-1 activity of novel polysaccharides from red algae Grateloupia longifolia and Grateloupia filicina. Int J Biol Macromol 2007, 41, 369–375. [Google Scholar]
- Queiroz, KCS; Medeiros, VP; Queiroz, LS; Abreu, LRD; Rocha, HAO; Ferreira, CV; Juca, MB; Aoyama, H; Leite, EL. Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. Biomed Pharmacother 2008, 62, 303–307. [Google Scholar]
- Trinchero, J; Ponce, NMA; Cordoba, OL; Flores, ML; Pampuro, S; Stortz, CA; Salomon, H; Turk, G. Antiretroviral activity of fucoidans extracted from the brown seaweed Adenocystis utricularis. Phytother Res 2009, 23, 707–712. [Google Scholar]
- Miao, B; Li, J; Fu, X; Gan, L; Xin, X; Geng, M. Sulfated polymannuroguluronate, a novel anti-AIDS drug candidate, inhibits T cell apoptosis by combating oxidative damage of mitochondria. Mol Pharmacol 2005, 68, 1716–1727. [Google Scholar]
- Sato, T; Hori, K. Cloning, expression, and characterization of a novel anti-HIV lectin from the cultured cyanobacterium Oscillatoria agardhii. Fish Sci 2009, 75, 743–753. [Google Scholar]
- Geyer, H; Holschbach, C; Hunsmann, G; Schneider, J. Carbohydrates of human immunodeficiency virus. Structures of oligosaccharides linked to the envelope glycoprotein 120. J Biol Chem 1988, 263, 11760–11767. [Google Scholar]
- Balzarini, J. Carbohydrate-binding agents: a potential future cornerstone for the chemotherapy of enveloped viruses. Antivir Chem Chemother 2007, 18, 1–11. [Google Scholar]
- Balzarini, J. Targeting the glycans of glycoproteins: a novel paradigm for antiviral therapy. Nat Rev Microbiol 2007, 5, 583–597. [Google Scholar]
- Balzarini, J; Laethem, KV; Daelemans, D; Hatse, S; Bugatti, A; Rusnati, M; Igarashi, Y; Oki, T; Schols, D. Pradimicin A, a carbohydrate-binding nonpeptidic lead compound for treatment of infections with viruses with highly glycosylated envelopes, such as human immunodeficiency virus. J Virol 2007, 81, 362–373. [Google Scholar]
- Mori, T; O’Keefe, BR; Sowder, RC, II; Bringans, S; Gardella, R; Berg, S; Cochran, P; Turpin, JA; Buckheit, RW, Jr; McMahon, JB; Boyd, MR. Isolation and characterization of Griffithsin, a novel HIV-inactivating protein, from the red alga Griffithsia sp. J Biol Chem 2005, 280, 9345–9353. [Google Scholar]
- Wang, JH; Kong, J; Li, W; Molchanova, V; Chikalovets, I; Belogortseva, N; Luk’yanov, P; Zheng, YT. A β-galactose-specific lectin isolated from the marine worm Chaetopterus variopedatus possesses anti-HIV-1 activity. Comp Biochem Physiol 2006, 142, 111–117. [Google Scholar]
- Molchanova, V; Chikalovets, I; Chernikov, O; Belogortseva, N; Li, W; Wang, JH; Ou Yang, DY; Zheng, YT; Lukyanov, P. A new lectin from the sea worm Serpula vermicularis: Isolation, characterization and anti-HIV activity. Comp Biochem Physiol 2007, 145, 184–193. [Google Scholar]
