Anti-Inflammatory and Neuroprotective Effects of Undaria pinnatifida Fucoidan
Abstract
1. Introduction
2. Materials and Methods
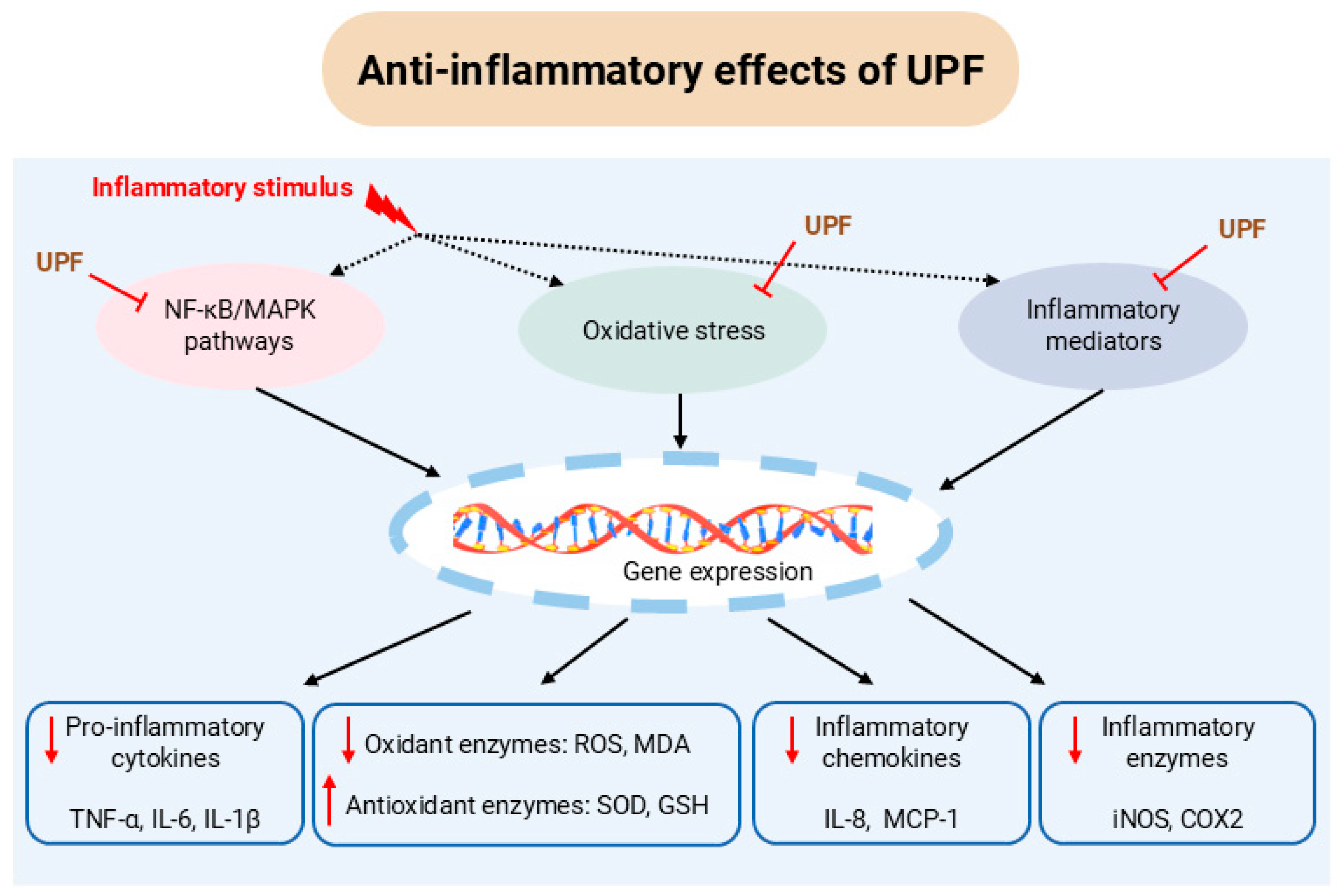
3. Anti-Inflammatory Activity of UPF
3.1. In Vitro Studies
3.2. In Vivo Studies
3.3. Clinical Trials
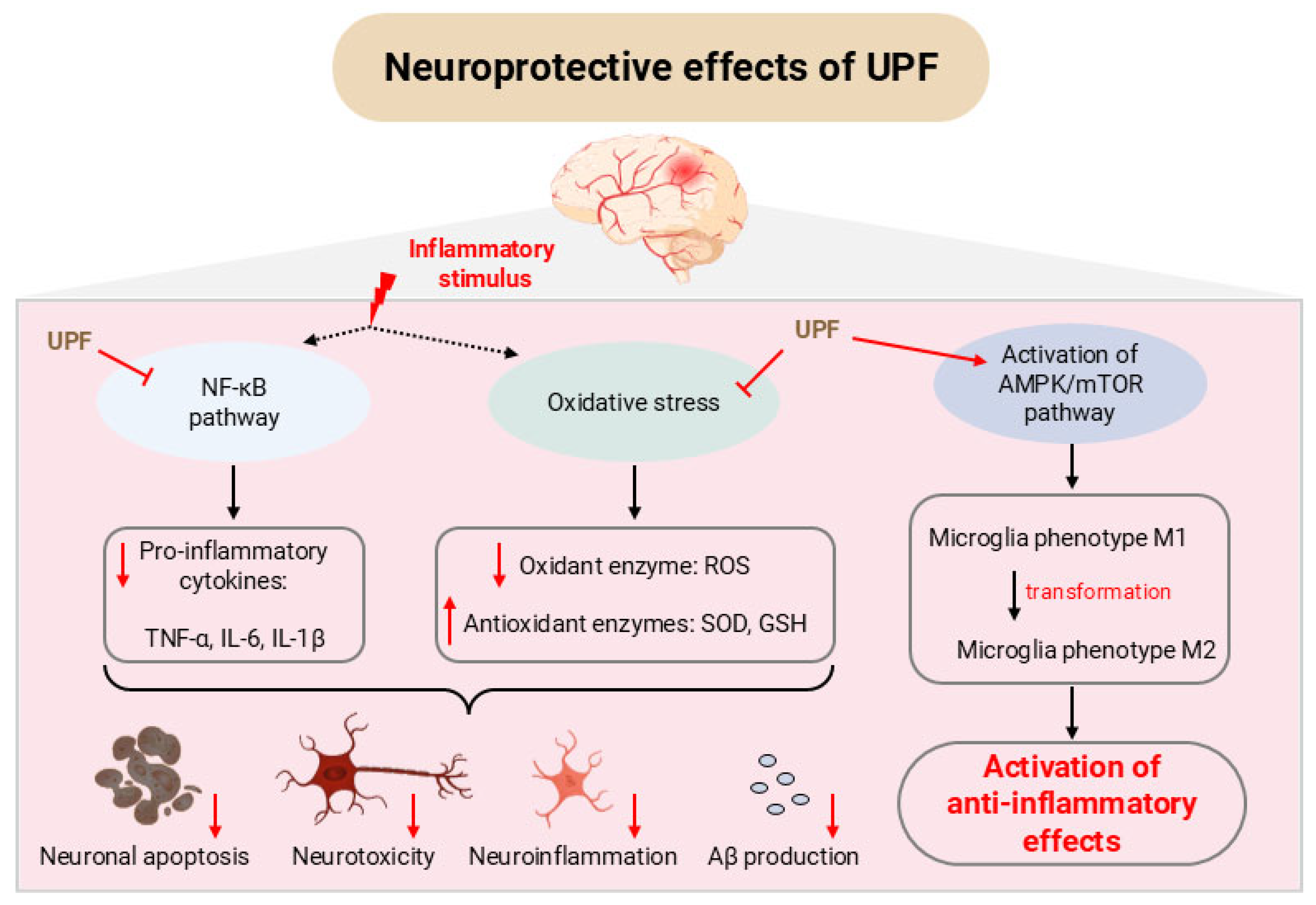
4. Neuroprotective Effect of UPF
4.1. In Vitro Studies
4.2. In Vivo Studies
5. Discussion
5.1. Toxicity Study of UPF
5.2. Pharmacokinetics of UPF
5.3. Structure–Activity Relationships of UPF
5.4. Limitation
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sanjeewa, K.K.A.; Herath, K.H.I.N.M.; Yang, H.-W.; Choi, C.S.; Jeon, Y.-J. Anti-Inflammatory Mechanisms of Fucoidans to Treat Inflammatory Diseases: A Review. Mar. Drugs 2021, 19, 678. [Google Scholar] [CrossRef] [PubMed]
- Apostolova, E.; Lukova, P.; Baldzhieva, A.; Katsarov, P.; Nikolova, M.; Iliev, I.; Peychev, L.; Trica, B.; Oancea, F.; Delattre, C.; et al. Immunomodulatory and Anti-Inflammatory Effects of Fucoidan: A Review. Polymers 2020, 12, 2338. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wang, H.; Liu, N.; Zhao, K.; Sheng, Y.; Pang, H.; Shao, K.; Zhang, M.; Li, S.; He, N. Algal polysaccharides and derivatives as potential therapeutics for obesity and related metabolic diseases. Food Funct. 2022, 13, 11387–11409. [Google Scholar] [CrossRef]
- Yao, Y.; Yim, E.K.F. Fucoidan for cardiovascular application and the factors mediating its activities. Carbohydr. Polym. 2021, 270, 118347. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.S.; Oliveira, C.; Reis, R.L.; Martins, A.; Silva, T.H. Marine-Inspired Drugs and Biomaterials in the Perspective of Pancreatic Cancer Therapies. Mar. Drugs 2022, 20, 689. [Google Scholar] [CrossRef]
- Miyata, Y.; Matsuo, T.; Ohba, K.; Mitsunari, K.; Mukae, Y.; Otsubo, A.; Harada, J.; Matsuda, T.; Kondo, T.; Sakai, H. Present Status, Limitations and Future Directions of Treatment Strategies Using Fucoidan-Based Therapies in Bladder Cancer. Cancers 2020, 12, 3776. [Google Scholar] [CrossRef]
- Jin, J.-O.; Yadav, D.; Madhwani, K.; Puranik, N.; Chavda, V.; Song, M. Seaweeds in the Oncology Arena: Anti-Cancer Potential of Fucoidan as a Drug—A Review. Molecules 2022, 27, 6032. [Google Scholar] [CrossRef]
- Ke, S.; Zhang, B.; Yu, Y.; Wang, S.; Jin, W.; Wu, J.; Chen, J.; Zhang, H.; Wei, B.; Wang, H. Structural characterization of sulfated galactofucan from Undaria pinnatifida and its effect on type 2 diabetic mice. J. Oceanol. Limnol. 2022, 41, 300–313. [Google Scholar] [CrossRef]
- Koh, H.S.A.; Lu, J.; Zhou, W. Structure characterization and antioxidant activity of fucoidan isolated from Undaria pinnatifida grown in New Zealand. Carbohydr. Polym. 2019, 212, 178–185. [Google Scholar] [CrossRef]
- Ale, M.T.; Meyer, A.S. Fucoidans from brown seaweeds: An update on structures, extraction techniques and use of enzymes as tools for structural elucidation. RSC Adv. 2013, 3, 8131–8141. [Google Scholar] [CrossRef]
- Gasilova, E.R.; Lapina, I.M.; Kulminskaya, A.A.; Skorik, Y.A. Branched architecture of fucoidan characterized by dynamic and static light scattering. Colloid Polym. Sci. 2020, 298, 1349–1359. [Google Scholar] [CrossRef]
- Yu, J.; Li, Q.; Wu, J.; Yang, X.; Yang, S.; Zhu, W.; Liu, Y.; Tang, W.; Nie, S.; Hassouna, A.; et al. Fucoidan Extracted From Sporophyll of Undaria pinnatifida Grown in Weihai, China – Chemical Composition and Comparison of Antioxidant Activity of Different Molecular Weight Fractions. Front. Nutr. 2021, 8, 636930. [Google Scholar] [CrossRef]
- Cheong, K.-L.; Chen, W.; Wang, M.; Zhong, S.; Veeraperumal, S. Therapeutic Prospects of Undaria pinnatifida Polysaccharides: Extraction, Purification, and Functional Activity. Mar. Drugs 2025, 23, 163. [Google Scholar] [CrossRef]
- Mak, W.; Hamid, N.; Liu, T.; Lu, J.; White, W.L. Fucoidan from New Zealand Undaria pinnatifida: Monthly variations and determination of antioxidant activities. Carbohydr. Polym. 2013, 95, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Oliveira, C.; Li, Q.; Ferreira, A.S.; Nunes, C.; Coimbra, M.A.; Reis, R.L.; Martins, A.; Wang, C.; Silva, T.H.; et al. Fucoidan from Fucus vesiculosus Inhibits Inflammatory Response, Both In Vitro and In Vivo. Mar. Drugs 2023, 21, 302. [Google Scholar] [CrossRef]
- Kopplin, G.; Rokstad, A.M.; Mélida, H.; Bulone, V.; Skjåk-Bræk, G.; Aachmann, F.L. Structural Characterization of Fucoidan from Laminaria hyperborea: Assessment of Coagulation and Inflammatory Properties and Their Structure–Function Relationship. ACS Appl. Bio Mater. 2018, 1, 1880–1892. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wu, S.; Ai, C.; Wen, C.; Liu, Z.; Wang, L.; Jiang, L.; Shen, P.; Zhang, G.; Song, S. Galactofucan from Laminaria japonica is not degraded by the human digestive system but inhibits pancreatic lipase and modifies the intestinal microbiota. Int. J. Biol. Macromol. 2021, 166, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, M.; Carvalho, L.G.; Vanegas, C.; Dimartino, S. A Comparative Review of Alternative Fucoidan Extraction Techniques from Seaweed. Mar. Drugs 2025, 23, 27. [Google Scholar] [CrossRef] [PubMed]
