Marine Bacterial Sialyltransferases
Abstract
:1. Introduction
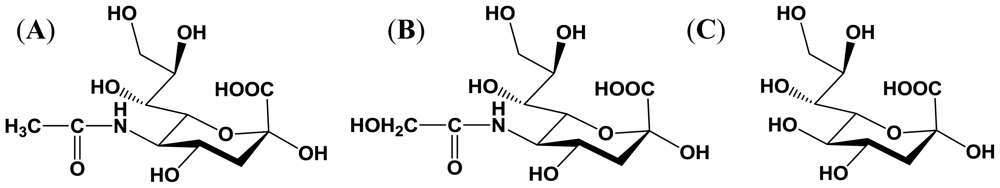
2. Sialic Acid
2.1. Structure and distribution
2.2. Importance of sialyloligosaccharides
3. Sialyltransferase
3.1. Classes and sources sialyltransferases
3.2. Classification of sialyltransferases produced by bacteria
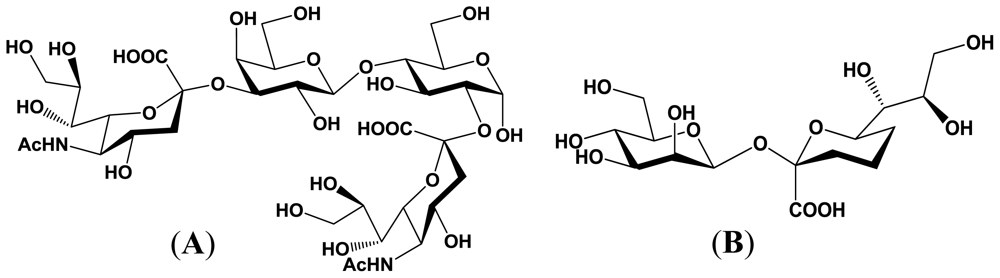
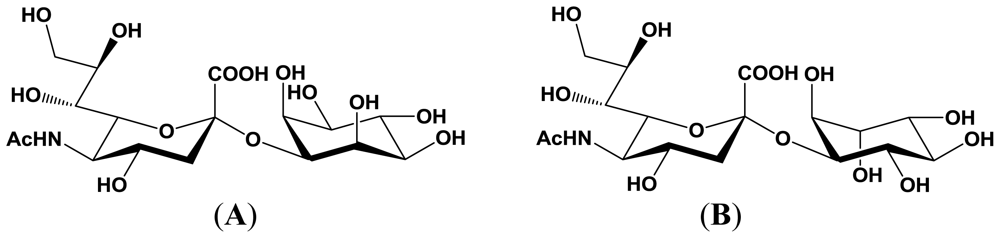
4. Enzymatic Properties of Marine Bacterial Sialyltransferases
4.1. An α2,6-sialyltransferase produced by P. damselae JT0160
4.2. An α2,6-sialyltransferase produced by Photobacterium sp. JT-ISH-224
4.3. An α2,3-sialyltransferase produced by P. phosphoreum JT-ISH-467
5. Application of Sialyltransferases
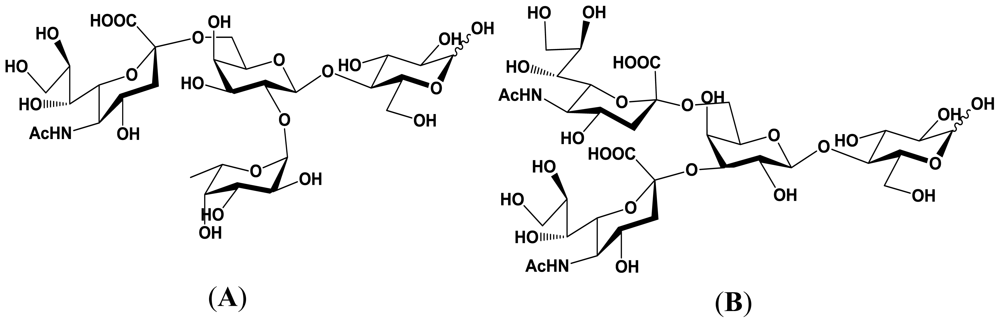
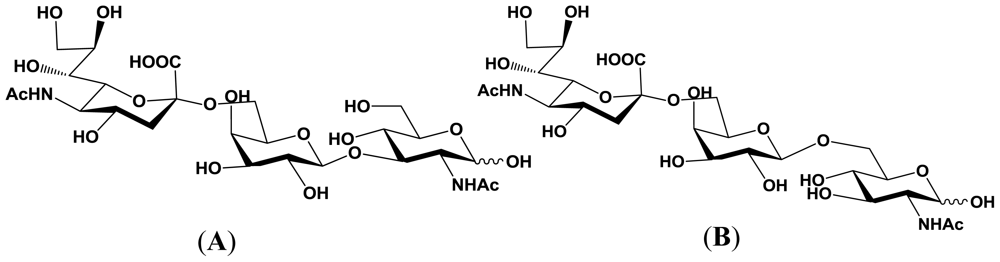
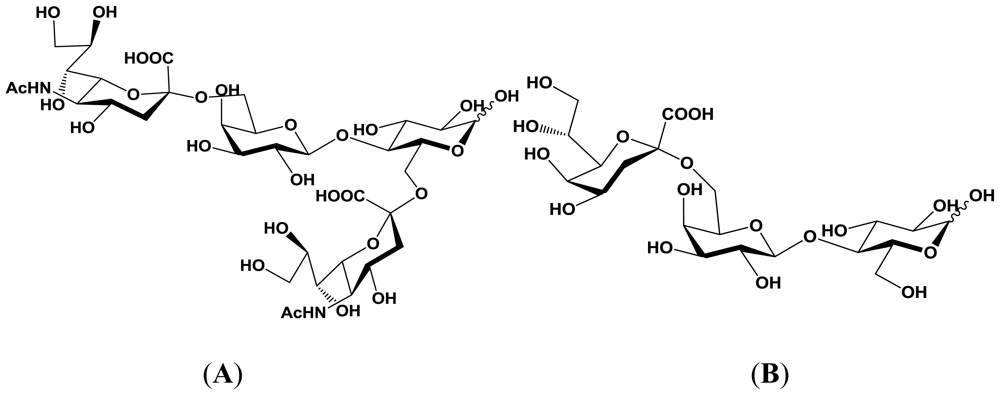
5.1. Production of sialyloligosaccharides
5.2. Application of sialyloligosaccharides
Acknowledgements
References
- Schauer, R. Sialic acid: fascinating sugars in higher animals and man. Zoology 2004, 107, 49–64. [Google Scholar]
- Angata, T; Varki, A. Chemical diversity in the sialic acids and related α-keto acids: an evolutionary perspective. Chem Rev 2002, 102, 439–469. [Google Scholar]
- Paulson, JC. Glycoproteins: What are the sugar chains for. Trends Biochem Sci 1989, 14, 272–276. [Google Scholar]
- Hakomori, S. Bifunctional role of glycosphingolipids. J Biol Chem 1990, 265, 18713–18716. [Google Scholar]
- Lasky, LA. Selectins: Interpreters of cell-specific carbohydrate information during inflammation. Science 1992, 258, 964–969. [Google Scholar]
- Kannagi, R. Regulatory roles of carbohydrate ligands for selectins in homing of lymphocytes. Curr Opin Struct Biol 2002, 12, 599–608. [Google Scholar]
- Suzuki, Y. Sialobiology of influenza: molecular mechanism of host range variation of influenza virus. Biol Pharm Bull 2005, 28, 399–408. [Google Scholar]
- Taniguchi, N; Honke, K; Fukuda, M. Handbook of Glycosyltransferases and Related Genes, 1st ed; Springer-Verlag: Tokyo, Japan, 2002; pp. 267–356. [Google Scholar]
- Schmidt, RR. New methods for the synthesis of glycosides and oligosaccharides—Are there alternatives to the Koenigs-Knorr method? New Synthetic Methods. Angew Chem Int Ed Engl 1986, 25, 212–235. [Google Scholar]
- Kanie, O; Hindsgaul, O. Synthesis of oligosaccharides, glycolipids and glycopeptides. Curr Opin Struct Biol 1992, 2, 674–681. [Google Scholar]
- Wang, Z; Zhang, X-F; Ito, Y; Nakahara, Y; Ogawa, T. A new strategy for stereoselective synthesis of sialic acid-containing glycopeptide fragment. Bioorg Med Chem 1996, 4, 1901–1908. [Google Scholar]