- Luk’yanov, PA; Chernikov, OV; Kobelev, SS; Chikalovets, IV; Molchanova, VI; Li, W. Carbohydrate-Binding Proteins of Marine Invertebrates. Russ J Bioorg Chem 2007, 33, 161–169. [Google Scholar]
- Je, JY; Qian, ZJ; Lee, SH; Byun, HG; Kim, SK. Purification and antioxidant properties of bigeye tuna (Thunnus obesus) dark muscle peptide on free radical-mediated oxidation systems. J Med Food 2008, 11, 629–637. [Google Scholar]
- Sheih, IC; Fang, TJ; Wu, TK. Isolation and characterization of a novel angiotensin I-converting enzyme (ACE) inhibitory peptide from the algae protein waste. Food Chem 2009, 115, 279–284. [Google Scholar]
- Slizyte, R; Mozuraityte, R; Martinez-Alvarez, O; Falch, E; Fouchereau-Peron, M; Rustad, T. Functional, bioactive and antioxidative properties of hydrolysates obtained from cod (Gadus morhua) backbones. Process Biochem 2009, 44, 668–677. [Google Scholar]
- Suetsuna, K; Chen, JR. Identification of antihypertensive peptides from peptic digest of two microalgae, Chlorella vulgaris and Spirulina platensis. Mar Biotechnol 2001, 3, 305–309. [Google Scholar]
- Zhao, Y; Li, B; Liu, Z; Dong, S; Zhao, X; Zeng, M. Antihypertensive effect and purification of an ACE inhibitory peptide from sea cucumber gelatin hydrolysate. Process Biochem 2007, 42, 1586–1591. [Google Scholar]
- Simpson, BK; Nayeri, G; Yaylayan, V; Ashie, INA. Enzymatic hydrolysis of shrimp meat. Food Chem 1998, 61, 131–138. [Google Scholar]
- Korhonen, H; Pihlanto, A. Bioactive peptides: Production and functionality. Int Dairy J 2006, 16, 945–960. [Google Scholar]
- Pihlanto-Leppala, A. Bioactive peptides derived from bovine whey proteins: opioid and ACE-inhibitory peptides. Trends Food Sci Technol 2001, 11, 347–356. [Google Scholar]
- Byun, HG; Kim, SK. Purification and characterization of angiotensin I converting enzyme (ACE) inhibitory peptides from Alaska Pollack (Theragra chalcogramma) skin. Process Biochem 2001, 36, 1155–1162. [Google Scholar]
- Kim, SY; Je, JY; Kim, SK. Purification and characterization of antioxidant peptide from hoki (Johnius balengerii) frame protein by gastrointestinal digestion. J Nutr Biochem 2007, 18, 31–38. [Google Scholar]
- Mendis, E; Rajapakse, N; Kim, SK. Antioxidant properties of a radical-scavenging peptide purified from enzymatically prepared fish skin gelatin hydrolysate. J Agric Food Chem 2005, 53, 581–587. [Google Scholar]
- Jo, HY; Jung, WK; Kim, SK. Purification and characterization of a novel anticoagulant peptide from marine echiuroid worm Urechis unicinctus. Process Biochem 2008, 43, 179–184. [Google Scholar]