- Saetan, U.; Nontasak, P.; Palasin, K.; Saelim, H.; Wonglapsuwan, M.; Mayakun, J.; Pongparadon, S.; Chotigeat, W. Potential health benefits of fucoidan from the brown seaweeds Sargassum plagiophyllum and Sargassum polycystum. J. Appl. Phycol. 2021, 33, 3357–3364. [Google Scholar] [CrossRef]
- Zayed, A.; Cao, H.T.T.; Trang, V.T.D.; Ulber, R. Structural tailoring of fucoidan backbones for maximizing their benefits: Enzymatic, chemical, and physical strategies. J. Appl. Phycol. 2023, 35, 2445–2462. [Google Scholar] [CrossRef]
- Hu, S.; Chen, S.; Zhu, H.; Du, M.; Jiang, W.; Liu, Y.; Gao, X.; Su, L.; Xu, Y. Low Molecular Weight, 4-O-Sulfation, and Sulfation at Meta-Fucose Positively Promote the Activities of Sea Cucumber Fucoidans on Improving Insulin Resistance in HFD-Fed Mice. Mar. Drugs 2021, 20, 37. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Dwan, C.; Wimmer, B.C.; Ronci, M.; Wilson, R.; Johnson, L.; Caruso, V. Neuroprotective and Anti-Inflammatory Activity of Undaria pinnatifida Fucoidan In Vivo—A Proteomic Investigation. Mar. Drugs 2025, 23, 189. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, B.; Lessa, R.C.; Baroutian, S.; Zhou, Q.; Chen, X.; Lu, J. Antioxidant and immunomodulatory activity of Perna canaliculus oil extract in combination with low molecular weight fucoidan extracted from Undaria pinnatifida. Futur. Foods 2025, 11, 100652. [Google Scholar] [CrossRef]
- Li, Q.; Wang, X.; Wan, Y.; Hu, X.; Liu, J.; Wang, J. In vivo immunomodulatory activity of fucoidan from brown alga Undaria pinnatifida in sarcoma 180-bearing mice. J. Funct. Foods 2023, 103, 105486. [Google Scholar] [CrossRef]
- Shi, F.-S.; Xie, Y.-H.; Yang, Y.-L.; Xu, L.-D.; Li, J.-J.; Wang, X.; Zhu, L.-Y.; Wang, W.-W.; Shen, P.-L.; Huang, Y.-W.; et al. Fucoidan from Ascophyllum nodosum and Undaria pinnatifida attenuate SARS-CoV-2 infection in vitro and in vivo by suppressing ACE2 and alleviating inflammation. Carbohydr. Polym. 2024, 332, 121884. [Google Scholar] [CrossRef]
- Giuliani, M.; Uboldi, C.; Dellatorre, F.G.; Latour, E.; Ponce, N.M.A.; Stortz, C.A.; Lassalle, V.L.; Ayala-Peña, V.B. Undaria pinnatifida fucoidan extract inhibits activation of the NF-κB signaling pathway by herpes simplex virus type 1 and prevents amyloid-β peptide synthesis in retinal pigment epithelium cells. Arch. Virol. 2025, 170, 27. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Eapen, M.S.; Ishaq, M.; Park, A.Y.; Karpiniec, S.S.; Stringer, D.N.; Sohal, S.S.; Fitton, J.H.; Guven, N.; Caruso, V.; et al. Anti-Inflammatory Activity of Fucoidan Extracts In Vitro. Mar. Drugs 2021, 19, 702. [Google Scholar] [CrossRef]
- Kim, K.J.; Yoon, K.Y.; Lee, B.Y. Low molecular weight fucoidan from the sporophyll of Undaria pinnatifida suppresses inflammation by promoting the inhibition of mitogen-activated protein kinases and oxidative stress in RAW264.7 cells. Fitoterapia 2012, 83, 1628–1635. [Google Scholar] [CrossRef]
- Flórez-Fernández, N.; Vaamonde-García, C.; Torres, M.D.; Buján, M.; Muíños, A.; Muiños, A.; Lamas-Vázquez, M.J.; Meijide-Faílde, R.; Blanco, F.J.; Domínguez, H. Relevance of the Extraction Stage on the Anti-Inflammatory Action of Fucoidans. Pharmaceutics 2023, 15, 808. [Google Scholar] [CrossRef]
- Vaamonde-García, C.; Flórez-Fernández, N.; Torres, M.D.; Lamas-Vázquez, M.J.; Blanco, F.J.; Domínguez, H.; Meijide-Faílde, R. Study of fucoidans as natural biomolecules for therapeutical applications in osteoarthritis. Carbohydr. Polym. 2021, 258, 117692. [Google Scholar] [CrossRef]
- Lim, J.M.; Yoo, H.J.; Lee, K.W. High Molecular Weight Fucoidan Restores Intestinal Integrity by Regulating Inflammation and Tight Junction Loss Induced by Methylglyoxal-Derived Hydroimidazolone-1. Mar. Drugs 2022, 20, 580. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Chang, M. Roles of PRR-Mediated Signaling Pathways in the Regulation of Oxidative Stress and Inflammatory Diseases. Int. J. Mol. Sci. 2021, 22, 7688. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Jia, J.; Zhang, C.; Zhang, P.; Song, S.; Ai, C. Undaria pinnatifida fucoidan ameliorates dietary fiber deficiency-induced inflammation and lipid abnormality by modulating mucosal microbiota and protecting intestinal barrier integrity. Int. J. Biol. Macromol. 2023, 247, 125724. [Google Scholar] [CrossRef]
- Phull, A.-R.; Majid, M.; Haq, I.-U.; Khan, M.R.; Kim, S.J. In vitro and in vivo evaluation of anti-arthritic, antioxidant efficacy of fucoidan from Undaria pinnatifida (Harvey) Suringar. Int. J. Biol. Macromol. 2017, 97, 468–480. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Kim, J.-H.; Kim, S.-M.; Kim, J.-Y.; Kim, J.-H.; Eom, S.-J.; Kang, M.-C.; Song, K.-M. The Antioxidant Activity of Undaria pinnatifida Sporophyll Extract Obtained Using Ultrasonication: A Focus on Crude Polysaccharide Extraction Using Ethanol Precipitation. Antioxidants 2023, 12, 1904. [Google Scholar] [CrossRef]
- Zhao, Y.; Zheng, Y.; Wang, J.; Ma, S.; Yu, Y.; White, W.L.; Yang, S.; Yang, F.; Lu, J. Fucoidan Extracted from Undaria pinnatifida: Source for Nutraceuticals/Functional Foods. Mar. Drugs 2018, 16, 321. [Google Scholar] [CrossRef]