- Izumi, M; Wong, C-H. Microbial sialyltransferases for carbohydrate synthesis. Trends Glycosci Glycotechnol 2001, 13, 345–360. [Google Scholar]
- Yamamoto, T; Takakura, Y; Tsukamoto, H. Bacterial sialyltransferase. Trends Glycosci Glycotechnol 2006, 18, 253–265. [Google Scholar]
- Connor, RJ; Kawaoka, Y; Webster, RG; Paulson, JC. Receptor specificity in human, avian, and equine H2 and H3 influenza virus isolates. Virology 1994, 205, 17–23. [Google Scholar]
- Rogers, GN; Paulson, JC. Receptor determinations of human and animal influenza virus isolates: differences in receptor specificity of the H3 hemagglutinin based on species of origin. Virology 1983, 127, 361–373. [Google Scholar]
- Suzuki, Y; Ito, T; Suzuki, T; Holland, RE, Jr; Chambers, TM; Kiso, M; Ishida, H; Kawaoka, Y. Sialic acid species as a determinant of the host range of influenza A virus. J Virol 2000, 74, 11825–11831. [Google Scholar]
- Yamada, S; Suzuki, Y; Suzuki, T; Li, MQ; Nidom, CA; Sakai-Tagawa, Y; Muramoto, Y; Ito, M; Kiso, M; Hiromoto, T; et al. Hemagglutinin mutations responsible for the binding of H5N1 influenza A viruses to human-type receptors. Nature 2006, 444, 378–382. [Google Scholar]
- Suzuki, Y. Gangliosides as influenza virus receptors. Variation of influenza viruses and their recognition of the receptor sialo-sugar chains. Prog Lipid Res 1994, 33, 429–457. [Google Scholar]
- Koizumi, S. Large-scale production of oligosaccharides using bacterial functions. Trends Glycosci Glycotechnol 2003, 15, 65–74. [Google Scholar]
- Sujino, K; Jackson, RJ; Chan, NWC; Tsuji, S; Palcic, MM. A novel viral α2,3-sialyltransferase (v-ST3Gal I): Transfer of sialic acid to fucosylated acceptors. Glycobiology 2000, 10, 313–320. [Google Scholar]
- Coutinho, PM; Deleury, E; Davies, GJ; Henrissat, B. An evolving hierarchical family classification for glycosyltransferases. J Mol Biol 2003, 328, 307–317. [Google Scholar]
- Datta, AK; Sinha, A; Paulson, JC. Mutation of the sialyltransferase S-sialylmotif alters the kinetics of the donor and acceptor substrates. J Biol Chem 1998, 273, 9608–9618. [Google Scholar]
- Sasaki, K. Molecular cloning and characterization of sialyltransferase. Trends Glycosci Glycotechnol 1996, 8, 195–215. [Google Scholar]
- Jeanneau, C; Chazalet, V; Augé, C; Soumpasis, DM; Harduin-Lepers, A; Delannoy, P; Imberty, A; Breton, C. Structure-function analysis of the human sialyltransferase ST3Gal I: role of N-glycosylation and a novel conserved sialylmotif. J Biol Chem 2004, 279, 13461–13468. [Google Scholar]
- Rao, FV; Rich, JR; Rakić, B; Buddai, S; Schwartz, MF; Johnson, K; Bowe, C; Wakarchuk, WW; Defrees, S; Withers, SG; Strynadka, NC. Structural insight into mammalian sialyltransferases. Nat Struct Mol Biol 2009, 16, 1186–1188. [Google Scholar]
