Attenuation of Nicotine Effects on A549 Lung Cancer Cells by Synthetic α7 nAChR Antagonists APS7-2 and APS8-2
Abstract
1. Introduction
2. Results
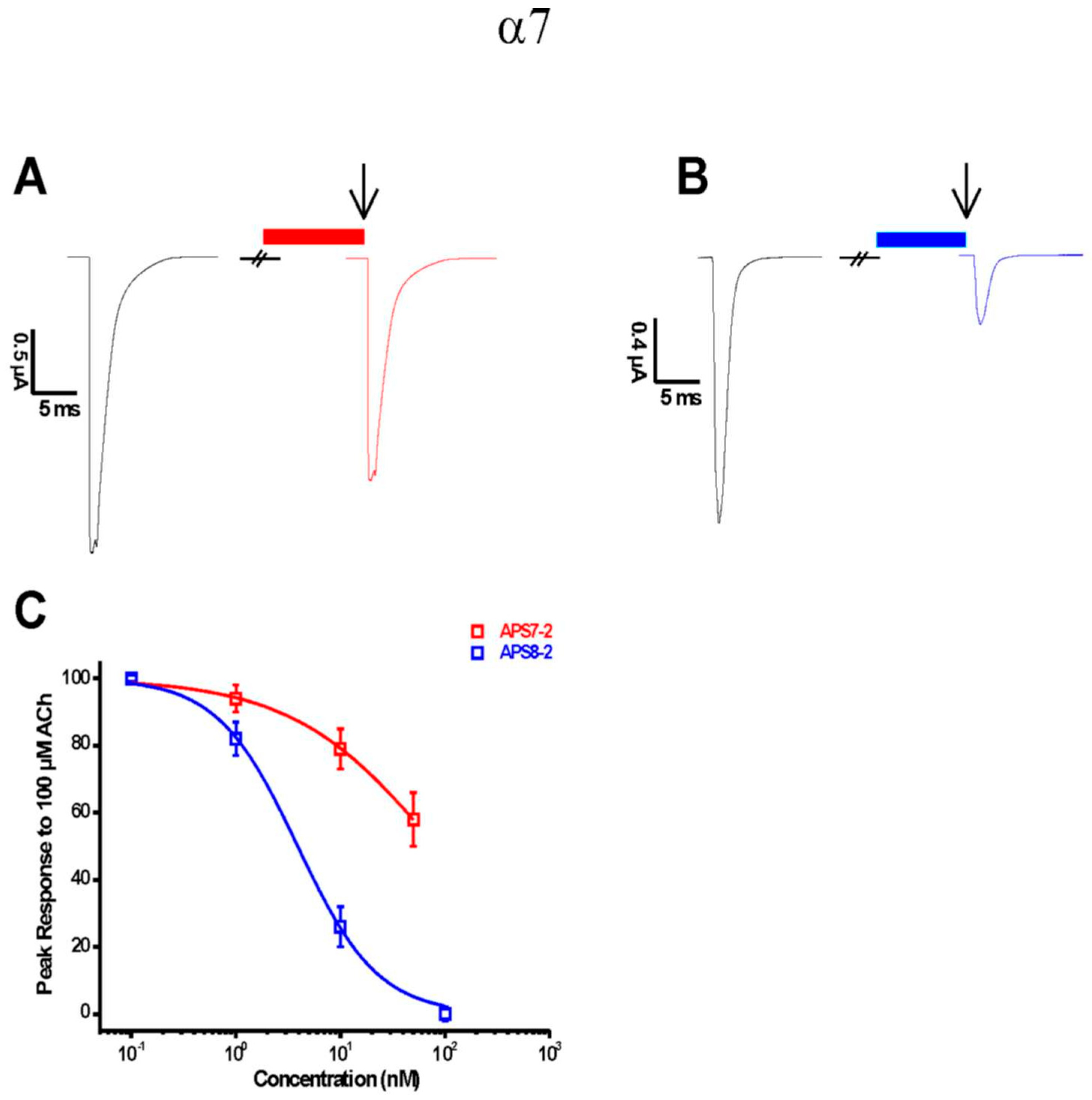
2.1. APS7-2 and APS8-2 Block α7 nAChRs
2.2. Cytotoxicity of APS7-2 and APS8-2 on A549 Lung Cancer Cells