- Rajapakse, N; Jung, WK; Mendis, E; Moon, SH; Kim, SK. A novel anticoagulant purified from fish protein hydrolysate inhibits factor XIIa and platelet aggregation. Life Sci 2005, 76, 2607–2619. [Google Scholar]
- Liu, Z; Dong, S; Xu, J; Zeng, M; Song, H; Zhao, Y. Production of cysteine-rich antimicrobial peptide by digestion of oyster (Crassostrea gigas) with alcalase and bromelin. Food Control 2008, 19, 231–235. [Google Scholar]
- Stensvag, K; Haug, T; Sperstad, SV; Rekdal, O; Indrevoll, B; Styrvold, OB. Arasin 1, a proline-arginine-rich antimicrobial peptide isolated from the spider crab Hyas araneus. Dev Comp Immuno 2008, 32, 275–285. [Google Scholar]
- Lee, TG; Maruyama, S. Isolation of HIV-1 protease-inhibiting peptides from Thermolysin hydrolysate of oyster proteins. Biochem Bioph Res Co 1998, 253, 604–608. [Google Scholar]
- Zampella, A; Valeria D’Auria, M; Paloma, LG; Casapullo, A; Minale, L; Debitus, C; Henin, Y. Callipeltin A, an anti-HIV cyclic depsipeptide from the new Caledonian lithistida sponge Callipelta sp. J Am Chem Soc 1996, 118, 6202–6209. [Google Scholar]
- Ford, PW; Gustafson, KR; McKee, TC; Shigematsu, N; Maurizi, LK; Pannell, LK; Williams, DE; Dilip de Silva, E; Lassota, P; Allen, TM; Soest, RV; Andersen, RJ; Boyd, MR. Papuamides A-D, HIV-inhibitory and cytotoxic depsipeptides from the sponges Theonella mirabilis and Theonella swinhoei collected in Papua new guinea. J Am Chem Soc 1999, 121, 5899–5909. [Google Scholar]
- Rashid, MA; Gustafson, KR; Cartner, LK; Shigematsu, N; Pannell, LK; Boyd, MR. Microspinosamide, a new HIV-inhibitory cyclic depsipeptide from the marine sponge Sidonops microspinosa. J Nat Prod 2001, 64, 117–121. [Google Scholar]
- Oku, N; Gustafson, KR; Cartner, LK; Wilson, JA; Shigematsu, N; Hess, S; Pannell, LK; Boyd, MR; McMahon, JB. Neamphamide A, a new HIV-inhibitory depsipeptide from the Papua new guinea marine sponge Neamphius huxleyi. J Nat Prod 2004, 67, 1407–1411. [Google Scholar]
- Plaza, A; Gustchina, E; Baker, HL; Kelly, M; Bewley, CA. Mirabamides A–D, depsipeptides from the sponge Siliquariaspongia mirabilis that inhibit HIV-1 fusion. J Nat Prod 2007, 70, 1753–1760. [Google Scholar]
- Plaza, A; Bifulco, G; Keffer, JL; Lloyd, JR; Baker, HL; Bewley, CA. Celebesides A-C and Theopapuamides B–D, depsipeptides from an Indonesian sponge that inhibit HIV-1 entry. J Org Chem 2009, 74 , 504–512. [Google Scholar]
- Zampella, A; Sepe, V; Luciano, P; Bellotta, F; Monti, MC; D’Auria, MV; Jepsen, T; Petek, S; Adeline, MT; Laprévôte, O; Aubertin, AM; Debitus, C; Poupat, C; Ahond, A. Homophymine A, an anti-HIV cyclodepsipeptide from the sponge Homophymia sp. J Org Chem 2008, 73, 5319–5327. [Google Scholar]