- Silva, M.M.C.L.; Lisboa, L.d.S.; Paiva, W.S.; Batista, L.A.N.C.; Luchiari, A.C.; Rocha, H.A.O.; Camara, R.B.G. Comparison of in vitro and in vivo antioxidant activities of commercial fucoidans from Macrocystis pyrifera, Undaria pinnatifida, and Fucus vesiculosus. Int. J. Biol. Macromol. 2022, 216, 757–767. [Google Scholar] [CrossRef]
- Kim, M.E.; Lee, J.S. Advances in the Regulation of Inflammatory Mediators in Nitric Oxide Synthase: Implications for Disease Modulation and Therapeutic Approaches. Int. J. Mol. Sci. 2025, 26, 1204. [Google Scholar] [CrossRef]
- Yang, H.; Xuefeng, Y.; Shandong, W.; Jianhua, X. COX-2 in liver fibrosis. Clin. Chim. Acta 2020, 506, 196–203. [Google Scholar] [CrossRef]
- Song, K.-M.; Ha, S.J.; Lee, J.-E.; Kim, S.-H.; Kim, Y.H.; Kim, Y.; Hong, S.P.; Jung, S.K.; Lee, N.H. High yield ultrasonication extraction method for Undaria pinnatifida sporophyll and its anti-inflammatory properties associated with AP-1 pathway suppression. LWT 2015, 64, 1315–1322. [Google Scholar] [CrossRef]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef]
- Chen, X.-W.; Long, J.; Zhang, Q.; Huang, L.-H.; Sun, X.-Y. Sulfated Undaria pinnatifida polysaccharides inhibit kidney stone formation through crystalline modulation and relieving cellular oxidative damage and inflammation. Biomater. Sci. 2025, 13, 1512–1528. [Google Scholar] [CrossRef]
- Kim, K.-J.; Lee, B.-Y. Fucoidan from the sporophyll of Undaria pinnatifida suppresses adipocyte differentiation by inhibition of inflammation-related cytokines in 3T3-L1 cells. Nutr. Res. 2012, 32, 439–447. [Google Scholar] [CrossRef]
- Vaamonde-García, C.; Capelo-Mera, E.; Flórez-Fernández, N.; Torres, M.D.; Rivas-Murias, B.; Mejide-Faílde, R.; Blanco, F.J.; Domínguez, H. In Vitro Study of the Therapeutic Potential of Brown Crude Fucoidans in Osteoarthritis Treatment. Int. J. Mol. Sci. 2022, 23, 14236. [Google Scholar] [CrossRef]
- Wimmer, B.C.; Dwan, C.; De Medts, J.; Duysburgh, C.; Rotsaert, C.; Marzorati, M. Undaria pinnatifida Fucoidan Enhances Gut Microbiome, Butyrate Production, and Exerts Anti-Inflammatory Effects in an In Vitro Short-Term SHIME® Coupled to a Caco-2/THP-1 Co-Culture Model. Mar. Drugs 2025, 23, 242. [Google Scholar] [CrossRef]
- Yang, J.-H. Topical Application of Fucoidan Improves Atopic Dermatitis Symptoms in NC/Nga Mice. Phytother. Res. 2012, 26, 1898–1903. [Google Scholar] [CrossRef]
- Li, X.; Li, J.; Li, Z.; Sang, Y.; Niu, Y.; Zhang, Q.; Ding, H.; Yin, S. Fucoidan from Undaria pinnatifida prevents vascular dysfunction through PI3K/Akt/eNOS-dependent mechanisms in the l-NAME-induced hypertensive rat model. Food Funct. 2016, 7, 2398–2408. [Google Scholar] [CrossRef]
- Herath, K.H.I.N.M.; Kim, H.J.; Kim, A.; Sook, C.E.; Lee, B.-Y.; Jee, Y. The Role of Fucoidans Isolated from the Sporophylls of Undaria pinnatifida against Particulate-Matter-Induced Allergic Airway Inflammation: Evidence of the Attenuation of Oxidative Stress and Inflammatory Responses. Molecules 2020, 25, 2869. [Google Scholar] [CrossRef] [PubMed]
- Shanmugasundaram, D.; Dwan, C.; Wimmer, B.C.; Srivastava, S. Fucoidan Ameliorates Testosterone-Induced Benign Prostatic Hyperplasia (BPH) in Rats. Res. Rep. Urol. 2024, 16, 283–297. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Zheng, W.; Sun, X.; Jiang, G.; Wu, S.; Xu, Y.; Song, S.; Ai, C. Sulfated polysaccharides from Undaria pinnatifida improved high fat diet-induced metabolic syndrome, gut microbiota dysbiosis and inflammation in BALB/c mice. Int. J. Biol. Macromol. 2021, 167, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.S.; Kim, I.D.; Kwon, R.H.; Ha, B.J. Undaria pinnatifida fucoidan extract protects against CCl4-induced oxidative stress. Biotechnol. Bioprocess Eng. 2008, 13, 168–173. [Google Scholar] [CrossRef]
- Oh, J.-Y.; Kim, E.-A.; Kang, S.I.; Yang, H.-W.; Ryu, B.; Wang, L.; Lee, J.-S.; Jeon, Y.-J. Protective Effects of Fucoidan Isolated from Celluclast-Assisted Extract of Undaria pinnatifida Sporophylls against AAPH-Induced Oxidative Stress In Vitro and In Vivo Zebrafish Model. Molecules 2020, 25, 2361. [Google Scholar] [CrossRef]
- Buttó, L.F.; Haller, D. Dysbiosis in intestinal inflammation: Cause or consequence. Int. J. Med. Microbiol. 2016, 306, 302–309. [Google Scholar] [CrossRef]
- Zeng, M.Y.; Inohara, N.; Nuñez, G. Mechanisms of inflammation-driven bacterial dysbiosis in the gut. Mucosal Immunol. 2017, 10, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Nagao-Kitamoto, H.; Kamada, N. Host-microbial Cross-talk in Inflammatory Bowel Disease. Immune Netw. 2017, 17, 1–12. [Google Scholar] [CrossRef]
- Liyanage, N.M.; Nagahawatta, D.P.; Jayawardena, T.U.; Jeon, Y.-J. The Role of Seaweed Polysaccharides in Gastrointestinal Health: Protective Effect against Inflammatory Bowel Disease. Life 2023, 13, 1026. [Google Scholar] [CrossRef]
- Liu, M.; Ma, L.; Chen, Q.; Zhang, P.; Chen, C.; Jia, L.; Li, H. Fucoidan alleviates dyslipidemia and modulates gut microbiota in high-fat diet-induced mice. J. Funct. Foods 2018, 48, 220–227. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Yuan, J.; Liu, Z.; Ye, C.; Qin, S. Undaria pinnatifida improves obesity-related outcomes in association with gut microbiota and metabolomics modulation in high-fat diet-fed mice. Appl. Microbiol. Biotechnol. 2020, 104, 10217–10231. [Google Scholar] [CrossRef]