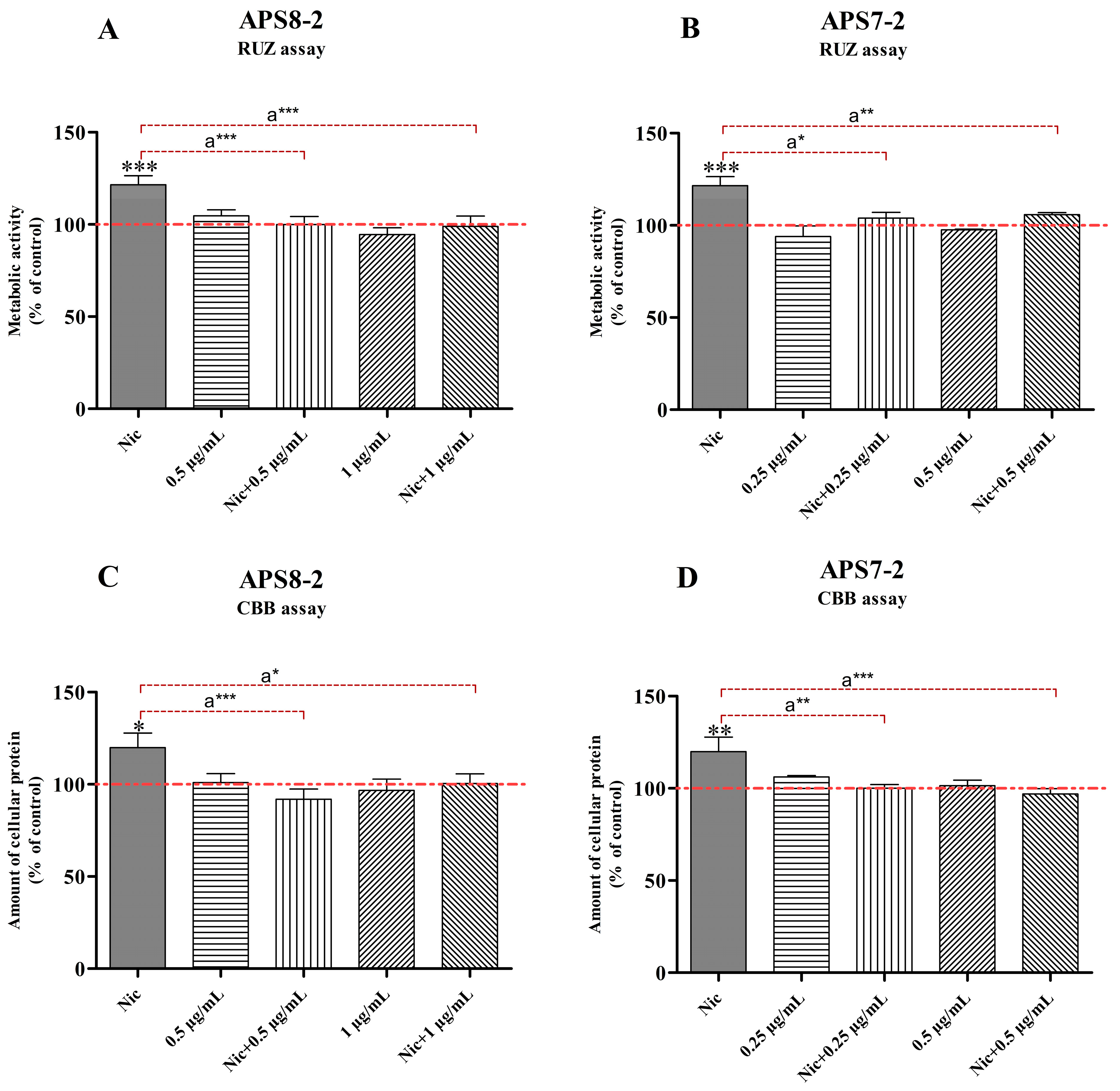
2.3. Attenuation of Nicotine-Induced Increased Cell Viability and Proliferation by APS8-2 and APS7-2
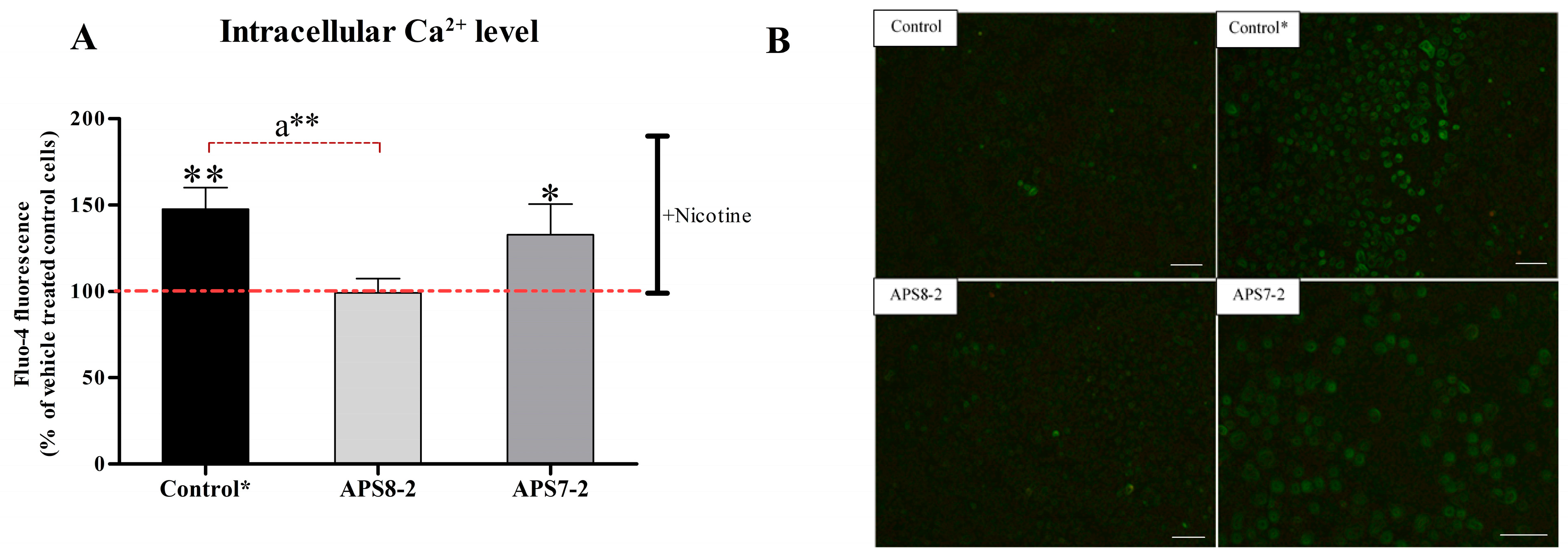
2.4. Prevention of Nicotine-Induced Intracellular Ca2+ Elevation in A549 Lung Cancer Cells by APS8-2
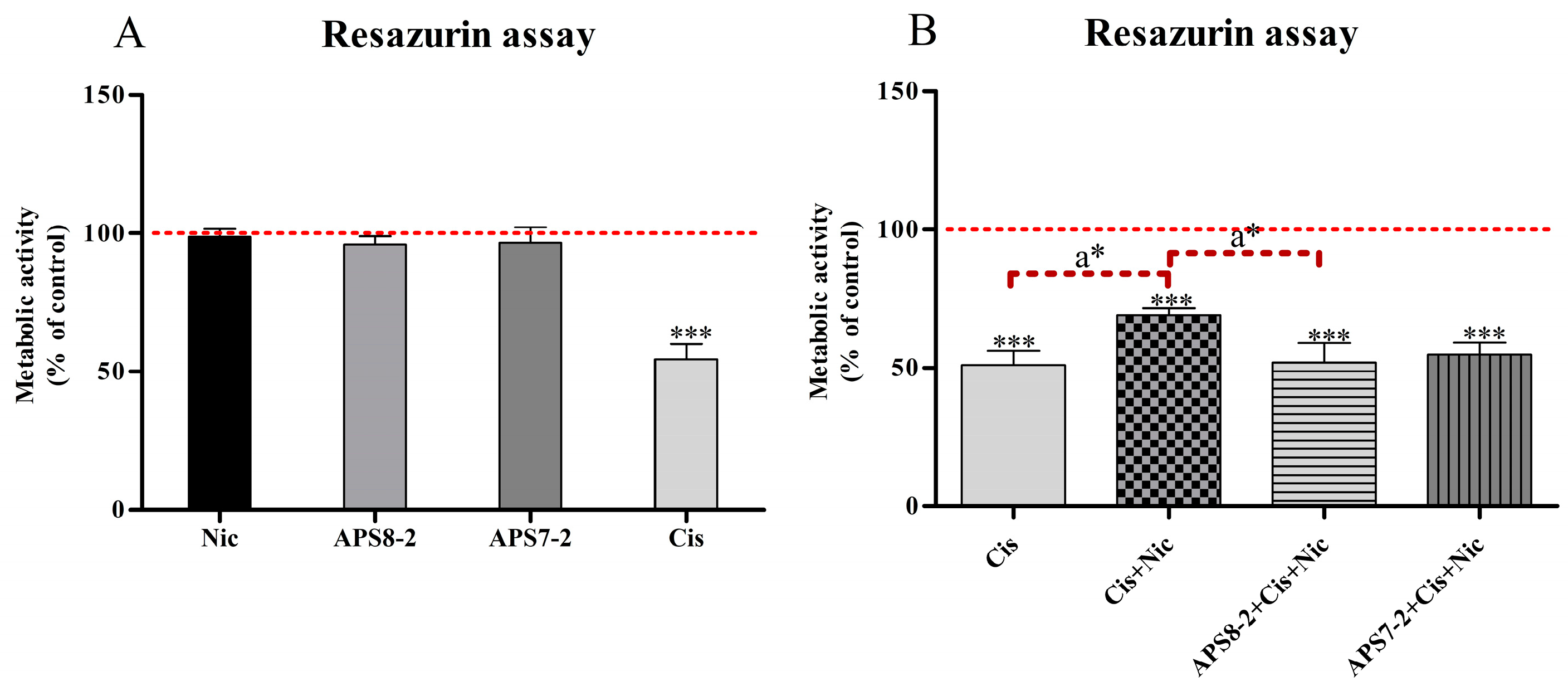