- Datta, AK; Paulson, JC. The sialyltransferase “Sialylmotif” participates in binding the donor substrate CMP-NeuAc. J Biol Chem 1995, 270, 1497–1500. [Google Scholar]
- Freiberger, F; Claus, H; Gunzel, A; Oltmann-Norden, I; Vionnet, J; Muhlenhoff, M; Vogel, U; Vann, WF; Gerardy-Schahn, R; Stummeyer, K. Biochemical characterization of a Neisseria meningitidis polysialyltransferase reveals novel functional motifs in bacterial sialyltransferases. Mol Microbiol 2007, 65, 1258–1275. [Google Scholar]
- Yamamoto, T; Ichikawa, M; Takakura, Y. Conserved amino acid sequences in the bacterial sialyltransferases belonging to Glycosyltransferase family 80. Biochem Biophys Res Commun 2008, 365, 340–343. [Google Scholar]
- Gilbert, M; Watson, DC; Cunningham, AM; Jennings, MP; Young, NM; Wakarchuk, WW. Cloning of the lipooligosaccharide α-2,3-sialyltransferase from the bacterial pathogens Neisseria meningitidis and Neisseria gonorrhoeae. J Biol Chem 1996, 271, 28271–28276. [Google Scholar]
- Edwards, U; Muller, A; Hammerschmidt, S; Gerardy-Schahn, R; Frosch, M. Molecular analysis of the biosynthesis pathway of the (α-2,8) polysialic acid capsule by Neisseria meningitidis serogroup B. Mol Microbiol 1994, 14, 141–149. [Google Scholar]
- Gilbert, M; Brisson, JR; Karwaski, MF; Michniewicz, J; Cunningham, AM; Wu, Y; Young, NM; Wakarchuk, WW. Biosynthesis of ganglioside mimics in Campylobacter jejuni OH 4384: Identification of the glycosyltransferase genes, enzymatic synthesis of model compounds, and characterization of nanomole amounts by 600-MHz 1H and 13C NMR analysis. J Biol Chem 2000, 275, 3896–3906. [Google Scholar]
- Shen, GJ; Datta, AK; Izumi, M; Koeller, KM; Wong, C-H. Expression of α2,8/2,9-Polysialyltransferase from Escherichia coli K92. characterization of the enzyme and its reaction products. J Biol Chem 1999, 274, 35139–35146. [Google Scholar]
- Yamamoto, T; Nakashizuka, M; Terada, I. Cloning and expression of a marine bacterial β-galactoside α2,6-sialyltransferase gene from Photobacterium damsela JT0160. J Biochem 1998, 123, 94–100. [Google Scholar]
- Tsukamoto, H; Takakura, Y; Yamamoto, T. Purification, cloning and expression of an α-/β-galactoside α2,3-sialyltransferase from a luminous marine bacterium Photobacterium phosphoreum. J Biol Chem 2007, 282, 29794–29802. [Google Scholar]
- Yamamoto, T; Hamada, Y; Ichikawa, M; Kajiwara, H; Mine, T; Tsukamoto, H; Takakura, Y. A β-galactoside α2,6-sialyltransferase produced by a marine bacterium, Photobacterium leiognathi JTSHIZ-145, is active at pH 8. Glycobiology 2007, 17, 1167–1174. [Google Scholar]