- Zhang, P.; Jia, J.; Jiang, P.; Zheng, W.; Li, X.; Song, S.; Ai, C. Polysaccharides from edible brown seaweed Undaria pinnatifida are effective against high-fat diet-induced obesity in mice through the modulation of intestinal microecology. Food Funct. 2022, 13, 2581–2593. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Dwan, C.; Wimmer, B.C.; Wilson, R.; Johnson, L.; Caruso, V. Fucoidan from Undaria pinnatifida Enhances Exercise Performance and Increases the Abundance of Beneficial Gut Bacteria in Mice. Mar. Drugs 2024, 22, 485. [Google Scholar] [CrossRef]
- Zheng, W.; Jia, J.; Tang, S.; Song, S.; Ai, C. Undaria pinnatifida fucoidan contributes to anti-inflammation activity of Bacteroides in fiber-deficient mice via modulation of gut microbiota and protection of intestinal barrier integrity. Int. J. Biol. Macromol. 2023, 252, 126256. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef]
- Spychala, M.S.; Venna, V.R.; Jandzinski, M.; Doran, S.J.; Durgan, D.J.; Ganesh, B.P.; Ajami, N.J.; Putluri, N.; Graf, J.; Bryan, R.M.; et al. Age-related changes in the gut microbiota influence systemic inflammation and stroke outcome. Ann. Neurol. 2018, 84, 23–36. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Kwon, H.; Kim, Y.J. The Firmicutes/Bacteroidetes Ratio as a Risk Factor of Breast Cancer. J. Clin. Med. 2023, 12, 2216. [Google Scholar] [CrossRef]
- Yin, Y.; Yang, T.; Tian, Z.; Shi, C.; Yan, C.; Li, H.; Du, Y.; Li, G. Progress in the investigation of the Firmicutes/Bacteroidetes ratio as a potential pathogenic factor in ulcerative colitis. J. Med Microbiol. 2025, 74, 001966. [Google Scholar] [CrossRef] [PubMed]
- Ren, P.; Liu, M.; Wei, B.; Tang, Q.; Wang, Y.; Xue, C. Fucoidan exerts antitumor effects by regulating gut microbiota and tryptophan metabolism. Int. J. Biol. Macromol. 2025, 300, 140334. [Google Scholar] [CrossRef]
- Park, E.-J.; Kim, J.-Y.; Jaiswal, V.; Park, H.-S.; Ki, D.-B.; Lee, Y.-S.; Lee, H.-J. High-molecular-weight Fucoidan exerts an immune-enhancing effect in RAW 264.7 cells and cyclophosphamide-induced immunosuppression rat by altering the gut microbiome. Int. Immunopharmacol. 2024, 139, 112677. [Google Scholar] [CrossRef]
- He, J.; Zhang, P.; Shen, L.; Niu, L.; Tan, Y.; Chen, L.; Zhao, Y.; Bai, L.; Hao, X.; Li, X.; et al. Short-Chain Fatty Acids and Their Association with Signalling Pathways in Inflammation, Glucose and Lipid Metabolism. Int. J. Mol. Sci. 2020, 21, 6356. [Google Scholar] [CrossRef]
- Vinolo, M.A.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of Inflammation by Short Chain Fatty Acids. Nutrients 2011, 3, 858–876. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.N.; Fan, Y.J.; Van Nguyen, T.; Piao, C.H.; Lee, B.-H.; Lee, S.-Y.; Shin, H.S.; Kim, T.-G.; Song, C.H.; Chai, O.H. Undaria pinnatifida extract attenuates combined allergic rhinitis and asthma syndrome by the modulation of epithelial cell dysfunction and oxidative stress. Acta Biochim. Biophys. Sin. 2024, 57, 792–804. [Google Scholar] [CrossRef]
- Men, Q.; Zhang, P.; Zheng, W.; Song, S.; Ai, C. Fucoidan alleviates Salmonella-induced inflammation and mortality by modulating gut microbiota and metabolites, protecting intestinal barrier, and inhibiting NF-κB pathway. Food Biosci. 2023, 56. [Google Scholar] [CrossRef]
- Zaharudin, N.; Tullin, M.; Pekmez, C.T.; Sloth, J.J.; Rasmussen, R.R.; Dragsted, L.O. Effects of brown seaweeds on postprandial glucose, insulin and appetite in humans – A randomized, 3-way, blinded, cross-over meal study. Clin. Nutr. 2021, 40, 830–838. [Google Scholar] [CrossRef]
- Yoshinaga, K.; Mitamura, R. Effects of Undaria pinnatifida (Wakame) on Postprandial Glycemia and Insulin Levels in Humans: A Randomized Crossover Trial. Plant Foods Hum. Nutr. 2019, 74, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Baldeón, M.E.; Chiriboga, D.E.; Davis, J.R.; Sarriés, A.J.; Braverman, L.E. Could dietary seaweed reverse the metabolic syndrome? Asia Pac. J. Clin. Nutr. 2009, 18, 145–154. [Google Scholar]
- Izaola, O.; Primo, D.; Rico Bargues, D.; Belen Martin-Diana, A.; Martinez Villaluenga, C.; Miranda, J.; de Luis, D.A. Effects of a snack enriched with carob and Undaria pinnatifida (wakame) on metabolic parameters in a double blind, randomized clinical trial in obese patients. Nutr. Hosp. 2020, 37, 465–473. [Google Scholar]
- Cooper, R.; Dragar, C.; Elliot, K.; Fitton, J.; Godwin, J.; Thompson, K. GFS, a preparation of Tasmanian Undaria pinnatifida is associated with healing and inhibition of reactivation of Herpes. BMC Complement. Altern. Med. 2002, 2, 11. [Google Scholar] [CrossRef]
- Gueven, N.; Spring, K.J.; Holmes, S.; Ahuja, K.; Eri, R.; Park, A.Y.; Fitton, J.H. Micro RNA Expression after Ingestion of Fucoidan; A Clinical Study. Mar. Drugs 2020, 18, 143. [Google Scholar] [CrossRef]
- Cox, A.J.; Cripps, A.W.; Taylor, P.A.; Fitton, J.H.; West, N.P. Fucoidan Supplementation Restores Fecal Lysozyme Concentrations in High-Performance Athletes: A Pilot Study. Mar. Drugs 2020, 18, 412. [Google Scholar] [CrossRef]