2.5. Modulation of Cisplatin-Induced Cytotoxicity in A549 Cells by Nicotine, APS8-2, or APS7-2
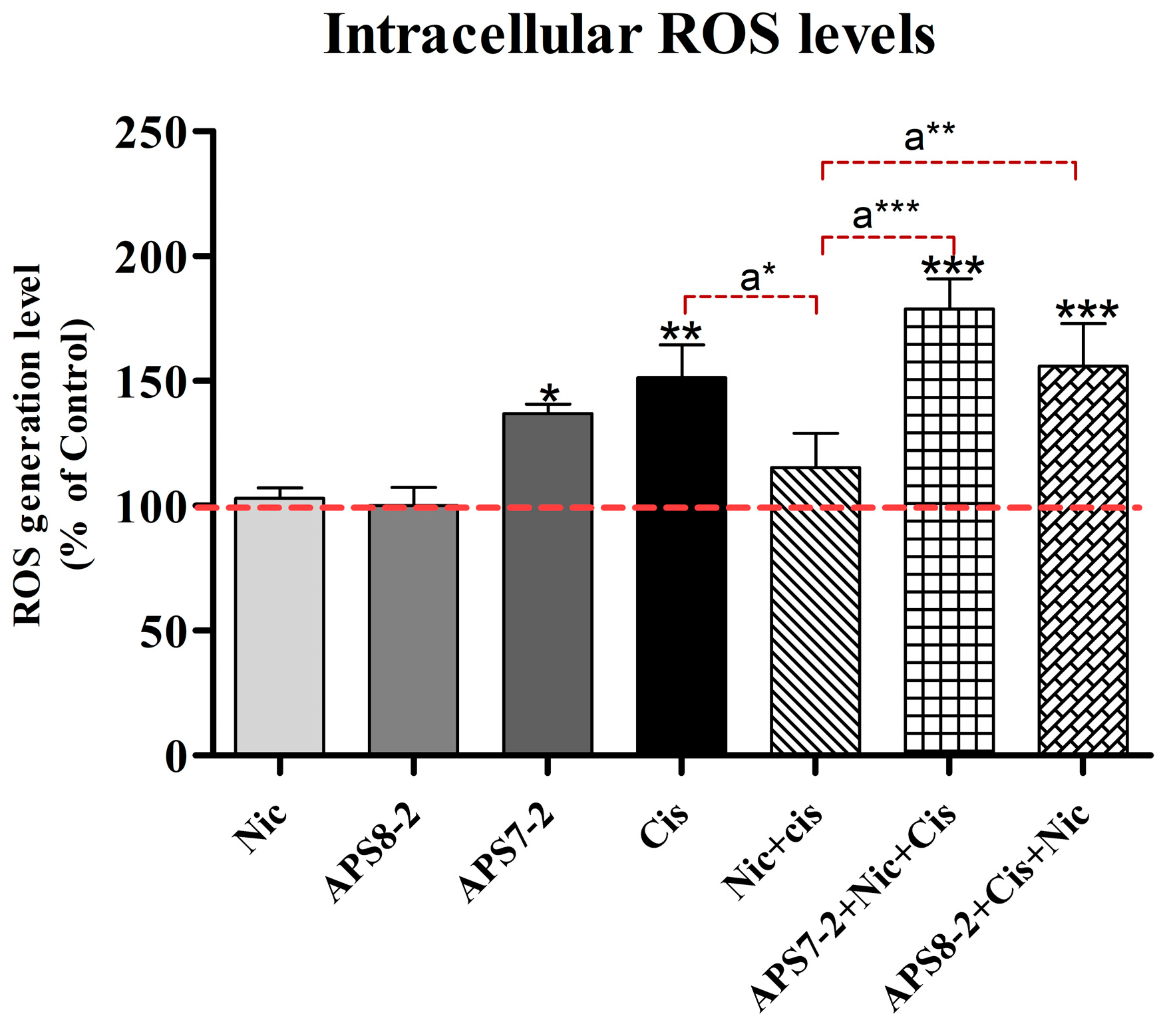
2.6. Modulation of Cisplatin-Induced Reactive Oxygen Species in A549 Cells by Nicotine, APS8-2, or APS7-2
3. Discussion
4. Materials and Methods