- Mine, T; Katayama, S; Kajiwara, H; Tsunashima, M; Tsukamoto, T; Takakura, Y; Yamamoto, T. An α2,6-sialyltransferase cloned from Photobacterium leiognathi strain JT-SHIZ-119 shows both sialyltransferase and neuraminidase activity. Glycobiology 2010, 20, 158–165. [Google Scholar]
- Tsukamoto, H; Takakura, Y; Mine, T; Yamamoto, T. Photobacterium sp. JT-ISH-224 Produces Two Sialyltransferases, α-/β-Galactoside α2,3-Sialyltransferase and β-Galactoside α2,6-Sialyltransferase. J Biochem 2008, 143, 187–197. [Google Scholar]
- Takakura, Y; Tsukamoto, H; Yamamoto, T. Molecular cloning, expression and properties of an α/β-galactoside α2,3-sialyltransferase from Vibrio sp. JT-FAJ-16. J Biochem 2007, 142, 403–412. [Google Scholar]
- Yu, H; Chokhawala, H; Karpel, R; Yu, H; Wu, B; Zhang, J; Zhang, Y; Jia, Q; Chen, X. A multifunctional Pasteurella multocida sialyltransferase: A powerful tool for the synthesis of sialoside libraries. J Am Chem Soc 2005, 127, 17618–17619. [Google Scholar]
- Hood, DW; Cox, AD; Gilbert, M; Makepeace, K; Walsh, S; Deadman, ME; Cody, A; Martin, A; Mansson, M; Schweda, EK; Brisson, JR; Richards, JC; Moxon, ER; Wakarchuk, WW. Identification of a lipopolysaccharide α-2,3-sialyltransferase from Haemophilus influenzae. Mol Microbiol 2001, 39, 341–350. [Google Scholar]
- Watanabe, M; Miyake, K; Yamamoto, S; Kataoka, Y; Koizumi, S; Endo, T; Ozaki, A; Iijima, S. Identification of sialyltransferases of Streptococcus agalactiae. J Biosci Bioeng 2002, 93, 610–613. [Google Scholar]
- Kajiwara, H; Toda, M; Mine, T; Nakada, H; Wariishi, H; Yamamoto, T. Visualization of sialic acid produced on bacterial cell surfaces by lectin staining. Microbes Environ 2010, 25, 152–155. [Google Scholar]
- Austin, B. Vibrios as causal agents of zoonoses. Vet Microbiol 2010, 140, 310–317. [Google Scholar]
- Jung, TS; Thompson, KD; Adams, A. A comparison of sialic acid between different isolates of Photobacterium damselae subsp. piscicida. Fish Pathol 2001, 36, 217–224. [Google Scholar]
- Love, M; Teebken-Fisher, D; Mecca, M. The marine bacterium Vibrio damselae sp. causes skin ulcers on the damselfish Chromis punctipinnis: Association with human infections. Science 1981, 214, 1139–1140. [Google Scholar]
- Yamamoto, T; Nagae, H; Kajihara, Y; Terada, I. Mass production of bacterial α2,6-sialyltransferase and enzymatic syntheses of sialyloligosaccharides. Biosci Biotechnol Biochem 1998, 62, 210–214. [Google Scholar]