- Olas, K.; Butterweck, H.; Teschner, W.; Schwarz, H.P.; Reipert, B. Immunomodulatory properties of human serum immunoglobulin A: Anti-inflammatory and pro-inflammatory activities in human monocytes and peripheral blood mononuclear cells. Clin. Exp. Immunol. 2005, 140, 478–490. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; Speer, H.; Radcliffe, C.R.; Masocha, K.; Ramsey, R.; West, N.P.; Pyne, D.B. Immunomodulatory effects of fucoidan in recreationally active adult males undertaking 3-weeks of intensified training. J. Sports Sci. 2023, 41, 1875–1882. [Google Scholar] [CrossRef]
- McFadden, B.A.; Vincenty, C.S.; Chandler, A.J.; Cintineo, H.P.; Lints, B.S.; Mastrofini, G.F.; Arent, S.M. Effects of fucoidan supplementation on inflammatory and immune response after high-intensity exercise. J. Int. Soc. Sports Nutr. 2023, 20, 2224751. [Google Scholar] [CrossRef]
- Tay, A.; Jiang, Y.; Signal, N.; O’BRien, D.; Chen, J.; Murphy, R.; Lu, J. Combining mussel with fucoidan as a supplement for joint pain and prediabetes: Study protocol for a randomized, double-blinded, placebo-controlled trial. Front. Nutr. 2022, 9, 1000510. [Google Scholar] [CrossRef]
- Alghazwi, M.; Smid, S.; Karpiniec, S.; Zhang, W. Comparative study on neuroprotective activities of fucoidans from Fucus vesiculosus and Undaria pinnatifida. Int. J. Biol. Macromol. 2019, 122, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Gao, Z.; Zheng, L.; Zhang, C.; Liu, Z.; Yang, Y.; Teng, H.; Hou, L.; Yin, Y.; Zou, X. Protective Effects of Fucoidan on Aβ25–35 and d-Gal-Induced Neurotoxicity in PC12 Cells and d-Gal-Induced Cognitive Dysfunction in Mice. Mar. Drugs 2017, 15, 77. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Moon, I.S.; Goo, T.-W.; Moon, S.-S.; Seo, M. Algae Undaria pinnatifida Protects Hypothalamic Neurons against Endoplasmic Reticulum Stress through Akt/mTOR Signaling. Molecules 2015, 20, 20998–21009. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Kaur, N.; Choubey, V.; Dhingra, N.; Kaur, T. Endoplasmic reticulum stress and its role in various neurodegenerative diseases. Brain Res. 2023, 1826, 148742. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wang, J.; Zhang, P.; Jiang, Z.; Chen, S.; Liang, S.; Ma, T.; Liao, H.; Tan, W.; Niu, C.; et al. Low molecular weight fucoidan induces M2 macrophage polarization to attenuate inflammation through activation of the AMPK/mTOR autophagy pathway. Eur. J. Pharmacol. 2024, 986, 177134. [Google Scholar] [CrossRef]
- Wu, Y.-Q.; Xiong, J.; He, Z.-L.; Yuan, Y.; Wang, B.-N.; Xu, J.-Y.; Wu, M.; Zhang, S.-S.; Cai, S.-F.; Zhao, J.-X.; et al. Metformin promotes microglial cells to facilitate myelin debris clearance and accelerate nerve repairment after spinal cord injury. Acta Pharmacol. Sin. 2021, 43, 1360–1371. [Google Scholar] [CrossRef]
- Jhamandas, J.H.; Wie, M.B.; Harris, K.; MacTavish, D.; Kar, S. Fucoidan inhibits cellular and neurotoxic effects of β-amyloid (Aβ) in rat cholinergic basal forebrain neurons. Eur. J. Neurosci. 2005, 21, 2649–2659. [Google Scholar] [CrossRef]
- Mohibbullah, M.; Hannan, M.A.; Choi, J.-Y.; Bhuiyan, M.M.H.; Hong, Y.-K.; Choi, J.-S.; Choi, I.S.; Moon, I.S. The Edible Marine Alga Gracilariopsis chorda Alleviates Hypoxia/Reoxygenation-Induced Oxidative Stress in Cultured Hippocampal Neurons. J. Med. Food 2015, 18, 960–971. [Google Scholar] [CrossRef] [PubMed]
- Mohibbullah, M.; Haque, M.N.; Khan, M.N.A.; Park, I.-S.; Moon, I.S.; Hong, Y.-K. Neuroprotective effects of fucoxanthin and its derivative fucoxanthinol from the phaeophyte Undaria pinnatifida attenuate oxidative stress in hippocampal neurons. J. Appl. Phycol. 2018, 30, 3243–3252. [Google Scholar] [CrossRef]
- Phuna, Z.X.; Madhavan, P. A reappraisal on amyloid cascade hypothesis: The role of chronic infection in Alzheimer’s disease. Int. J. Neurosci. 2023, 133, 1071–1089. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Zhang, G.; Zhao, Y.T. Fucoidan attenuates the existing allodynia and hyperalgesia in a rat model of neuropathic pain. Neurosci. Lett. 2014, 571, 66–71. [Google Scholar] [CrossRef]
- Che, N.; Ma, Y.; Xin, Y. Protective Role of Fucoidan in Cerebral Ischemia-Reperfusion Injury through Inhibition of MAPK Signaling Pathway. Biomol. Ther. 2017, 25, 272–278. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, M.; He, Z.Z. Low-Molecular-Weight Fucoidan Attenuates Mitochondrial Dysfunction and Improves Neurological Outcome After Traumatic Brain Injury in Aged Mice: Involvement of Sirt3. Cell. Mol. Neurobiol. 2016, 36, 1257–1268. [Google Scholar] [CrossRef]
- Wang, X.; Yi, K.; Zhao, Y. Fucoidan inhibits amyloid-β-induced toxicity in transgenic Caenorhabditis elegans by reducing the accumulation of amyloid-β and decreasing the production of reactive oxygen species. Food Funct. 2018, 9, 552–560. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. GRAS Notice No. 565: Fucoidan from Undaria pinnatifida; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2015.
- U.S. Food and Drug Administration. GRAS Notice No. 661: Fucoidan from Fucus vesiculosus; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2017.