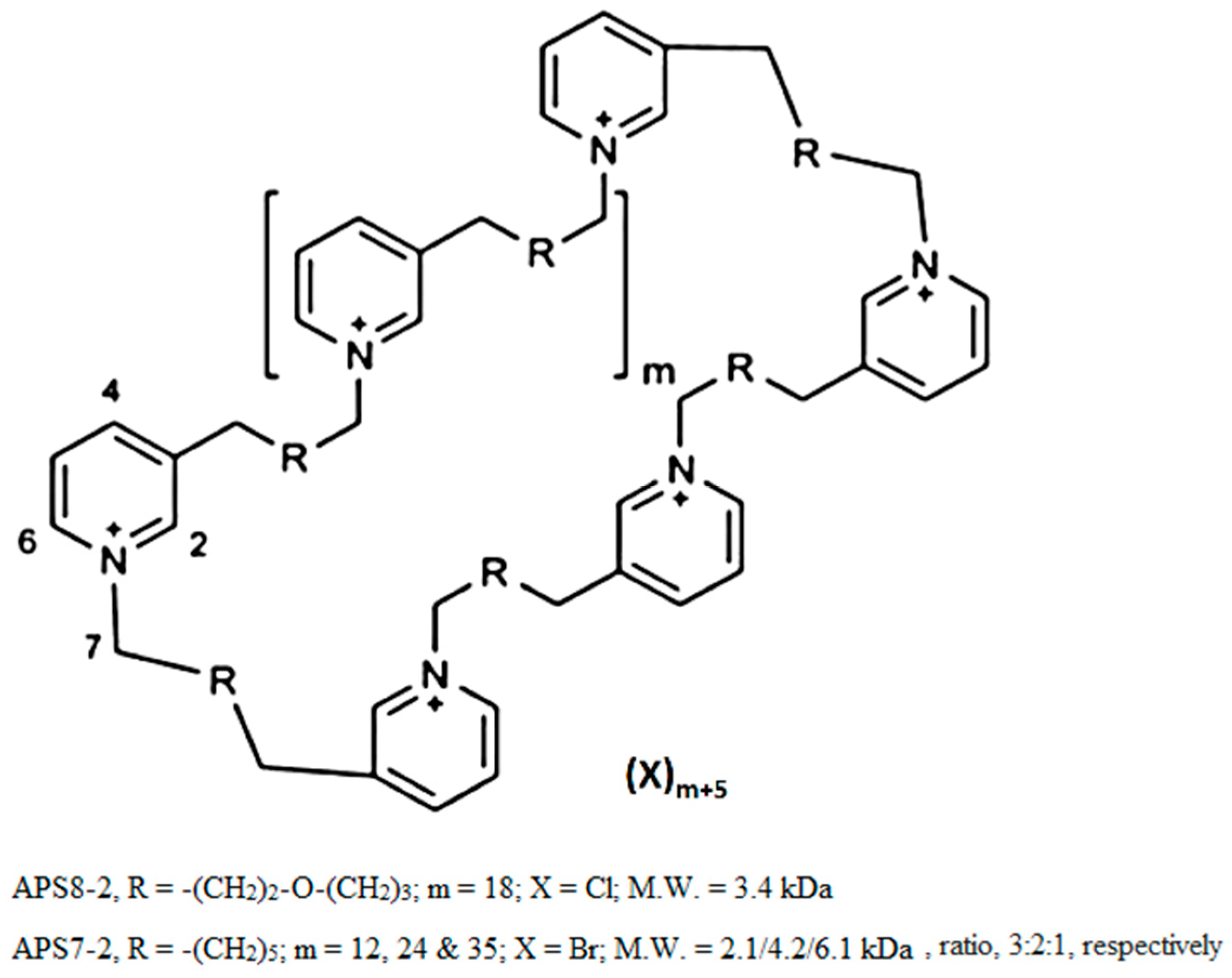
4.1. Chemicals
4.2. Cell Culture
4.3. Expression of nAChRs Xenopus Laevis Oocytes and Electrophysiological Recordings
4.4. Cell Viability and Proliferation Measurements
4.4.1. Cytotoxicity of APS8-2 and APS7-2
4.4.2. Modulation of Cisplatin-Induced Cytotoxicity in A549 Cells by Nicotine, APS8-2, or APS7-2
4.4.3. Attenuation of Nicotine-Induced Increased Cell Viability and Proliferation by APS8-2 and APS7-2
4.5. Calcium Imaging
4.6. Intracellular ROS Measurement
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/cancer/lung/basic_info/risk_factors.htm (accessed on 15 March 2023).