- Kushi, Y; Kamimiya, H; Hiratsuka, H; Nozaki, H; Fukui, H; Yanagida, M; Hashimoto, M; Nakamura, K; Watarai, S; Kasama, T; Kajiwara, H; Yamamoto, T. Sialyltransferases of marine bacteria efficiently utilize glycosphingolipid substrates. Glycobiology 2010, 20, 187–198. [Google Scholar]
- Yamamoto, T; Nakashizuka, M; Kodama, H; Kajihara, Y; Terada, I. Purification and characterization of a marine bacterial β-galactoside α2,6-sialyltransferase from Photobacterium damsela JT0160. J Biochem 1996, 120, 104–110. [Google Scholar]
- Kajihara, Y; Yamamoto, T; Nagae, H; Nakashizuka, M; Sakakibara, T; Terada, I. A Novel α2,6-Sialyltransferase: Transfer of Sialic Acid to Fucosyl and Sialyl Trisaccharides. J Org Chem 1996, 61, 8632–8635. [Google Scholar]
- Mine, T; Miyazaki, T; Kajiwara, H; Naito, K; Ajisaka, K; Yamamoto, T. Enzymatic synthesis of unique sialyloligosaccharides using marine bacterial α-(2→3)- and α-(2→6)-sialyltransferases. Carbohydr Res 2010, 345, 1417–1421. [Google Scholar]
- Kajihara, Y; Akai, S; Nakagawa, T; Sato, R; Ebata, T; Kodama, H; Sato, K. Enzymatic synthesis of Kdn oligosaccharides by a bacterial α-(2→6)-sialyltransferase. Carbohydr Res 1999, 315, 137–141. [Google Scholar]
- Yu, H; Huang, S; Chokhawala, H; Sun, M; Zheng, H; Chen, X. Highly efficient chemoenzymatic synthesis of naturally occurring and non-natural alpha-2,6-linked sialosides: A P. damsela alpha-2,6-sialyltransferase with extremely flexible donor-substrate specificity. Angew Chem Int Ed Engl 2006, 45, 3938–3944. [Google Scholar]
- Drouillard, S; Mine, T; Kajiwara, H; Yamamoto, T; Samain, E. Efficient synthesis of 6′-sialyllactose, 6′,6-disialyllactose and 6′-KDO-lactose by metabolically engineered Escherihiae coli expressing a multifunctional sialyltransferase from the Photobacterium sp. JT-ISH-224. Carbohydr Res 2010, 345, 1394–1399. [Google Scholar]
- Mine, T; Kajiwara, H; Murase, T; Kajihara, Y; Yamamoto, T. An α2,3-sialyltransferase cloned from Photobacterium sp. JT-ISH-224 transfers N-acetylneuraminic acid to both O-2 and O-3′ hydroxyl groups of lactose. J Carbohydr Chem 2010, 29, 51–60. [Google Scholar]
- Mine, T; Miyazaki, T; Kajiwara, H; Tateda, N; Ajisaka, K; Yamamoto, T. A recombinant α-(2→3)-sialyltransferase with an extremely broad acceptor substrate specificity from Photobacterium sp. JT-ISH-224 can transfer N-acetylneuraminic acid to inositols. Carbohydr Res 2010, 345, 2485–2490. [Google Scholar]
- Preim, B; Gilbert, M; Wakarchuk, WW; Heyraud, A; Samain, E. A new fermentation process allows large scale production of human milk oligosaccharides by metabolically engineered bacteria. Glycobiology 2002, 12, 235–240. [Google Scholar]