- Kim, K.-J.; Lee, O.-H.; Lee, H.-H.; Lee, B.-Y. A 4-week repeated oral dose toxicity study of fucoidan from the Sporophyll of Undaria pinnatifida in Sprague–Dawley rats. Toxicology 2010, 267, 154–158. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Q.; Song, J. Toxicological evaluation of fucoidan extracted from Laminaria japonica in Wistar rats. Food Chem. Toxicol. 2005, 43, 421–426. [Google Scholar] [CrossRef]
- Kim, K.-J.; Lee, O.-H.; Lee, B.-Y. Genotoxicity studies on fucoidan from Sporophyll of Undaria pinnatifida. Food Chem. Toxicol. 2010, 48, 1101–1104. [Google Scholar] [CrossRef] [PubMed]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M. Pilot clinical study to evaluate the anticoagulant activity of fucoidan. Blood Coagul. Fibrinolysis 2009, 20, 607–610. [Google Scholar] [CrossRef]
- Phull, A.R.; Kim, S.J. Fucoidan as bio-functional molecule: Insights into the anti-inflammatory potential and associated molecular mechanisms. J. Funct. Foods 2017, 38, 415–426. [Google Scholar] [CrossRef]
- Dockal, M.; Till, S.; Knappe, S.; Ehrlich, H.J.; Scheiflinger, F. Anticoagulant Activity and Mechanism of Non-Anticoagulant Sulfated Polysaccharides. Blood 2011, 118, 1208. [Google Scholar] [CrossRef]
- Mathew, L.; Burney, M.; Gaikwad, A.; Nyshadham, P.; Nugent, E.K.; Gonzalez, A.; Smith, J.A. Preclinical Evaluation of Safety of Fucoidan Extracts From Undaria pinnatifida and Fucus vesiculosus for Use in Cancer Treatment. Integr. Cancer Ther. 2016, 16, 572–584. [Google Scholar] [CrossRef]
- Nagamine, T.; Nakazato, K.; Tomioka, S.; Iha, M.; Nakajima, K. Intestinal Absorption of Fucoidan Extracted from the Brown Seaweed, Cladosiphon okamuranus. Mar. Drugs 2014, 13, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.; Chu, F.; Xu, L.; Liang, H.; Song, S.; Ji, A. Use of fluorescein isothiocyanate isomer I to study the mechanism of intestinal absorption of fucoidan sulfate in vivo and in vitro. Biopharm. Drug Dispos. 2018, 39, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Pozharitskaya, O.N.; Shikov, A.N.; Faustova, N.M.; Obluchinskaya, E.D.; Kosman, V.M.; Vuorela, H.; Makarov, V.G. Pharmacokinetic and Tissue Distribution of Fucoidan from Fucus vesiculosus after Oral Administration to Rats. Mar. Drugs 2018, 16, 132. [Google Scholar] [CrossRef]
- Tan, J.; Song, Y.; Wang, J.; Wu, N.; Yue, Y.; Zhang, Q. Pharmacokinetics of fucoidan and low molecular weight fucoidan from Saccharina japonica after oral administration to mice. J. Oceanol. Limnol. 2023, 41, 1900–1909. [Google Scholar] [CrossRef]
- Bai, X.; Zhang, E.; Hu, B.; Liang, H.; Song, S.; Ji, A. Study on Absorption Mechanism and Tissue Distribution of Fucoidan. Molecules 2020, 25, 1087. [Google Scholar] [CrossRef]
- Warttinger, U.; Giese, C.; Harenberg, J.; Krämer, R. Direct quantification of brown algae-derived fucoidans in human plasma by a fluorescent probe assay. arXiv 2016, arXiv:1608.00108. [Google Scholar] [CrossRef]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M.; Kongtawelert, P. A quantitative method to detect fucoidan in human plasma using a novel antibody. Methods Find. Exp. Clin. Pharmacol. 2005, 27, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Tokita, Y.; Nakajima, K.; Mochida, H.; Iha, M.; Nagamine, T. Development of a Fucoidan-Specific Antibody and Measurement of Fucoidan in Serum and Urine by Sandwich ELISA. Biosci. Biotechnol. Biochem. 2010, 74, 350–357. [Google Scholar] [CrossRef]
- Chen, Y.; Li, X.; Gan, X.; Qi, J.; Che, B.; Tai, M.; Gao, S.; Zhao, W.; Xu, N.; Hu, Z. Fucoidan from Undaria pinnatifida Ameliorates Epidermal Barrier Disruption via Keratinocyte Differentiation and CaSR Level Regulation. Mar. Drugs 2019, 17, 660. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Shikov, A.N.; Obluchinskaya, E.D.; Vuorela, H. The Pharmacokinetics of Fucoidan after Topical Application to Rats. Mar. Drugs 2019, 17, 687. [Google Scholar] [CrossRef]
- Chen, B.-R.; Hsu, K.-T.; Li, T.-L.; Chan, Y.-L.; Wu, C.-J. Topical application of fucoidan derived from Cladosiphon okamuranus alleviates atopic dermatitis symptoms through immunomodulation. Int. Immunopharmacol. 2021, 101, 108362. [Google Scholar] [CrossRef]
- Fitton, J.H.; Dell’Acqua, G.; Gardiner, V.-A.; Karpiniec, S.S.; Stringer, D.N.; Davis, E. Topical Benefits of Two Fucoidan-Rich Extracts from Marine Macroalgae. Cosmetics 2015, 2, 66–81. [Google Scholar] [CrossRef]
- Chen, B.-R.; Hsu, K.-T.; Hsu, W.-H.; Lee, B.-H.; Li, T.-L.; Chan, Y.-L.; Wu, C.-J. Immunomodulation and mechanisms of fucoidan from Cladosiphon okamuranus ameliorates atopic dermatitis symptoms. Int. J. Biol. Macromol. 2021, 189, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Obluchinskaya, E.D.; Pozharitskaya, O.N.; Flisyuk, E.V.; Shikov, A.N. Formulation, Optimization and In Vivo Evaluation of Fucoidan-Based Cream with Anti-Inflammatory Properties. Mar. Drugs 2021, 19, 643. [Google Scholar] [CrossRef] [PubMed]
- Bachelet, L.; Bertholon, I.; Lavigne, D.; Vassy, R.; Jandrot-Perrus, M.; Chaubet, F.; Letourneur, D. Affinity of low molecular weight fucoidan for P-selectin triggers its binding to activated human platelets. Biochim. Biophys. Acta Gen. Subj. 2009, 1790, 141–146. [Google Scholar] [CrossRef]
- Zayed, A.; Al-Saedi, D.A.; Mensah, E.O.; Kanwugu, O.N.; Adadi, P.; Ulber, R. Fucoidan’s Molecular Targets: A Comprehensive Review of Its Unique and Multiple Targets Accounting for Promising Bioactivities Supported by In Silico Studies. Mar. Drugs 2023, 22, 29. [Google Scholar] [CrossRef]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Hemmingson, J.A.; Falshaw, R.; Furneaux, R.H.; Thompson, K. Structure and Antiviral Activity of the Galactofucan Sulfates Extracted from Undaria Pinnatifida (Phaeophyta). J. Appl. Phycol. 2006, 18, 185–193. [Google Scholar] [CrossRef]
- Lee, J.; Lee, S.; Synytsya, A.; Capek, P.; Lee, C.W.; Choi, J.W.; Cho, S.; Kim, W.J.; Park, Y.I. Low Molecular Weight Mannogalactofucans Derived from Undaria pinnatifida Induce Apoptotic Death of Human Prostate Cancer Cells In Vitro and In Vivo. Mar. Biotechnol. 2018, 20, 813–828. [Google Scholar] [CrossRef]
- Zheng, W.; Tang, S.; Ren, X.; Song, S.; Ai, C. Fucoidan alleviated colitis aggravated by fiber deficiency through protecting the gut barrier, suppressing the MAPK/NF-κB pathway, and modulating gut microbiota and metabolites. Front. Nutr. 2025, 11, 1462584. [Google Scholar] [CrossRef] [PubMed]
- Obluchinskaya, E.D.; Pozharitskaya, O.N.; Shikov, A.N. In Vitro Anti-Inflammatory Activities of Fucoidans from Five Species of Brown Seaweeds. Mar. Drugs 2022, 20, 606. [Google Scholar] [CrossRef] [PubMed]
| Cell Line | Model | Compound | Tested Concentration | Effective Concentration | Activity | Reference |
|---|---|---|---|---|---|---|
| Murine RAW264.7 cells | LPS stimulation | Low molecular weight UPF | 1, 10 and 100 µg/mL | 1, 10 and 100 µg/mL | Reduced ROS, COX-2 and iNOS; suppressed phosphorylation of p38, ERK1/2, and JNK; and downregulated TNF-α, IL-6, and IL-1β | [28] |