- Kunda, N.K. Antimicrobial Peptides as Novel Therapeutics for Non-Small Cell Lung Cancer. Drug Discov. Today 2020, 25, 238–247. [Google Scholar] [CrossRef]
- Woodman, C.; Vundu, G.; George, A.; Wilson, C.M. Applications and Strategies in Nanodiagnosis and Nanotherapy in Lung Cancer. Semin. Cancer Biol. 2021, 69, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Wang, X.; Nie, S.; Chen, Z.G.; Shin, D.M. Therapeutic Nanoparticles for Drug Delivery in Cancer. Clin. Cancer Res. 2008, 14, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Ashrafizadeh, M.; Zarrabi, A.; Hushmandi, K.; Hashemi, F.; Rahmani, E.; Owrang, M.; Hashemi, F.; Makvandi, P. Lung Cancer Cells and Their Sensitivity/Resistance to Cisplatin Chemotherapy: Role of MicroRNAs and Upstream Mediators. Cell Signal 2021, 78, 109871. [Google Scholar] [CrossRef]
- Vasconcellos, V.F.; Marta, G.N.; da Silva, E.M.K.; Gois, A.F.T.; de Castria, T.B.; Riera, R. Cisplatin versus Carboplatin in Combination with Third-generation Drugs for Advanced Non-small Cell Lung Cancer. Cochrane Database Syst. Rev. 2020, 8, CD009256. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, J.; Liu, Z.Q.; Xiao, R.; Xu, Q.P.; Xiong, W.; Xu, L.; Cai, L.; He, F. Association between Smoking and Environmental Tobacco Smoke with Lung Cancer Risk: A Case–Control Study in the Fujian Chinese Population. J. Public Health 2022, 30, 2047–2057. [Google Scholar] [CrossRef]
- Talhout, R.; Schulz, T.; Florek, E.; van Benthem, J.; Wester, P.; Opperhuizen, A. Hazardous Compounds in Tobacco Smoke. Int. J. Environ. Res. Public Health 2011, 8, 613–628. [Google Scholar] [CrossRef]
- Mucchietto, V.; Fasoli, F.; Pucci, S.; Moretti, M.; Benfante, R.; Mclntosh, M.; Clementi, F.; Gotti, C. A9- and A7-Containing Receptors Mediate the pro-Proliferative Effects of Nicotine in the A549 Adenocarcinoma Cell Line. Br. J. Pharmacol. 2018, 175, 1957–1972. [Google Scholar] [CrossRef]
- Zovko, A.; Viktorsson, K.; Lewensohn, R.; Kološa, K.; Filipič, M.; Xing, H.; Kem, W.R.; Paleari, L.; Turk, T. APS8, a Polymeric Alkylpyridinium Salt Blocks A7 NAChR and Induces Apoptosis in Non-Small Cell Lung Carcinoma. Mar. Drugs 2013, 11, 2574–2594. [Google Scholar] [CrossRef]
- Hsu, C.; Tsai, K.-Y.; Su, Y.; Chien, C.; Chen, Y.; Wu, Y.; Liu, S.-Y.; Shieh, Y.-S. A7-Nicotine Acetylcholine Receptor Mediated Nicotine Induced Cell Survival and Cisplatin Resistance in Oral Cancer. Arch. Oral. Biol. 2020, 111, 104653. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Lee, C.H.; Ho, Y.S. Nicotinic Acetylcholine Receptor-Based Blockade: Applications of Molecular Targets for Cancer Therapy. Clin. Cancer Res. 2011, 17, 3533–3541. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, P.; Kabbani, N. Ionotropic and Metabotropic Responses by Alpha 7 Nicotinic Acetylcholine Receptors. Pharmacol. Res. 2023, 197, 106975. [Google Scholar] [CrossRef]
- Schuller, H.M. Regulatory Role of the A7nAChR in Cancer. Curr. Drug Targets 2012, 13, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Schaal, C.; Chellappan, S. Nicotine-Mediated Regulation of Nicotinic Acetylcholine Receptors in Non-Small Cell Lung Adenocarcinoma by E2F1 and STAT1 Transcription Factors. PLoS ONE 2016, 11, e0156451. [Google Scholar] [CrossRef] [PubMed]