- Antoine, T; Priem, B; Heyraud, A; Greffe, L; Gilbert, M; Wakarchuk, WW; Lam, JS; Samain, E. Large-scale in vivo synthesis of the carbohydrate moieties of gangliosides GM1 and GM2 by metabolically engineered Escherichia coli. Chembiochem 2003, 4, 406–412. [Google Scholar]
- Drouillard, S; Driguez, H; Samain, E. Large-scale synthesis of H-antigen oligosaccharides by expressing Helicobacter pylori alpha1,2-fucosyltransferase in metabolically engineered Escherichia coli cells. Angew Chem Int Ed Engl 2006, 45, 1778–1780. [Google Scholar]
- Fierfort, N; Samain, E. Genetic engineering of Escherichia coli for the economical production of sialylated oligosaccharides. J Biotechnol 2008, 134, 261–265. [Google Scholar]
- Tsuchida, A; Kobayashi, K; Matsubara, N; Muramatsu, T; Suzuki, T; Suzuki, Y. Simple synthesis of sialyllactose-carrying polystyrene and its binding with influenza virus. Glycoconj J 1998, 15, 1047–1054. [Google Scholar]
- Sigal, GB; Mammen, M; Dahmann, G; Whitesides, GM. Polyacrylamides bearing pendant α-sialoside groups strongly inhibit agglutination of erythrocytes by influenza virus: The strong inhibition reflects enhances binding through cooperative polyvalent interactions. J Am Chem Soc 1996, 118, 3789–3800. [Google Scholar]
- Makimura, Y; Watanabe, S; Suzuki, T; Suzuki, Y; Ishida, H; Kiso, M; Katayama, T; Kumagai, H; Yamamoto, K. Chemoenzymatic synthesis and application of a sialoglycopolymer with a chitosan backbone as a potent inhibitor of human influenza virus hemagglutination. Carbohydr Res 2006, 341, 1803–1808. [Google Scholar]
- Choi, S-K; Mammen, M; Whitesides, GM. Generation and in situ evaluation of libraries of poly(acrylic acid) presenting sialosides as side chaina as polyvalent inhibitors of influenza-mediated hemagglutination. J Am Chem Soc 1997, 119, 4103–4111. [Google Scholar]
- Umemura, M; Itoh, M; Makimura, Y; Yamazaki, K; Umekawa, M; Masui, A; Matahira, Y; Shibata, M; Ashida, H; Yamamoto, K. Design of a sialylglycopolymer with a chitosan backbone having efficient inhibitory activity against influenza virus infection. J Med Chem 2008, 51, 4496–4503. [Google Scholar]
- Totani, K; Kubota, T; Kuroda, T; Murata, T; Hidari, KI-PJ; Suzuki, T; Suzuki, Y; Kobayashi, K; Ashida, H; Yamamoto, K; Usui, T. Chemoenzymatic synthesis and application of glycopolymers containing multivalent sialyloligosaccharides with a poly(L-glutamic acid) backbone for inhibition of infection by influenza viruses. Glycobiology 2003, 13, 315–326. [Google Scholar]
- Furuike, T; Aiba, S; Suzuki, T; Takahashi, T; Suzuki, Y; Yamada, K; Nishimura, S-I. Synthesis and anti-influenza virus activity of novel glycopolymers having triantennary oligosaccharide branches. J Chem Soc Perkin Trans I 2000, 1, 3000–3005. [Google Scholar]