| UPF | 12.5, 25, and 50 µg/mL | 50 µg/mL | Inhibited COX-2 and iNOS, and attenuated production of NO and PGE2 | [40] | ||
| Human osteoarthritic chondrocytes | IL-1β- induced inflammation | UPF | 5, 30, and 100 µg/mL | 5 µg/mL | Inhibited NF-κB activation; and reduced production of NO, PGE2, and IL-6 | [30] |
| UPF | 1, 5, and 30 µg/mL | 5 µg/mL | Downregulated IL-6 and IL-8 (CXCL8); upregulated Nrf-2, HO-1, and SOD-2. | [44] | ||
| THP-1 cells and PBMCs | LPS-induced inflammation | UPF | 10, 50, 100, and 200 µg/mL | 10, 50, and 100 µg/mL | Reduced expression of TNF-α, IL-1β, and IL-6 | [27] |
| Caco-2-Nint cells | SARS-CoV-2 infection | UPF | 0–1000 µg/mL | 200 µg/mL | Reduced IL-6, IFN-α, IFN-γ, and TNF-α | [25] |
| Caco-2 cells | MG-H1-induced inflammation | High molecular weight UPF | 0–1000 µg/mL | 500 µg/mL | Inhibited NF-κB signalling; downregulated IL-6 and TNF-α; and suppressed COX-2 and iNOS expression | [31] |
| IEC-6 cells | H2O2-induced oxidative stress | UPF | 10, 20, 50, and 100 µg/mL | 100 µg/mL | Decreased levels of MDA, and increased CAT, T-SOD, and GSH | [33] |
| Rabbit articular chondrocytes | Antioxidant assays | UPF | 0–500 µg/mL | 2.5–100 µg/mL | Reduced COX-2; scavenged DPPH, nitric oxide and hydroxyl radicals; and exhibited iron chelating activity | [34] |
| Vero cells | H2O2-induced viability reduction | Water-ultrasonicated UPE | 50, 100, 200, and 400 µg/mL | 200 and 400 µg/mL | Suppressed ROS production; decreased Bax; and increased Bcl-2 | [35] |
| Human renal cells | Oxalate crystal-induced inflammation | sulphated Undaria pinnatifida polysaccharides | 50, 100, 150, 200, and 250 µg/mL | 200 µg/mL | Reduced ROS and MCP-1 production; increased SOD content; and decreased secretion of TNF-α and IL-1β | [42] |
| 3T3-L1 adipocytes | Adipogenesis | UPF | 1, 10, and 100 µg/mL | 100 µg/mL | Reduced production of ROS, SOD, and GPx; and downregulated expression of TNF-α, MCP-1 and PAI-1 | [43] |
| Caco-2/THP-1 coculture | Microbial stimulation | UPF | 2.5 g/L | 2.5 g/L | Reduced secretion of IL-8 and MCP-1; decreased TNF-α; and increased IL-6 and IL-10 | [45] |
| Human epidermal keratinocyte cell line | TNF-α or IFN-γ-induced inflammation | UPF | 400 µg/mL | 400 µg/mL | Inhibited expression of TARC, MDC, and RANTES (CCL5) | [46] |
| Model | Animal | Compound | Dose | Treatment | Tissue | Result | Reference |
|---|---|---|---|---|---|---|---|
| HFD-induced obesity | Male and female C57BL/6J mice | UPF | 400 mg/kg/day | Oral supplementation for 10 weeks | Skeletal Muscle | Reduced TNF-α, IL-1β, and IL-6 | [22] |
| Small Intestine | Reduced TNF-α, IL-1β, IL-6, NF-κB, Tjp1, GPR41, and GPR43 | ||||||
| Plasma | Reduced IL-1α and IL-6 | ||||||
| Male and female C57BL/6J mice | UPF | 400 mg/kg/day | Oral supplementation for 10 weeks | Faeces | Increased abundance of Bacteroidetes, Bacteroides/Prevotella, Akkermansia muciniphila, and Lactobacillus; and reduced F/B ratio | [60] | |
| Male BALB/c mice | Sulphated polysaccharides from Undaria pinnatifida | 150 and 300 mg/kg/day | Oral gavage for 10 weeks | Serum | Reduced levels of TC, TG, and LDL-c; increased HDL-c; suppressed FITC and LPS | [59] | |
| Liver | Increased expression of ABCG8, PPAR-γ, PGC-1α and CAT; reduced content of TC, TG, and MDA; and inhibited LPS production | ||||||
| Colon | Increased IL-10 expression; and reduced IL-6 | ||||||
| Faeces | Increased abundance of Bacteroidetes, Bacteroidaceae, and Prevotellaceae; decreased Firmicutes, and Proteobacteria; increased levels acetate, propionate, and butyrate; and reduced F/B ratio | ||||||
| HFD-induced obesity | Male BALB/c mice | Sulphated polysaccharides from Undaria pinnatifida | 100, 300, and 500 mg/kg/day | Oral gavage for 10 weeks | Serum | Reduced levels of TC, TG, LDL-c, LPS, and FITC; and increased HDL-c | [50] |
| Liver | Suppressed levels of LDL-c and MDA; and increased SOD | ||||||
| Colon | Decreased TNF-α; and increased IL-10 | ||||||
| Faeces | Increased Bacteroidetes abundance; reduced Firmicutes, Desulfovibrionales, and Clostridia; and increased levels acetate, propionate, and butyrate | ||||||
| Male C57BL/6J mice | Undaria pinnatifida powder | 10% (w/w) | Oral supplementation for 10 weeks | Faeces | Increased acetic acid, propionic acid, and butyric acid; increased Bacteroidetes, Bacteroidaceae, and Bacteroides; and reduced Firmicutes, Lachnospiraceae, Streptococcaceae, Marinifilaceae | [58] | |
| HFD-induced dyslipidaemia | Male BALB/c mice | UPF | 50 and 100 mg/kg/day | Oral gavage for 8 weeks | Serum | Suppressed levels of TC and LDL-c | [57] |
| Liver | Attenuated levels of TG and CHO | ||||||
| Faeces | Increased Bacteroidetes; and reduced Firmicutes | ||||||
| l-NAME-induced hypertension | Male SD rats | UPF | 20 and 100 mg/kg/day | Oral gavage for 4 weeks | Thoracic aorta | Increased phosphorylation of eNOS and Akt; and decreased levels of iNOS and NO | [47] |
| Serum | Decreased levels of TNF-α and IL-1β | ||||||
| Particulate-matter-induced allergic airway inflammation | Female BALB/c mice | UPF | 100 and 400 mg/kg/day | Oral gavage for 7 days | Lung | Suppressed MDA level; attenuated eosinophils, Gr-1+ cells, F4/80+ macrophage, and CD4+ T cell infiltration; and reduced IL-4, IL-17a, and IL-33 | [48] |
| Trachea | Attenuated eosinophils, Gr-1+ cells, F4/80+ macrophage, and CD4+ T cell infiltration | ||||||
| Serum | Inhibited MDA level; attenuated total IgE; and reduced IL-4 | ||||||
| Testosterone-induced BPH | Male SD rats | UPF | 40 and 400 mg/kg/day | Oral administration for 4 weeks | Prostate | Reduced levels of testosterone and DHT; increased Bax; and reduced Bcl-2 expression | [49] |
| Serum | Decreased levels of IL-1β, TNF-α, testosterone, DHT, and PSA | ||||||
| Fibre deficiency-induced intestinal inflammation | Male BALB/c mice | UPF | 100 and 400 mg/kg/day | Oral supplementation for 4 weeks | Colon | Increased levels of occludin, ZO-1, and claudin-3; reduced expression of TNF-α, IL-6, and IL-1β; increased IL-10; suppressed MDA, MPO, and LPS; promoted CAT and T-SOD; and increased production of acetate, propionate, and butyrate | [33] |
| Male BALB/c mice | UPF | 300 mg/kg/day | Oral gavage for 4 weeks | Colon | Reduced expression of TNF-α and IL-1β; elevated occludin and IL-10; increased levels of T-SOD and CAT; and decreased COX-2, iNOS, and LPS | [61] | |
| Faeces | Increased abundance of Bacteroidetes and Bacteroidales; and decreased Firmicutes, Clostridiales, and Ruminococcaceae | ||||||
| SARS-CoV-2 infection | Female Syrian hamsters | UPF | 100 and 200 mg/kg/day | Oral administration for 6 days | Lung | Downregulated ACE2, IL-6, and TNF-α | [25] |
| Colon | Reduced levels of ACE2, IL-6, and TNF-α | ||||||
| Faeces | Decreased Firmicutes, Limosillactobacter; increased Bacteroidota, Patescibacteria, Allobaculum, Candidatus Saccharimonas, and Ileibacteria; and increased levels of acetate and propionate | ||||||
| MG-H1-induced intestinal inflammation | Male ICR mice | High molecular weight UPF | 25 and 75 mg/kg/day | Oral administration for 4 weeks | Colon | Inhibited MPO activity; and decreased expression of ZO-1, RAGE, and TNF-α | [31] |