- Grandič, M.; Bajuk, B.P.; Sepčić, K.; Košorok, M.D.; Frangež, R. Effects of Synthetic Analogues of Poly-APS on Contractile Response of Porcine Coronary Arteries. Toxicol. Vitr. 2013, 27, 627–631. [Google Scholar] [CrossRef]
- Piazza, V.; Dragić, I.; Sepčić, K.; Faimali, M.; Garaventa, F.; Turk, T.; Berne, S. Antifouling Activity of Synthetic Alkylpyridinium Polymers Using the Barnacle Model. Mar. Drugs 2014, 12, 1959–1976. [Google Scholar] [CrossRef]
- Grandič, M.; Sepčič, K.; Turk, T.; Juntes, P.; Frangež, R. In Vivo Toxic and Lethal Cardiovascular Effects of a Synthetic Polymeric 1,3-Dodecylpyridinium Salt in Rodents. Toxicol. Appl. Pharmacol. 2011, 255, 86–93. [Google Scholar] [CrossRef]
- Berne, S.; Čemažar, M.; Frangež, R.; Juntes, P.; Kranjc, S.; Berne, S.; Cemažar, M.; Turk, T. APS8 Delays Tumor Growth in Mice by Inducing Apoptosis of Lung Adenocarcinoma Cells Expressing High Number of A7 Nicotinic Receptors. Mar. Drugs 2018, 16, 367. [Google Scholar] [CrossRef]
- Friedman, J.R.; Richbart, S.D.; Merritt, J.C.; Brown, K.C.; Nolan, N.A.; Akers, A.T.; Jamie, K.; Robateau, Z.R.; Miles, S.L.; Dasgupta, P.; et al. Acetylcholine Signaling System in Progression of Lung Cancers. Pharmacol. Ther. 2019, 194, 222–254. [Google Scholar] [CrossRef]
- Mei, D.; Zhao, L.; Chen, B.; Zhang, X.; Wang, X.; Yu, Z.; Ni, X.; Zhang, Q. α-Conotoxin ImI-Modified Polymeric Micelles as Potential Nanocarriers for Targeted Docetaxel Delivery to A7-NAChR Overexpressed Non-Small Cell Lung Cancer. Drug Deliv. 2018, 25, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Grando, S.A. Connections of Nicotine to Cancer. Nat. Rev. Cancer 2014, 14, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Zovko, A.; Vaukner Gabrič, M.; Sepčić, K.; Pohleven, F.; Jaklič, D.; Gunde-Cimerman, N.; Lu, Z.; Edrada-Ebel, R.A.; Houssen, W.E.; Mancini, I.; et al. Antifungal and Antibacterial Activity of 3-Alkylpyridinium Polymeric Analogs of Marine Toxins. Int. Biodeterior. Biodegradation 2012, 68, 71–77. [Google Scholar] [CrossRef]
- Grandič, M.; Frangež, R. Pathophysiological Effects of Synthetic Derivatives of Polymeric Alkylpyridinium Salts from the Marine Sponge, Reniera Sarai. Mar. Drugs 2014, 12, 2408–2421. [Google Scholar] [CrossRef]
- Bai, S.; Zhi, Y.; Zhou, H.; Wen, W.; Lv, Y.; Dong, Y.; Hou, X.; Tan, X.; Wu, J.; Liu, L.; et al. Inhibitory Effect of Sinomenine on Lung Cancer Cells via Negative Regulation of A7 Nicotinic Acetylcholine Receptor. J. Leukoc. Biol. 2021, 109, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Witayateeraporn, W.; Arunrungvichian, K.; Pothongsrisit, S.; Doungchawee, J.; Vajragupta, O.; Pongrakhananon, V. A7-Nicotinic Acetylcholine Receptor Antagonist QND7 Suppresses Non-Small Cell Lung Cancer Cell Proliferation and Migration via Inhibition of Akt/MTOR Signaling. Biochem. Biophys. Res. Commun. 2020, 521, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Cheuk, I.W.Y.; Shin, V.Y.; Kwong, A.; Kong, H. Acetylcholine Receptors: Key Players in Cancer Development. Surg. Oncol. 2019, 31, 46–53. [Google Scholar] [CrossRef]
- Hong, W.; Peng, G.; Hao, B.; Liao, B.; Zhao, Z.; Zhou, Y.; Peng, F.; Ye, X.; Huang, L.; Zheng, M.; et al. Nicotine-Induced Airway Smooth Muscle Cell Proliferation Involves TRPC6-Dependent Calcium Influx via A7 NAChR. Cell. Physiol. Biochem. 2017, 43, 986–1002. [Google Scholar] [CrossRef]