- Aneiros, A; Garateix, A. Bioactive peptides from marine sources: pharmacological properties and isolation procedures. J Chromatogr B 2004, 803, 41–53. [Google Scholar]
- Andjelic, CD; Planelles, V; Barrows, LR. Characterizing the anti-HIV activity of Papuamide A. Mar Drugs 2008, 6, 528–549. [Google Scholar]
- Bhadury, P; Mohammad, BT; Wright, PC. The current status of natural products from marine fungi and their potential as anti-infective agents. J Ind Microbiol Biotechnol 2006, 33, 325–337. [Google Scholar]
- Yasuhara-Bella, J; Lu, Y. Marine compounds and their antiviral activities. Antiviral Res 2010, 86, 231–240. [Google Scholar]
Abbreviations
| AIDS | acquired immune deficiency syndrome |
| SCOS | sulfated chitooligosaccharide |
| SPs | sulfated polysaccharides |
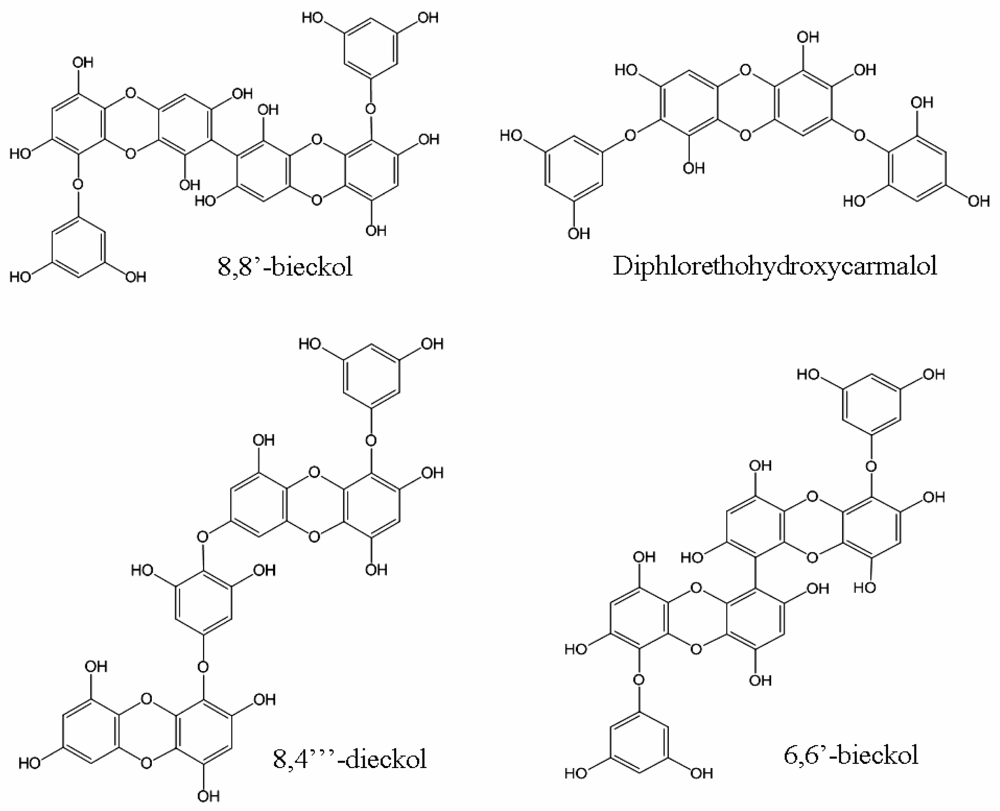
| Compound | IC50a (μM) | Ref. | ||
|---|---|---|---|---|
| RT | Integrase | Protease | ||
| 8,8′-bieckol | 0.51 | 81.5 | [24] | |
| 8,4‴-dieckol | 5.31 | 36.9 | [24] | |
| diphlorethohydroxycarmalol | 9.1 | 25.2 | [25] | |
| 6,6′-bieckol | 1.07 | [26] | ||
| Compound | Mw (kDa) | EC50a (μg/mL) | CC50b (μg/mL) |
|---|---|---|---|
| C-6S | 58 | 57.0 | >1000 |
| C-3S | 28 | 9.6 | >1000 |
| C-2,3S | 16 | 0.28 | >1000 |
| Curdlan sulfate | 79 | 0.10 | >1000 |
| SPs | Major units | Sources | Ref. |
|---|---|---|---|
| Sulfated glucuronogalactan | Galactose | Red algae Schizymenia dubyi | [77] |
| Kakelokelose | Mannose | Pacific tunicate Didemnum molle | [78] |
| Sulfated β-galactan | Galactose | Clam Meretrix petechialis | [79] |
| Sulfated polymannoroguluronate | Mannuronate | Brown algae | [80] |
| Sulfated polymannuronate | Mannuronate | Brown algae | [81] |
| Naviculan | Fucose, Xylose, Galactose, Mannose, Rhamnose | Diatom Navicula directa | [82] |
| Sulfated galactans | Galactose, Xylose | Red algae Grateloupia filicina, Grateloupia longifolia | [83] |
| Sulfated fucans | Fucose | Brown algae Dictyota mertensii, Lobophora variegate, Fucus vesiculosus, Spatoglossum schröederi | [84] |
| Galactofucan | Fucose, Galactose | Brown algae Adenocystis utricularis | [85] |
| Species | Lectin | Carbohydrate specificity | Activity | EC50a | Ref. |
|---|---|---|---|---|---|
| Red algae Griffithsia sp. | GRFT | Man/Glc-specific lectin | Against T cell tropic and macrophage tropic strains of HIV-1 | Ranging from 0.043–0.63 μM | [92] |
| Abort cell-to-cell fusion and transmission of HIV-1 infection | Ranging from 0.043–0.63 μM | ||||
| Marine worm Chaetopterus variopedatus | CVL | β-galactose-specific lectin | Inhibit HIV-induced syncytium formation | 0.0043 μM | [93] |
| Inhibit HIV-1 p24 production | 0.057 μM | ||||