| Carrageenan induced inflammation | Male SD rats | UPF | 50 and 150 mg/kg/day | Oral gavage for 25 days | Serum | Decreased production of CAT, POD, and SOD | [34] |
| CCL4-induced oxidative stress | Female SD rats | UPF | 100 mg/kg/day | Intraperitoneal injection for 2 weeks | Serum | Reduced levels of GOT, GPT, ALP, and LDH | [51] |
| Liver | Decreased MDA production; and increased SOD, CAT, and GPx | ||||||
| Broad-spectrum antibiotics (ABX)-induced tumour model | Male C57BL/6 mice | UPF | 400 mg/kg/day | Oral gavage for 3 weeks | Tumour tissue | Reduced levels of CD31+, Bcl2; increased Bax level and CD8+ cells; and decreased CD4+ cells and IDO1 expression | [67] |
| Faeces | Increased abundance of Akkermansia, Bifidobacterium, and Lactobacillus | ||||||
| Cyclophosphamide-induced immunosuppression | Male SD rats | High molecular weight UPF | 50, 100, and 200 mg/kg/day | Oral administration for 4 weeks | Faeces | Increased abundance of Papillibacter cinnamivorans and Desulfomicrobium orale; and reduced Marvinbryantia formatexigens | [68] |
| Ovalbumin-induced CARAS | Male BALB/c mice | Ethanol-extracted UPE | 50, 100, and 200 mg/kg/day | Oral administration for 16 days | Serum | Attenuated IgE and IgG1 levels; and increased IgG2a | [71] |
| Nasal lavage fluid | Increased expression of IFN-γ, SOD, and HO-1; reduced IL-4, IL-5, IL-13, and MDA; and enhanced ZO-1 and occludin | ||||||
| Bronchoalveolar lavage fluid | Decreased levels of IL-4, IL-5, IL-13, and MDA; and increased HO-1 and occludin production | ||||||
| Lung | Increased expression of occludin and ZO-1 | ||||||
| Salmonella typhimurium-induced inflammation | Male BALB/c mice | UPF | 200 and 500 mg/kg/day | Oral administration for 21 days | Colon | Increased expression of occludin and claudin-1; reduced TNF-α, IKBα, p-IKBα, p65, and p-p65; elevated levels of CAT and SOD; and decreased MDA and iNOS | [72] |
| Faeces | Reduced abundance of Proteobacteria, Colidextribacter, and Oscillibacter; increased Parabacteroides, Lactobacillus, Akkermansia, Lachnospiraceae_NK4A136 group and Muribaculum; and enhanced levels of acetate and butyrate |
| Cell Line | Model | Compound | Tested Concentration | Effective Concentration | Activity | Reference |
|---|---|---|---|---|---|---|
| Human RPE cell line | HSV-1-induced Aβ production | HCl- extracted UPF | 100 µg/mL | 100 µg/mL | Inhibited NF-κB phosphorylation, IL-6 expression, and Aβ42 synthesis; and reduced DPPH scavenging and ROS production | [26] |
| Rat PC-12 cells | Aβ-induced neurotoxicity | UPF | 3.125–100 µg/mL | 3.125–100 µg/mL | Increased cell viability; reduced Aβ1–42 aggregation and cell apoptosis; and enhanced neurite outgrowth | [84] |
| PC12 cells | Aβ25–35 and d-Gal-induced neurotoxicity | Water-extracted UPF | 100, 200, and 400 µg/mL | 100, 200, and 400 µg/mL | Improved cell viability; prevented cell apoptosis; reduced levels of cleaved caspase-3, caspase-8, caspase-9, and cytochrome c; increased livin and X-linked apoptosis inhibitor protein expression; and elevated levels of SOD and GSH | [85] |
| Hypothalamic neurons (GT1-7 cells) | Tunicamycin- induced ER stress | Ethanol-extracted UPE | 5–40 µg/mL | 5 µg/mL | Increased cell viability; reduced expression of CHOP and ATF-6; decreased levels of cleaved-PARP and cleaved-caspase-3; and modulated AKT/mTOR signalling | [86] |
| BMDMs | LPS-induced macrophage inflammation | Low molecular weight UPF | 0.0625, 0.125, 0.25, 0.5 mg/mL | 0.125 mg/mL | Reduced CD86+ proportion; increased CD206+ proportion; regulated AMPK/mTOR pathway | [88] |
| Rat basal forebrain cholinergic neurons | Aβ-induced neurotoxicity | UPF | 50 nM–1 µM | 1 µM | Improved neuronal survival; inhibited ROS generation and PKC phosphorylation; and blocked cleavage of caspases 9 and 3 | [90] |
| Rat hippocampal neurons | Hypoxia-mediated oxidative injury | Ethanol- extracted UPE | 5, 15, 30 µg/mL | 15 µg/mL | Reduced ROS formation; increased cell viability; and decreased cytotoxicity | [92] |
| Model | Animal | Compound | Dose | Treatment | Tissue | Result | Reference |
|---|---|---|---|---|---|---|---|
| HFD- induced obesity | Male and female C57BL/6J mice | UPF | 400 mg/kg/day | Oral supplementation for 10 weeks | Hypothalamus | Reduced TNF-α, IL-1β, IL-6, and IFN-γ | [22] |
| Nucleus accumbens | Suppressed Lrrk2, Wfs1, and Ngb | ||||||
| SNL-induced neuropathic pain | Male SPF SD rats | UPF | 15, 50, and 100 mg/kg/day | Intrathecal injection for 10 days | Lumbar spinal cord | Inhibited microglia and astrocyte activation; and reduced expression of GFAP and mac-1 | [94] |
| Spinal dorsal horn | Downregulated expression of TNF-α, IL-1β, and IL-6; and attenuated phosphorylation of ERK | ||||||
| IRI-caused neurological impairment | Male SD rats | UPF | 80 and 160 mg/kg/day | Intraperitoneal injection for 7 days | Ischemic brain | Reduced levels of TNF-α, IL-1β, IL-6, MPO, SOD, MDA, p-p53, p-p38, p-ERK, p-JNK, and Bax; and increased Bcl-2 | [95] |
| D-Gal-induced AD model | Male ICR mice | UPF | 50, 100, and 200 mg/kg/day | Oral administration for 21 days | Brain | Increased levels of Ach, ChAT, and GSH; reduced AChE activity; and decreased Aβ deposition | [85] |
| Serum | Increased levels of SOD and GSH | ||||||
| Controlled cortical impact-induced TBI | Male C57BL/6 mice | Low molecular weight UPF | 10 and 50 mg/kg | Intraperitoneal injection | Brain | Decreased brain oedema and cell apoptosis; reduced generation of MDA, 4-HNE, and ROS; increased levels of CAT, SOD, and GPx; suppressed cytochrome c release; and upregulated Sirt3 expression | [96] |
| Aβ-induced AD model | Caenorhabditis elegans | UPF | 50–500 ng/mL | Bath immersion method | Entire organism | Decreased Aβ deposition, aggregation, and fibrillization; increased expression of pbs-2 and pbs-5; and reduced ROS production | [97] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.; Dwan, C.; Wimmer, B.C.; Ahamed, S.K.; James, F.; Thinley, J.; Wilson, R.; Johnson, L.; Caruso, V. Anti-Inflammatory and Neuroprotective Effects of Undaria pinnatifida Fucoidan. Mar. Drugs 2025, 23, 350. https://doi.org/10.3390/md23090350
Yang C, Dwan C, Wimmer BC, Ahamed SK, James F, Thinley J, Wilson R, Johnson L, Caruso V. Anti-Inflammatory and Neuroprotective Effects of Undaria pinnatifida Fucoidan. Marine Drugs. 2025; 23(9):350. https://doi.org/10.3390/md23090350
Chicago/Turabian StyleYang, Cheng, Corinna Dwan, Barbara C. Wimmer, Sayed Koushik Ahamed, Fionnghuala James, Jigme Thinley, Richard Wilson, Luke Johnson, and Vanni Caruso. 2025. "Anti-Inflammatory and Neuroprotective Effects of Undaria pinnatifida Fucoidan" Marine Drugs 23, no. 9: 350. https://doi.org/10.3390/md23090350
APA StyleYang, C., Dwan, C., Wimmer, B. C., Ahamed, S. K., James, F., Thinley, J., Wilson, R., Johnson, L., & Caruso, V. (2025). Anti-Inflammatory and Neuroprotective Effects of Undaria pinnatifida Fucoidan. Marine Drugs, 23(9), 350. https://doi.org/10.3390/md23090350