- Mammen, M; Dahmann, G; Whitesides, GM. Effective inhibitors of hemagglutination by influenza virus synthesized from polymers having active ester groups. Insight into mechanism of inhibition. J Med Chem 1995, 38, 4179–4190. [Google Scholar]
- Reuter, JD; Myc, A; Hayes, MM; Gan, Z; Roy, R; Qin, D; Yin, R; Piehler, LT; Esfand, R; Tomalia, DA; Baker, J, Jr. Inhibition of viral adhesion and infection by sialic-conjugated dendritic polymers. Bioconjug Chem 1999, 10, 271–278. [Google Scholar]
- Ikeuchi, T; Asaka, T; Saito, M; Tanaka, H; Higuchi, S; Tanaka, K; Saida, K; Uyama, E; Mizusawa, H; Fukuhara, N; Nonaka, I; Takemori, M; Tsuji, S. Gene locus autosomal recessive distal myopathy with rimmed vacuoles maps to chromosome 9. Ann Neurol 1997, 41, 432–437. [Google Scholar]
- Eisenberg, I; Avidan, N; Potikha, T; Hochner, H; Chen, M; Olender, T; Barash, M; Shemesh, M; Sadeh, M; Grabov-Nardini, G; et al. The UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase gene is mutated in recessive hereditary inclusion body myopathy. Nat Genet 2001, 29, 83–87. [Google Scholar]
- Nishino, I; Noguchi, S; Murayama, K; Driss, A; Sugie, K; Oya, Y; Nagata, T; Chida, K; Takahashi, T; Takusa, Y; et al. Distal myopathy with rimmed vacuoles is allelic to hereditary inclusion body pyopathy. Neurology 2002, 59, 1689–1693. [Google Scholar]
- Malicdan, MCV; Noguchi, S; Hayashi, YK; Nonaka, I; Nishino, I. Prophylactic treatment with sialic acid metabolites precludes the development of the myopathic phenotype in the DMRV-hIBM mouse model. Nat Med 2009, 15, 690–695. [Google Scholar]
| Enzyme origin | Enzyme activities | |
|---|---|---|
| Sialyltransferase activity | Neuraminidase activity | |
| Photobacterium phosphoreum JT-ISH-467 | α2,3-sialyltransferase | + |
| Photobacterium sp. JT-ISH-224 | + | |
| Vibrio sp. JT-FAJ-16 | − | |
| Photobacterium damselae JT0160 | α2,6-sialyltransferase | − |
| Photobacterium leiognathi JT-SHIZ-119 | + | |
| Photobacterium leiognathi JT-SHIZ-145 | − | |
| Photobacterium sp. JT-ISH-224 | − | |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yamamoto, T. Marine Bacterial Sialyltransferases. Mar. Drugs 2010, 8, 2781-2794. https://doi.org/10.3390/md8112781
Yamamoto T. Marine Bacterial Sialyltransferases. Marine Drugs. 2010; 8(11):2781-2794. https://doi.org/10.3390/md8112781
Chicago/Turabian StyleYamamoto, Takeshi. 2010. "Marine Bacterial Sialyltransferases" Marine Drugs 8, no. 11: 2781-2794. https://doi.org/10.3390/md8112781
APA StyleYamamoto, T. (2010). Marine Bacterial Sialyltransferases. Marine Drugs, 8(11), 2781-2794. https://doi.org/10.3390/md8112781