| Marine worm Serpula vermicularis | SVL | GlcNAc-specific lectin | Inhibit HIV-induced syncytium formation | 0.15 μg/mL | [94] |
| Inhibit HIV-1 p24 production | 0.23 μg/mL | ||||
| Marine mussel Crenomytilus grayanus | CGL | High affinity to the glycoproteins of mucin type | Inhibit HIV-replication | 45.7 μg/mL | [95] |
| Ascidium Didemnum ternatanum | DTL | GlcNAc-specific lectin | Inhibit HIV-replication | 0.006 μg/mL | [95] |
| Ascidium Didemnum ternatanum | DTL-A | GlcNAc/GalNAc and heparin-binding lectin | Inhibit HIV-replication | 0.59 μg/mL | [95] |
| Marine worm Serpula vermicularis | SVL-1 | Mannan-binding lectin | Inhibit HIV-replication | 89.1 μg/mL | [95] |
| Marine worm Serpula vermicularis | SVL-2 | GlcNAc-specific lectin | Inhibit HIV-replication | 0.23 μg/mL | [95] |
| Sources | Peptide name | Activity | Potency | Ref. |
|---|---|---|---|---|
| Oyster Crassostrea gigas | Peptide 1 Peptide 2 | Inhibit HIV-1 protease | IC50: 20 nM (1) 15 nM (2) | [111] |
| Marine sponge Callipelta | Callipeltin A | Inhibit cytopathic effects induced by HIV-1 | EC50: 0.01 μg/mL | [112] |
| Marine sponge Theonella mirabilis Theonella swinhoei | Papuamides A Papuamides B | Inhibit HIV-1 infection | EC50: 4 ng/mL | [113] |
| Marine sponge Sidonops microspinosa | Microspinosamide | Inhibit cytopathic effect of HIV-1 infection | EC50: 0.2 μg/mL | [114] |
| Marine sponge Neamphius huxleyi | Neamphamide A | Against HIV-1 infection | EC50: 28 nM | [115] |
| Marine sponge Siliquariaspongia mirabilis | Mirabamide A Mirabamide C Mirabamide D | Inhibit HIV-1 neutralization and fusion | IC50: 0.04 and 0.14 μM (A) 0.14 and 1.3 μM (C) 0.19 and 3.9 μM (D) | [116] |
| Marine sponge Siliquariaspongia mirabilis | Celebesides A Theopapuamide B | Block HIV-1 entry (A) Neutralize HIV-1 (B) | IC50: 1.9 μg/mL (A) 0.8 μg/mL (B) | [117] |
| Marine sponge Homophymia sp | Homophymine A | Against HIV-1 infection | IC50: 75 nM | [118] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vo, T.-S.; Kim, S.-K. Potential Anti-HIV Agents from Marine Resources: An Overview. Mar. Drugs 2010, 8, 2871-2892. https://doi.org/10.3390/md8122871
Vo T-S, Kim S-K. Potential Anti-HIV Agents from Marine Resources: An Overview. Marine Drugs. 2010; 8(12):2871-2892. https://doi.org/10.3390/md8122871
Chicago/Turabian StyleVo, Thanh-Sang, and Se-Kwon Kim. 2010. "Potential Anti-HIV Agents from Marine Resources: An Overview" Marine Drugs 8, no. 12: 2871-2892. https://doi.org/10.3390/md8122871
APA StyleVo, T.-S., & Kim, S.-K. (2010). Potential Anti-HIV Agents from Marine Resources: An Overview. Marine Drugs, 8(12), 2871-2892. https://doi.org/10.3390/md8122871