- Ma, X.; Jia, Y.; Zu, S.; Li, R.; Jia, Y.; Zhao, Y.; Xiao, D.; Dang, N.; Wang, Y. Alpha5 Nicotinic Acetylcholine Receptor Mediates Nicotine-Induced HIF-1α and VEGF Expression in Non-Small Cell Lung Cancer. Toxicol. Appl. Pharmacol. 2014, 278, 172–179. [Google Scholar] [CrossRef]
- Dasgupta, P.; Rizwani, W.; Pillai, S.; Kinkade, R.; Kovacs, M.; Rastogi, S.; Banerjee, S.; Carless, M.; Kim, E.; Coppola, D.; et al. Nicotine Induces Cell Proliferation, Invasion and Epithelial-mesenchymal Transition in a Variety of Human Cancer Cell Lines. Int. J. Cancer 2009, 124, 36–45. [Google Scholar] [CrossRef]
- Dasgupta, P.; Rastogi, S.; Pillai, S.; Ordonez-Ercan, D.; Morris, M.; Haura, E.; Chellappan, S. Nicotine Induces Cell Proliferation by β-Arrestin–Mediated Activation of Src and Rb–Raf-1 Pathways. J. Clin. Investig. 2006, 116, 2208–2217. [Google Scholar] [CrossRef]
- Kyte, S.L.; Gewirtz, D.A. The Influence of Nicotine on Lung Tumor Growth, Cancer Chemotherapy, and Chemotherapy-Induced Peripheral Neuropathy. J. Pharmacol. Exp. Ther. 2018, 366, 303–313. [Google Scholar] [CrossRef]
- Choi, Y.M.; Kim, H.K.; Shim, W.; Anwar, M.A.; Kwon, J.W.; Kwon, H.K.; Kim, H.J.; Jeong, H.; Kim, H.M.; Hwang, D.; et al. Mechanism of Cisplatin-Induced Cytotoxicity Is Correlated to Impaired Metabolism Due to Mitochondrial ROS Generation. PLoS ONE 2015, 10, e0135083. [Google Scholar] [CrossRef]
- Dong, Y.; Bi, W.; Zheng, K.; Zhu, E.; Shaoxiang, W.; Xiong, Y.; Chang, J.; Jiang, J.; Liu, B.; Lu, H.; et al. Nicotine Prevents Oxidative Stress-Induced Hippocampal Neuronal Injury Through A7-NAChR/Erk1/2 Signaling Pathway. Front. Mol. Neurosci. 2020, 13, 204. [Google Scholar] [CrossRef]
- Kononenko, V.; Drobne, D. In Vitro Cytotoxicity Evaluation of the Magnéli Phase Titanium Suboxides (TixO2x−1) on A549 Human Lung Cells. Int. J. Mol. Sci. 2019, 20, 196. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joukhan, A.; Kononenko, V.; Bele, T.; Sollner Dolenc, M.; Peigneur, S.; Pinheiro-Junior, E.L.; Tytgat, J.; Turk, T.; Križaj, I.; Drobne, D. Attenuation of Nicotine Effects on A549 Lung Cancer Cells by Synthetic α7 nAChR Antagonists APS7-2 and APS8-2. Mar. Drugs 2024, 22, 147. https://doi.org/10.3390/md22040147
Joukhan A, Kononenko V, Bele T, Sollner Dolenc M, Peigneur S, Pinheiro-Junior EL, Tytgat J, Turk T, Križaj I, Drobne D. Attenuation of Nicotine Effects on A549 Lung Cancer Cells by Synthetic α7 nAChR Antagonists APS7-2 and APS8-2. Marine Drugs. 2024; 22(4):147. https://doi.org/10.3390/md22040147
Chicago/Turabian StyleJoukhan, Ahmad, Veno Kononenko, Tadeja Bele, Marija Sollner Dolenc, Steve Peigneur, Ernesto Lopes Pinheiro-Junior, Jan Tytgat, Tom Turk, Igor Križaj, and Damjana Drobne. 2024. "Attenuation of Nicotine Effects on A549 Lung Cancer Cells by Synthetic α7 nAChR Antagonists APS7-2 and APS8-2" Marine Drugs 22, no. 4: 147. https://doi.org/10.3390/md22040147
APA StyleJoukhan, A., Kononenko, V., Bele, T., Sollner Dolenc, M., Peigneur, S., Pinheiro-Junior, E. L., Tytgat, J., Turk, T., Križaj, I., & Drobne, D. (2024). Attenuation of Nicotine Effects on A549 Lung Cancer Cells by Synthetic α7 nAChR Antagonists APS7-2 and APS8-2. Marine Drugs, 22(4), 147. https://doi.org/10.3390/md22040147








